1. Introduction
Hydrothermal systems in the deep ocean have been proposed to be among the most credible settings for the biological origins of life [
1,
2]. Indeed, such alkaline systems have been suggested as the source of the evolution of primordial metabolism [
1,
3]. The marine environment possesses a rich pool of bioactive ingredients with numerous purported health benefits [
4]. Moreover, novel marine-based bioactive compounds might also augment human performance, especially in those who engage in physical activity [
4]. Administration of deep ocean minerals (DOM) has previously demonstrated positive effects on exercise performance in animal [
5,
6] and human models [
7,
8,
9]. For example, Hou et al. [
7] reported that DOM extracted from 662 m below the ocean surface facilitated substantially quicker recovery from a prolonged bout of exercise at 30 °C, which induced 3% reduction in body mass of healthy males. More specifically, maximal oxygen uptake (V
·O
2MAX) increased by ~2% after 4 h compared to baseline with DOM, whilst it was reduced by ~11% with placebo. After 24 h, V
·O
2MAX increased by ~6% compared to baseline with DOM, whilst it was ~13% lower with placebo. The authors opined that specific elements in DOM, such as boron, magnesium and rubidium, were likely to have contributed to the observed ergogenic effects. Interestingly, using a similar protocol, Stasiule et al. [
9] reported comparable differences in aerobic capacity of healthy females 4 h after supplementation with (deep) mineral water (DMW) extracted from a well at a depth of 689 m or placebo (purified tap water). Similarly, these authors also postulated that the minerals and trace elements that constituted DMW may have worked collaboratively to recover normal human performance [
9].
Given the importance of optimising recovery for individuals who train/compete multiple times per day with limited time between bouts of exercise [
10,
11], and the fact that it is presently unknown whether DOM can improve high-intensity intermittent running capacity, which is integral to many team sports such as soccer [
12], this study examined the effects of DOM on high-intensity intermittent running capacity in soccer players after short-term recovery from an initial bout of prolonged high-intensity exercise in thermoneutral environmental conditions. As such, the primary performance outcome measure for this study was high-intensity intermittent running endurance capacity with secondary outcomes including various metabolic (i.e., blood glucose and blood lactate), anthropometric (i.e., body mass), and physiological (i.e., urine osmolality) markers. We hypothesised that DOM would augment high-intensity intermittent running endurance capacity with other variables remaining similar between treatments.
3. Discussion
Oral ingestion of DOM has previously demonstrated positive effects on exercise performance in humans [
7,
8,
9], although such benefits have been observed during exercise after recovery from an initial bout of prolonged dehydrating exercise in the heat (i.e., 30 °C). Therefore, the aim of this study was to examine whether DOM facilitated ergogenic benefits during exercise after recovery from an initial bout of prolonged exercise in more thermoneutral conditions (i.e., 20 °C). In support of our hypothesis, DOM increased high-intensity intermittent running capacity in recreationally active soccer players at the group level by 56%. However, assuming a conservative daily variation of ~10% for a high-intensity exercise capacity test [
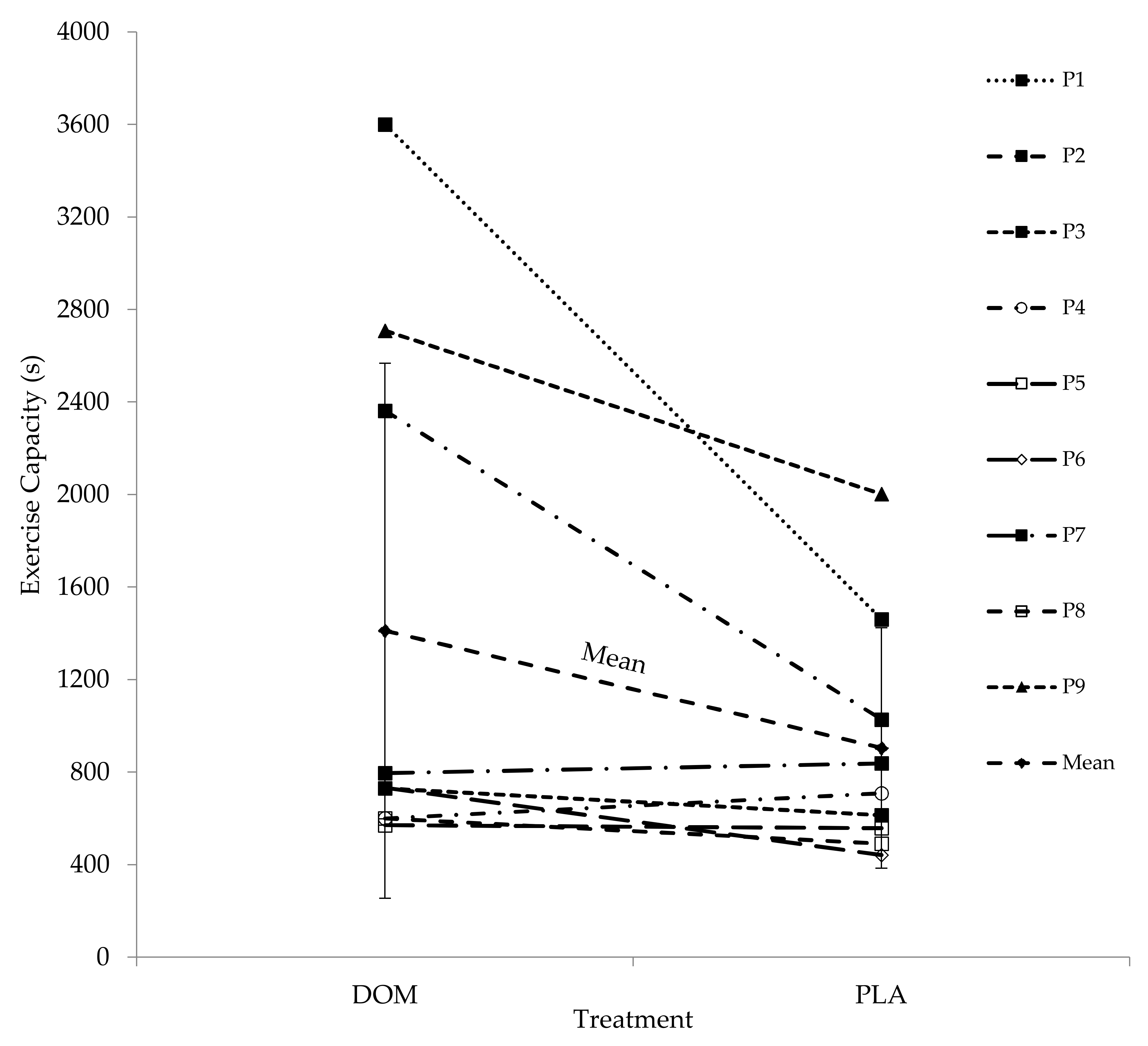
13], and after accounting for any potential order effects between Trials 1 and 2 of ~20%, we suggest the minimum likely ergogenic effects after DOM at the group level to be ~25%. In summary, oral ingestion of DOM seems likely to promote ergogenic benefits for recreationally active soccer players after recovery from prolonged exercise in thermoneutral environmental conditions; however, given the somewhat variable results, an individual approach to supplementation is warranted.
In the present study, body mass was greater at baseline compared to after 60 min running at 75% V
·O
2PEAK after 2 h recovery and at the end of the high-intensity intermittent exercise capacity test. The majority of this reduction in body mass over time is most likely indicative of sweat losses. As prolonged endurance exercise might induce an increase in magnesium (Mg) excretion via sweat and urine [
6], it is plausible that participants in the present study exhibited marked Mg losses. Additionally, amongst several other factors such as decreases in food crop magnesium content and the availability of refined and processed foods, the vast majority of people in modern societies are at risk for magnesium deficiency regardless. Furthermore, a normal serum Mg level does not necessarily rule out Mg deficiency [
14]. As Mg is involved with numerous fundamental biological processes, such as energy production, electrolyte regulation and oxygen uptake, and with evidence that marginal Mg deficiency impairs performance and augments the negative consequences of strenuous exercise [
15], it seems likely that the high Mg concentration in DOM (
Table 6) has played some role in the observed ergogenic effects. Indeed, the mean amount of Mg consumed from DOM in the present study (~236 mg) was ~80% of the participant’s 300 mg recommended daily allowance (RDA) [
16]. Moreover, a recent review provides further support for the potential of magnesium supplementation to improve aspects related to both aerobic and anaerobic exercise [
17].
Boron (B) has been shown to boost Mg absorption, reduce levels of inflammatory biomarkers such as high-sensitivity C-reactive protein (hs-CRP), tumour necrosis factor α (TNF-α), and interleukin 6 (IL-6), as well as contribute to the synthesis and activity of crucial biomolecules such as nicotinamide adenine dinucleotide (NAD
+; [
18]). Therefore, the B in DOM could work synergistically with elevated levels of Mg, or it could work individually through the mediation of exercise-induced inflammation and/or through facilitating key biochemical reactions where NAD
+ is crucial, such as ATP production and calcium signalling [
18]. Albeit in an older population and using a different exercise modality than the present study, we have previously shown that DOM can mediate the post-exercise neutrophil to lymphocyte ratio (NLR; [
19]), which is associated with various cytokines including IL-6 [
20]. Furthermore, both acute (one day) and chronic (seven days) B supplementation has been shown to be tolerated well in humans with chronic supplementation displaying large reductions in inflammatory cytokines and a large increase in free testosterone [
21]. However, it should be added that the mean dosage used in the present study (~0.74 mg) was substantially less than the acute dosage used in the aforementioned study (10 mg; [
21]), and that using similar hardness of DOM (713) to that of the present study (704), Hou et al. [
7] reported no differences in testosterone between DOM and PLA.
Rubidium (Rb), present in all human tissues and ranging from 8 to 30 mg·kg
−1 [
22], has been referred to as a ‘biological proxy’ for potassium (K
+) [
23]. It is well known that there is a rapid and marked increase of extracellular K
+ and considerable loss of K
+ from active muscle during high-intensity exercise. Thus, it is likely that the resultant electrical changes induced by ionic shifts across the muscle surface membrane are principal contributors of fatigue [
24]. Therefore, it is possible that the high levels of Rb in DOM (
Table 6) might proxy for K
+ and facilitate a prolonged status of optimal electrical activity and, hence, contraction in the muscle, thus delaying fatigue. Furthermore, Rb has been found to be 49% and 34% greater in professional footballers than sedentary controls and long-distance runners, respectively [
25]. However, although Rb is associated with the K cycle, it does not necessarily substitute for it [
22,
25] and, thus, other mechanisms might be responsible. Besides, Rb has been implicated in brain function, although specific roles are yet to be identified [
22,
25].
In support of our hypothesis, the present study found no differences in urine osmolality between treatments. This is in contrast to Keen et al. [
8] who reported that DOM returned salivary osmolality to baseline values approximately two-fold quicker than spring water and a carbohydrate-based sports drink. However, a variety of methodological differences between studies are likely to explain the observed difference. For example, the participants in the Keen et al. [
8] study had exercised in the heat to achieve approximately −3% reduction in body mass, and subsequent rehydration was based on prescriptive rather than ad libitum intake. Furthermore, salivary osmolality demonstrates considerable day-to-day measurement variability and in magnitude of response to hypohydration; thus, its use as a method of assessing hydration status has been questioned [
26]. However, despite differences in urine osmolality between studies, Keen et al. [
8] also reported physical performance improvement with DOM. Ingesting DOM facilitated the greatest recovery of post-exercise knee extension peak torque, although participants failed to fully recover baseline peak torque regardless of treatment. In contrast to our original hypothesis, we observed a difference in body mass at baseline between DOM and PLA. However, the difference of 0.28 kg was well below the typical daily variation of body mass measurement in young males (0.6 kg) previously reported [
27]. Thus, we do not believe differences in body mass had any major influence on our results.
In support of our hypothesis, there were no differences in blood lactate and blood glucose between DOM and PLA. This was similar to Wei et al. [
19], who reported no differences in either metabolite after 15 min cycling at 75% V
·O
2MAX. Importantly the absolute values of blood lactate after 60 min running at 75% V
·O
2PEAK in the present study were similar to those observed in various other studies examining blood lactate responses in the second half of a soccer match (3.7 to 4.7 mmol L
−1; [
28]), highlighting that our protocol had a similar metabolic profile. However, with various methodological differences between studies and a lack of other data, the influence of DOM on blood lactate and blood glucose requires further investigation.
Arguably the most intriguing finding from the metabolic data relates to a specific participant who, after completion of this study, was diagnosed with type I diabetes. The participant confirmed there was a familial history of this, which was undisclosed prior to participation (P2;
Table 5, bottom panel). Strikingly, at the end of the recovery period, blood glucose had increased by 65% (+3.69 mmol L
−1) after DOM but 117% for PLA (+8.23 mmol L
−1) when compared to values at the end of 60 min running at 75% V
·O
2PEAK. Excluding P2, the mean increase in blood glucose between these time points was 18% and 22% for DOM and PLA, respectively. Furthermore, only part of the difference observed for P2 might be explained by the differences between treatments of −1.39 mmol L
−1 at baseline and of +1.35 mmol L
−1 after completion of 60 min running at 75% V
·O
2PEAK for PLA vs. DOM, respectively. The potential of DOM to facilitate greater glucose uptake is supported by Ha et al. [
29], who reported that DOM improved glucose tolerance and suppressed hyperglycaemia via modulation of glucose metabolism in streptozotocin-induced diabetic mice. The potential antidiabetic effects of DOM (or derivatives of) have also been reported in various other animal models [
30,
31,
32]. Furthermore, a number of the elements contained in DOM, such as vanadium [
33] and chromium [
34], have also been strongly linked to improved glucose metabolism.
This research has a number of strengths. Firstly, we are the first to report ergogenic benefits after oral ingestion of DOM in humans in thermoneutral environmental conditions; thus, we expand on the potential pool of individuals who might benefit from this approach. Additionally, by incorporating fluid consumption based on an individual participant’s preference in familiarisation trials, we feel this provides wider applied ecological validity. This research is also not without limitation. Even though the sample size is consistent with seminal papers in this field [
7,
9], we acknowledge the relative lack of statistical power. Moreover, we acknowledge that the analysis on the type I diabetic participant is somewhat speculative and, thus, should be treated with caution. That said, we believe it could serve as useful initial case study data and therefore could be a translational platform to research examining the potential effects of DOM on (dysfunctional) glucose metabolism in humans during/post exercise.
In summary, in support of our hypothesis, DOM increased high-intensity intermittent running capacity in soccer players after short-term recovery from an initial bout of prolonged exercise at the group level by ~25%. To date, the mechanism for this effect is unclear, and further work is required to establish the mechanism(s) behind the ergogenic effects of DOM to maximise its potential performance and health benefits in the wider community.