Marine Bacteria, A Source for Alginolytic Enzyme to Disrupt Pseudomonas aeruginosa Biofilms
Abstract
1. Introduction
2. Results
2.1. Isolation of Bacterial Strains with Alginolytic Activity
2.2. Antibiofilm Activity of Cell-Free Supernatants of Seven Isolates
2.3. Alginate Lyase Production and Purification
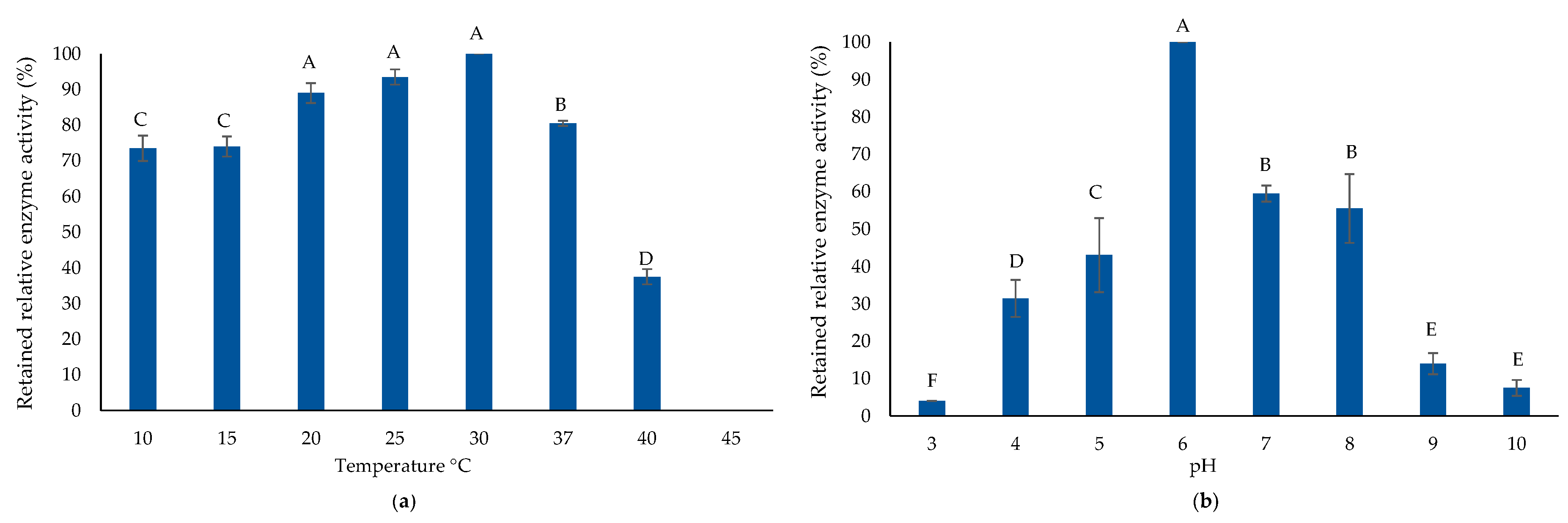
2.4. Biochemical Properties of Purified AlyP1400
2.5. Liquid Chromatography Tandem-Mass Spectrometry (LC-MS/MS) Enzyme Analysis
2.6. Inhibitory Effect of AlyP1400 against P. aeruginosa PA14 Biofilm
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Growth Conditions
4.2. Isolation of Alginate Lyase Producer Strains
4.3. Bacterial Identification
4.4. Alginolytic Activity and Substrate Specificity of Cell-Free Supernatants
4.5. Pseudomonas aeruginosa PA14 Biofilm Cultivation in 96-Microtiter Plate
4.6. Pseudomonas aeruginosa PA14 Biofilm Inhibition by Cell-Free Supernatants of Pseudoalteromonas and Cellulophaga spp.
4.7. AlyP1400 Production and Purification
4.8. AlyP1400 Characterization
4.9. Liquid Chromatography Tandem-Mass Spectrometry (LC-MS/MS) Enzyme Analysis
4.10. Inhibitory Effect of AlyP1400 against P. aeruginosa PA14 Biofilm
4.11. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Costerton, J.W. Introduction to biofilm. Int. J. Antimicrob. Agents 1999, 11, 217–221. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Stoodley, P. Biofilm formation and dispersal and the transmission of human pathogens. Trends Microbiol. 2005, 13, 7–10. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Klaus, K.M.; Peter, Ø.J.; Kit, G.M.; Richard, P.; Karen, K.; Niels, H.; Michael, G. Why chronic wounds will not heal: A novel hypothesis. Wound Repair Regen. 2008, 16, 2–10. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Jensen, P.; Fiandaca, M.J.; Pedersen, J.; Hansen, C.R.; Andersen, C.B.; Pressler, T.; Givskov, M.; Høiby, N. Pseudomonas aeruginosa biofilms in the respiratory tract of cystic fibrosis patients. Pediatr. Pulmonol. 2009, 44, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, M.M.; Whiteley, M. Pseudomonas aeruginosa attachment and biofilm development in dynamic environments. Mol. Microbiol. 2004, 53, 1075–1087. [Google Scholar] [CrossRef]
- Karatan, E.; Watnick, P. Signals, regulatory networks, and materials that build and break bacterial biofilms. Microbiol. Mol. Biol. Rev. 2009, 73, 310–347. [Google Scholar] [CrossRef] [PubMed]
- Bernardes, E.T.; Charron-Mazenod, L.; Reading, D.J.; Reckseidler-Zenteno, S.L.; Lewenza, S. Exopolysaccharide-repressing small molecules with antibiofilm and antivirulence activity against Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2017, 61, e01997-16. [Google Scholar] [CrossRef]
- Davies, D. Understanding biofilm resistance to antibacterial agents. Nat. Rev. Drug Discov. 2003, 2, 114–122. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Jensen, P.Ø.; Jakobsen, T.H.; Phipps, R.; Nielsen, A.K.; Morten, T.R.; Tim, T.N.; Michael, G.; Niels, H.; Oana, C. Quorum sensing and virulence of Pseudomonas aeruginosa during lung infection of cystic fibrosis patients. PLoS ONE 2010, 5, e10115. [Google Scholar] [CrossRef]
- Chua, S.L.; Louise, D.H.; Mingjun, Y.; Morten, R.; Thomas, E.N.; Michael, G.; Tim, T.N.; Liang, Y. In vitro and in vivo generation and characterization of Pseudomonas aeruginosa biofilm-dispersed cells via c-di-GMP manipulation. Nat. Protoc. 2015, 10, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Gnanadhas, D.P.; Monalisha, E.; Akshay, D.; Dipshikha, C. Chronic lung infection by Pseudomonas aeruginosa biofilm is cured by L-Methionine in combination with antibiotic therapy. Sci. Rep. 2015, 5, 16043. [Google Scholar] [CrossRef]
- Francolini, I.; Donelli, G. Prevention of biofilm-based medical-device-related infections. FEMS Immunol. Med. Microbiol. 2010, 59, 227–238. [Google Scholar] [CrossRef]
- Drenkard, E. Antimicrobial resistance of Pseudomonas aeruginosa biofilms. Microbes Infect. 2003, 5, 1213–1219. [Google Scholar] [CrossRef]
- Reffuveille, F.; de la Fuente-Nùñez, C.; Mansour, S.; Hancock, R.E. A broad-spectrum antibiofilm peptide enhances antibiotic action against bacterial biofilms. Antimicrob. Agents Chemother. 2014, 58, 5363–5371. [Google Scholar] [CrossRef] [PubMed]
- Bjarnsholt, T.; Jensen, P.; Burmølle, M.; Morten, H.; Janus, A.J.H.; Hans, P.H.; Henrik, C.; Kit, G.M.; Claus, M.; Søren, M.; et al. Pseudomonas aeruginosa tolerance to tobramycin, hydrogen peroxide and polymorphonuclear leukocytes is quorum-sensing dependent. Microbiology 2005, 151, 373–383. [Google Scholar] [CrossRef]
- Williams, B.J.; Dehnbostel, J.; Blackwell, T.S. Pseudomonas aeruginosa: Host defense in lung diseases. Respirology 2010, 15, 1037–1056. [Google Scholar] [CrossRef]
- Hassett, D.J.; Cuppoletti, J.; Trapnell, B.; Lymar, S.V.; Rowe, J.J.; Yoon, S.S.; Hilliard, G.M.; Parvatiyar, K.; Kamani, M.C.; et al. Anaerobic metabolism and quorum sensing by Pseudomonas aeruginosa biofilms in chronically infected cystic fibrosis airways: Rethinking antibiotic treatment strategies and drug targets. Adv. Drug Deliv. Rev. 2002, 54, 1425–1443. [Google Scholar] [CrossRef]
- Wagner, V.E.; Iglewski, B.H. Pseudomonas aeruginosa biofilms in CF Infection. Clin. Rev. Allergy Immunol. 2008, 35, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Rowntree, R.K.; Ann, H. The phenotypic consequences of CFTR mutations. Ann. Hum. Genet. 2003, 67, 471–485. [Google Scholar] [CrossRef]
- Boucher, R.C. New concepts of the pathogenesis of cystic fibrosis lung disease. Eur. Respir. J. 2004, 23, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Kirov, S.M.; Webb, J.S.; Kjelleberg, S. Clinical significance of seeding dispersal in biofilms. Microbiology 2005, 151, 3452–3453. [Google Scholar] [CrossRef][Green Version]
- Kirov, S.M.; Webb, J.S.; O’May, C.Y.; Reid, D.W.; Woo, J.; Rice, S.A.; Kjelleberg, S. Biofilm differentiation and dispersal in mucoid Pseudomonas aeruginosa isolates from patients with cystic fibrosis. Microbiology 2007, 153, 3264–3274. [Google Scholar] [CrossRef]
- Anderson, G.G.; Moreau-Marquis, S.; Stanton, B.A.; O’Toole, G.A. In vitro analysis of tobramycin-treated Pseudomonas aeruginosa biofilms on cystic fibrosis-derived airway epithelial cells. Infect. Immun. 2008, 1423–1433. [Google Scholar] [CrossRef]
- Woo, J.K.K.; Webb, J.S.; Kirov, S.M.; Kjelleberg, S.; Rice, S.A. Biofilm dispersal cells of a cystic fibrosis Pseudomonas aeruginosa isolate exhibit variability in functional traits likely to contribute to persistent infection. FEMS Immunol. Med. Microbiol. 2012, 66, 251–264. [Google Scholar] [CrossRef]
- Murphy, T.F.; Brauer, A.L.; Eschberger, K.; Lobbins, P.; Grove, L.; Cai, X.; Sethi, S. Pseudomonas aeruginosa in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2008, 177, 853–860. [Google Scholar] [CrossRef]
- Hengzhuang, W.; Wu, H.; Ciofu, O.; Song, Z.; Høiby, N. Pharmacokinetics/pharmacodynamics of colistin and imipenem on mucoid and nonmucoid Pseudomonas aeruginosa Biofilms. Antimicrob. Agents Chemother. 2011, 5, 4469–4474. [Google Scholar] [CrossRef]
- Trizna, E.; Bogachev, M.I.; Kayumov, A. Degrading of the Pseudomonas aeruginosa biofilm by extracellular levanase SacC from Bacillus subtilis. BioNanoScience 2018, 9, 48–52. [Google Scholar] [CrossRef]
- Nijland, R.; Hall, M.J.; Burgess, J.G. Dispersal of biofilms by secreted, matrix degrading, bacterial DNase. PLoS ONE 2010, 5, e15668. [Google Scholar] [CrossRef]
- Baker, P.; Hill, P.J.; Snarr, B.D.; Alnabelseya, N.; Pestrak, M.; Lee, M.J.; Jennings, L.K.; Tam, J.; Melnyk, R.A.; Parsek, M.R.; et al. Exopolysaccharide biosynthetic glycoside hydrolases can be utilized to disrupt and prevent Pseudomonas aeruginosa biofilms. Sci. Adv. 2016, 2, e1501632. [Google Scholar] [CrossRef]
- Alipour, M.; Suntres, Z.E.; Omri, A. Importance of DNase and alginate lyase for enhancing free and liposome encapsulated aminoglycoside activity against Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2009, 64, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liyun, W.; Yanhong, C.; Hui, N.; Anfeng, X.; Huinong, C. Characterization of an extracellular biofunctional alginate lyase from marine Microbulbifer sp. ALW1 and antioxidant activity of enzymatic hydrolysates. Microbiol. Res. 2016, 182, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Dufourcq, R.; Chalkiadakis, E.; Fauchon, M.; Deslandes, E.; Kerjean, V.; Chanteau, S.; Petit, E.; Guezennec, J.; Dupont-Rouzeyrol, M. Isolation and partial characterization of bacteria (Pseudoalteromonas sp.) with potential antibacterial activity from a marine costal environment from New Caledonia. Lett. Appl. Microbiol. 2014, 58, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Dheilly, A.; Soum-Soutéra, E.; Klein, G.L.; Bazire, A.; Compère, C.; Haras, D.; Dufour, A. Antibiofilm activity of the marine bacterium Pseudoalteromonas sp. strain 3J6. Appl. Environ. Microbiol. 2010, 76, 3452–3461. [Google Scholar] [CrossRef] [PubMed]
- Høiby, N.; Ciofu, O.; Johansen, H.K.; Song, Z.J.; Moser, C.; Jensen, P.; Molin, S.; Givskov, M.; Tolker-Nielsen, T.; Bjarnsholt, T. The clinical impact of bacterial biofilms. Int. J. Oral Sci. 2011, 3, 55–65. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Ciofu, O.; Molin, S.; Givskov, M.; Høiby, N. Applying insights from biofilm biology to drug development―Can a new approach be developed? Nat. Rev. Drug Discov. 2013, 12, 791–808. [Google Scholar] [CrossRef] [PubMed]
- Lamppa, J.W.; Karl, E.G. Alginate lyase exhibits catalysis-independent biofilm dispersion and antibiotic synergy. Antimicrob. Agents Chemother. 2013, 57, 137–145. [Google Scholar] [CrossRef]
- Nithya, C.; Shunmugiah, K.P. The In vitro antibiofilm activity of selected marine bacterial culture supernatants against Vibrio spp. Arch. Microbiol. 2010, 192, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Xiaolu, J.; Hueymin, H.; Shiliang, L.; Huashi, G. Antitumor activities of alginate-derived oligosaccharides and their sulphated substitution derivatives. Eur. J. Phycol. 2004, 39, 67–71. [Google Scholar] [CrossRef]
- Hu, X.; Xiaolu, J.; Jun, G.; Hueymin, H.; Yan, L.; Huashi, G. Antibacterial activity of lyase-depolymerized products of alginate. J. Appl. Phycol. 2005, 17, 57–60. [Google Scholar] [CrossRef]
- Wong, T.Y.; Preston, L.A.; Schiller, N.L. Alginate lyase: Review of major sources and enzyme characteristics, structure-function analysis, biological roles, and application. Annu. Rev. Microbiol. 2000, 54, 289–340. [Google Scholar] [CrossRef]
- Ertesvåg, H. Alginate-modifying enzymes: Biological roles and biotechnological uses. Front. Microbiol. 2015, 6, 523. [Google Scholar] [CrossRef]
- Rendueles, O.; Kaplan, J.B.; Ghigo, J.M. Antibiofilm polysaccharides. Environ. Microbiol. 2013, 15, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Papa, R.; Ermenegilda, P.; Filomena, S.; Gaetano, B.; Maria, L.T.; Marco, A.; Laura, S. Anti-biofilm activity of the Antarctic marine bacterium Pseudoalteromonas haloplanktis TAC125. Res. Microbiol. 2013, 164, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Richards, G.P.; Watson, M.A.; Needleman, D.S.; Uknalis, J.; Boyd, E.F.; Fay, J.P. Mechanisms for Pseudoalteromonas piscicida induced killing of vibrios and other bacterial pathogens. Appl. Environ. Microbiol. 2017, 83, e00175-17. [Google Scholar] [CrossRef]
- Klein, G.L.; Soum-Soutéra, E.; Guedec, Z.; Bazirea, A.; Compere, C.; Dufoura, A. The anti-biofilm activity secreted by a marine Pseudoalteromonas strain. Biofouling 2011, 27, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Walters, M.C.; Roe, F.; Bugnicourt, A.; Franklin, M.J.; Stewart, P.S. Contributions of antibiotic penetration, oxygen limitation, and low metabolic activity to tolerance of Pseudomonas aeruginosa biofilms to ciprofloxacin and tobramycin. Antimicrob. Agents Chemother. 2003, 47, 317–323. [Google Scholar] [CrossRef]
- Hall, C.W.; Hinz, A.J.; Gagnon, L.B.; Zhang, L.; Nadeau, J.P.; Copeland, S.; Saha, B.; Mah, T.F. Pseudomonas aeruginosa biofilm antibiotic resistance gene ndvB expression requires the RpoS stationary-phase sigma factor. Appl. Environ. Microbiol. 2018, 19, e02762-17. [Google Scholar] [CrossRef]
- Leid, J.G.; Willson, C.J.; Shirtliff, M.E.; Hassett, D.J.; Parsek, M.R.; Jeffers, A.K. The exopolysaccharide alginate protects Pseudomonas aeruginosa biofilm bacteria from IFNγ- mediated macrophage killing. J. Immunol. 2005, 175, 7512–7518. [Google Scholar] [CrossRef]
- Lamppa, J.W.; Ackerman, M.E.; Lai, J.I.; Scanlon, T.C.; Griswold, K.E. Genetically engineered alginate lyase-PEG conjugates exhibit enhanced catalytic function and reduced immunoreactivity. PLoS ONE 2011, 6, e17042. [Google Scholar] [CrossRef] [PubMed]
- Robles-Price, A.; Wong, T.Y.; Sletta, H.; Valla, S.; Schiller, N.L. AlgX is a periplasmic protein required for alginate biosynthesis in Pseudomonas aeruginosa. J. Bacteriol. 2004, 186, 7369–7377. [Google Scholar] [CrossRef] [PubMed]
- O’May, C.Y.; Reid, D.W.; Kirov, S.M. Anaerobic culture conditions favor biofilm-like phenotypes in Pseudomonas aeruginosa isolates from patients with cystic fibrosis. FEMS Immunol. Med. Microbiol. 2006, 48, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xiaolu, J.; Huashi, G.; Peng, W. Preparation, purification and characterization of alginate oligosaccharides degraded by alginate lyase from Pseudomonas sp. HZJ 216. Carbohydr. Res. 2011, 346, 794–800. [Google Scholar] [CrossRef]
- Weisburg, W.G.; Barns, S.M.; Pelletier, D.A.; Lane, D.J. 16S ribosomal DNA amplification for phylogenetic study. J. Bacteriol. 1991, 173, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Babalola, O.O.; Bronwyn, M.K.; Marilize, L.R.; Andrew, E.C.; Cary, C.S.; Stephanie, G.B.; Don, A.C. Phylogenetic analysis of actinobacterial populations associated with Antarctic Dry Valley mineral soils. Environ. Microbiol. 2009, 11, 566–576. [Google Scholar] [CrossRef]
- Boratyn, G.M.; Christiam, C.; Peter, S.C.; George, C.; Amelia, F.; Ning, M.; Thomas, L.M.; Wayne, T.M.; Scott, D.M.; Yuri, M.; Yan, R.; Eric, W.S.; Tao, T.; Jian, Y.; Irena, Z. BLAST: A more efficient report with usability improvements. Nucleic Acids Res. 2013, 41, W29–W33. [Google Scholar] [CrossRef]
- Pendyala, V.; Baburao, C.; Chandrasekhar, K. Studies on some physicochemical properties of Leucaena leucocephala bark gum. J. Adv. Pharm. Technol. Res. 2010, 1, 253–259. [Google Scholar]
- Zhu, Z.; Xiangqian, L.; Hao, S.; Jia, Z.; Zhongbiao, T.; Mengdi, Y.; Peng, Y.; Xiaoyan, L. Characterization of a novel alginate lyase from marine bacterium Vibrio furnissii H1. Mar. Drugs 2018, 16, 30. [Google Scholar] [CrossRef]
- Nakagawa, A.; Ozaki, T.; Chubachi, K.; Hosoyama, T.; Okubo, T.; Iyobe, S.; Suzuki, T. An effective method for isolating alginate lyase-producing Bacillus sp. ATB-1015 strain and purification and characterization of the lyase. J. Appl. Microbiol. 1998, 84, 328–335. [Google Scholar] [CrossRef]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011, 47, e2437. [Google Scholar] [CrossRef] [PubMed]
- Stepanovic, S.; Dragana, V.; Veronika, H.; Giovanni, D.B.; Slobodanka, D.; Ivana, C.; Filip, R. Quantification of biofilm in microtiter plates: Overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS 2007, 115, 891–899. [Google Scholar] [CrossRef]
- O’Toole, G.A.; Kolter, R. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signaling pathways: A genetic analysis. Mol. Microbiol. 1998, 28, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Li, J.W.; Sheng, D.; Jie, S.; Chun-Bo, L.; Xiu-Lan, C.; Bin-Bin, X.; Yu-Zhong, Z. Purification and characterization of a bifunctional alginate lyase from Pseudoalteromonas sp. SM0524. Mar. Drugs 2011, 9, 109–123. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Ana, P.; Antonio, P. Detection of alginate lyase activity staining after sodium dodecyl sulfate-polyacrylamide gel electrophoresis and subsequent renaturation. Analyt. Biochem. 1994, 217, 124–127. [Google Scholar] [CrossRef]
- Chen, X.L.; Zhang, Y.Z.; Gao, P.J.; Luan, X.W. Two different proteases produced by a deep-sea psychrotrophic strain Pseudoaltermonas sp. SM9913. Mar. Biol. 2003, 143, 989–993. [Google Scholar] [CrossRef]
- Lukas, K.; Jesse, D.C.; Jason, W.; William, S.N.; Michael, J.M. Semi-supervised learning for peptide identification from shotgun proteomics datasets. Nat. Methods 2007, 4, 923–925. [Google Scholar] [CrossRef]
- Shevchenko, A.; Tomas, H.; Havliš, J.; Olsen, J.V.; Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 2006, 1, 2856–2860. [Google Scholar] [CrossRef]
- Zaidi, T.; Pier, G.B. Prophylactic and therapeutic efficacy of a fully human immunoglobulin G1 monoclonal antibody to Pseudomonas aeruginosa alginate in murine keratitis infection. Infect. Immun. 2008, 76, 4720–4725. [Google Scholar] [CrossRef]
- Heffernan, B.; Cormac, D.M.; Eoin, C. Comparison of planktonic and biofilm cultures of Pseudomonas fluorescens DSM 8341 cells grown on fluoroacetate. J. Appl. Environ. Microbiol. 2009, 75, 2899–2907. [Google Scholar] [CrossRef] [PubMed]
- Heydorn, A.; Nielsen, A.T.; Hentzer, M.; Sternberg, C.; Givskov, M.; Ersboll, B.K.; Molin, S. Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology 2000, 146, 2395–2407. [Google Scholar] [CrossRef]
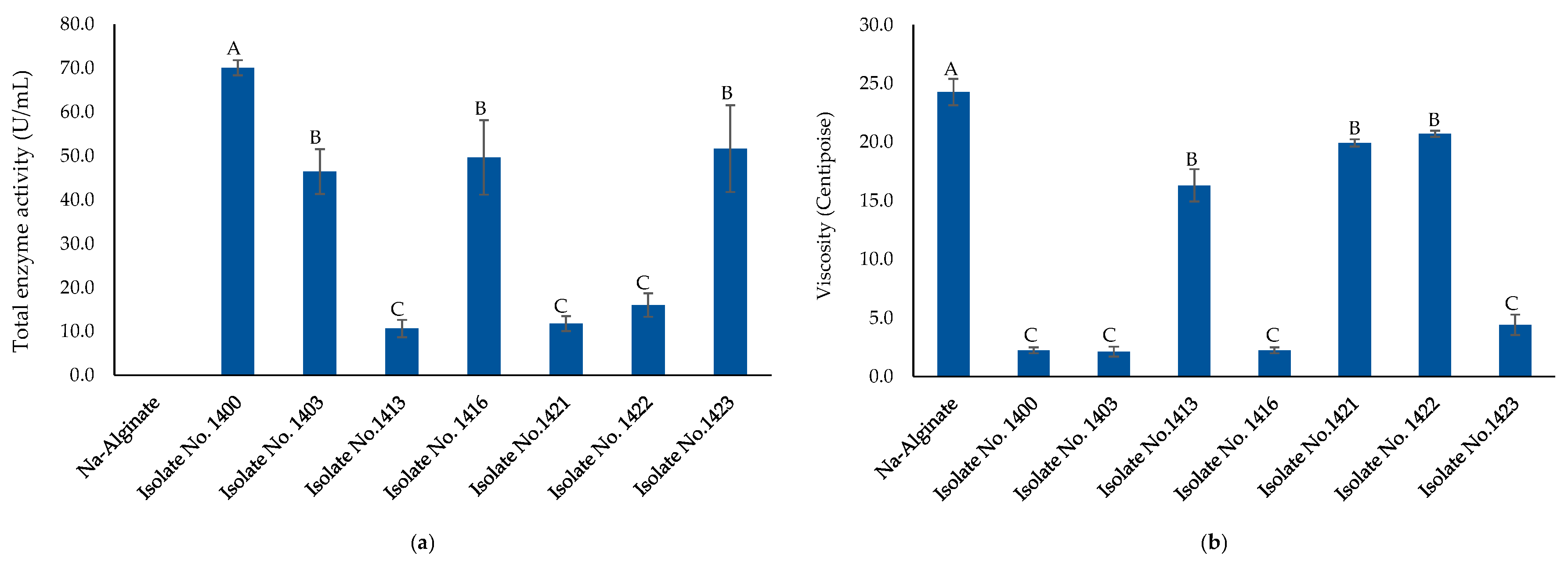
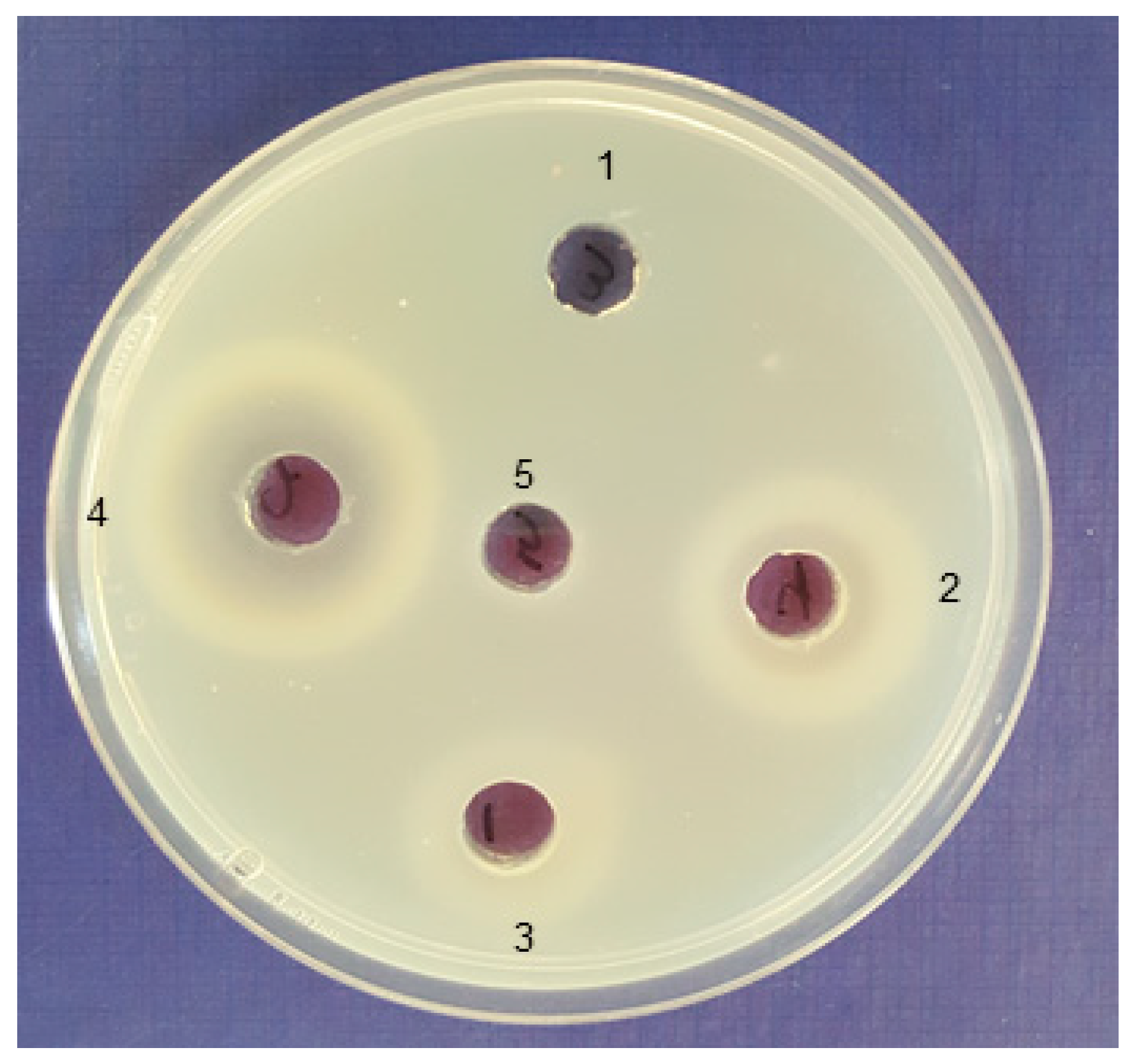
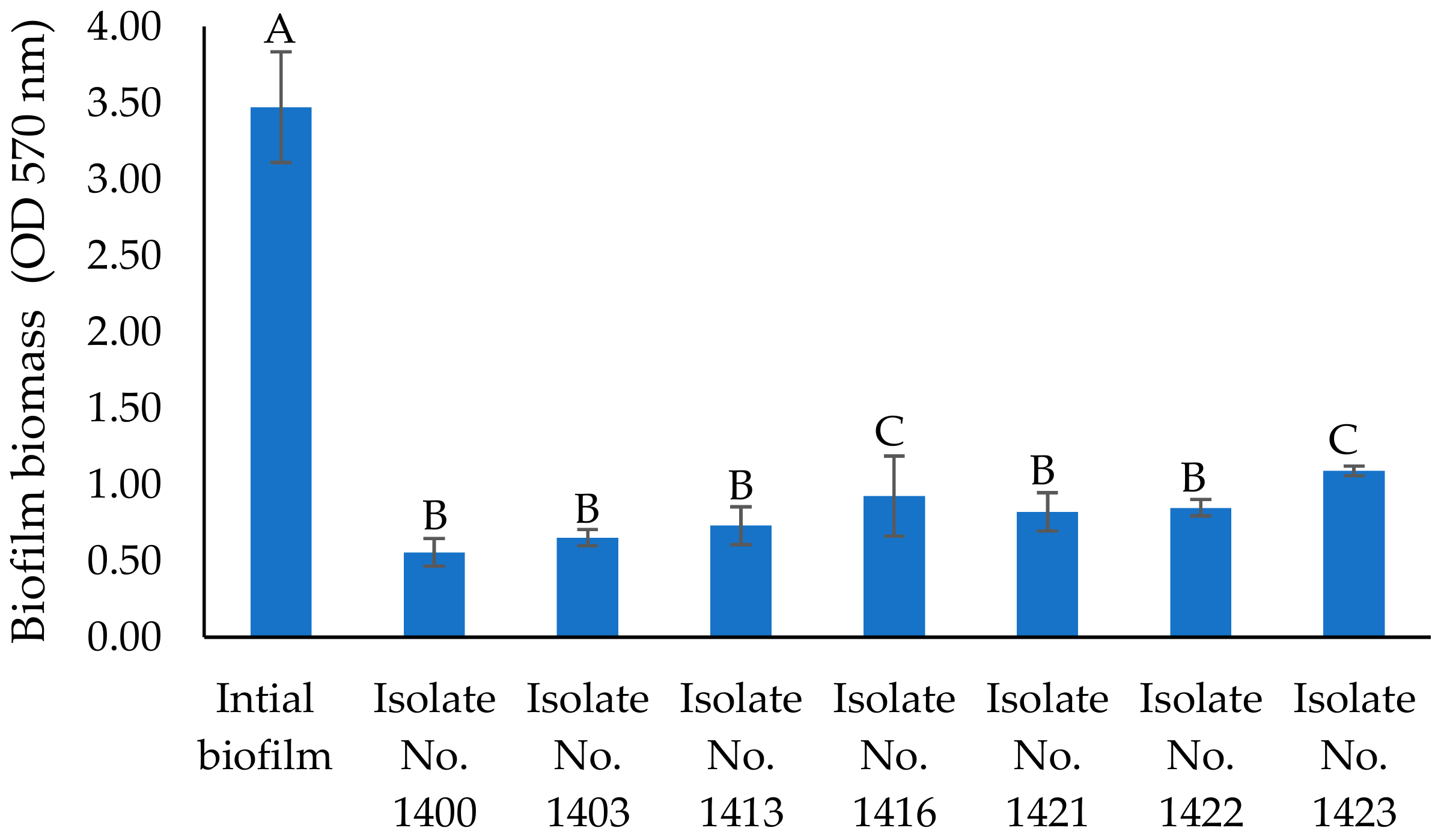

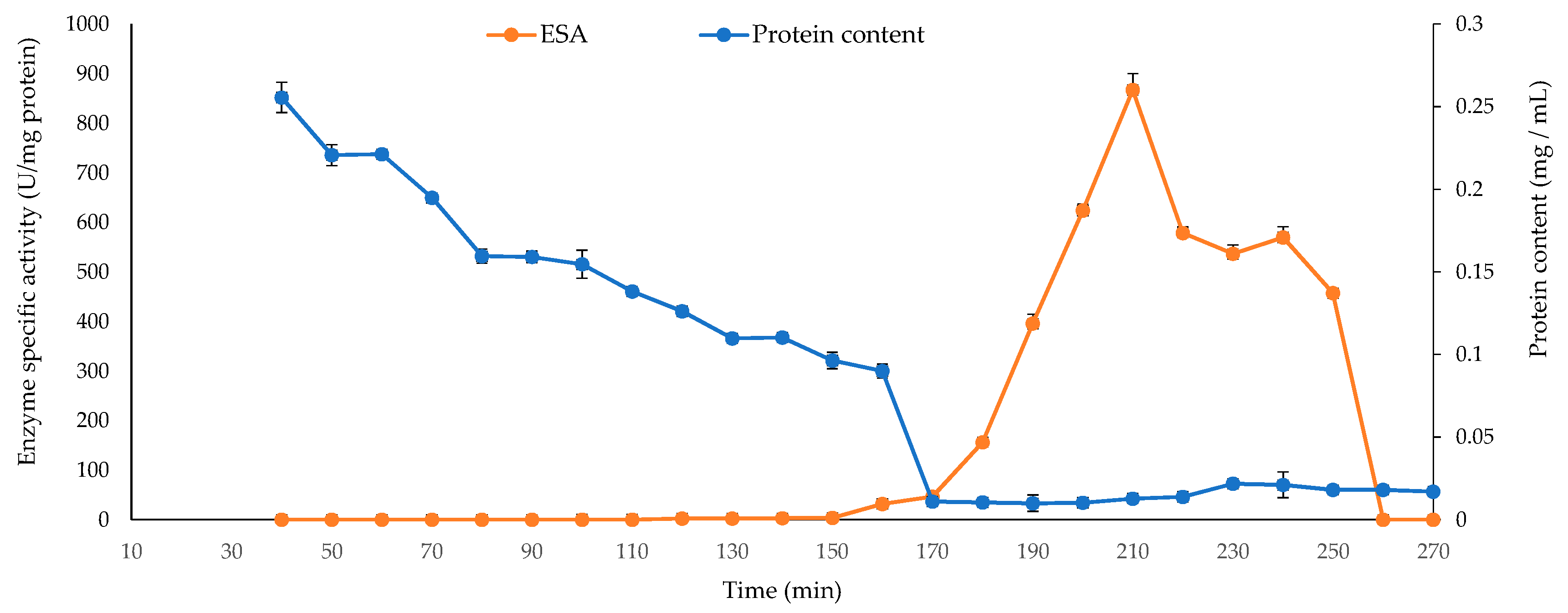


| No. | The Most Closely Related Bacterial Species Name and Sequence ID from GenBank | Alginate Lyase Activity (Clearing Zone, mm) * |
|---|---|---|
| 101 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 12.0 ± 1.4 |
| 1002 | Celeribacter halophilus strain Xmb039, KT986167.1 | 10.5 ± 2.1 |
| 2001 | Shewanella algae strain SFH3, MG738264.1 | 9.5 ± 0.7 |
| 2005 | Vibrio parahaemolyticus strain HH101313, MG386398.1 | 14.5 ± 0.7 |
| 2006 | Shewanella algae strain KC-Na-R1, CP033575.1 | 6.5 ± 0.7 |
| 4001 | Vibrio natriegens strain AUCASVE5, JQ277719.1 | 7.0 ± 1.4 |
| 4002 | Vibrio alginolyticus strain QY170324, MF101235.1 | 6.0 ± 1.4 |
| 4003 | Vibrio alginolyticus strain QY170324, MF101235.1 | 8.0 ± 1.4 |
| 4005 | Vibrio parahaemolyticus strain SC2, MK308579.1 | 10.0 ± 1.4 |
| 6002 | Vibrio azureus strain Xmb005, KT986135.1 | 10.5 ± 2.1 |
| 6006 | Vibrio alginolyticus strain FDAARGOS_114, CP014045.1 | 3.0 ± 1.4 |
| 8009 | Vibrio alginolyticus strain QY170324, MF101235.1 | 11.0 ± 1.4 |
| 9001 | Vibrio parahaemolyticus strain CHB-33, KR347290.1 | 5.5 ± 2.1 |
| 9002 | Vibrio alginolyticus strain CX-72, MH368392.1 | 4.5 ± 0.7 |
| 9003 | Vibrio alginolyticus strain Val-3, MH879822.1 | 6.5 ± 2.1 |
| 9004 | Vibrio diabolicus strain FDAARGOS_96, CP014094.1 | 7.5 ± 0.7 |
| 1400 | Pseudoalteromonas tetraodonis strain GFC, CP011041.1 | 30.0 ± 1.4 |
| 1401 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 2.5 ± 0.7 |
| 1403 | Pseudoalteromonas agarivorans strain SDRB-Py1, MG456901.1 | 27.5 ± 0.7 |
| 1404 | Pseudoalteromonas distincta strain 20KNS10Z3, MH478310.1 | 4.0 ± 1.4 |
| 1405 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 11.0 ± 1.4 |
| 1406 | Pseudoalteromonas agarivorans strain DSM 14585, NR_025509.1 | 6.5 ± 0.7 |
| 1407 | Pseudoalteromonas tetraodonis strain GFC, CP011041.1 | 2.5 ± 0.7 |
| 1408 | Pseudoalteromonas tetraodonis strain GFC, CP011041.1 | 11.0 ± 1.4 |
| 1410 | Pseudoalteromonas tetraodonis strain GFC, CP011041.1 | 12.0 ± 1.4 |
| 1412 | Pseudoalteromonas atlantica strain ECSMB14104, CP023464.1 | 5.0 ± 0.0 |
| 1413 | Cellulophaga fucicola strain NN015860, NR_025287.1 | 17.5 ± 1.4 |
| 1414 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 14.5 ± 0.7 |
| 1416 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 20.5 ± 0.7 |
| 1417 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 9.5 ± 0.7 |
| 1418 | Alteromonas stellipolaris strain PQQ-44, CP015346.1 | 10.0 ± 1.4 |
| 1419 | Pseudoalteromonas tetraodonis strain GFC, CP011041.1 | 13.5 ± 2.1 |
| 1421 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 24.0 ± 1.4 |
| 1422 | Pseudoalteromonas carrageenovora IAM 12662 strain ATCC43555T, LT965929.1 | 24.0 ± 1.4 |
| 1423 | Cellulophaga fucicola strain NN015860, NR_025287.1 | 21.5 ± 2.1 |
| 1427 | Pseudoalteromonas espejiana strain ATCC 29659, CP011028.1 | 7.5 ± 0.7 |
| Purification Proceedings | Total Protein (mg) | Total Enzyme Activity (U)* | Enzyme Specific Activity (U/mg Protein) | Purification (Fold) | Yield (%) |
|---|---|---|---|---|---|
| Crude enzyme | 5126.11 ± 5.35 | 90565 ± 5.0 | 17.67 ± 0.018 | 1.00 | 100 |
| Fractionation I 60% (NH4)2SO4 | 1551.33 ± 4.16 | 53375.33 ± 4.51 | 34.41 ± 0.92 | 1.95 | 59 |
| Fractionation II 80% (NH4)2SO4 | 85.9 ± 1.13 | 9123 ± 3.0 | 106.47 ± 1.4 | 6.03 | 10 |
| Anion exchange chromatography (DEAE Sepharose) | 32.166 ± 1.78 | 3902.33 ± 6.81 | 125.94 ± 7.32 | 7.13 | 4.0 |
| Gel-filtration chromatography (Sephadex G-100) | 1.17 ± 0.06 | 410 ± 2.0 | 342.16 ± 3.84 | 19.36 | 0.5 |
| Treatments | Average Thickness (µm) | Biovolume (µm3/µm2) | Surface Area (105 µm2) |
|---|---|---|---|
| Non-treated | 37.2 ± 3.74 | 10.27 ± 3.77 | 5.0 ± 3.5 |
| Carbenicillin (CB) | 27.9 ± 4.45 * | 10.28 ± 1.26 | 6.3 ± 4.0 |
| Ciprofloxacin (Cip) | 27.7 ± 4.14 * | 10.70 ± 1.0 | 6.1 ± 0.1 |
| AlyP1400 | 25.3 ± 2.41 * | 7.71 ± 3.79 * | 2.6 ± 1.6 * |
| AlyP1400 + CB | 16.8 ± 2.20 ** | 5.27 ± 1.34 ** | 0.29 ± 0.2 ** |
| AlyP1400 + Cip | 13.2 ± 3.42 ** | 3.37 ± 0.64 ** | 0.24 ± 0.1 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daboor, S.M.; Raudonis, R.; Cohen, A.; Rohde, J.R.; Cheng, Z. Marine Bacteria, A Source for Alginolytic Enzyme to Disrupt Pseudomonas aeruginosa Biofilms. Mar. Drugs 2019, 17, 307. https://doi.org/10.3390/md17050307
Daboor SM, Raudonis R, Cohen A, Rohde JR, Cheng Z. Marine Bacteria, A Source for Alginolytic Enzyme to Disrupt Pseudomonas aeruginosa Biofilms. Marine Drugs. 2019; 17(5):307. https://doi.org/10.3390/md17050307
Chicago/Turabian StyleDaboor, Said M., Renee Raudonis, Alejandro Cohen, John R. Rohde, and Zhenyu Cheng. 2019. "Marine Bacteria, A Source for Alginolytic Enzyme to Disrupt Pseudomonas aeruginosa Biofilms" Marine Drugs 17, no. 5: 307. https://doi.org/10.3390/md17050307
APA StyleDaboor, S. M., Raudonis, R., Cohen, A., Rohde, J. R., & Cheng, Z. (2019). Marine Bacteria, A Source for Alginolytic Enzyme to Disrupt Pseudomonas aeruginosa Biofilms. Marine Drugs, 17(5), 307. https://doi.org/10.3390/md17050307





