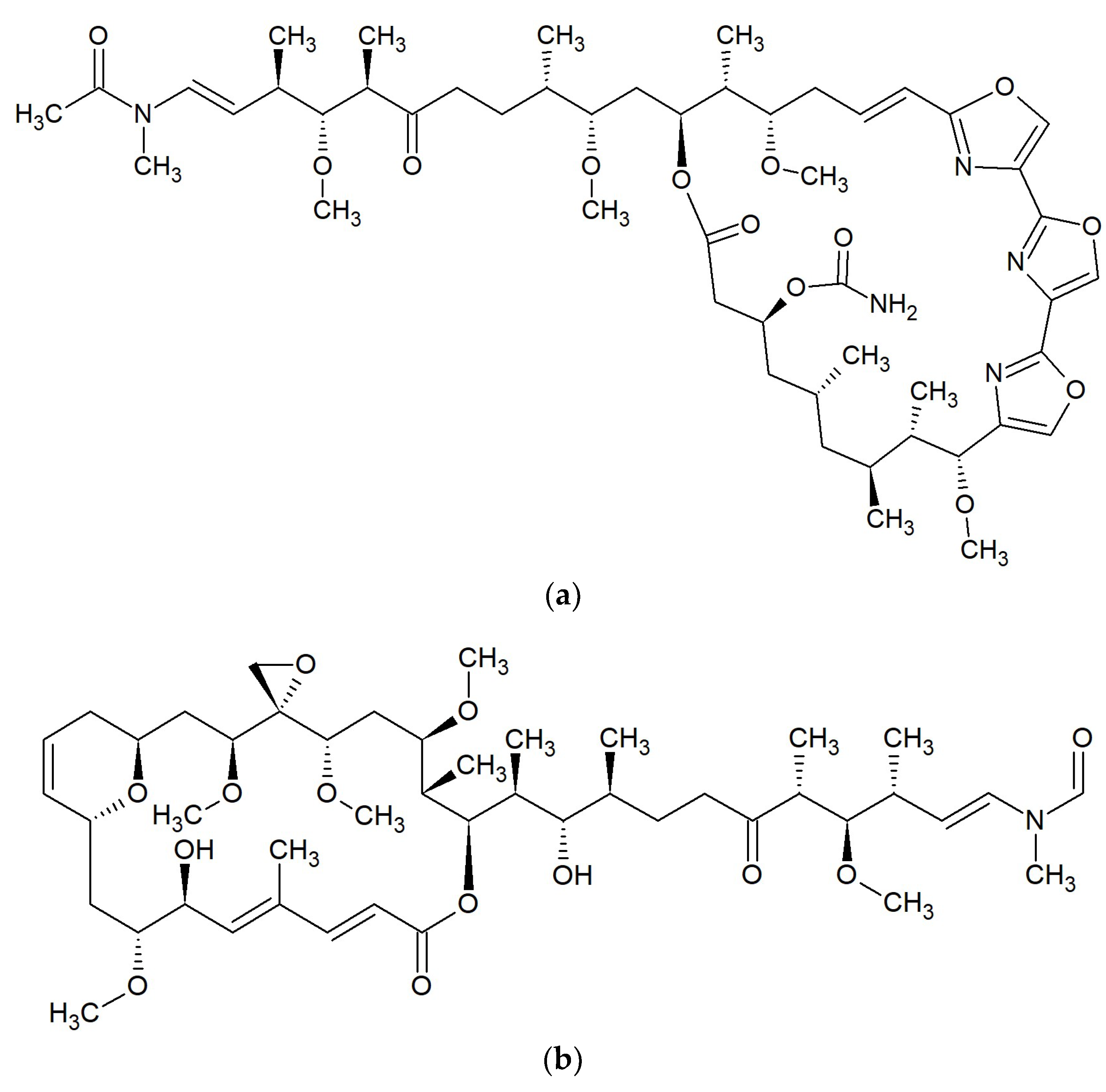
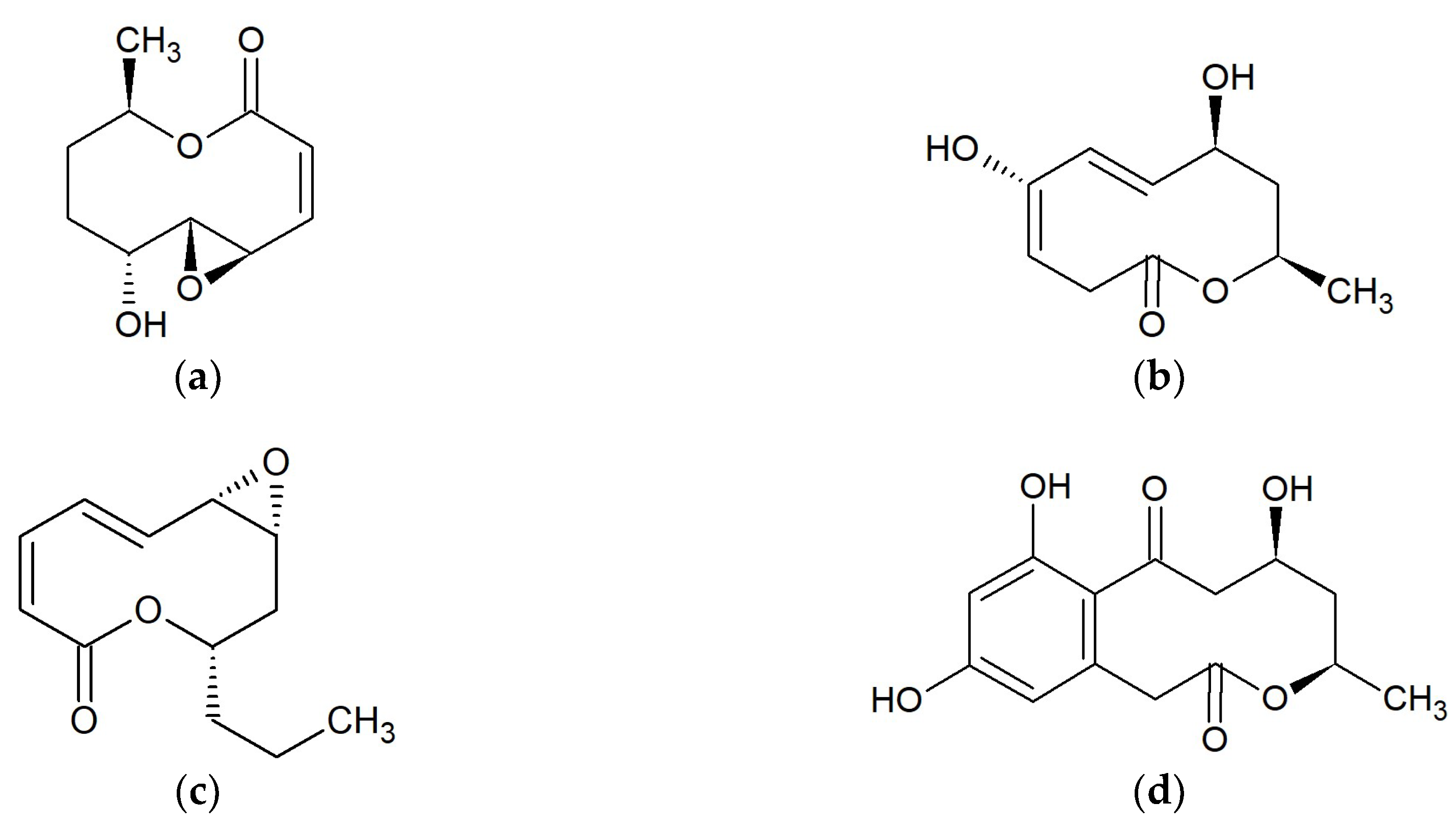
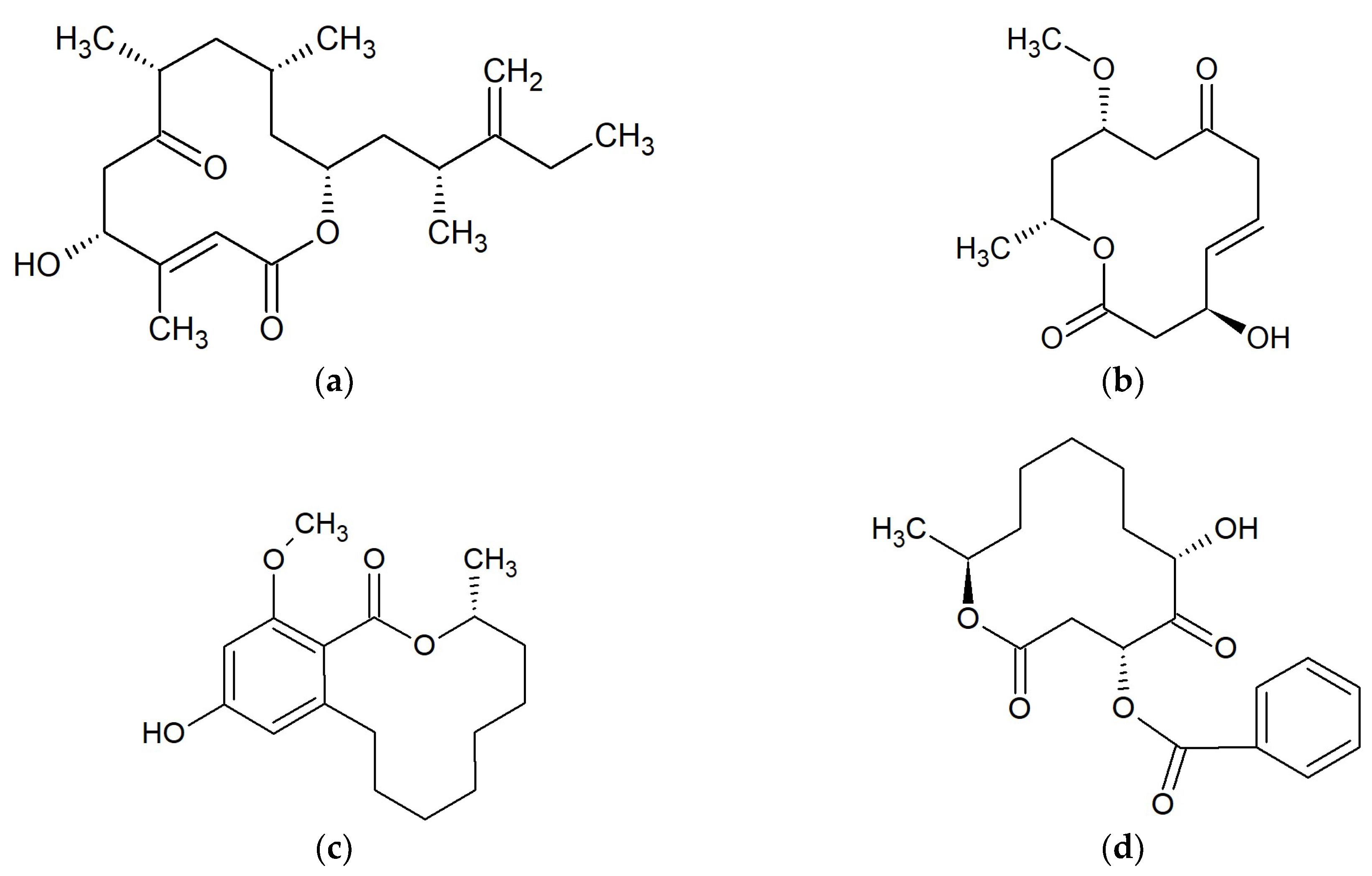
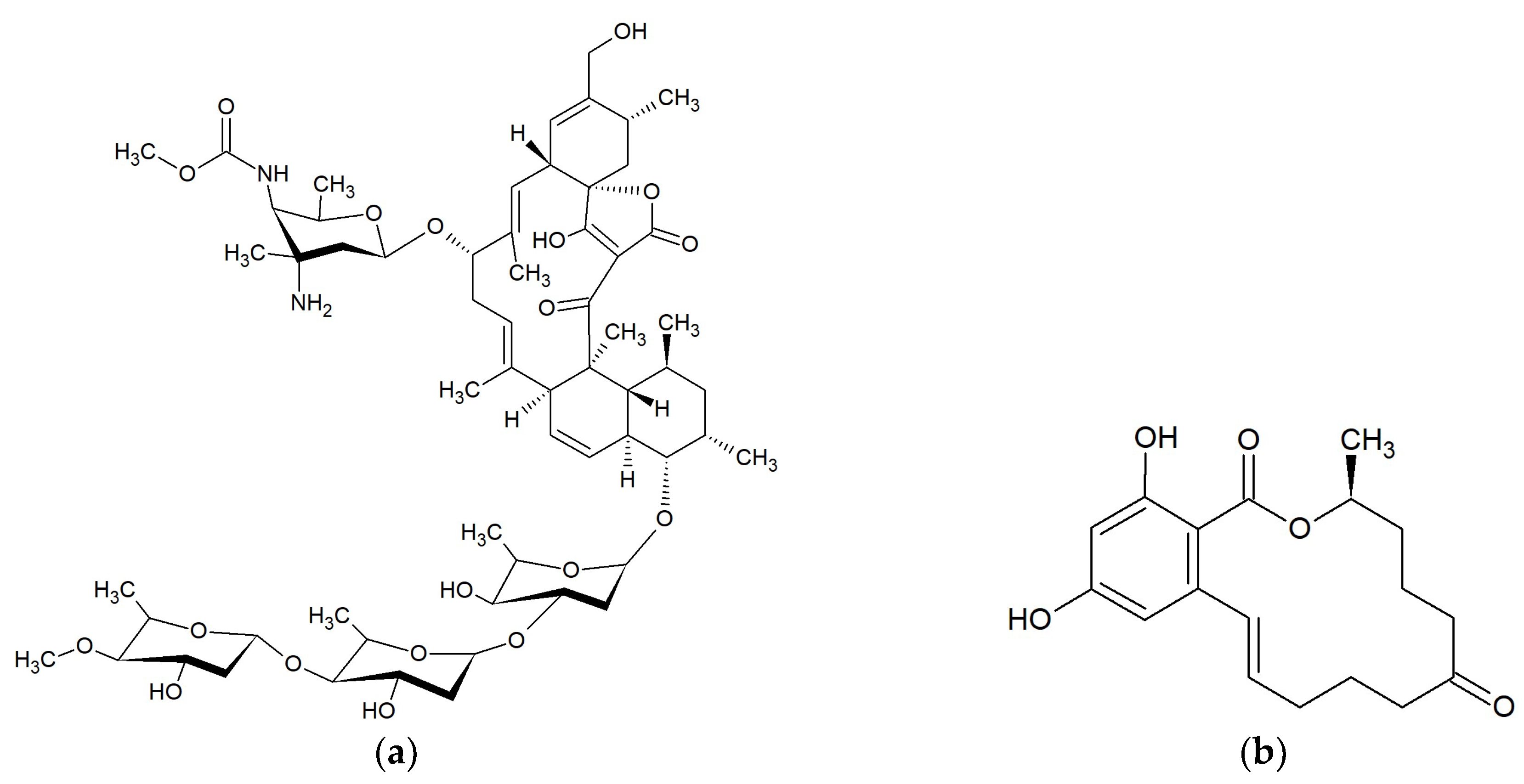
Marine Macrolides with Antibacterial and/or Antifungal Activity
Abstract
1. Introduction
2. Antimicrobial Activity of Marine Macrolides
2.1. Macrolides 10-Membered
2.1.1. Curvulides
2.1.2. Modiolides
2.1.3. Phomolides
2.1.4. Xestodecalactones
2.2. Macrolides 12-Membered
2.2.1. Amphidinolides
2.2.2. Dendrodolides
2.2.3. Lasiodiplodins
2.2.4. Sporiolides
2.3. Macrolides 14-Membered
2.3.1. Lobophorins
2.3.2. Zearalanones
2.4. Macrolides 15- and 16-Membered
Bromophycolides
2.5. Macrolides 16-Membered
2.5.1. Butremycin
2.5.2. Chalcomycins
2.5.3. Neurymenolides
2.6. Macrolides 18-Membered
2.6.1. Borrelidins
2.6.2. Leucascandrolides
2.6.3. Tedanolides
2.7. Macrolides 20-Membered
2.7.1. Macrocyclic Polyesters
2.7.2. Misakinolides
2.8. Macrolides 22-Membered
2.8.1. Kabiramides
2.8.2. Scytophycins
2.9. Macrolides 22–25-Membered
2.9.1. Gageomacrolactins
2.9.2. Halichondramides
2.9.3. Macrolactins
2.9.4. Maduralide
2.10. Macrolides 26-Membered
2.10.1. Neomaclafungins
2.10.2. Phorboxazoles
2.11. Macrolides 31-Membered
Reedsmycins
2.12. Macrolides 34-Membered
Marinisporolides
2.13. Macrolides 36-Membered
2.13.1. Azalomycins
2.13.2. Bahamaolides
2.13.3. Polyhydroxyl Macrolides
2.14. Macrolides 40-Membered
Amantelides
2.15. Macrolides 42-Membered
Spongistatins
3. Conclusions
Funding
Conflicts of Interest
References
- Burja, A.M.; Banaigs, B.; Abou-Mansour, E.; Burgess, J.G.; Wright, P.C. Marine cyanobacteria - a prolific source of natural products. Tetrahedron 2001, 57, 9347–9377. [Google Scholar] [CrossRef]
- El-Demerdash, A.; Tammam, M.A.; Atanasov, A.G.; Hooper, J.N.A.; Al-Mourabit, A.; Kijjoa, A. Chemistry and biological activities of the marine sponges of the genera Mycale (Arenochalina), Biemna and Clathria. Mar. Drugs 2018, 16, 214. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, J.; He, S.; Yan, X. A review study on macrolides isolated from cyanobacteria. Mar. Drugs 2017, 15, 126. [Google Scholar] [CrossRef]
- Swain, S.S.; Paidesetty, S.K.; Padhy, R.N. Antibacterial, antifungal and antimycobacterial compounds from cyanobacteria. Biomed. Pharmacother. 2017, 90, 760–776. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.-A.; Zheng, J.-J.; Gu, Y.-C.; Wang, C.-Y.; Shao, C.-L. Chapter 7. The chemistry and bioactivity of macrolides from marine microorganisms. Stud. Nat. Prod. Chem. 2015, 44, 353–401. [Google Scholar]
- Jelić, D.; Antolović, R. From erythromycin to azithromycin and new potential ribosome-binding antimicrobials. Antibiotics 2016, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Campoy, S.; Adrio, J.L. Antifungals. Biochem. Pharmacol. 2017, 133, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Zuckerman, J.M.; Qamar, F.; Bono, B.R. Review of macrolides (azithromycin, clarithromycin), ketolides (telithromycin) and glycylcyclines (tigecycline). Med. Clin. North Am. 2011, 95, 761–791. [Google Scholar] [CrossRef]
- Dinos, G.P. The macrolide antibiotic renaissance. Br. J. Pharmacol. 2017, 174, 2967–2983. [Google Scholar] [CrossRef]
- Karpiński, T.M.; Andrzejewska, E.; Eder, P.; Linke, K.; Szkaradkiewicz, A. Evaluation of antimicrobial resistance of Helicobacter pylori in the last 15 years in West Poland. Acta Microbiol. Immunol. Hung. 2015, 62, 287–293. [Google Scholar] [CrossRef]
- Bolhuis, M.S.; Panday, P.N.; Pranger, A.D.; Kosterink, J.G.; Alffenaar, J.C. Pharmacokinetic drug interactions of antimicrobial drugs: A systematic review on oxazolidinones, rifamycines, macrolides, fluoroquinolones, and beta-lactams. Pharmaceutics. 2011, 3, 865–913. [Google Scholar] [CrossRef] [PubMed]
- Mesa-Arango, A.C.; Scorzoni, L.; Zaragoza, O. It only takes one to do many jobs: Amphotericin B as antifungal and immunomodulatory drug. Front. Microbiol. 2012, 3, 286. [Google Scholar] [CrossRef] [PubMed]
- OECD. Antimicrobial Resistance. Policy Insights. 2016. Available online: https://www.oecd.org/health/health-systems/AMR-Policy-Insights-November2016.pdf (accessed on 24 January 2019).
- Alekshun, M.N.; Levy, S.B. Molecular mechanisms of antibacterial multidrug resistance. Cell 2007, 128, 1037–1050. [Google Scholar] [CrossRef]
- Sanjit Singh, A.; Lekshmi, M.; Prakasan, S.; Nayak, B.B.; Kumar, S. Multiple antibiotic-resistant, extended spectrum-β-lactamase (ESBL)-producing enterobacteria in fresh seafood. Microorganisms 2017, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, F.A.M.; Helal, Z.H.; Khan, M.I. High prevalence of blaNDM-1, blaVIM, qacE, and qacEΔ1 genes and their association with decreased susceptibility to antibiotics and common hospital biocides in clinical isolates of Acinetobacter baumannii. Microorganisms 2017, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Jayarao, B.; Almeida, R.; Oliver, S.P. Antimicrobial resistance on dairy farms. Foodborne Pathog. Dis. 2019, 16, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Florez-Cuadrado, D.; Moreno, M.A.; Ugarte-Ruíz, M.; Domínguez, L. Antimicrobial resistance in the food chain in the European Union. Adv. Food Nutr. Res. 2018, 86, 115–136. [Google Scholar] [PubMed]
- Bernd, S.; Kunz, O. Bidirectional cross metathesis and ring-closing metathesis/ring opening of a C2-symmetric building block: A strategy for the synthesis of decanolide natural products. Beilstein, J. Org. Chem. 2013, 9, 2544–2555. [Google Scholar]
- Greve, H.; Schupp, P.J.; Eguereva, E.; Kehraus, S.; König, G.M. Ten-membered lactones from the marine-derived fungus Curvularia sp. J. Nat. Prod. 2008, 71, 1651–1653. [Google Scholar] [CrossRef]
- Mondol, M.A.; Farthouse, J.; Islam, M.T.; Schüffler, A.; Laatsch, H. Metabolites from the endophytic fungus Curvularia sp. M12 act as motility inhibitors against Phytophthora capsici zoospores. J. Nat. Prod. 2017, 80, 347–355. [Google Scholar] [CrossRef]
- Ha, T.M.; Ko, W.; Lee, S.J.; Kim, Y.C.; Son, J.Y.; Sohn, J.H.; Yim, J.H.; Oh, H. Anti-inflammatory effects of curvularin-type metabolites from a marine-derived fungal strain Penicillium sp. SF-5859 in lipopolysaccharide-induced RAW264.7 macrophages. Mar. Drugs 2017, 15, 282. [Google Scholar] [CrossRef]
- Greve, H.; Schupp, P.J.; Eguereva, E.; Kehraus, S.; Kelter, G.; Maier, A.; Fiebig, H.H.; König, G.M. Apralactone A and a new stereochemical class of curvularins from the marine-derived fungus Curvularia sp. Eur. J. Org. Chem. 2008, 2008, 5085–5092. [Google Scholar] [CrossRef]
- Ye, X.; Anjum, K.; Song, T.; Wang, W.; Yu, S.; Huang, H.; Lian, X.Y.; Zhang, Z. A new curvularin glycoside and its cytotoxic and antibacterial analogues from marine actinomycete Pseudonocardia sp. HS7. Nat. Prod. Res. 2016, 30, 1156–1161. [Google Scholar] [CrossRef]
- Xie, L.W.; Ouyang, Y.C.; Zou, K.; Wang, G.H.; Chen, M.J.; Sun, H.M.; Dai, S.K.; Li, X. Isolation and difference in anti-Staphylococcus aureus bioactivity of curvularin derivates from fungus Eupenicillium sp. Appl. Biochem. Biotechnol. 2009, 159, 284–293. [Google Scholar] [CrossRef]
- Trisuwan, K.; Rukachaisirikul, V.; Phongpaichit, S.; Preedanon, S.; Sakayaroj, J. Modiolide and pyrone derivatives from the sea fan-derived fungus Curvularia sp. PSU-F22. Arch. Pharm. Res. 2011, 34, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, M.; Mugishima, T.; Komatsu, K.; Sone, T.; Tanaka, M.; Mikami, Y.; Kobayashi, J. Modiolides A and B, two new 10-membered macrolides from a marine-derived fungus. J. Nat. Prod. 2003, 66, 412–415. [Google Scholar] [CrossRef]
- Du, X.; Lu, C.; Li, Y.; Zheng, Z.; Su, W.; Shen, Y. Three new antimicrobial metabolites of Phomopsis sp. J. Antibiot. (Tokyo) 2008, 61, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, D.K.; Reddy, D.P.; Dash, U.; Yadav, J.S. Total synthesis of Z-isomer of phomolide B. Tetrahedron Lett. 2011, 52, 151–154. [Google Scholar] [CrossRef]
- Lin, X.; Lu, C.-H.; Shen, Y.-M. One new ten-membered lactone from Phomopsis sp. B27, an endophytic fungus of Annona squamosa. Chin. J. Nat. Med. 2008, 6, 391–394. [Google Scholar] [CrossRef]
- Ito, A.; Maeda, H.; Tonouchi, A.; Hashimoto, M. Relative and absolute structure of phomolide C. Biosci. Biotechnol. Biochem. 2015, 79, 1067–1069. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Edrada, R.A.; Heubes, M.; Brauers, G.; Wray, V.; Berg, A.; Gräfe, U.; Wohlfarth, M.; Mühlbacher, J.; Schaumann, K.; Bringmann, G.; et al. Online analysis of xestodecalactones A-C, novel bioactive metabolites from the fungus Penicillium cf montanense and their subsequent isolation from the sponge Xestospongia exigua. J. Nat. Prod. 2002, 65, 1598–1604. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Zhang, J.; Quan, W.; Sun, Y.; She, X.; Pan, X. The first asymmetric total syntheses and determination of absolute configurations of xestodecalactones B and C. J. Org. Chem. 2007, 72, 2694–2697. [Google Scholar] [CrossRef]
- Ebrahim, W.; Aly, A.H.; Mándi, A.; Totzke, F.; Kubbutat, M.H.G.; Wray, V.; Lin, W.-H.; Dai, H.; Proksch, P.; Kurtán, T.; et al. Decalactone derivatives from Corynespora cassiicola, an endophytic fungus of the mangrove plant Laguncularia racemosa. Eur. J. Org. Chem. 2012, 18, 3476–3484. [Google Scholar] [CrossRef]
- Kubota, T.; Iwai, T.; Sakai, K.; Gonoi, T.; Kobayashi, J. Amphidinins C-F, amphidinolide Q analogues from marine dinoflagellate Amphidinium sp. Org. Lett. 2014, 16, 5624–5627. [Google Scholar] [CrossRef] [PubMed]
- Cao, F.; Yang, Q.; Shao, C.L.; Kong, C.J.; Zheng, J.J.; Liu, Y.F.; Wang, C.Y. Bioactive 7-oxabicyclic[6.3.0]lactam and 12-membered macrolides from a gorgonian-derived Cladosporium sp. fungus. Mar. Drugs 2015, 13, 4171–4178. [Google Scholar] [CrossRef]
- Xu, J.; Jiang, C.S.; Zhang, Z.L.; Ma, W.Q.; Guo, Y.W. Recent progress regarding the bioactivities, biosynthesis and synthesis of naturally occurring resorcinolic macrolides. Acta Pharmacol. Sin. 2014, 35, 316–330. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.Y.; Li, C.Y.; Lin, Y.C.; Peng, G.T.; She, Z.G.; Zhou, S.N. Lactones from a brown alga endophytic fungus (No. ZZF36) from the South China Sea and their antimicrobial activities. Bioorg. Med. Chem. Lett. 2006, 16, 4205–4208. [Google Scholar] [CrossRef] [PubMed]
- Shigemori, H.; Kasai, Y.; Komatsu, K.; Tsuda, M.; Mikami, Y.; Kobayashi, J. Sporiolides A and B, new cytotoxic twelve-membered macrolides from a marine-derived fungus Cladosporium species. Mar. Drugs 2004, 2, 164–169. [Google Scholar] [CrossRef]
- Du, Y.; Chen, Q.; Linhardt, R.J. The first total synthesis of sporiolide A. J. Org. Chem. 2006, 71, 8446–8451. [Google Scholar] [CrossRef]
- Jiang, Z.D.; Jensen, P.R.; Fenical, W. Lobophorins A and B, new antiinflammatory macrolides produced by a tropical marine bacterium. Bioorg. Med. Chem. Lett. 1999, 9, 2003–2006. [Google Scholar] [CrossRef]
- Li, S.; Xiao, J.; Zhu, Y.; Zhang, G.; Yang, C.; Zhang, H.; Ma, L.; Zhang, C. Dissecting glycosylation steps in lobophorin biosynthesis implies an iterative glycosyltransferase. Org. Lett. 2013, 15, 1374–1377. [Google Scholar] [CrossRef]
- Niu, S.; Li, S.; Chen, Y.; Tian, X.; Zhang, H.; Zhang, G.; Zhang, W.; Yang, X.; Zhang, S.; Ju, J.; et al. Lobophorins E and F, new spirotetronate antibiotics from a South China Sea-derived Streptomyces sp. SCSIO 01127. J. Antibiot. (Tokyo) 2011, 64, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.Q.; Zhang, S.Y.; Wang, N.; Li, Z.L.; Hua, H.M.; Hu, J.C.; Wang, S.J. New spirotetronate antibiotics, lobophorins H and I, from a South China Sea-derived Streptomyces sp. 12A35. Mar. Drugs 2013, 11, 3891–3901. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Khong, T.T.; Chen, L.; Choi, H.D.; Kang, J.S.; Son, B.W. 8’-Hydroxyzearalanone and 2’-hydroxyzearalanol: Resorcyclic acid lactone derivatives from the marine-derived fungus Penicillium sp. Chem. Pharm. Bull (Tokyo) 2008, 56, 1355–1356. [Google Scholar] [CrossRef] [PubMed]
- Hitchcock, S.A.; Pattenden, G. Synthesis of macrocycles via allylic radical intermediates. A total synthesis of (−)-zearalenone. Tetrahedron Lett. 1990, 31, 3641–3644. [Google Scholar] [CrossRef]
- Zhao, L.L.; Gai, Y.; Kobayashi, H.; Hu, C.Q.; Zhang, H.P. 5′-Hydroxyzearalenol, a new β-resorcylic macrolide from Fusarium sp. 05ABR26. Chin. Chem. Lett. 2008, 19, 1089–1092. [Google Scholar] [CrossRef]
- Arunpanichlert, J.; Rukachaisirikul, V.; Sukpondma, Y.; Phongpaichit, S.; Supaphon, O.; Sakayaroj, J. A β-resorcylic macrolide from the seagrass-derived fungus Fusarium sp. PSU-ES73. Arch. Pharm. Res. 2011, 34, 1633–1637. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.L.; Stout, E.P.; Lin, A.S.; Prudhomme, J.; Le Roch, K.; Fairchild, C.R.; Franzblau, S.G.; Hay, M.E.; Aalbersberg, W.; Kubanek, J. Antimalarial bromophycolides J-Q from the Fijian red alga Callophycus serratus. J. Org. Chem. 2009, 74, 2736–2742. [Google Scholar] [CrossRef] [PubMed]
- Kyeremeh, K.; Acquah, K.S.; Sazak, A.; Houssen, W.; Tabudravu, J.; Deng, H.; Jaspars, M. Butremycin, the 3-hydroxyl derivative of ikarugamycin and a protonated aromatic tautomer of 5’-methylthioinosine from a Ghanaian Micromonospora sp. K310. Mar. Drugs 2014, 12, 999–1012. [Google Scholar] [CrossRef]
- Asolkar, R.N.; Maskey, R.P.; Helmke, E.; Laatsch, H. Chalcomycin B, a new macrolide antibiotic from the marine isolate Streptomyces sp. B7064. J. Antibiot. (Tokyo) 2002, 55, 893–898. [Google Scholar] [CrossRef]
- Jiang, S.; Zhang, L.; Pei, X.; Deng, F.; Hu, D.; Chen, G.; Wang, C.; Hong, K.; Yao, X.; Gao, A.H. Chalcomycins from marine-derived Streptomyces sp. and their antimicrobial activities. Mar. Drugs 2017, 15, 153. [Google Scholar] [CrossRef]
- Stout, E.P.; Hasemeyer, A.P.; Lane, A.L.; Davenport, T.M.; Engel, S.; Hay, M.E.; Fairchild, C.R.; Prudhomme, J.; Le Roch, K.; Aalbersberg, W.; et al. Antibacterial neurymenolides from the Fijian red alga Neurymenia fraxinifolia. Org. Lett. 2009, 11, 225–228. [Google Scholar] [CrossRef]
- Motuhi, S.E.; Feizbakhsh, O.; Foll-Josselin, B.; Baratte, B.; Delehouzé, C.; Cousseau, A.; Fant, X.; Bulinski, J.C.; Payri, C.E.; Ruchaud, S.; et al. Neurymenolide A, a novel mitotic spindle poison from the new caledonian rhodophyta Phacelocarpus neurymenioides. Mar. Drugs 2019, 17, 93. [Google Scholar] [CrossRef]
- Kim, J.; Shin, D.; Kim, S.H.; Park, W.; Shin, Y.; Kim, W.K.; Lee, S.K.; Oh, K.B.; Shin, J.; Oh, D.C. Borrelidins C-E: New antibacterial macrolides from a saltern-derived halophilic Nocardiopsis sp. Mar. Drugs 2017, 15, 166. [Google Scholar] [CrossRef]
- Hunter, T.J.; Zheng, J.; O’Doherty, G.A. Approach to the synthesis of the C1-C11 and C14-C18 portion of leucascandrolide A. Org. Chem. Front. 2016, 3, 1120–1125. [Google Scholar] [CrossRef]
- D’Ambrosio, M.; Guerriero, A.; Pietra, F.; Debitus, C. Leucascandrolide A, a new type of macrolide: The first powerfully bioactive metabolite of calcareous sponges (Leucascandra caveolata, a new genus from the Coral Sea). Helvet. Chim. Acta. 1996, 79, 51–60. [Google Scholar] [CrossRef]
- Fusetani, N.; Sugawara, T.; Matsunaga, S. Cytotoxic metabolites of the marine sponge Mycale adhaerens Lambe. J. Org. Chem. 1991, 56, 4971. [Google Scholar] [CrossRef]
- Nishimura, S.; Matsunaga, S.; Yoshida, S.; Nakao, Y.; Yokoyama, S.; Fusetani, N. 13-Deoxytedanolide, a marine sponge-derived antitumor macrolide, binds to the 60S large ribosomal subunit. Bioorg. Med. Chem. 2005, 13, 449. [Google Scholar] [CrossRef]
- Schlingmann, G.; Milne, L.; Williams, D.R.; Carter, G.T. Cell wall active antifungal compounds produced by the marine fungus Hypoxylon oceanicum LL-15G256. II. Isolation and structure determination. J. Antibiot. (Tokyo) 1998, 51, 303–316. [Google Scholar] [CrossRef]
- Schlingmann, G.; Milne, L.; Carter, G.T. Isolation and identification of antifungal polyesters from the marine fungus Hypoxylon oceanicum LL-15G256. Tetrahedron. 2002, 58, 6825–6835. [Google Scholar] [CrossRef]
- Sakai, R.; Higa, T.; Kashma, Y. Misakinolide-A, an antitumor macrolide from the marine sponge Theonella sp. Chem. Lett. 1986, 1499–1502. [Google Scholar] [CrossRef]
- Kato, Y.; Fusetani, N.; Matsunaga, S.; Hashimoto, K.; Sakai, R.; Higa, T.; Kashman, Y. Antitumor macrodiolides isolated from a marine sponge sp.: Structure revision of misakinolide A. Tetrahedron Lett. 1987, 28, 6225–6228. [Google Scholar] [CrossRef]
- Matsunaga, S.; Fusetani, N.; Hashimoto, K.; Koseki, K.; Noma, M. Kabiramide C, a novel antifungal macrolide from nudibranch eggmasses. J. Am. Chem. Soc. 1986, 108, 847–849. [Google Scholar] [CrossRef]
- Sirirak, T.; Kittiwisut, S.; Janma, C.; Yuenyongsawad, S.; Suwanborirux, K.; Plubrukarn, A. Kabiramides J and K, trisoxazole macrolides from the sponge Pachastrissa nux. J. Nat. Prod. 2011, 74, 1288–1292. [Google Scholar] [CrossRef]
- Ishibashi, M.; Moore, R.E.; Patterson, G.M.L. Scytophycins, cytotoxic and antimycotic agents from the cyanophyte Scytonema pseudohofmanni. J. Org. Chem. 1986, 51, 5300–5306. [Google Scholar] [CrossRef]
- Jung, J.H.; Moore, R.E.; Patterson, G.M.L. Scytophycins from a blue-green alga belonging to the Nostocaceae. Phytochemistry. 1991, 30, 3615–3616. [Google Scholar] [CrossRef]
- Carmeli, S.; Moore, R.E.; Patterson, G.M. Tolytoxin and new scytophycins from three species of Scytonema. J. Nat. Prod. 1990, 53, 1533–1542. [Google Scholar] [CrossRef] [PubMed]
- Patterson, G.M.; Carmeli, S. Biological effects of tolytoxin (6-hydroxy-7-O-methyl-scytophycin b), a potent bioactive metabolite from cyanobacteria. Arch. Microbiol. 1992, 157, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Shishido, T.K.; Humisto, A.; Jokela, J.; Liu, L.; Wahlsten, M.; Tamrakar, A.; Fewer, D.P.; Permi, P.; Andreote, A.P.; Fiore, M.F.; et al. Antifungal compounds from cyanobacteria. Mar. Drugs 2015, 13, 2124–2140. [Google Scholar] [CrossRef]
- Tareq, F.S.; Kim, J.H.; Lee, M.A.; Lee, H.S.; Lee, J.S.; Lee, Y.J.; Shin, H.J. Antimicrobial gageomacrolactins characterized from the fermentation of the marine-derived bacterium Bacillus subtilis under optimum growth conditions. J. Agric. Food Chem. 2013, 61, 3428–3434. [Google Scholar] [CrossRef]
- Ciavatta, M.L.; Lefranc, F.; Carbone, M.; Mollo, E.; Gavagnin, M.; Betancourt, T.; Dasari, R.; Kornienko, A.; Kiss, R. Marine mollusk-derived agents with antiproliferative activity as promising anticancer agents to overcome chemotherapy resistance. Med. Res. Rev. 2017, 37, 702–801. [Google Scholar] [CrossRef]
- Kernan, M.R.; Faulkner, D.J. Halichondramide, an antifungal macrolide from the sponge Halichondria sp. Tetrahedron Lett. 1987, 28, 2809–2812. [Google Scholar] [CrossRef]
- Kernan, M.R.; Molinski, T.F.; Faulkner, D.J. Macrocyclic antifungal metabolites from the Spanish dancer nudibranch Hexabranchus sanguineus and sponges of the genus Halichondria. J. Org. Chem. 1988, 53, 5014–5020. [Google Scholar] [CrossRef]
- Shin, J.; Lee, H.S.; Kim, J.Y.; Shin, H.J.; Ahn, J.W.; Paul, V.J. New macrolides from the sponge Chondrosia corticata. J. Nat. Prod. 2004, 67, 1889–1892. [Google Scholar] [CrossRef]
- Chung, S.C.; Lee, S.H.; Jang, K.H.; Park, W.; Jeon, J.E.; Oh, H.; Shin, J.; Oh, K.B. Actin depolymerizing effect of trisoxazole-containing macrolides. Bioorg. Med. Chem. Lett. 2011, 21, 3198–3201. [Google Scholar] [CrossRef] [PubMed]
- Nagao, T.; Adachi, K.; Sakai, M.; Nishijima, M.; Sano, H. Novel macrolactins as antibiotic lactones from a marine bacterium. J. Antibiot. (Tokyo) 2001, 54, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Jaruchoktaweechai, C.; Suwanborirux, K.; Tanasupawatt, S.; Kittakoop, P.; Menasveta, P. New macrolactins from a marine Bacillus sp. Sc026. J. Nat. Prod. 2000, 63, 984–986. [Google Scholar] [CrossRef] [PubMed]
- Romero-Tabarez, M.; Jansen, R.; Sylla, M.; Lünsdorf, H.; Häussler, S.; Santosa, D.A.; Timmis, K.N.; Molinari, G. 7-O-malonyl macrolactin A, a new macrolactin antibiotic from Bacillus subtilis active against methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci, and a small-colony variant of Burkholderia cepacia. Antimicrob. Agents Chemother. 2006, 50, 1701–1709. [Google Scholar] [CrossRef]
- Timmis, K.N.; Molinari, G.; Jansen, R.; Romero-Tabarez, M.; Santosa, D.A. Antibiotic and Method for Production Thereof. EP1791823A1 European Patent Office. Available online: https://patents.google.com/patent/EP1791823A1/en (accessed on 10 February 2019).
- Yoo, J.S.; Zheng, C.J.; Lee, S.; Kwak, J.H.; Kim, W.G. Macrolactin N, a new peptide deformylase inhibitor produced by Bacillus subtilis. Bioorg. Med. Chem. Lett. 2006, 16, 4889–4892. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.L.; Xu, Q.Z.; Shen, Y.H.; Liu, X.Y.; Jiao, B.H.; Zhang, W.D.; Ni, K.Y. Macrolactin S, a novel macrolactin antibiotic from marine Bacillus sp. Nat. Prod. Res. 2008, 22, 342–347. [Google Scholar] [CrossRef]
- Xue, C.; Tian, L.; Xu, M.; Deng, Z.; Lin, W. A new 24-membered lactone and a new polyene delta-lactone from the marine bacterium Bacillus marinus. J. Antibiot. (Tokyo) 2008, 61, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.H.; Tian, X.P.; Qi, S.H.; Luo, X.M.; Wang, P.; Zhang, S. Antibacterial and antilarval compounds from marine gorgonian-associated bacterium Bacillus amyloliquefaciens SCSIO 00856. J. Antibiot. (Tokyo) 2010, 63, 191–193. [Google Scholar] [CrossRef] [PubMed]
- Mondol, M.A.; Kim, J.H.; Lee, H.S.; Lee, Y.J.; Shin, H.J. Macrolactin W, a new antibacterial macrolide from a marine Bacillus sp. Bioorg. Med. Chem. Lett. 2011, 21, 3832–3835. [Google Scholar] [CrossRef] [PubMed]
- Mondol, M.A.; Tareq, F.S.; Kim, J.H.; Lee, M.; Lee, H.S.; Lee, Y.J.; Lee, J.S.; Shin, H.J. Cyclic ether-containing macrolactins, antimicrobial 24-membered isomeric macrolactones from a marine Bacillus sp. J. Nat. Prod. 2011, 74, 2582–2587. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, K.; Thilakan, B.; Kizhakkekalam, V.K. Antibacterial aryl-crowned polyketide from Bacillus subtilis associated with seaweed Anthophycus longifolius. J. Appl. Microbiol. 2018, 124, 108–125. [Google Scholar] [CrossRef]
- Pathirana, C.; Tapiolas, D.; Jensen, P.R.; Dwight, R.; Fenical, W. Structure determination of maduralide: A new 24-membered ring macrolide glycoside produced by a marine bacterium (actinomycetales). Tetrahedron Lett. 1991, 32, 2323–2326. [Google Scholar] [CrossRef]
- Sato, S.; Iwata, F.; Yamada, S.; Katayama, M. Neomaclafungins A-I: Oligomycin-class macrolides from a marine-derived actinomycete. J. Nat. Prod. 2012, 75, 1974–1982. [Google Scholar] [CrossRef]
- Searle, P.A.; Molinski, T.F. Phorboxazoles A and B: Potent cytostatic macrolides from marine sponge Phorbas sp. J. Am. Chem. Soc. 1995, 117, 8126–8131. [Google Scholar] [CrossRef]
- Yao, T.; Liu, Z.; Li, T.; Zhang, H.; Liu, J.; Li, H.; Che, Q.; Zhu, T.; Li, D.; Li, W. Characterization of the biosynthetic gene cluster of the polyene macrolide antibiotic reedsmycins from a marine-derived Streptomyces strain. Microb. Cell Fact. 2018, 17, 98. [Google Scholar] [CrossRef]
- Che, Q.; Li, T.; Liu, X.; Yao, T.; Li, J.; Gu, Q.; Li, D.; Li, W.; Zhu, T. Genome scanning inspired isolation of reedsmycins A-F, polyene-polyol macrolides from Streptomyces sp. CHQ-64. RSC Adv. 2015, 5, 22777–22782. [Google Scholar] [CrossRef]
- Kwon, H.C.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Marinisporolides, polyene-polyol macrolides from a marine actinomycete of the new genus Marinispora. J. Org. Chem. 2009, 74, 675–684. [Google Scholar] [CrossRef]
- Yuan, G.; Lin, H.; Wang, C.; Hong, K.; Liu, Y.; Li, J. 1H and 13C assignments of two new macrocyclic lactones isolated from Streptomyces sp. 211726 and revised assignments of azalomycins F3a, F4a and F5a. Magn. Reson. Chem. 2011, 49, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Yuan, G.; Hong, K.; Lin, H.; She, Z.; Li, J. New azalomycin F analogs from mangrove Streptomyces sp. 211726 with activity against microbes and cancer cells. Mar. Drugs 2013, 11, 817–829. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.G.; Moon, K.; Kim, S.H.; Park, S.H.; Park, S.; Lee, S.K.; Oh, K.B.; Shin, J.; Oh, D.C. Bahamaolides A and B, antifungal polyene polyol macrolides from the marine actinomycete Streptomyces sp. J. Nat. Prod. 2012, 75, 959–967. [Google Scholar] [CrossRef]
- Pérez, M.; Schleissner, C.; Fernández, R.; Rodríguez, P.; Reyes, F.; Zuñiga, P.; de la Calle, F.; Cuevas, C. PM100117 and PM100118, new antitumor macrolides produced by a marine Streptomyces caniferus GUA-06-05-006A. J. Antibiot. (Tokyo) 2016, 69, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Salcedo, R.G.; Olano, C.; Gómez, C.; Fernández, R.; Braña, A.F.; Méndez, C.; de la Calle, F.; Salas, J.A. Characterization and engineering of the biosynthesis gene cluster for antitumor macrolides PM100117 and PM100118 from a marine actinobacteria: Generation of a novel improved derivative. Microb. Cell Fact. 2016, 15, 44. [Google Scholar] [CrossRef]
- Salvador-Reyes, L.A.; Sneed, J.; Paul, V.J.; Luesch, H. Amantelides A and B, polyhydroxylated macrolides with differential broad-spectrum cytotoxicity from a Guamanian marine cyanobacterium. J. Nat. Prod. 2015, 78, 1957–1962. [Google Scholar] [CrossRef]
- Pettit, G.R.; Cichacz, Z.A.; Herald, C.L.; Boyd, M.R.; Schmidt, J.M.; Hooper, J.N.A. Isolation and structure of spongistatin 1. J. Org. Chem. 1993, 58, 1302–1304. [Google Scholar] [CrossRef]
- Pettit, G.R.; Cichacz, Z.A.; Gao, F.; Herald, C.L.; Boyd, M.R. Isolation and structure of the remarkable human cancer cell growth inhibitors spongistatins 2 and 3 from an Eastern Indian Ocean Spongia sp. J. Chem. Soc. Chem. Commun. 1993, 14, 1166–1168. [Google Scholar] [CrossRef]
- Pettit, G.R.; Herald, C.L.; Cichacz, Z.A.; Gao, F.; Schmidt, J.M.; Boyd, M.R.; Christie, N.D.; Boettner, F.E. Isolation and structure of the powerful human cancer cell growth inhibitors spongistatins 4 and 5 from an African Spirastrella spinispirulifera (Porifera). J. Chem. Soc. Chem. Commun. 1993, 24, 1805–1807. [Google Scholar] [CrossRef]
- Pettit, G.R.; Herald, C.L.; Cichacz, Z.A.; Gao, F.; Schmidt, J.M.; Boyd, M.R.; Christie, N.D.; Boettner, F.E. Antineoplastic agents 293. The exceptional human cancer cell growth inhibitors spongistatins 6 and 7. Nat. Prod. Lett. 1993, 3, 239–244. [Google Scholar] [CrossRef]
- Pettit, R.K.; McAllister, S.C.; Pettit, G.R.; Herald, C.L.; Johnson, J.M.; Cichacz, Z.A. A broad-spectrum antifungal from the marine sponge Hyrtios erecta. Int. J. Antimicrob. Agents. 1997, 9, 147–152. [Google Scholar] [CrossRef]
- Kasanah, N.; Hamann, M.T. Development of antibiotics and the future of marine microorganisms to stem the tide of antibiotic resistance. Curr. Opin. Investig. Drugs. 2004, 5, 827–837. [Google Scholar]
- Fair, R.J.; Tor, Y. Antibiotics and bacterial resistance in the 21st century. Perspect. Medicin. Chem. 2014, 6, 25–64. [Google Scholar] [CrossRef]
- Abdelmohsen, U.R.; Balasubramanian, S.; Oelschlaeger, T.A.; Grkovic, T.; Pham, N.B.; Quinn, R.J.; Hentschel, U. Potential of marine natural products against drug-resistant fungal, viral, and parasitic infections. Lancet Infect. Dis. 2017, 17, e30–e41. [Google Scholar] [CrossRef]
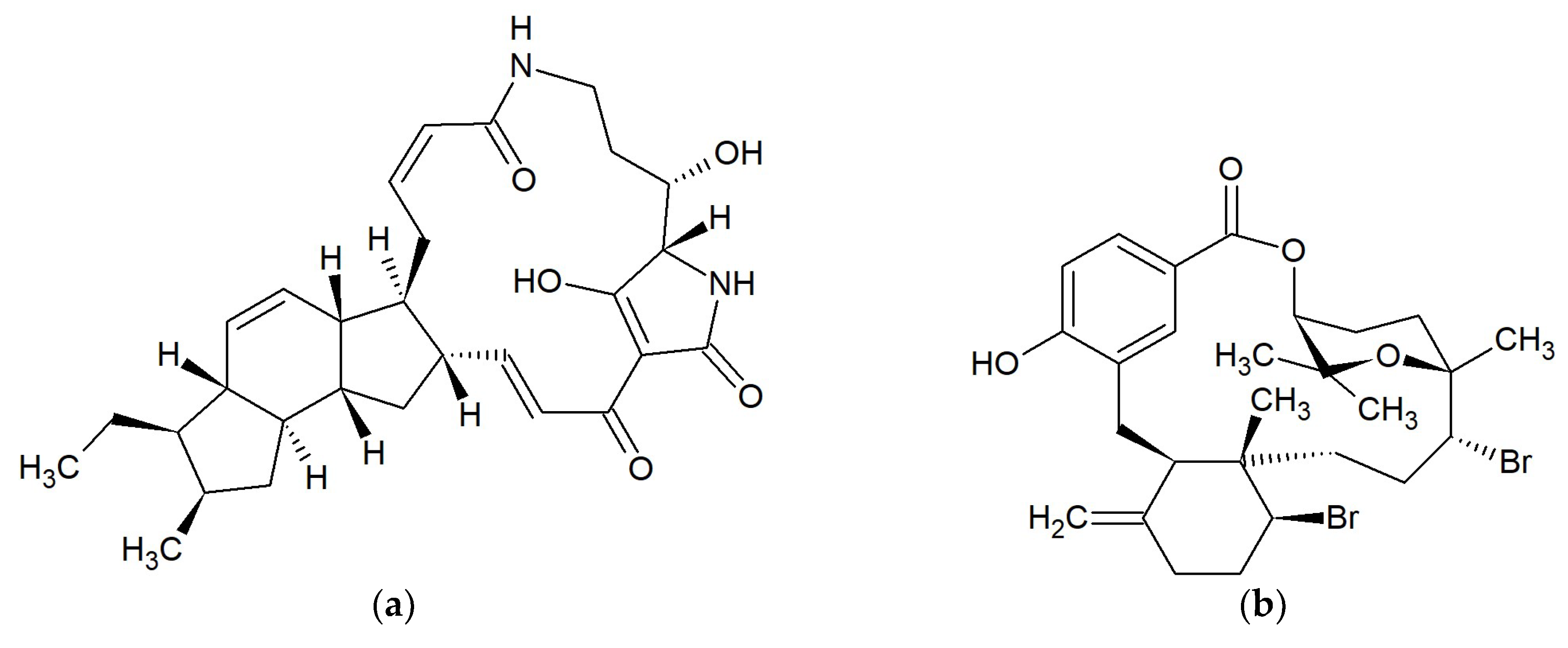

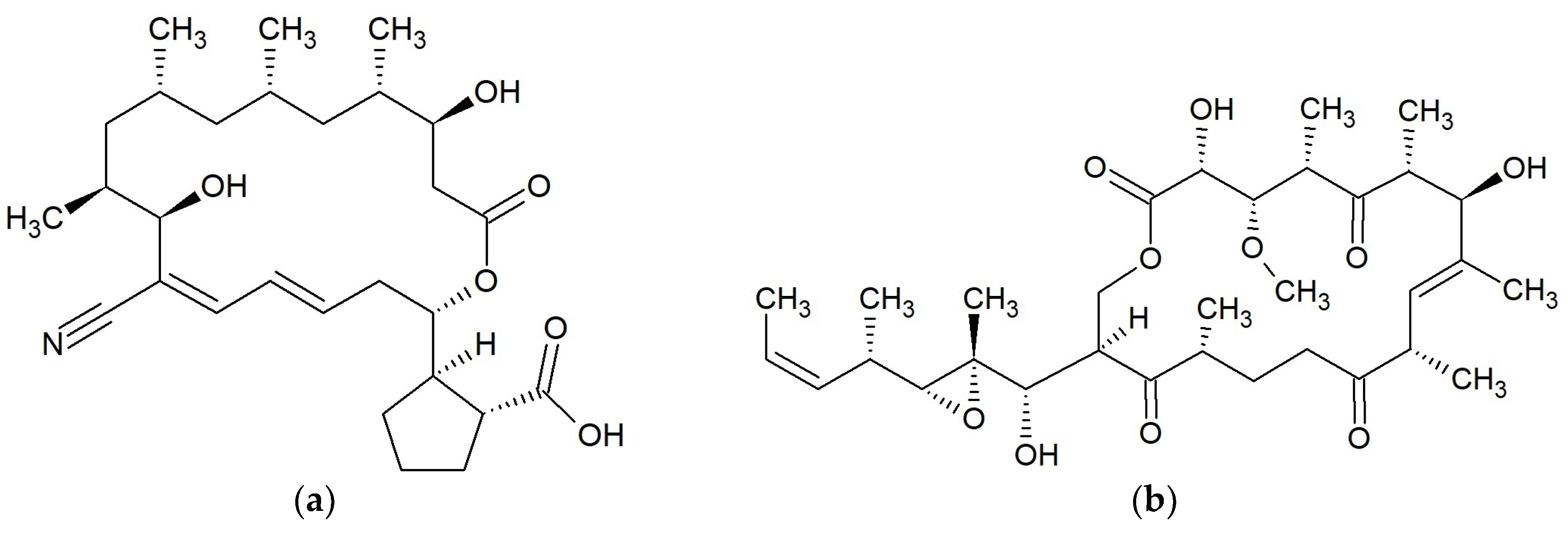
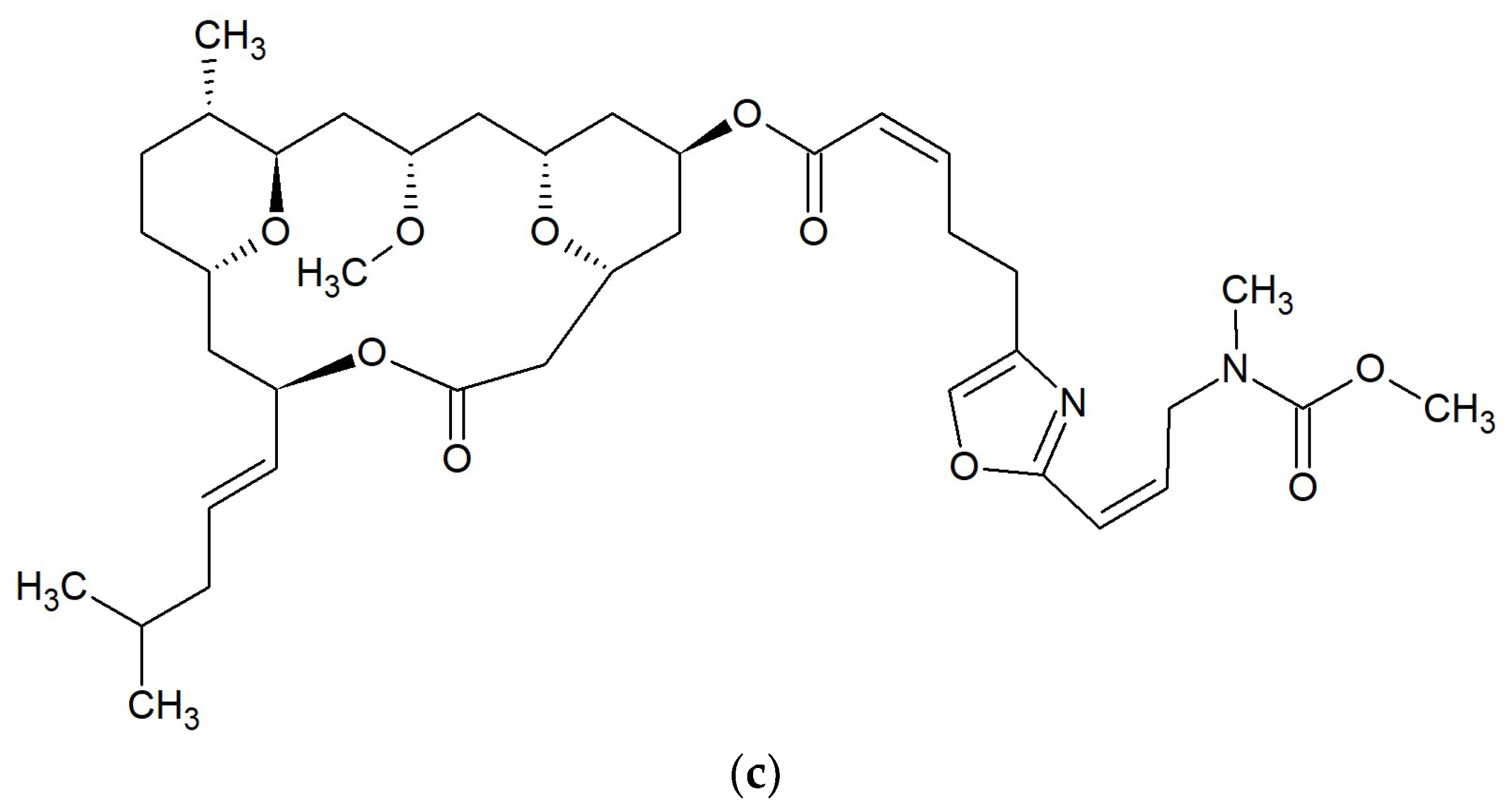
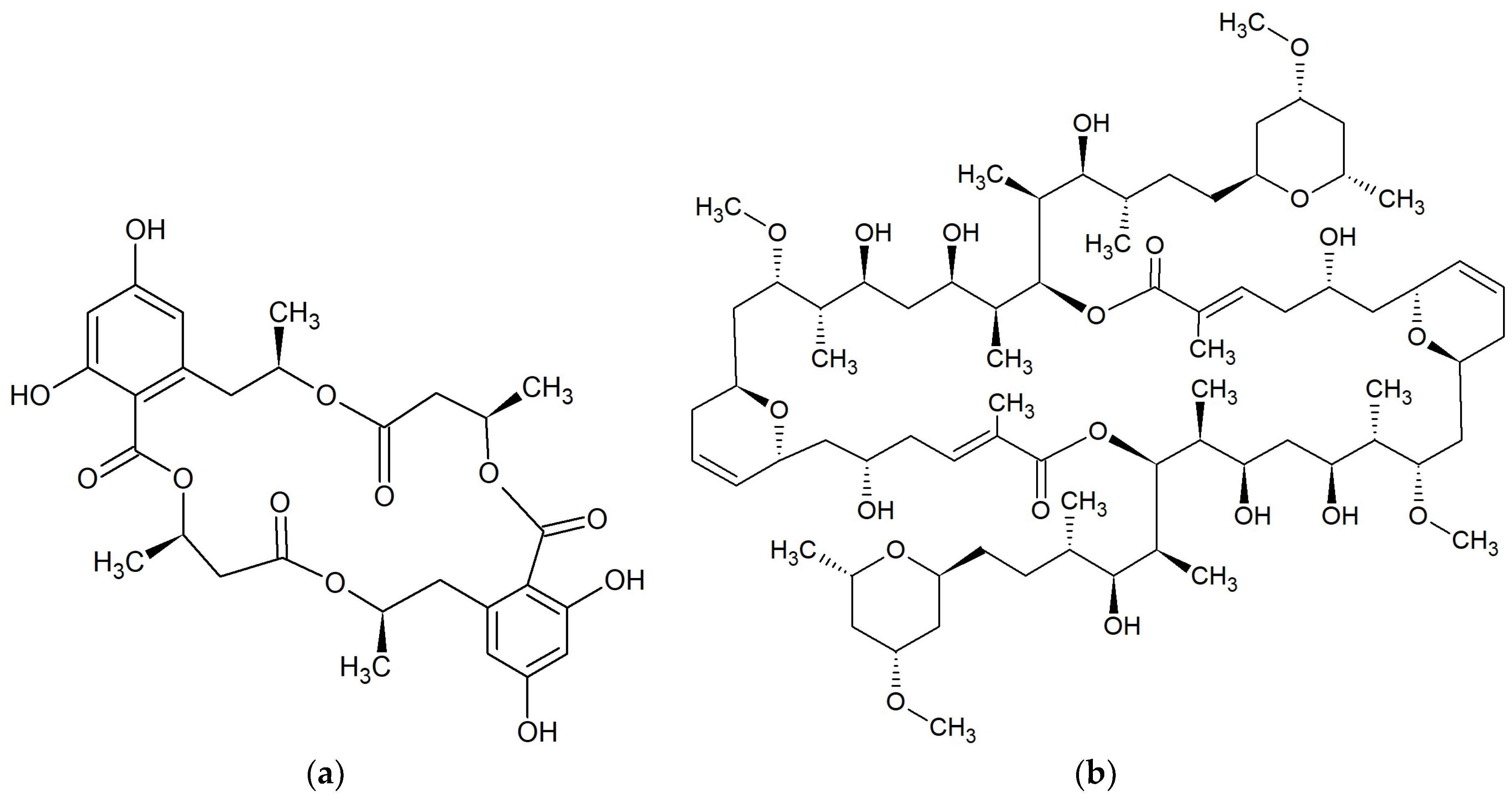
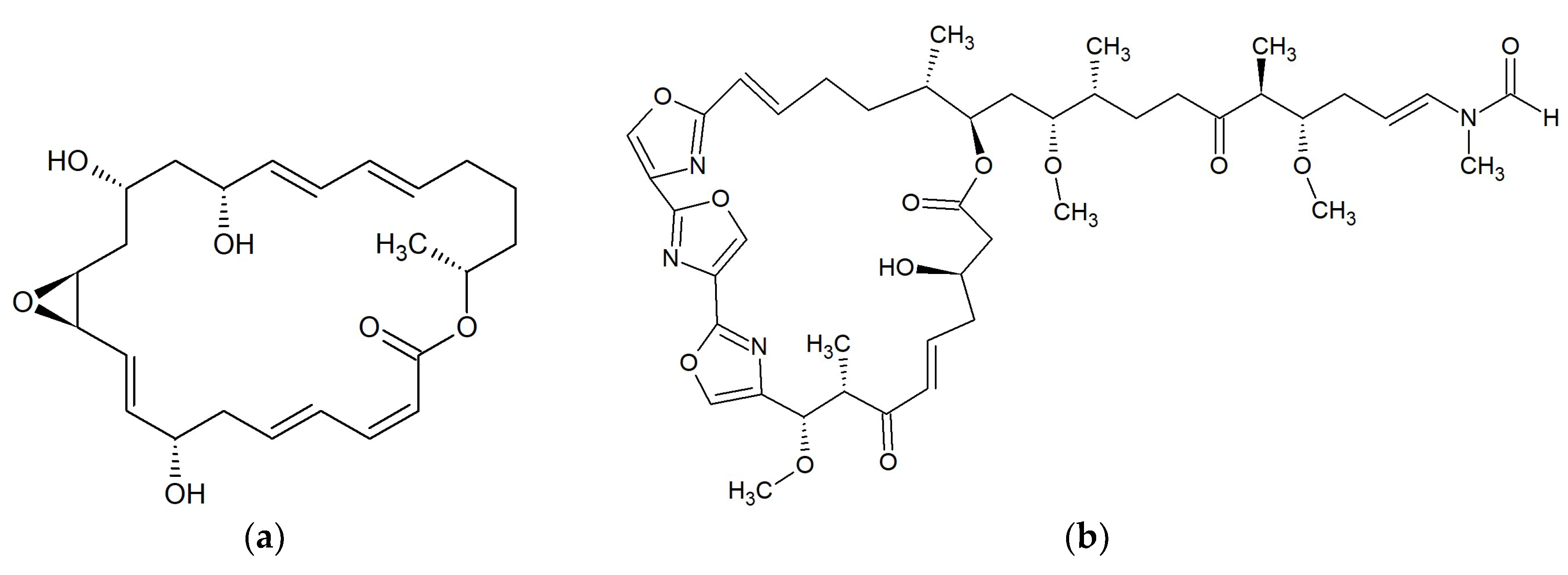
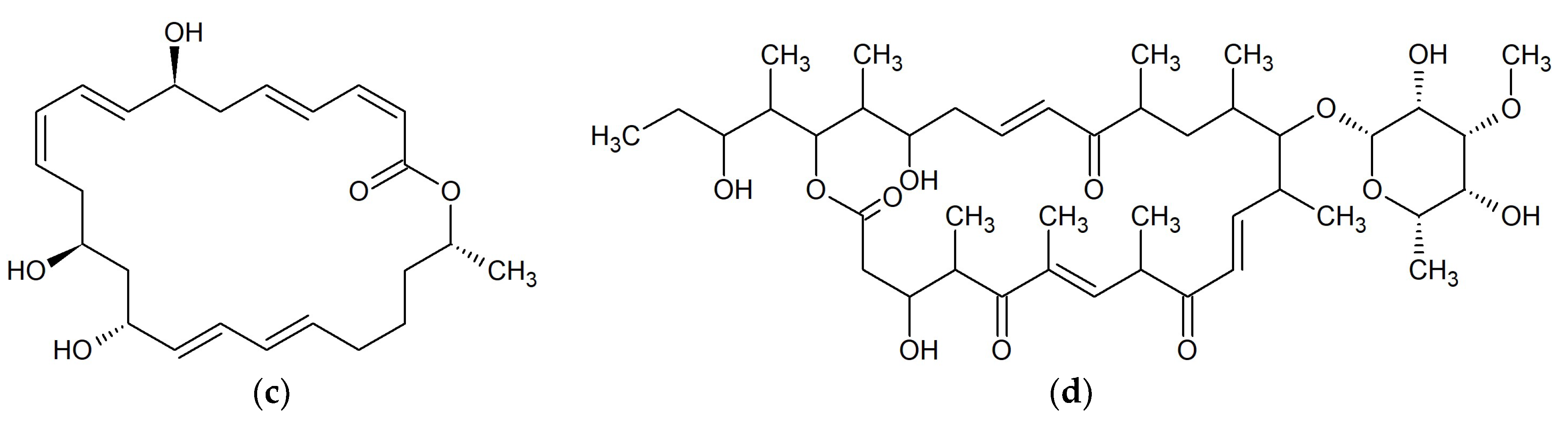
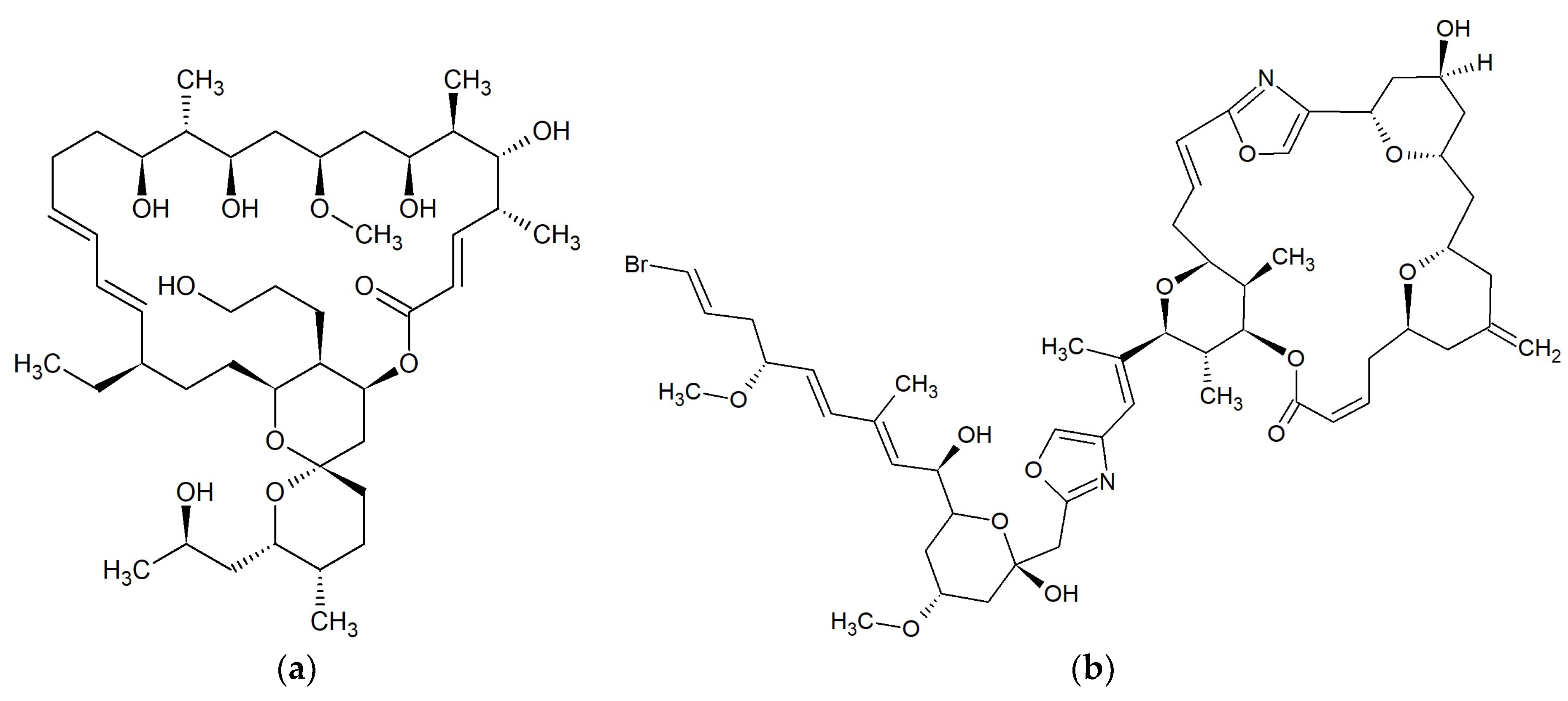


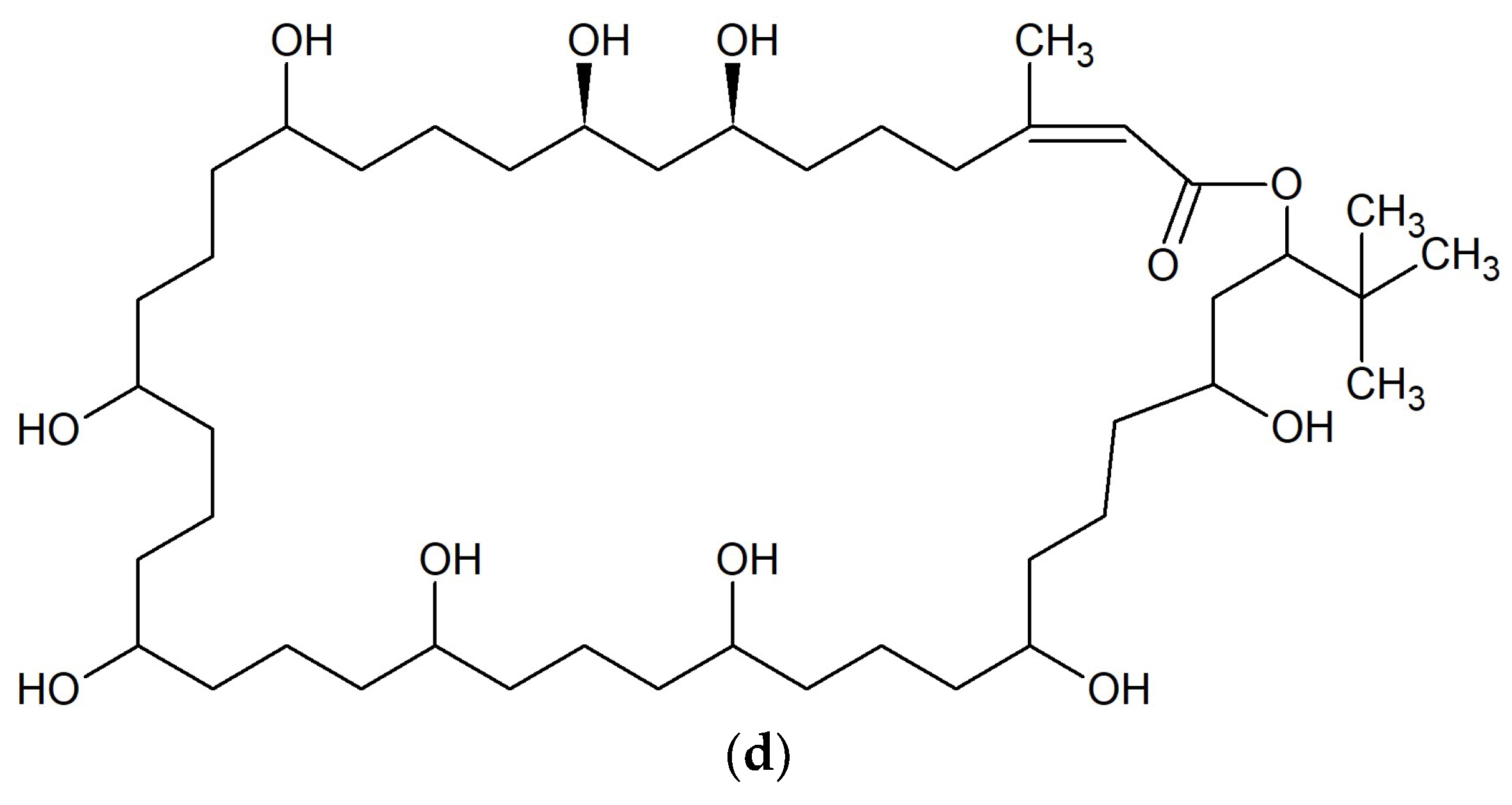
| No. | Macrolide | Source | Target | References |
|---|---|---|---|---|
| 1 | (19Z)-halichondramide | sponge Chondrosia corticata | Fungi: Candida albicans, Aspergillus niger, Aspergillus fumigatus, Trichophyton rubrum, T. mentagrophytes | [75,76] |
| 2 | (S)-dehydrocurvularin | fungi Curvularia sp. | Fungi: Phytophthora capsici | [21] |
| 3 | 11-hydroxycurvularin | actinomycete Pseudonocardia sp. | Bacteria: Escherichia coli | [24] |
| 4 | 13-Deoxytedanolide | sponge Mycale adhaerens | Fungi: Saccharomyces cerevisiae | [59] |
| 5 | 15G256w | fungus Hypoxylon oceanicum | Fungi: Neurospora crassa | [60,61] |
| 6 | 15G256ι | fungus Hypoxylon oceanicum | Fungi: Neurospora crassa | [60,61] |
| 7 | 19-O-demethylscytophycin C | algae Scytonema mirabile, S. burmanicum, S. ocellatum | Fungi: Aspergillus oryzae, Candida albicans, Penicillium notatum, Saccharomyces cerevisiae | [68] |
| 8 | 5-hydroxy-de-O-methyllasiodiplodin | fungus No. ZZF36 | Bacteria: Staphylococcus aureus | [38] |
| 9 | 6-hydroxyscytophycin B | algae Cylindrospermum muscicola, Scytonema mirabile, S. burmanicum, S. ocellatum | Fungi: Aspergillus oryzae, Candida albicans, Penicillium notatum, Saccharomyces cerevisiae | [67,68] |
| 10 | 7-O-malonylmacrolactin A | bacteria Bacillus subtilis | Bacteria: Staphylococcus aureus, Enterococcus sp., Burkholderia cepacia; Fungi: Candida crusei | [79,80] |
| 11 | 7-O-succinylmacrolactin A and F | bacteria Bacillus sp. | Bacteria: Bacillus subtilis, Staphylococcus aureus | [78] |
| 12 | 8’-hydroxyzearalanone | fungi Penicillium sp. | Fungi: Pyricularia oryzae | [45,47] |
| 13 | Amantelide A | cyanobacterium from family Oscillatoriales | Bacteria: Staphylococcus aureus, Pseudomonas aeruginosa; Fungi: Dendryphiella salina, Lindra thalassiae, Fusarium sp. | [99] |
| 14 | Amantelide B | cyanobacterium from family Oscillatoriales | Fungi: Dendryphiella salina | [99] |
| 15 | Amphidinolide Q | dinoflagellate Amphidinium sp. | Bacteria: Staphylococcus aureus, Bacillus subtilis, Escherichia coli; Fungi: Candida albicans | [35] |
| 16 | Aryl-crowned polyketide macrolactin | bacterium Bacillus subtilis | Bacteria: Escherichia coli, Aeromonas hydrophilla, Pseudomonas aeruginosa, Klebsiella pneumoniae, Vibrio sp. | [87] |
| 17 | Azalomycin F analogs | bacteria Streptomyces sp. | Bacteria: Staphylococcus aureus, Bacillus subtilis, Escherichia coli; Fungi: Candida albicans | [94,95] |
| 18 | Bahamaolide A | actinomycete Streptomyces sp. | Fungi: Candida albicans, Aspergillus fumigatus, Trichophyton rubrum, T. mentagrophytes | [95] |
| 19 | Borrelidin | actinomycete Nocardiopsis sp. | Bacteria: Enterococcus faecalis, E. faecium, Proteus hauseri, Klebsiella pneumoniae, Salmonella enterica | [53] |
| 20 | Borrelidins C and D | actinomycete Nocardiopsis sp. | Bacteria: Salmonella enterica | [55] |
| 21 | Bromophycolides P and Q | alga Callophycus serratus | Bacteria: Staphylococcus aureus, Enterococcus faecium | [50] |
| 22 | Butremycin | bacteria Micromonospora sp. | Bacteria: Staphylococcus aureus; Escherichia coli | [49] |
| 23 | Chalcomycins A and B | bacteria Streptomyces sp. | Bacteria: Staphylococcus aureus, Bacillus subtilis, Escherichia coli | [51,52] |
| 24 | Curvularin | fungi Curvularia sp., Eupenicillium sp. | Bacteria: Bacillus subtilis; Fungi: Phytophthora capsici, Saccharomyces cerevisiae, Sclerotinia sclerotiorum | [21,25] |
| 25 | Dendrodolides A, C and M | fungi Cladosporium sp. | Bacteria: Bacillus cereus, Tetragenococcus halophilus, Staphylococcus epidermidis, Staphylococcus aureus, Escherichia coli, Pseudomonas putida, Nocardia brasiliensis, Vibrio parahaemolyticus | [36] |
| 26 | de-O-methyllasiodiplodin | fungus No. ZZF36 | Bacteria: Staphylococcus aureus, Bacillus subtilis, Salmonella enteritidis; Fungi: Candida albicans, Fusarium oxysporum f.sp. cubense | [38] |
| 27 | Dihydrochalcomycin | bacteria Streptomyces sp. | Bacteria: Staphylococcus aureus | [52] |
| 28 | Dihydrohalichondramide | sponge Halichondria sp. | Fungi: Candida albicans | [74] |
| 29 | Gageomacrolactins | bacterium Bacillus subtilis | Bacteria: Staphylococcus aureus, Bacillus subtilis, B. cereus, Escherichia coli, Salmonella typhi, Pseudomonas aeruginosa; Fungi: Aspergillus niger, Botrytis cinerea, Colletotrichum acutatum, Candida albicans, Rhizoctonia solani | [71] |
| 30 | Halichondramide | sponge Halichondria sp. | Fungi: Candida albicans, Trichophyton mentagrophytes, Aspergillus fumigatus, Trichophyton rubrum, T. mentagrophytes | [73,76] |
| 31 | Isohalichondramide | sponge Halichondria sp. | Fungi: Candida albicans | [74] |
| 32 | Kabiramide C | unidentified nudibranch | Fungi: Candida albicans, Aspergillus niger, Penicillium citrium, Trichophyton interdigitae | [64] |
| 33 | Lasiodiplodin | fungus No. ZZF36 | Bacteria: Staphylococcus aureus, Bacillus subtilis; Fungi: Fusarium oxysporum | [38] |
| 34 | Leucascandrolide A | sponge Leucascandra caveolata | Fungi: Fusarium oxysporum, Helminthosporium sativum, Phytophtora hevea, Botrytis cinerea, Pyricularia oryzae, Candida albicans | [57] |
| 35 | Lobophorin A | bacteria actinomycete | Bacteria: Bacillus thuringensis | [41,43] |
| 36 | Lobophorin B | bacteria actinomycete | Bacteria: Bacillus thuringensis, Bacillus subtilis | [41,44] |
| 37 | Lobophorin E | bacteria Streptomyces sp. | Bacteria: Bacillus thuringensis, Bacillus subtilis | [43,44] |
| 38 | Lobophorin F | bacteria Streptomyces sp. | Bacteria: Bacillus thuringensis, Bacillus subtilis, Staphylococcus aureus, Enterococcus faecalis | [43,44] |
| 39 | Lobophorin H | bacteria Streptomyces sp. | Bacteria: Bacillus subtilis, Staphylococcus aureus | [44] |
| 40 | Lobophorin I | bacteria Streptomyces sp. | Bacteria: Bacillus subtilis | [44] |
| 41 | Macrolactin A | bacteria Bacillus sp., B. subtilis, B. marinus | Bacteria: Staphylococcus aureus, Bacillus subtilis, Escherichia coli; Fungi: Aspergillus niger, Botrytis cinerea, Colletotrichum acutatum, Candida albicans, Rhizoctonia solani | [71,77,82,83] |
| 42 | Macrolactin B | bacteria Bacillus subtilis, B. marinus | Bacteria: Staphylococcus aureus, Escherichia coli; Fungi: Aspergillus niger, Botrytis cinerea, Colletotrichum acutatum, Candida albicans, Rhizoctonia solani, Pyricularia oryzae, Alternaria solani | [71,82,83] |
| 43 | Macrolactin D | bacterium Bacillus marinus | Bacteria: Staphylococcus aureus; Fungi: Pyricularia oryzae, Alternaria solani | [83] |
| 44 | Macrolactin F | bacteria Bacillus sp., B. subtilis | Bacteria: Staphylococcus aureus, Bacillus subtilis; Fungi: Aspergillus niger, Botrytis cinerea, Colletotrichum acutatum, Candida albicans, and Rhizoctonia solani | [71,77,78] |
| 45 | Macrolactin K | bacteria Bacillus sp. | Bacteria: Staphylococcus aureus, Bacillus subtilis | [77] |
| 46 | Macrolactin N | bacteria Bacillus subtilis | Bacteria: Escherichia coli, Staphylococcus aureus, Bacillus subtilis | [81] |
| 47 | Macrolactin S | bacteria Bacillus sp., B. marinus, B. amyloliquefaciens | Bacteria: Escherichia coli, Bacillus subtilis, Staphylococcus aureus | [82,83,84] |
| 48 | Macrolactin V | bacterium Bacillus amyloliquefaciens | Bacteria: Escherichia coli, Bacillus subtilis, Staphyloccocus aureus | [84] |
| 49 | Macrolactin W | bacteria Bacillus sp., B. subtilis | Bacteria: Bacillus subtilis, Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa; Fungi: Aspergillus niger, Botrytis cinerea, Colletotrichum acutatum, Candida albicans, Rhizoctonia solani | [71,85] |
| 50 | Macrolactins G, H, I, J, L and M | bacteria Bacillus sp. | Bacteria: Staphylococcus aureus, Bacillus subtilis | [77] |
| 51 | Maduralide | bacteria actinomycete | Bacteria: Bacillus subtilis | [88] |
| 52 | Marinisporolides A and B | actinomycete Marinispora sp. | Fungi: Candida albicans | [93] |
| 53 | Misakinolide A | sponge Theonella sp. | Fungi: Candida albicans | [62] |
| 54 | Modiolide A | fungi Paraphaeosphaeria sp., Curvularia sp. | Bacteria: Micrococcus luteus, Staphylococcus aureus; Fungi: Neurospora crassa, Phytophthora capsici, Microsporum gypseum | [21,26,27] |
| 55 | Modiolide B | fungi Paraphaeosphaeria sp. | Bacteria: Micrococcus luteus; Fungi: Neurospora crassa | [27] |
| 56 | Neohalichondramide | sponge Chondrosia corticata | Bacteria: Fungi: Candida albicans, Aspergillus niger | [75] |
| 57 | Neomaclafungins A-I | bacteria Actinoalloteichus sp. | Fungi: Trichophyton mentagrophytes | [89] |
| 58 | Neurymenolide A | alga Neurymenia fraxinifolia | Bacteria: Staphylococcus aureus, Enterococcus faecium | [53] |
| 59 | Phomolide A | fungi Phomopsis sp. | Bacteria: Escherichia coli; Fungi: Candida albicans, Saccharomyces cerevisiae | [28] |
| 60 | Phomolide B | fungi Phomopsis sp. | Bacteria: Escherichia coli; Fungi: Candida albicans, Saccharomyces cerevisiae | [28] |
| 61 | Phorboxazoles A and B | sponge Phorbas sp. | Fungi: Candida albicans, Saccharomyces carlsbergensis | [90] |
| 62 | PM100117 | bacterium Streptomyces caniferus | Fungi: Candida albicans, Saccharomyces cerevisiae | [97,98] |
| 63 | PM100118 | bacterium Streptomyces caniferus | Fungi: Candida albicans | [97] |
| 64 | Reedsmycins A-E | bacteria Streptomyces sp., S. youssoufiensis | Fungi: Candida albicans | [91,92] |
| 65 | Scytophycins | algae Scytonema sp., S. pseudohofmanni, Cylindrospermum muscicola, Anabaena sp., Nostoc sp. | Fungi: Candida albicans, Aspergillus flavus | [66,67,71] |
| 66 | Secohalichondramide | sponge Chondrosia corticata | Fungi: Candida albicans, Aspergillus niger | [75] |
| 67 | Spongistatin 1 | porifera Spongia sp., Hyrtios erecta | Fungi: Candida albicans, Cryptococcus neoformans, Issatchenkia orientalis, Rhodotorula mucilaginosa, Aspergillus fumigatus, Rhizopus oligosporus | [100,101,104] |
| 68 | Spongistatins 2-7 | porifera Hyrtios erecta, Spirastrella spinispirulifera | Fungi: Candida albicans, Cryptococcus neoformans | [101,102,103,104] |
| 69 | Sporiolide A | fungi Cladosporium sp. | Bacteria: Micrococcus luteus; Fungi: Aspergillus niger, Candida albicans, Cryptococcus neoformans, Neurospora crassa | [39] |
| 70 | Sporiolide B | fungi Cladosporium sp. | Bacteria: Micrococcus luteus | [39] |
| 71 | Tolytoxin (6-hydroxy-7-O-methylscytophycin B) | algae Cylindrospermum muscicola, Scytonema mirabile, S. burmanicum, S. ocellatum, Tolypothrix conglutinata var. colorata | Fungi: Aspergillus oryzae, Candida albicans, Penicillium notatum, Saccharomyces cerevisiae Alternaria alternata, Bipolaris incurvata, Calonectria critalarae, Colletotrichum coccodes, Phyllosticta capitalensis, Phytophtora nicotianae, Rhizoctonia solani, Sclerotium rofsii, Thielaviopsis paradoxa. Trichophyton mentagrophytes | [67,68,69] |
| 72 | Xestodecalactone B | fungus Penicillium cf. montanense | Fungi: Candida albicans | [32] |
| 73 | Zearalanone | fungi Penicillium sp., Fusarium sp. | Bacteria: Staphylococcus aureus; Fungi: Pyricularia oryzae Cryptococcus neoformans | [45,47,48] |
| 74 | αβ-dehydrocurvularin | fungi Eupenicillium sp. | Bacteria: Bacillus subtilis, Staphylococcus aureus; Fungi: Saccharomyces cerevisiae, Sclerotinia sclerotiorum | [25] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karpiński, T.M. Marine Macrolides with Antibacterial and/or Antifungal Activity. Mar. Drugs 2019, 17, 241. https://doi.org/10.3390/md17040241
Karpiński TM. Marine Macrolides with Antibacterial and/or Antifungal Activity. Marine Drugs. 2019; 17(4):241. https://doi.org/10.3390/md17040241
Chicago/Turabian StyleKarpiński, Tomasz M. 2019. "Marine Macrolides with Antibacterial and/or Antifungal Activity" Marine Drugs 17, no. 4: 241. https://doi.org/10.3390/md17040241
APA StyleKarpiński, T. M. (2019). Marine Macrolides with Antibacterial and/or Antifungal Activity. Marine Drugs, 17(4), 241. https://doi.org/10.3390/md17040241