C-phycocyanin from Limnothrix Species KNUA002 Alleviates Cisplatin-Induced Ototoxicity by Blocking the Mitochondrial Apoptotic Pathway in Auditory Cells
Abstract
1. Introduction
2. Results and Discussion
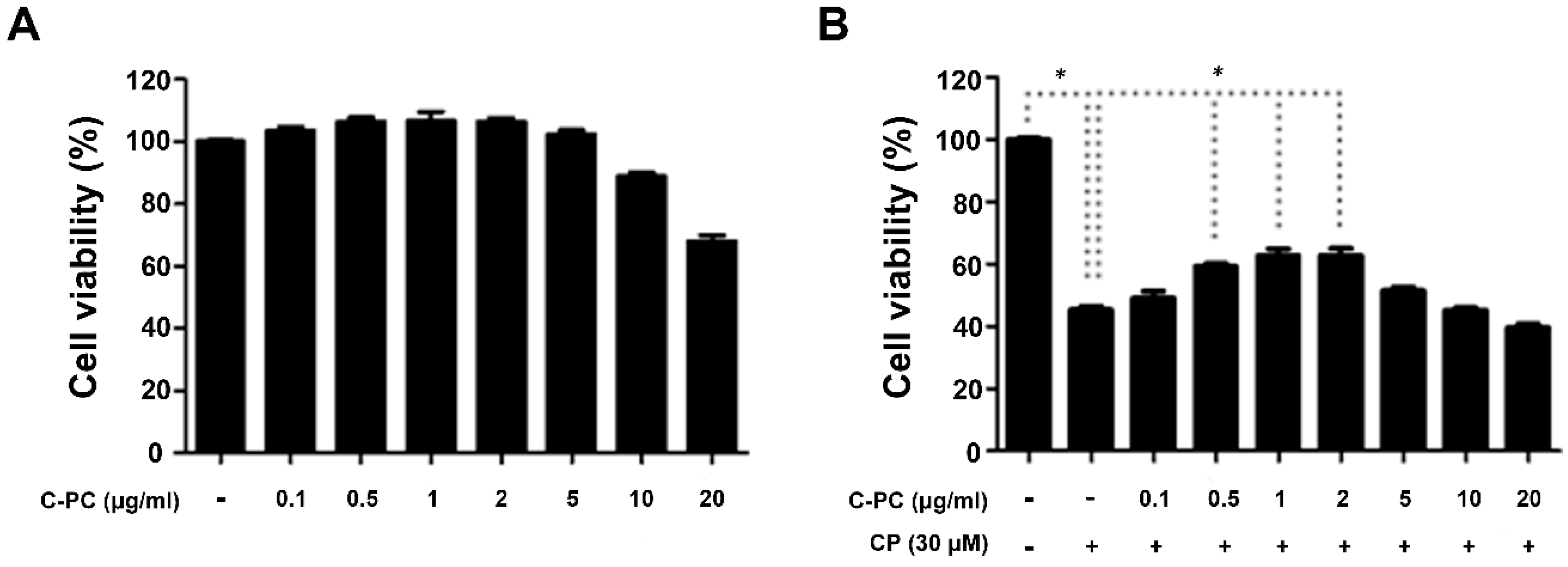
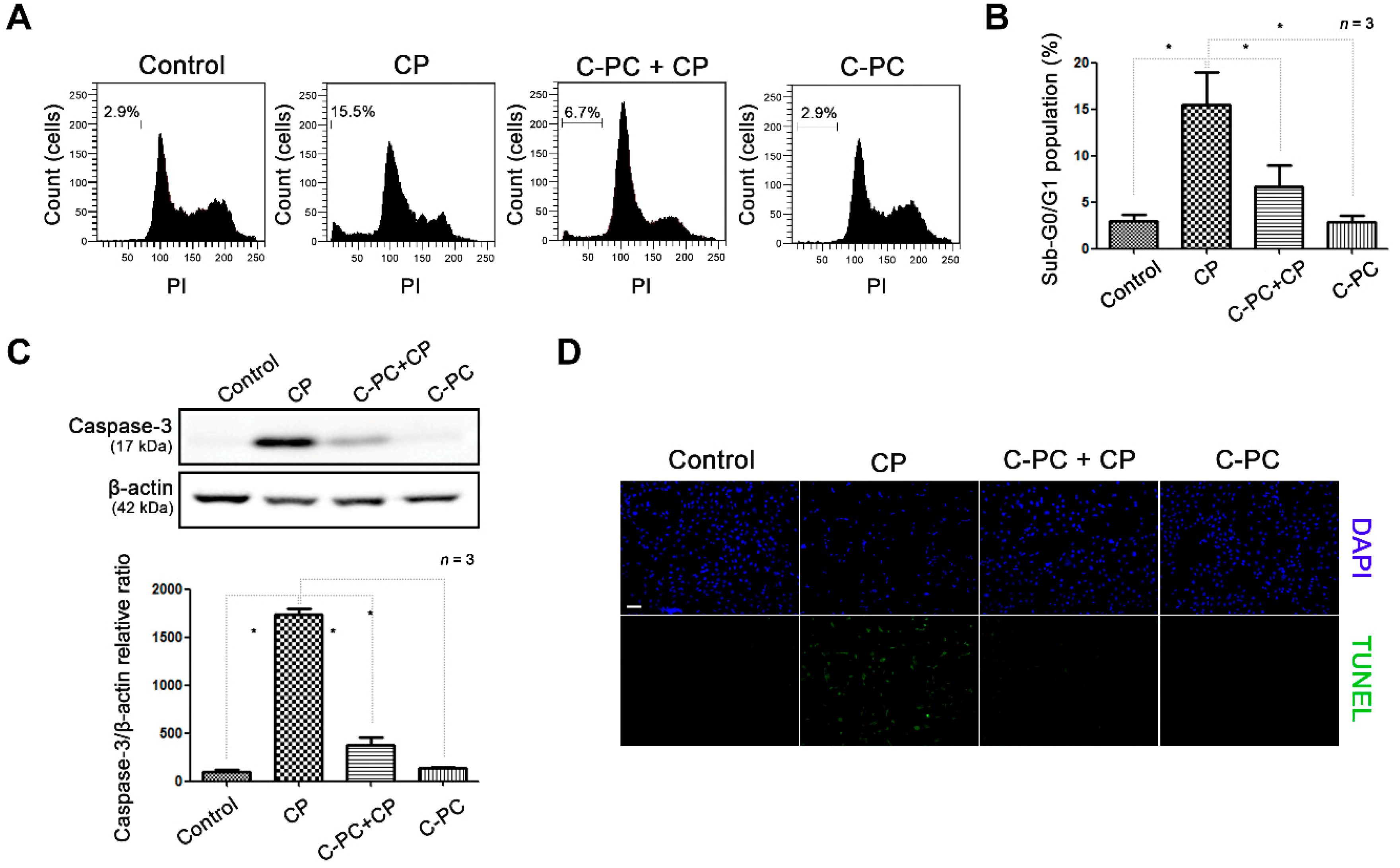
2.1. C-PC Alleviates Cisplatin-Induced Apoptosis in HEI-OC1 Cells
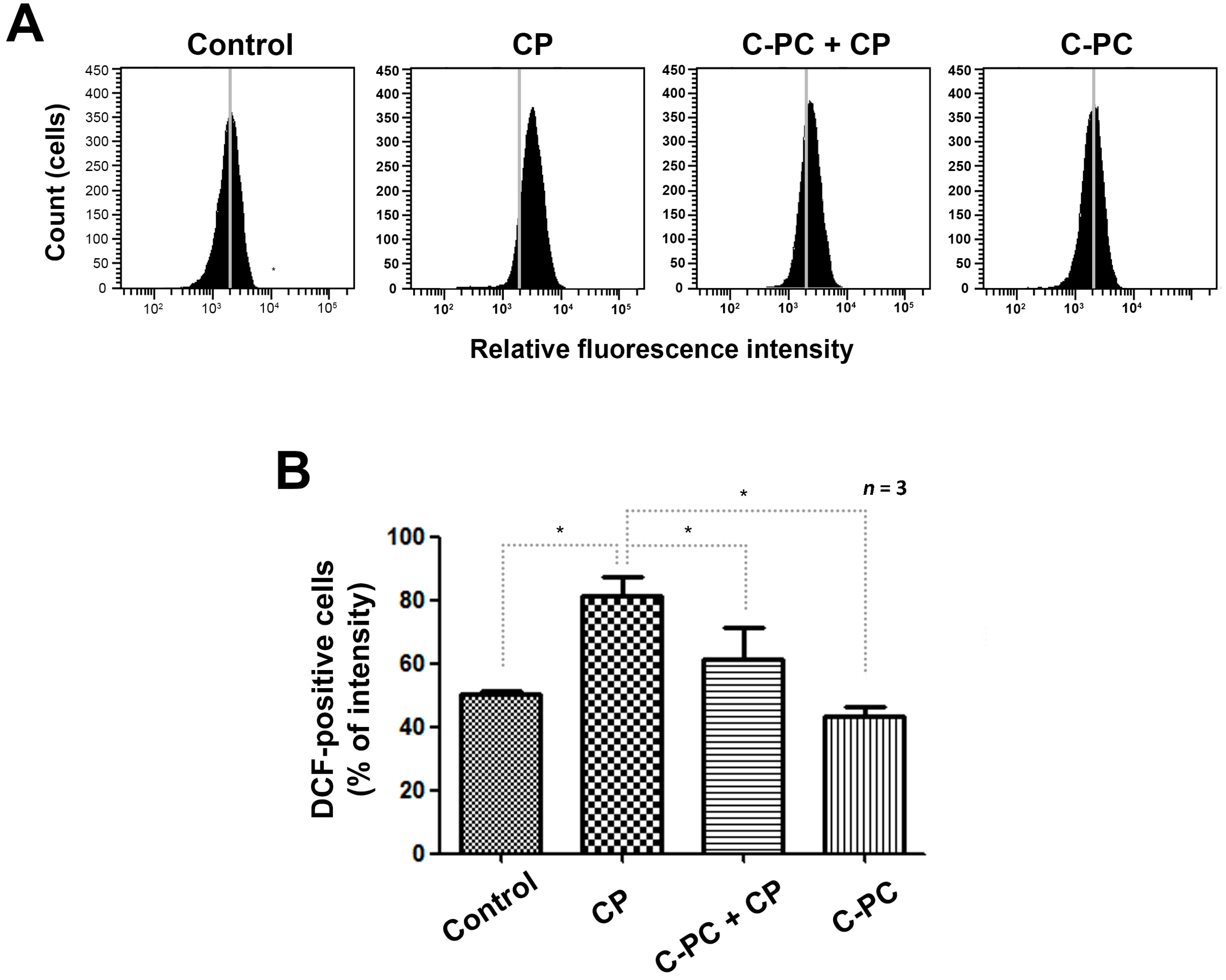
2.2. C-PC Effectively Reduces Intracellular ROS Levels in HEI-OC1 Cells
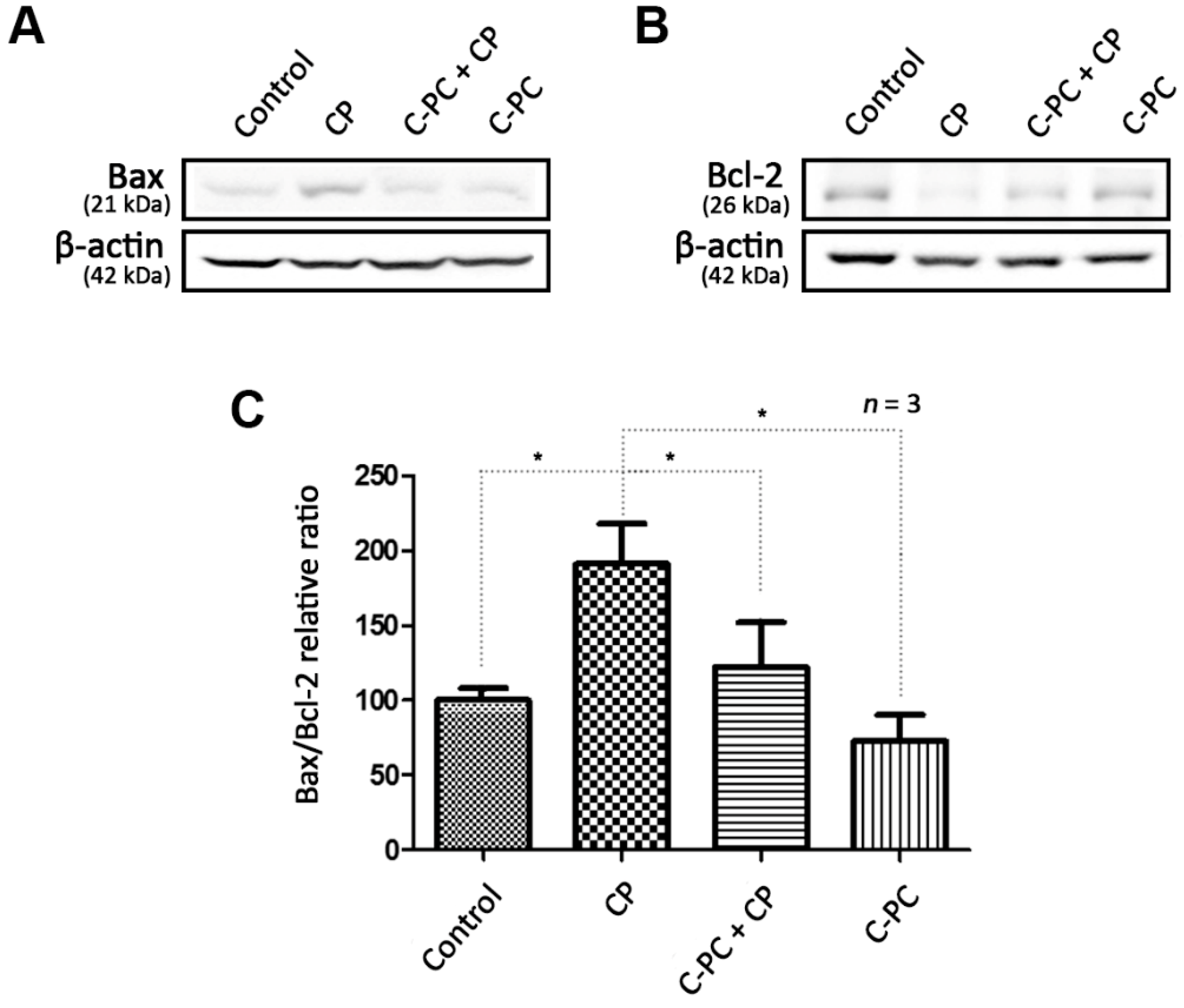
2.3. C-PC Regulates the Changes in Bax and Bcl-2 Protein Levels Caused by Cisplatin-induced ROS Accumulation
3. Materials and Methods
3.1. Culture Conditions of Limnothrix sp. KNUA002
3.2. Acetate Buffer Extraction of C-PC from Limnothrix sp. KNUA002
3.3. Cell Culture and Viability Assay
3.4. Cell Cycle Analysis
3.5. Detection of DNA Fragmentation
3.6. Measurement of Intracellular ROS Production
3.7. Protein Preparation
3.8. Western Blot Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Prasanna, R.; Sood, A.; Suresh, A.; Nayak, S.; Kaushik, B.D. Potentials and applications of algal pigments in biology and industry. Acta Botanica Hungarica 2007, 49, 131–156. [Google Scholar] [CrossRef]
- Romay, C.; Gonzalez, R.; Ledon, N.; Remirez, D.; Rimbau, V. C-phycocyanin: A biliprotein with antioxidant, anti-inflammatory and neuroprotective effects. Curr. Protein Pept. Sci. 2003, 4, 207–216. [Google Scholar] [CrossRef]
- Romay, C.; Ledon, N.; Gonzalez, R. Further studies on anti-inflammatory activity of phycocyanin in some animal models of inflammation. Inflamm. Res. 1998, 47, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Arslan, E.; Orzan, E.; Santarelli, R. Global problem of drug-induced hearing loss. Ann. N. Y. Acad. Sci. 1999, 884, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Reavis, K.M.; McMillan, G.; Austin, D.; Gallun, F.; Fausti, S.A.; Gordon, J.S.; Helt, W.J.; Konrad-Martin, D. Distortion-product otoacoustic emission test performance for ototoxicity monitoring. Ear Hear 2011, 32, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Rybak, L.P.; Mukherjea, D.; Jajoo, S.; Ramkumar, V. Cisplatin ototoxicity and protection: clinical and experimental studies. Tohoku J. Exp. Med. 2009, 219, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.Y.; Hara, K.; Kim, J.W.; Sato, E.F.; Shim, E.B.; Cho, K.H. Minimal systems analysis of mitochondria-dependent apoptosis induced by cisplatin. Korean J. Physiol. Pharmacol. 2016, 20, 367–378. [Google Scholar] [CrossRef]
- Goncalves, M.S.; Silveira, A.F.; Teixeira, A.R.; Hyppolito, M.A. Mechanisms of cisplatin ototoxicity: Theoretical review. J. Laryngol. Otol. 2013, 127, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Rybak, L.P. Mechanisms of cisplatin ototoxicity and progress in otoprotection. Curr. Opin. Otolaryngol. Head Neck Surg. 2007, 15, 364–369. [Google Scholar] [CrossRef]
- Lim, B.J.; Jeong, J.Y.; Chang, Y.K.; Na, K.R.; Lee, K.W.; Shin, Y.T.; Choi, D.E. C-phycocyanin attenuates cisplatin-induced nephrotoxicity in mice. Ren Fail 2012, 34, 892–900. [Google Scholar] [CrossRef]
- Fernandez-Rojas, B.; Medina-Campos, O.N.; Hernandez-Pando, R.; Negrette-Guzman, M.; Huerta-Yepez, S.; Pedraza-Chaverri, J. C-phycocyanin prevents cisplatin-induced nephrotoxicity through inhibition of oxidative stress. Food Funct. 2014, 5, 480–490. [Google Scholar] [CrossRef]
- Wu, Q.; Liu, L.; Miron, A.; Klimova, B.; Wan, D.; Kuca, K. The antioxidant, immunomodulatory, and anti-inflammatory activities of Spirulina: An overview. Arch. Toxicol. 2016, 90, 1817–1840. [Google Scholar] [CrossRef]
- Bermejo-Bescos, P.; Pinero-Estrada, E.; Villar del Fresno, A.M. Neuroprotection by Spirulina platensis protean extract and phycocyanin against iron-induced toxicity in SH-SY5Y neuroblastoma cells. Toxicol. In Vitro 2008, 22, 1496–1502. [Google Scholar] [CrossRef]
- Abd El-Baky, H.H.; El Baz, F.K.; El-Baroty, G.S. Enhancement of antioxidant production in Spirulina platensis under oxidative stress. Acta Physiol. Plantarum 2009, 31, 623–631. [Google Scholar] [CrossRef]
- Mohan, I.K.; Khan, M.; Shobha, J.C.; Naidu, M.U.; Prayag, A.; Kuppusamy, P.; Kutala, V.K. Protection against cisplatin-induced nephrotoxicity by Spirulina in rats. Cancer Chemother. Pharmacol. 2006, 58, 802–808. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Mishra, S.; Ghosh, P.K. Antioxidant potential of C-phycocyanin isolated from cyanobacterial species Lyngbya, Phormidium and Spirulina spp. Indian J. Biochem. Biophys. 2006, 43, 25–31. [Google Scholar]
- Gantar, M.; Simovic, D.; Djilas, S.; Gonzalez, W.W.; Miksovska, J. Isolation, characterization and antioxidative activity of C-phycocyanin from Limnothrix sp. strain 37-2-1. J. Biotechnol. 2012, 159, 21–26. [Google Scholar] [CrossRef]
- Gonzalez-Gonzalez, S. The role of mitochondrial oxidative stress in hearing loss. Neurol. Disord. Ther. 2017, 1, 1–5. [Google Scholar] [CrossRef]
- Pugazhenthi, S.; Nesterova, A.; Jambal, P.; Audesirk, G.; Kern, M.; Cabell, L.; Eves, E.; Rosner, M.R.; Boxer, L.M.; Reusch, J.E. Oxidative stress-mediated down-regulation of bcl-2 promoter in hippocampal neurons. J. Neurochem. 2003, 84, 982–996. [Google Scholar] [CrossRef]
- Han, C.; Someya, S. Mouse models of age-related mitochondrial neurosensory hearing loss. Mol. Cell Neurosci. 2013, 55, 95–100. [Google Scholar] [CrossRef]
- Farooq, S.M.; Boppana, N.B.; Devarajan, A.; Sekaran, S.D.; Shankar, E.M.; Li, C.; Gopal, K.; Bakar, S.A.; Karthik, H.S.; Ebrahim, A.S. C-phycocyanin confers protection against oxalate-mediated oxidative stress and mitochondrial dysfunctions in MDCK cells. PLoS ONE 2014, 9, e93056. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Rojas, B.; Rodriguez-Rangel, D.S.; Granados-Castro, L.F.; Negrette-Guzman, M.; Leon-Contreras, J.C.; Hernandez-Pando, R.; Molina-Jijon, E.; Reyes, J.L.; Zazueta, C.; Pedraza-Chaverri, J. C-phycocyanin prevents cisplatin-induced mitochondrial dysfunction and oxidative stress. Mol. Cell Biochem. 2015, 406, 183–197. [Google Scholar] [CrossRef]
- Kumar, D.; Dhar, D.W.; Pabbi, S.; Kumar, N.; Walia, S. Extraction and purification of C-phycocyanin from Spirulina platensis (CCC540). Indian J. Plant Physiol. 2014, 19, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.; Bogorad, L. Complementary chromatic adaptation in a filamentous blue-green alga. J. Cell Biol. 1973, 58, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Kalinec, G.M.; Webster, P.; Lim, D.J.; Kalinec, F. A cochlear cell line as an in vitro system for drug ototoxicity screening. Audiol. Neurootol. 2003, 8, 177–189. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.-R.; Do, J.-M.; Kim, K.H.; Stoica, A.R.; Jo, S.-W.; Kim, U.-K.; Yoon, H.-S. C-phycocyanin from Limnothrix Species KNUA002 Alleviates Cisplatin-Induced Ototoxicity by Blocking the Mitochondrial Apoptotic Pathway in Auditory Cells. Mar. Drugs 2019, 17, 235. https://doi.org/10.3390/md17040235
Kim Y-R, Do J-M, Kim KH, Stoica AR, Jo S-W, Kim U-K, Yoon H-S. C-phycocyanin from Limnothrix Species KNUA002 Alleviates Cisplatin-Induced Ototoxicity by Blocking the Mitochondrial Apoptotic Pathway in Auditory Cells. Marine Drugs. 2019; 17(4):235. https://doi.org/10.3390/md17040235
Chicago/Turabian StyleKim, Ye-Ri, Jeong-Mi Do, Kyung Hee Kim, Alexandra R. Stoica, Seung-Woo Jo, Un-Kyung Kim, and Ho-Sung Yoon. 2019. "C-phycocyanin from Limnothrix Species KNUA002 Alleviates Cisplatin-Induced Ototoxicity by Blocking the Mitochondrial Apoptotic Pathway in Auditory Cells" Marine Drugs 17, no. 4: 235. https://doi.org/10.3390/md17040235
APA StyleKim, Y.-R., Do, J.-M., Kim, K. H., Stoica, A. R., Jo, S.-W., Kim, U.-K., & Yoon, H.-S. (2019). C-phycocyanin from Limnothrix Species KNUA002 Alleviates Cisplatin-Induced Ototoxicity by Blocking the Mitochondrial Apoptotic Pathway in Auditory Cells. Marine Drugs, 17(4), 235. https://doi.org/10.3390/md17040235



