Ethnopharmacology, Phytochemistry, and Global Distribution of Mangroves―A Comprehensive Review
Abstract
1. Introduction
2. Review Methodology
3. Terms, Origin, and Definition
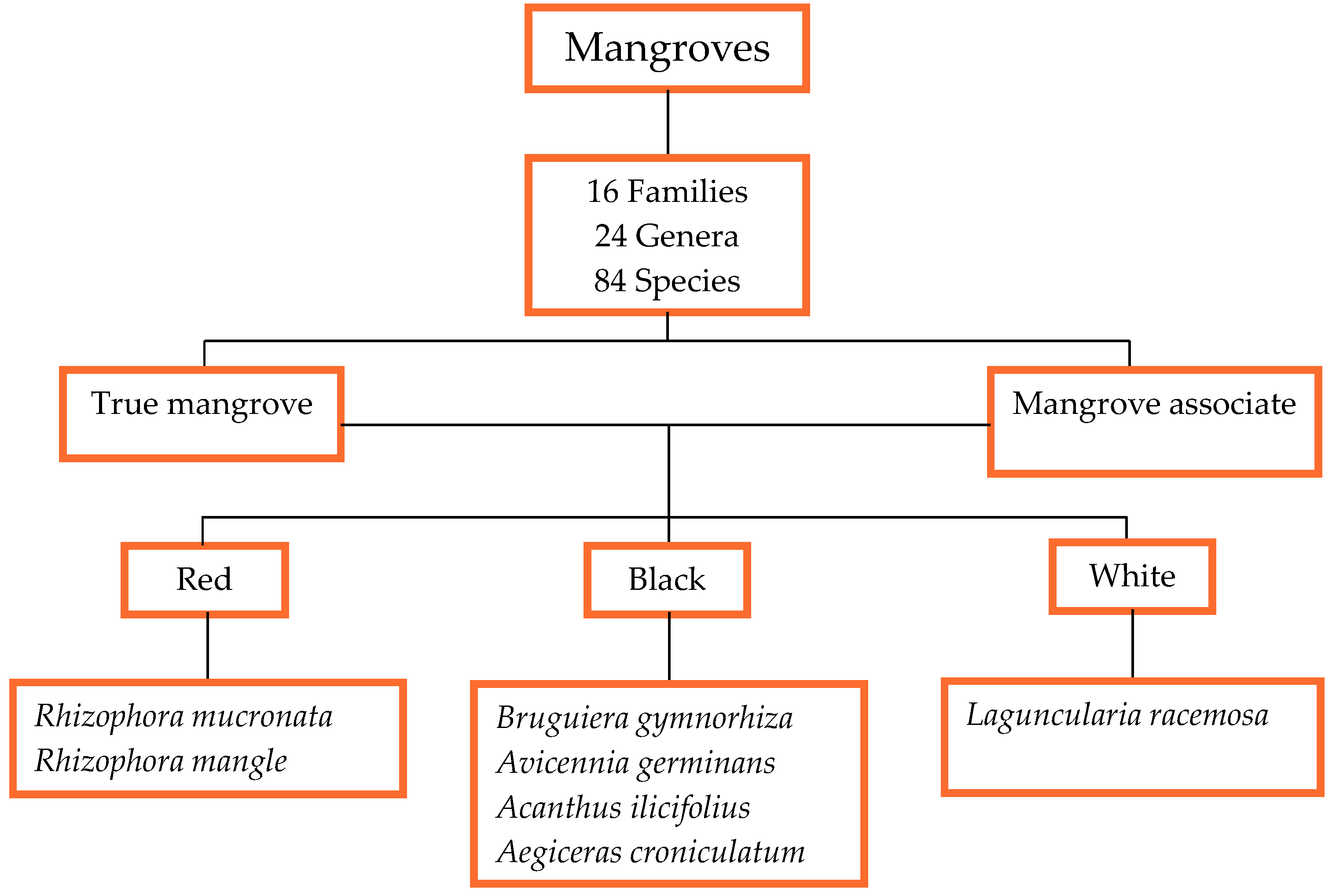
4. Botanical Classification and Types of Mangroves
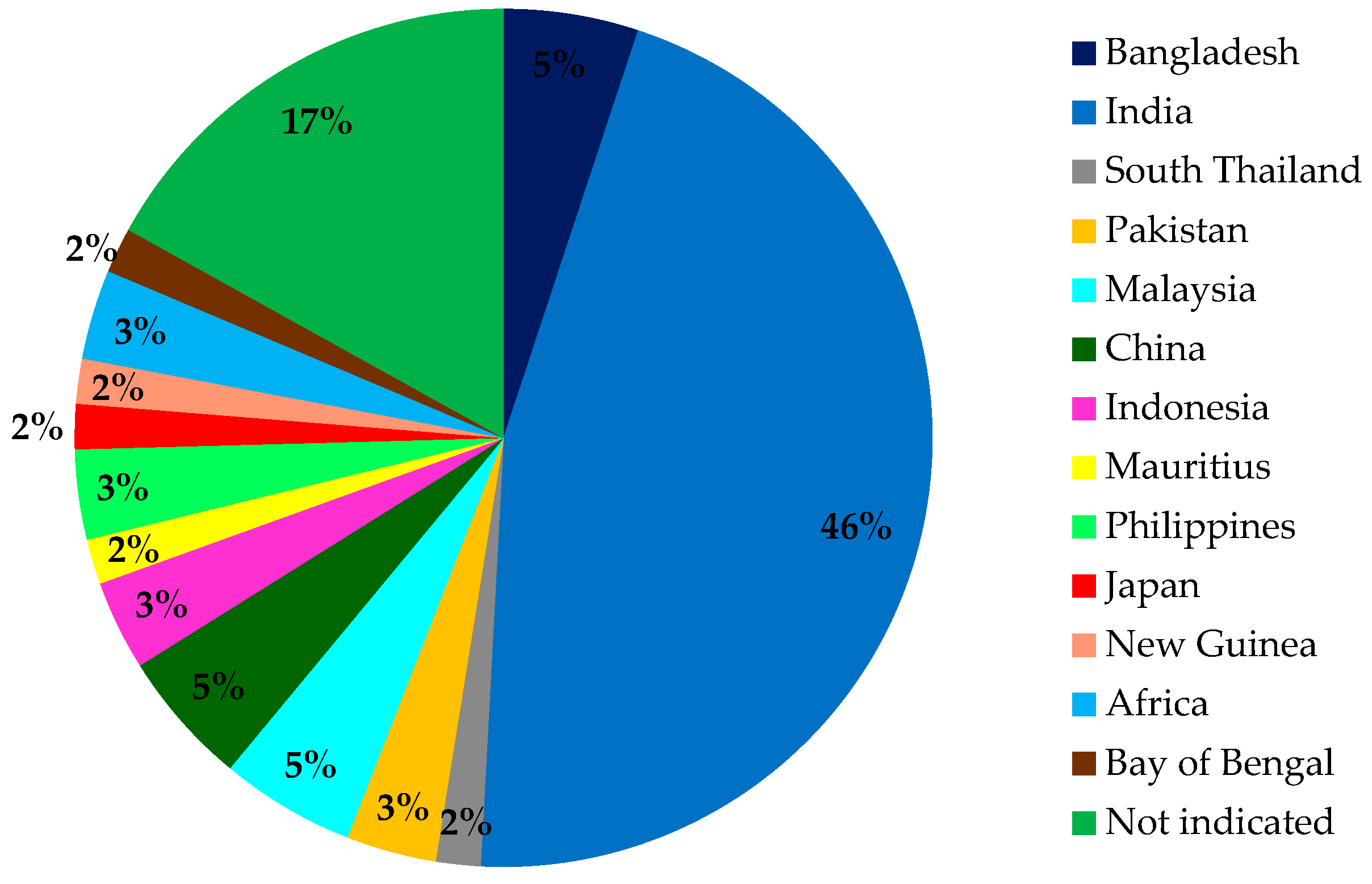
5. Biogeographical Distribution of Mangroves
6. Morphological Characteristics
7. Ethnopharmacological Uses
8. Pharmacological Activities
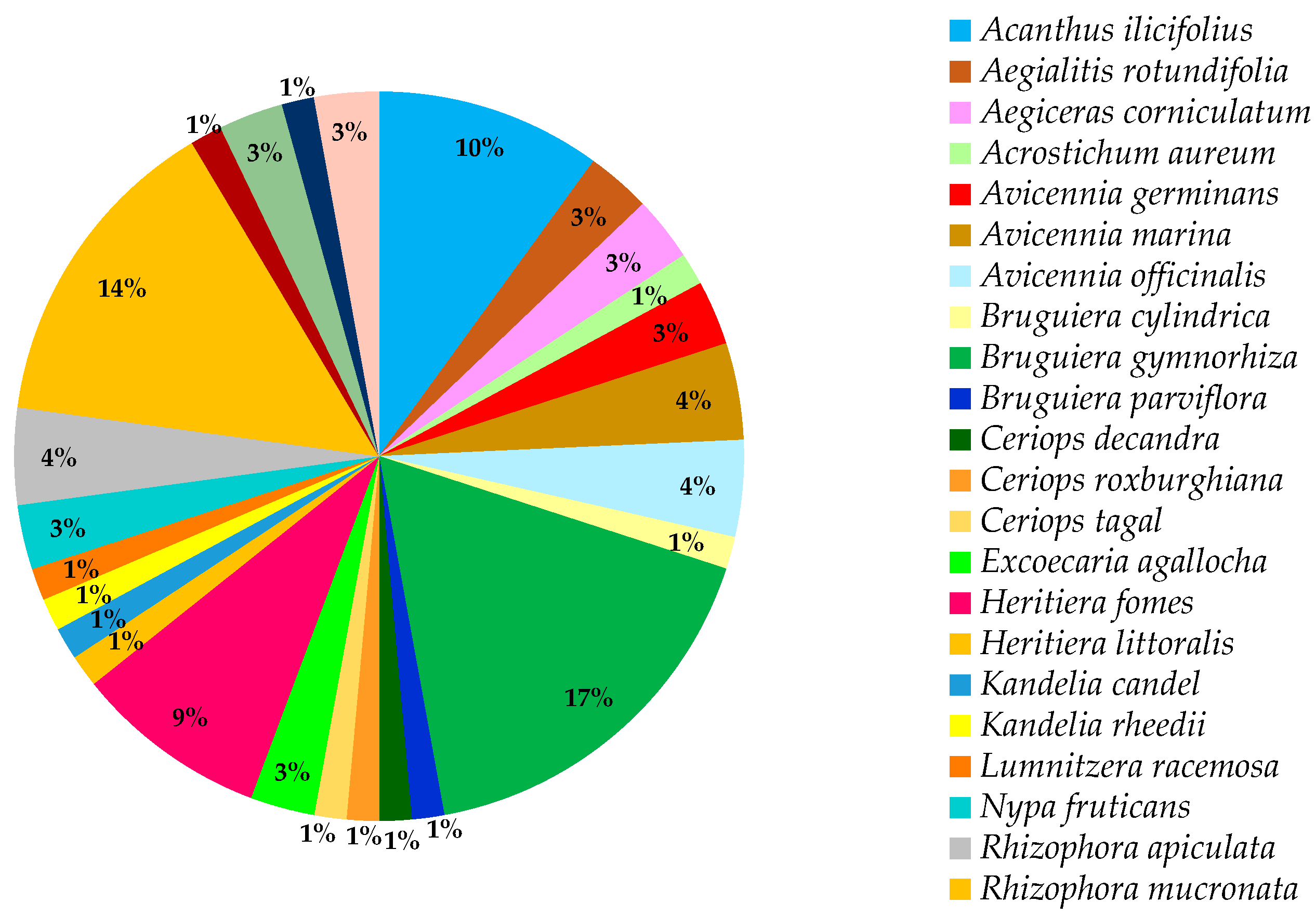
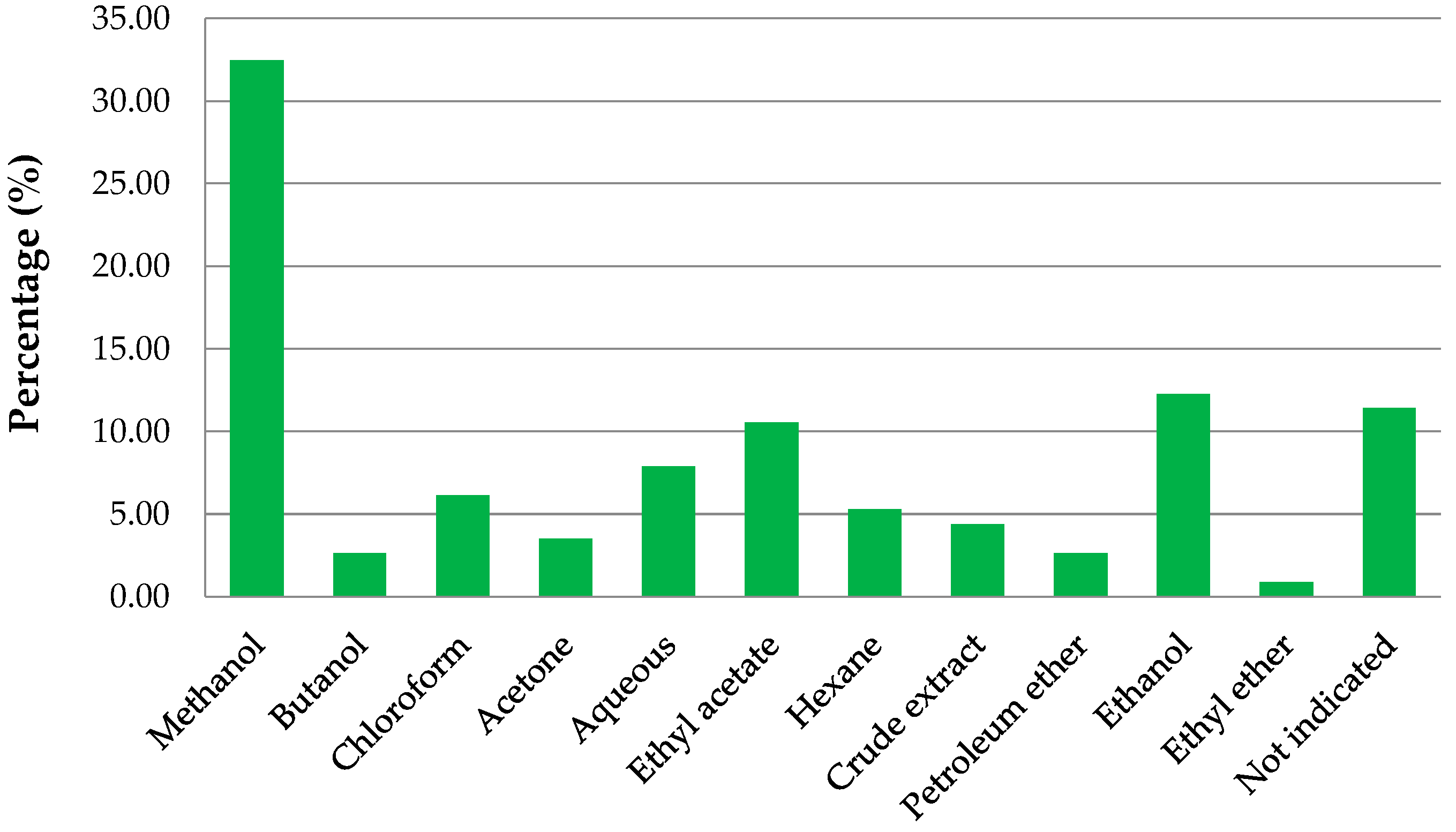
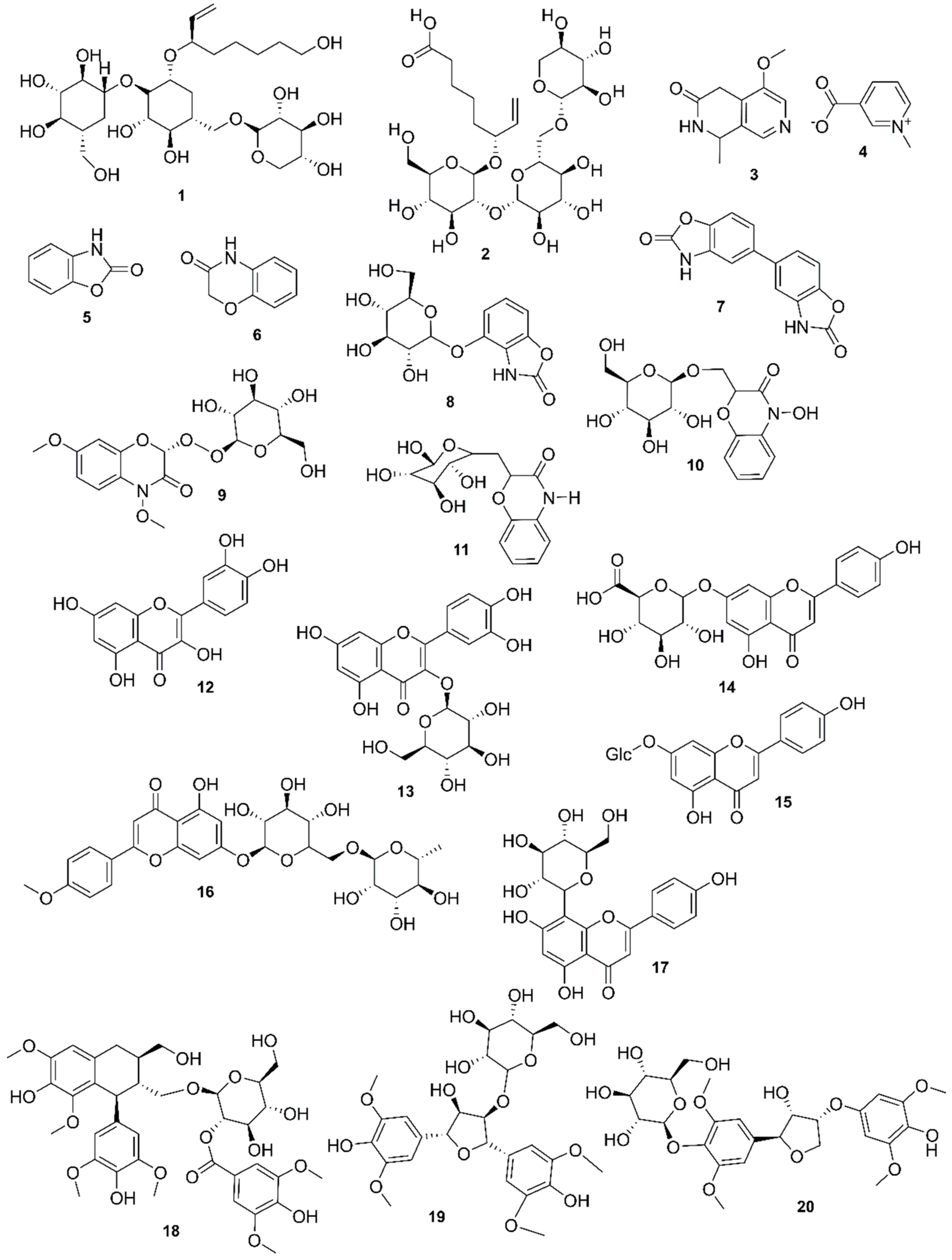
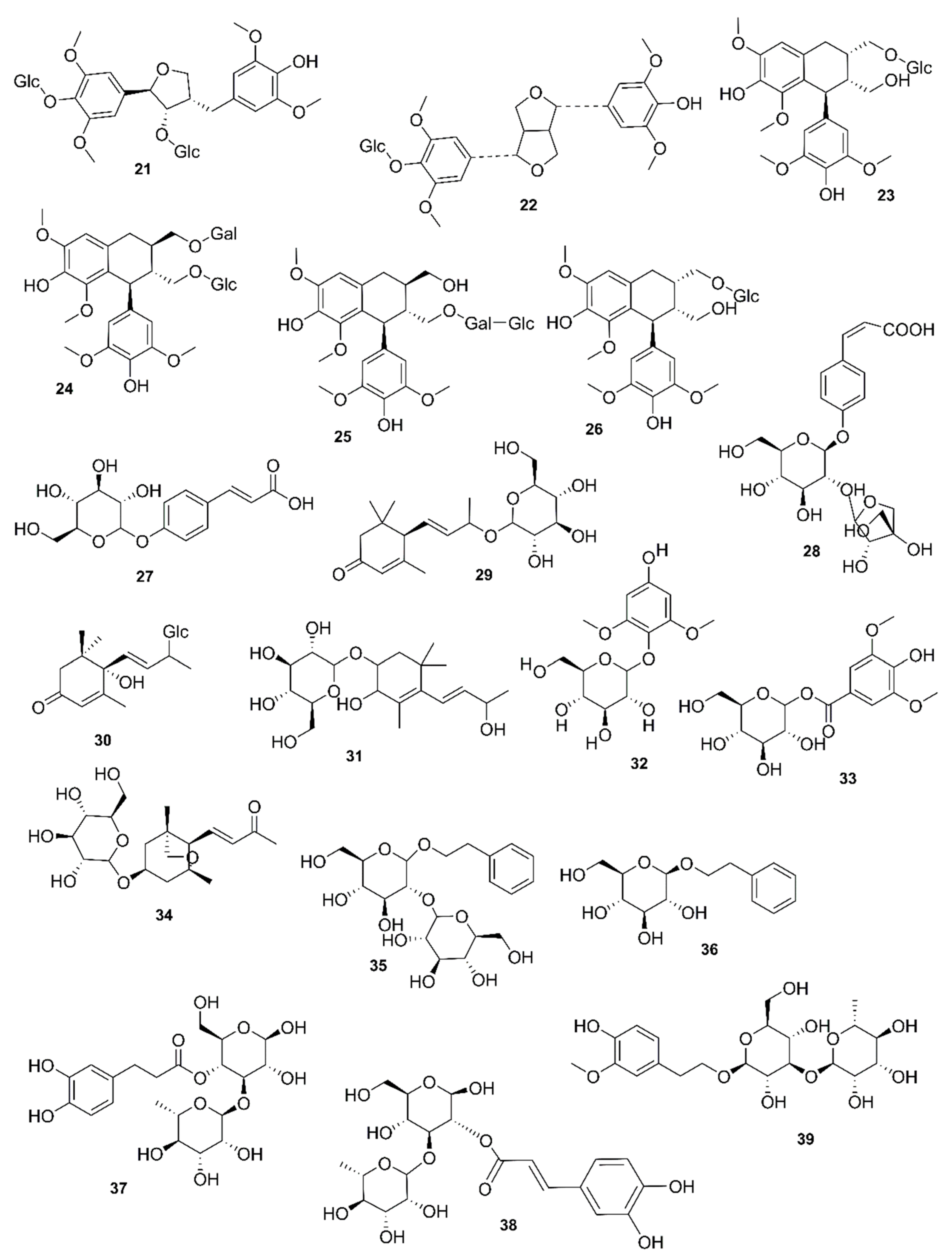
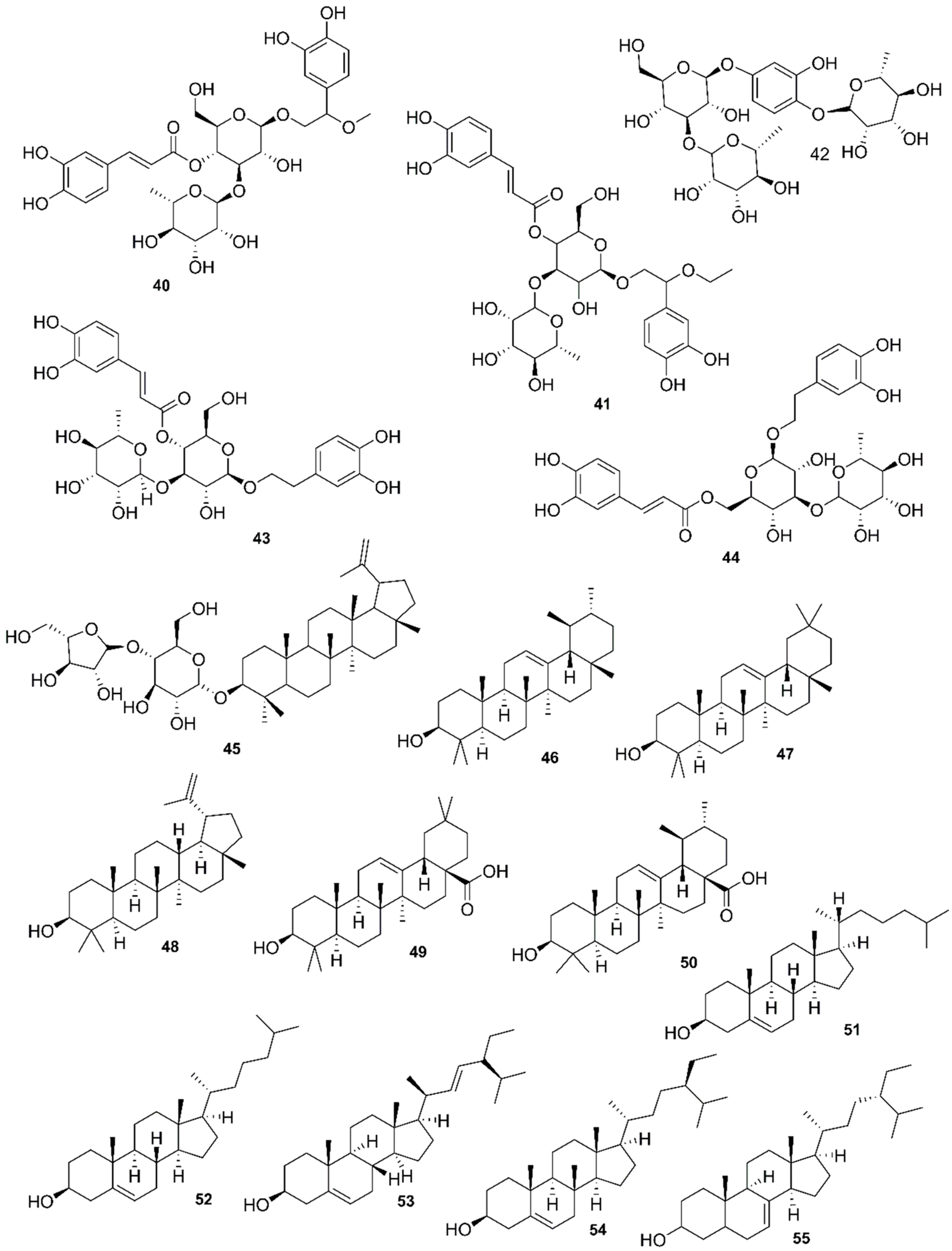
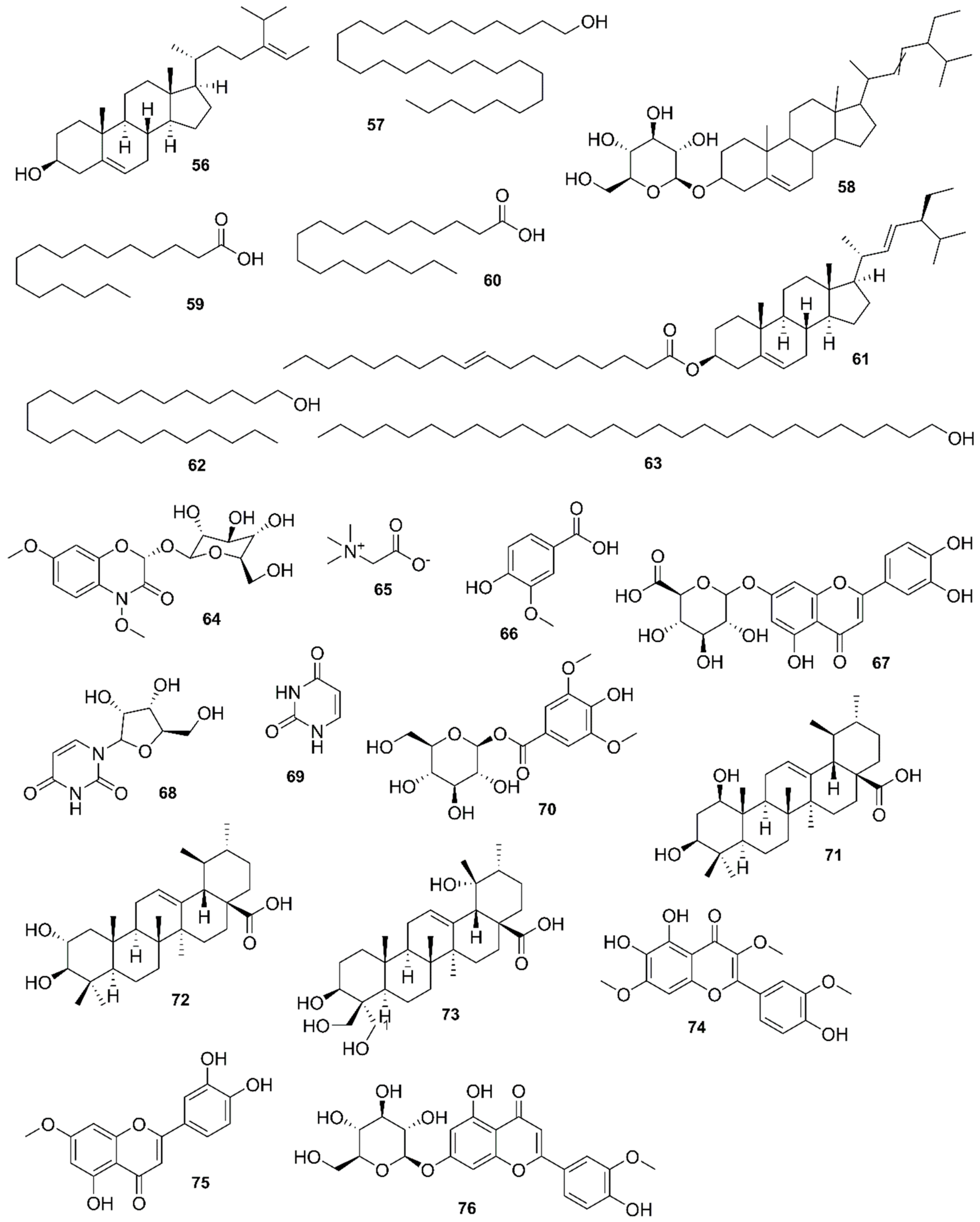
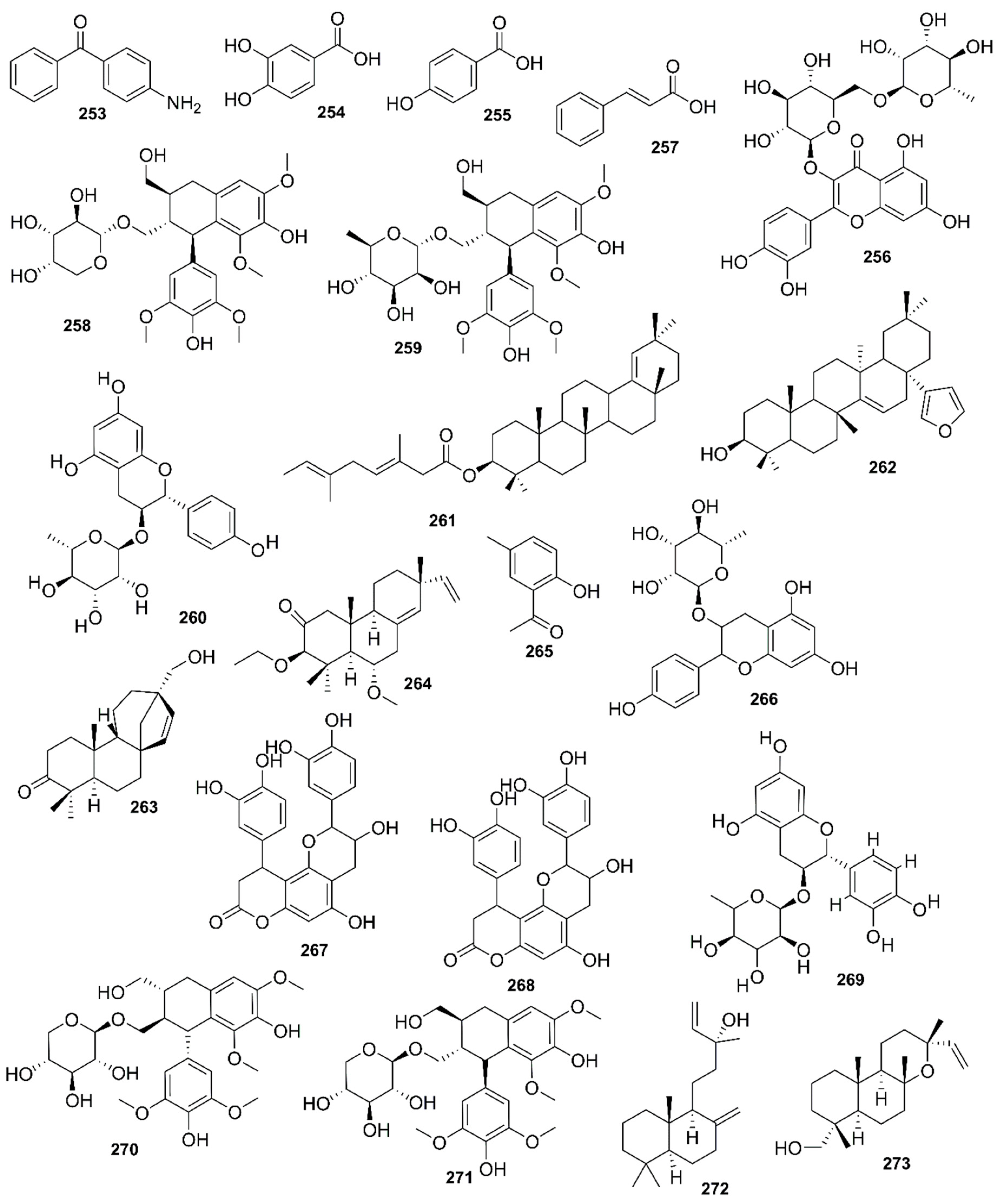
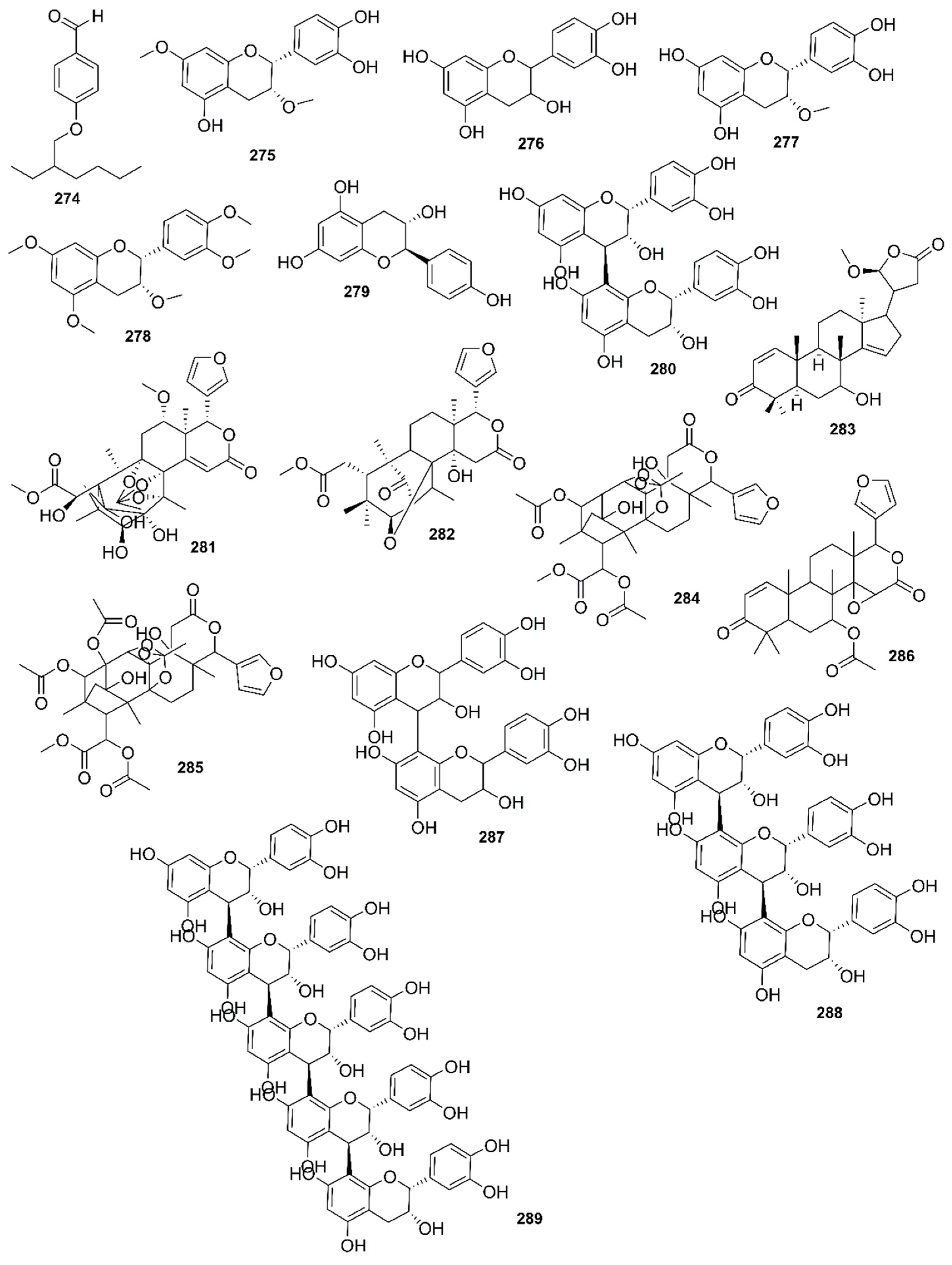
9. Phytochemistry of Mangroves
10. Results and Discussion
10.1. Acanthus ilicifolius
10.2. Aegialitis rotundifolia
10.3. Aegiceras corniculatum
10.4. Acrostichum aureum
10.5. Avicennia
10.6. Bruguiera
10.7. Ceriops
10.8. Excoecaria agallocha
10.9. Heritiera
10.10. Lumnitzera racemosa
10.11. Kandelia
10.12. Nypa fruticans
10.13. Pelliciera rhizophorae
10.14. Rhizophora
10.15. Xylocarpus granatum
11. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| APCI/HR/MS | Atmospheric pressure chemical ionisation high resolution mass spectrometry |
| ABTS | 2,2-azino-bis-3-ethyl benzthiazoline-6-sulfonic acid radical |
| COX-2 | Cyclooxigenase 2 |
| DPPH | 1-diphenyl-2-picryhydrazyl |
| EBSCO | Elton B. Stephens Co |
| FRAP | Ferric reducing antioxidant power |
| GC/MS | Gas chromatography mass spectrometry |
| HRESI/MS | High resolution electrospray ionisation mass spectrometry |
| HO | Hydroxyl |
| IC50 | Inhibitory concentration 50 |
| NR | No result |
| NI | Not indicated |
| PROSEA | Plant resources south-east Asia |
| REMA | Resazurin microtitre assay |
| SI | Selective index |
| SO | Superoxide |
| UPLC/DAD/MS | Ultra high performance liquid chromatography diode array detector tandem mass spectrometry |
References
- Ernst, M.; Saslis-Lagoudakis, C.H.; Grace, O.M.; Nilsson, N.; Simonsen, H.T.; Horn, J.W.; Rønsted, N. Evolutionary prediction of medicinal properties in the genus Euphorbia L. Sci. Rep. 2016, 6, 30531. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, J.G. Medicinal potential of willow: A chemical perspective of aspirin discovery. J. Saudi. Chem. Soc. 2010, 14, 317–322. [Google Scholar] [CrossRef]
- Daugan, M.; Wojcicki, A.D.; d’Hayer, B.; Boudy, V. Metformin: An anti-diabetic drug to fight cancer. Pharmacol. Res. 2016, 113, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Surapaneni, M.S.; Das, S.K.; Das, N.G. Designing paclitaxel drug delivery systems aimed at improved patient outcomes: Current status and challenges. Int. Sch. Res. Notices 2012, 623139, 1–15. [Google Scholar] [CrossRef]
- Willis, K.J. State of the World’s Plants 2017; Royal Botanic Gardens, Kew: London, UK, 2017. [Google Scholar]
- Kathiresan, K.; Bingham, B.L. Biology of mangroves and mangrove ecosystems. Adv. Mar. Biol. 2001, 40, 81–251. [Google Scholar]
- Thatoi, H.; Samantaray, D.; Das, S.K. The genus Avicennia, a pioneer group of dominant mangrove plant species with potential medicinal values: A review. Front. Life Sci. 2016, 9, 267–291. [Google Scholar] [CrossRef]
- Hamilton, S.E.; Casey, D. Creation of a high spatio-temporal resolution global database of continuous mangrove forest cover for the 21st century (cgmfc-21). Glob. Ecol. Biogeogr. 2016, 25, 729–738. [Google Scholar] [CrossRef]
- Yeo, S. Save Mangroves for People, Planet and the Economy, Says UN. 2014. Available online: https://www.climatechangenews.com/2014/09/30/save-mangroves-for-people-planet-and-the-economy-says-un/ (accessed on 02 August 2018).
- Van Lavieren, H.; Spalding, M.; Alongi, D.M.; Kainuma, M.; Clüsener-Godt, M.; Adeel, Z. Securing the Future of Mangroves; United Nations University, Institute for Water, Environment and Health: Hamilton, ON, Canada, 2012. [Google Scholar]
- Naidoo, G. The mangroves of South Africa: An ecophysiological review. S. Afr. J. Bot. 2016, 107, 101–113. [Google Scholar] [CrossRef]
- MacNae, W. A general account of the fauna and flora of mangrove swamps and forests in the indo-west-pacific region. In Advances in Marine Biology; Elsevier: Amsterdam, The Netherlands, 1969; Volume 6, pp. 73–270. [Google Scholar]
- Gurib-Fakim, A.; Brendler, T. Medicinal and Aromatic Plants of Indian Ocean Islands: Madagascar, Comoros, Seychelles and Mascarenes; Medpharm GmbH Scientific Publishers: Stuttgart, Germany, 2004. [Google Scholar]
- Spalding, M.; Kainuma, M.; Collins, L. World Atlas of Mangroves. A Collaborative Project of Itto, Isme, Fao, Unep-Wcmc; Earthscan: London, UK, 2010. [Google Scholar]
- Schneider, P. The discovery of tropical mangroves in graeco-roman antiquity: Science and wonder. J. Hakluyt Soc. 2011. 1–16. Available online: https://www.academia.edu/7635491/The_Discovery_of_Tropical_Mangroves_in_Graeco-Roman_Antiquity_Science_and_Wonder (accessed on 16 April 2019).
- Gardner, C.J. Use of mangroves by lemurs. Int. J. Primatol. 2016, 37, 317–332. [Google Scholar] [CrossRef]
- Yong, J. Origin of Mangroves & Mangrove Diversity 2018. Available online: http://mangroveactionproject.org/origin-of-mangroves-mangrove-diversity/#more-2692 (accessed on 02 May 2018).
- COLLINS (2018) Definition of ‘Mangrove’. Available online: https://www.collinsdictionary.com/dictionary/english/mangrove (accessed on 31 July 2018).
- Merriam-Webster. Definition of Mangrove. Available online: https://www.merriam-webster.com/dictionary/mangrove (accessed on 31 July 2018).
- Portuguese Translation of ‘Mangrove’. Available online: https://www.collinsdictionary.com/dictionary/english-portuguese/mangrove (accessed on 21 March 2018).
- Dale, N. Flowering Plants: The Santa Monica Mountains, Coastal & Chaparral Regions of Southern California; Capra Pr: Bakersfield, CA, USA, 1986. [Google Scholar]
- WELSH, S.L. 2003 Flora of North America. Available online: http://www.efloras.org/florataxon.aspx?flora_id=1&taxon_id=103110 (accessed on 26 July 2018).
- Jayatissa, L.P.; Dahdouh-Guebas, F.; Koedam, N. A review of the floral composition and distribution of mangroves in Sri Lanka. Bot. J. Linn. Soc. 2002, 138, 29–43. [Google Scholar] [CrossRef]
- Guide to the Mangroves of Singapore. Available online: http://mangrove.nus.edu.sg/guidebooks/contents.htm (accessed on 16 April 2019).
- Saenger, P.; Hegerl, E.; Davie, J.D. Global Status of Mangrove Ecosystems; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 1983. [Google Scholar]
- Tomlinson, P.B. The Botany of Mangroves, 2nd ed.; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Mu, M.R.; Jiang, Q.L.; Wang, W.Q. Comparison of leaf chloride content and leaf traits between true mangrove plants and semimangrove plants. J. Plant. Ecol. 2007, 497–504. [Google Scholar]
- Mukherjee, A.; Acharya, L.; Mattagajasingh, I.; Panda, P.; Mohapatra, T.; Das, P. Molecular characterization of three heritiera species using aflp markers. Biol. Plant. 2003, 47, 445–448. [Google Scholar] [CrossRef]
- Wang, L.; Mu, M.; Li, X.; Lin, P.; Wang, W. Differentiation between true mangroves and mangrove associates based on leaf traits and salt contents. J. Plant. Ecol. 2010, 4, 292–301. [Google Scholar] [CrossRef]
- A Working List of All Plant Species. Available online: http://www.theplantlist.org/browse/A/Rhizophoraceae/ (accessed on 30 November 2017).
- Nebula, M.; Harisankar, H.; Chandramohanakumar, N. Metabolites and bioactivities of Rhizophoraceae mangroves. Nat. Prod. Bioprospect. 2013, 3, 207–232. [Google Scholar] [CrossRef]
- Ricklefs, R.E.; Schwarzbach, A.E.; Renner, S.S. Rate of lineage origin explains the diversity anomaly in the world’s mangrove vegetation. Am. Nat. 2006, 168, 805–810. [Google Scholar] [PubMed]
- Bandaranayake, W. Traditional and medicinal uses of mangroves. Mangroves Salt Marshes 1998, 2, 133–148. [Google Scholar] [CrossRef]
- Duke, N.C. Mangrove floristics and biogeography. In Tropical Mangrove Ecosystems; Wiley: Hoboken, NJ, USA, 1992. [Google Scholar]
- Selvam, V.E.P.; Karunagaran, V.M.; Ravishankar, T.; Ramasuburamanian, R. Mangrove Plants of Tamil Nadu; M. S. Swaminathan Research Foundation: Chennai, India, 2004. [Google Scholar]
- Saranraj, P.; Sujitha, D. Mangrove medicinal plants: A review. Am.-Eurasian J. Toxicol. Sci. 2015, 7, 146–156. [Google Scholar]
- Giri, C.; Ochieng, E.; Tieszen, L.L.; Zhu, Z.; Singh, A.; Loveland, T.; Masek, J.; Duke, N. Status and distribution of mangrove forests of the world using earth observation satellite data. Glob. Ecol. Biogeogr. 2011, 20, 154–159. [Google Scholar] [CrossRef]
- Thomas, N.; Lucas, R.; Bunting, P.; Hardy, A.; Rosenqvist, A.; Simard, M. Distribution and drivers of global mangrove forest change, 1996–2010. PLoS ONE 2017, 12, e0179302. [Google Scholar] [CrossRef]
- Appadoo, C. Status of mangroves in Mauritius. J. Coast. Dev. 2003, 7, 1–4. [Google Scholar]
- Sauer, J.D. Effects of recent tropical cyclones on the coastal vegetation of Mauritius. J. Ecol. 1962, 50, 275–290. [Google Scholar] [CrossRef]
- 4th National Report to the Convention on Biological Diversity. Available online: https://www.cbd.int/doc/world/sg/sg-nr-04-en.pdf (accessed on 25 March 2018).
- Saranya, A.; Ramanathan, T.; Kesavanarayanan, K.S.; Adam, A. Traditional medicinal uses, chemical constituents and biological activities of a mangrove plant, Acanthus ilicifolius linn. A brief review. Am. Eurasian J. Agric. Environ. Sci. 2015, 15, 243–250. [Google Scholar]
- Flora and Fauna Web. Available online: https://florafaunaweb.nparks.gov.sg/ (accessed on 20 November 2017).
- López, D.; Cherigo, L.; Spadafora, C.; Loza-Mejía, M.A.; Martínez-Luis, S. Phytochemical composition, antiparasitic and α–glucosidase inhibition activities from Pelliciera rhizophorae. Chem. Cent. J. 2015, 9, 53. [Google Scholar] [CrossRef]
- Hasan, I.; Hussain, M.S.; Millat, M.S.; Sen, N.; Rahman, M.A.; Rahman, M.A.; Islam, S.; Moghal, M.M.R. Ascertainment of pharmacological activities of Allamanda neriifolia hook and Aegialitis rotundifolia roxb used in bangladesh: An in vitro study. J. Tradit. Complement. Med. 2018, 8, 107–112. [Google Scholar] [CrossRef]
- Rippey, E.; Rowland, B. Coastal Plants: Perth and the South-West Region; ISBS: Pimpri-Chinchwad, India, 2004. [Google Scholar]
- Sahoo, G.; Ansari, Z.; Shaikh, J.B.; Varik, S.U.; Gauns, M. Epibiotic communities (microalgae and meiofauna) on the pneumatophores of Avicennia officinalis (l.). Estuar. Coast. Shelf. 2018, 207, 391–401. [Google Scholar] [CrossRef]
- Putih, B. Bruguiera cylindrica. 2013. Available online: http://www.wildsingapore.com/wildfacts/plants/mangrove/bruguiera/cylindrica.htm (accessed on 16 December 2017).
- Soepadmo, E.; Saw, L.; Chung, R. Tree Flora of Sabah and Sarawak: Volume 5; Forest Research Institute Malaysia (FRIM): Selangor, Malaysia, 2004.
- Mondal, S.; Ghosh, D.; Ramakrishna, K. A complete profile on blind-your-eye mangrove Excoecaria agallocha l.(euphorbiaceae): Ethnobotany, phytochemistry, and pharmacological aspects. Pharmacogn. Rev. 2016, 10, 123. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, I.; Islam, M.K.; Saha, S.; Barman, A.K.; Rahman, M.M.; Anisuzzman, M.; Rahman, T.; Al-Nahain, A.; Jahan, R.; Rahmatullah, M. Pharmacological and ethnomedicinal overview of Heritiera fomes: Future prospects. Int. Sch. Res. Notices 2014, 2014, 938543. [Google Scholar] [CrossRef] [PubMed]
- China, F.O. Nypa fruticans. 1779. Available online: http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=220009317 (accessed on 12 December 2017).
- Mangroves. Available online: http://www.mangrove.at/pelliciera-rhizophorae_tea-mangrove.html (accessed on 15 January 2018).
- Yi, X.-X.; Deng, J.-G.; Gao, C.-H.; Hou, X.-T.; Li, F.; Wang, Z.-P.; Hao, E.-W.; Xie, Y.; Du, Z.-C.; Huang, H.-X. Four new cyclohexylideneacetonitrile derivatives from the hypocotyl of mangrove (Bruguiera gymnorrhiza). Molecules 2015, 20, 14565–14575. [Google Scholar] [CrossRef]
- Rhizophora apiculata (PROSEA). Available online: https://uses.plantnet-project.org/en/Rhizophora_apiculata_(PROSEA) (accessed on 20 December 2018).
- Duke, J. Rhizophora mangle l. 1983. Available online: https://www.hort.purdue.edu/newcrop/duke_energy/Rhizophora_mangle.html (accessed on 18 December 2017).
- Soepadmo, E.W. Tree Flora of Sabah and Sarawak, 1st ed.; Forest Research Institute Malaysia: Kuala Lumpur, Malaysia, 1995.
- Ravindran, K.; Venkatesan, K.; Balakrishnan, V.; Chellappan, K.; Balasubramanian, T. Ethnomedicinal studies of pichavaram mangroves of east coast, Tamil nadu. Indian J. Tradit. Knowl. 2005, 4, 409–411. [Google Scholar]
- Liebezeit, G.; Rau, M.T. New Guinean mangroves—Traditional usage and chemistry of natural products. Senck. Marit. 2006, 36, 1–10. [Google Scholar] [CrossRef]
- Nurdiani, R.; Firdaus, M.; Prihanto, A.A. Phytochemical screening and antibacterial activity of methanol extract of mangrove plant (Rhizophora mucronata) from porong river estuary. J. Basic Sci. Technol. 2012, 1, 27–29. [Google Scholar]
- Rollet, B. Bibliography on Mangrove Research 1600–1975; UNESCO: Paris, France, 1981. [Google Scholar]
- Seepana, R.; Perumal, K.; Kada, N.M.; Chatragadda, R.; Raju, M.; Annamalai, V. Evaluation of antimicrobial properties from the mangrove Rhizophora apiculata and bruguiera gymnorrhiza of burmanallah coast, south andaman, india. J. Coast. Life Med. 2016, 4, 475–478. [Google Scholar] [CrossRef]
- Krishnamoorthy, M.; Sasikumar, J.; Shamna, R.; Pandiarajan, C.; Sofia, P.; Nagarajan, B. Antioxidant activities of bark extract from mangroves, Bruguiera cylindrica (l.) blume and Ceriops decandra perr. Indian J. Pharmacol. 2011, 43, 557. [Google Scholar] [PubMed]
- Sur, T.K.; Hazra, A.; Hazra, A.K.; Bhattacharyya, D. Antioxidant and hepatoprotective properties of Indian sunderban mangrove Bruguiera gymnorrhiza l. Leave. J. Basic Clin. Pharm. 2016, 7, 75. [Google Scholar] [CrossRef] [PubMed]
- Bandaranayake, W.M. Bioactivities, bioactive compounds and chemical constituents of mangrove plants. Wetl. Ecol. Manag. 2002, 10, 421–452. [Google Scholar] [CrossRef]
- Kumar, K.M.S.; Gorain, B.; Roy, D.K.; Samanta, S.K.; Pal, M.; Biswas, P.; Roy, A.; Adhikari, D.; Karmakar, S.; Sen, T. Anti-inflammatory activity of Acanthus ilicifolius. J. Ethnopharmacol. 2008, 120, 7–12. [Google Scholar] [CrossRef]
- Babu, B.; Shylesh, B.; Padikkala, J. Antioxidant and hepatoprotective effect of Acanthus ilicifolius. Fitoterapia 2001, 72, 272–277. [Google Scholar] [CrossRef]
- Islam, M.A.; Saifuzzaman, M.; Ahmed, F.; Rahman, M.M.; Sultana, N.A.; Naher, K. Antinociceptive activity of methanolic extract of Acanthus ilicifolius linn. Leaves. Turk. J. Pharm. Sci. 2012, 9, 51–60. [Google Scholar]
- Rahmatullah, M.; Sadeak, I.; Bachar, S.C.; Hossain, T.; Jahan, N.; Chowdhury, M.H.; Jahan, R.; Nasrin, D.; Rahman, M.; Rahman, S. Brine shrimp toxicity study of different bangladeshi medicinal plants. Adv. Nat. Appl. Sci. 2010, 4, 163–174. [Google Scholar]
- Bose, S.; Bose, A. Antimicrobial activity of Acanthus ilicifolius (l.). Indian J. Pharm. Sci. 2008, 70, 821. [Google Scholar] [CrossRef] [PubMed]
- Miles, D.H.; Kokpol, U.; Chittawong, V.; Tip-Pyang, S.; Tunsuwan, K.; Nguyen, C. Mangrove forests-the importance of conservation as a bioresource for ecosystem diversity and utilization as a source of chemical constituents with potential medicinal and agricultural value. IUPAC 1998, 70, 1–9. [Google Scholar]
- Ray, T. Customary use of mangrove tree as a folk medicine among the sundarban resource collectors. Int. J. Res. Hum. Arts Lit. 2014, 2, 43–48. [Google Scholar]
- Raju, G.S.; RahmanMoghal, M.M.; Hossain, M.S.; Hassan, M.M.; Billah, M.M.; Ahamed, S.K.; Rana, S.M. Assessment of pharmacological activities of two medicinal plant of bangladesh: Launaea sarmentosa and Aegialitis rotundifolia roxb in the management of pain, pyrexia and inflammation. Biol. Res. 2014, 47, 55. [Google Scholar] [CrossRef] [PubMed]
- Roome, T.; Dar, A.; Naqvi, S.; Choudhary, M.I. Evaluation of antinociceptive effect of Aegiceras corniculatum stems extracts and its possible mechanism of action in rodents. J. Ethnopharmacol. 2011, 135, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Roome, T.; Dar, A.; Naqvi, S.; Ali, S.; Choudhary, M.I. Aegiceras corniculatum extract suppresses initial and late phases of inflammation in rat paw and attenuates the production of eicosanoids in rat neutrophils and human platelets. J. Ethnopharmacol. 2008, 120, 248–254. [Google Scholar] [CrossRef]
- Thomas, T. In vitro evaluation of antibacterial activity of Acrostichum aureum linn. Indian J. Nat. Prod. Resour. 2012, 31, 135–138. [Google Scholar]
- Rajeshwari, E.; Gajendiran; Elamathy, S. Study of preliminary phytochemical analysis and antibacterial activity of selected medicinal plants (Avicennia germinans). Int. J. 2013, 1, 952–954. [Google Scholar]
- Khajehzadeh, S.; Behbahani, M. Activity of Avicennia marina methanol extracts on proliferation of lymphocytes and their mutagenicity using ames test and in silico method. J. Mazandaran Univ. Med. Sci. 2016, 26, 32–42. [Google Scholar]
- Sumithra, M.; Anbu, J.; Nithya, S.; Ravichandiran, V. Anticancer activity of methanolic leaves extract of Avicennia officinalis on ehrlich ascitis carcinoma cell lines in rodents. Int. J. Pharm. Tech. Res. 2011, 3, 1290–1292. [Google Scholar]
- Thirunavukkarasu, P.; Ramanathan, T.; Ramkumar, L.; Shanmugapriya, R.; Renugadevi, G. The antioxidant and free radical scavenging effect of Avicennia officinalis. J. Med. Plants Res. 2011, 5, 4754–4758. [Google Scholar]
- Ahmed, F.; Shahid, I.; Gain, N.; Reza, M.; Sadhu, S. Antinociceptive and antidiarrhoeal activities of Bruguiera gymnorrhiza. Orient. Pharm. Exp. Med. 2007, 7, 280–285. [Google Scholar] [CrossRef]
- Karimulla, S.; Kumar, B. Antidiabetic and antihyperlipidemic activity of bark of Bruguiera gymnorrhiza on streptozotocin induced diabetic rats. AJPS 2011, 1, 4–7. [Google Scholar]
- Haq, M.; Sani, W.; Hossain, A.; Taha, R.M.; Monneruzzaman, K. Total phenolic contents, antioxidant and antimicrobial activities of Bruguiera gymnorrhiza. J. Med. Plants Res. 2011, 5, 4112–4118. [Google Scholar]
- Bamroongrugsa, N. Bioactive substances from the mangrove resource. Songklanakarin J. Sci. Technol. 1999, 21, 377–386. [Google Scholar]
- Siemonsma, J.; Piluek, K. Plant resources of south-east Asia. No. 8: Vegetables. 1993. Available online: http://agris.fao.org/agris-search/search.do?recordID=XF2015019284 (accessed on 25 January 2018).
- Ravi, A.V.; Kathiresan, K. Seasonal-variation in gallotannin from mangroves. IJMS 1990, 19, 224–225. [Google Scholar]
- Premanathan, M.; Nokashima, H.; Kathiresan, K.; Rajendran, N.; Yamamoto, N. In vitro anti human immunodeficiency virus activity of mangrove plants. Indian J. Med. Res. 1996, 103, 278–281. [Google Scholar]
- Revathi, P.; Senthinath, T.J.; Thirumalaikolundusubramanian, P.; Prabhu, N. An overview of antidiabetic profile of mangrove plants. Int. J. Pharm. Pharm. Sci. 2014, 6, 1–5. [Google Scholar]
- Hossain, M.A.; Panthi, S.; Asadujjaman, M.; Khan, S.A.; Ferdous, F.; Sadhu, S.K. Phytochemical and pharmacological assessment of the ethanol leaves extract of Heritiera fomes buch. Ham.(family-sterculiaceae). J. Porphyr. Phthalocyanines. 2013, 2, 95–101. [Google Scholar]
- Patra, J.K.; Thatoi, H. Anticancer activity and chromatography characterization of methanol extract of Heritiera fomes buch. Ham.; a mangrove plant from bhitarkanika, india. OPEM 2013, 13, 133–142. [Google Scholar] [CrossRef]
- Ali, M.; Nahar, K.; Sintaha, M.; Khaleque, H.N.; Jahan, F.I.; Biswas, K.R.; Swarna, A.; Monalisa, M.N.; Jahan, R.; Rahmatullah, M. An evaluation of antihyperglycemic and antinociceptive effects of methanol extract of Heritiera fomes buch-ham.(sterculiaceae) barks in swiss albino mice. Adv. Nat. Appl. Sci. 2011, 5, 116–121. [Google Scholar]
- Ge, L.; Li, Y.; Yang, K.; Pan, Z. Chemical constituents of the leaves of Heritiera littoralis. Chem. Nat. Compd. 2016, 52, 702–703. [Google Scholar] [CrossRef]
- Wei, S.-D.; Zhou, H.-C.; Lin, Y.-M. Antioxidant activities of extract and fractions from the hypocotyls of the mangrove plant Kandelia candel. Int. J. Mol. Sci. 2010, 11, 4080–4093. [Google Scholar] [CrossRef] [PubMed]
- Zaman, A. Docking studies and network analyses reveal capacity of compounds from Kandelia rheedii to strengthen cellular immunity by interacting with host proteins during tuberculosis infection. Bioinformation 2012, 8, 1012. [Google Scholar] [CrossRef] [PubMed]
- Pattanaik, C.; Reddy, C.; Dhal, N.; Das, R. Utilisation of mangrove forests in bhitarkanika wildlife sanctuary, orissa. Indian J. Tradit. Knowl. 2008, 7, 598–603. [Google Scholar]
- Yusoff, N.A.; Yam, M.F.; Beh, H.K.; Razak, K.N.A.; Widyawati, T.; Mahmud, R.; Ahmad, M.; Asmawi, M.Z. Antidiabetic and antioxidant activities of Nypa fruticans wurmb. Vinegar sample from Malaysia. Asian Pac. J. Trop. Biomed. 2015, 8, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, V.; Guruvayoorappan, C. Protective effect of marine mangrove Rhizophora apiculata on acetic acid induced experimental colitis by regulating anti-oxidant enzymes, inflammatory mediators and nuclear factor-kappa b subunits. Int. J. Immunopharmacol. 2014, 18, 124–134. [Google Scholar]
- Premanathan, M.; Arakaki, R.; Izumi, H.; Kathiresan, K.; Nakano, M.; Yamamoto, N.; Nakashima, H. Antiviral properties of a mangrove plant, Rhizophora apiculata blume, against human immunodeficiency virus. Antiviral Res. 1999, 44, 113–122. [Google Scholar] [CrossRef]
- Kusuma, S.; Kumar, P.A.; Boopalan, K. Potent antimicrobial activity of Rhizophora mucronata. J. Ecobiotechnol. 2011, 3, 40–41. [Google Scholar]
- Suganthy, N.; Pandima Devi, K. In vitro antioxidant and anti-cholinesterase activities of Rhizophora mucronata. Pharm. Biol. 2016, 54, 118–129. [Google Scholar] [CrossRef]
- Arora, K.; Nagpal, M.; Jain, U.; Jat, R.; Jain, S. Mangroves: A novel gregarious phyto medicine for diabetes. Int. J. Res. Dev. Pharm. Lif. Sci. 2014, 3, 1244–1257. [Google Scholar]
- Angalabiri-Owei, B.; Isirima, J. Evaluation of the lethal dose of the methanol extract of Rhizophora racemosa leaf using karbers method. Afr. J. Cell Pathol. 2014, 2, 65–68. [Google Scholar] [CrossRef]
- Dossou-Yovo, H.; Fifanou, G.V.; Sinsin, B. Ethnobotanical survey of mangrove plant species used as medicine from ouidah to grand-popo districts, southern Benin. Am. J. Ethnomed. 2017, 4, 1–6. [Google Scholar]
- Das, S.; Samantaray, D.; Thatoi, H. Ethnomedicinal, antimicrobial and antidiarrhoeal studies on the mangrove plants of the genus Xylocarpus: A mini review. J. Bioanal. Biomed. 2014, 12, 004. [Google Scholar]
- Banerjee, D.; Chakrabarti, S.; Hazra, A.K.; Banerjee, S.; Ray, J.; Mukherjee, B. Antioxidant activity and total phenolics of some mangroves in sundarbans. Afr. J. Biotechnol. 2008, 7, 805–810. [Google Scholar]
- Bakshi, M.; Chaudhuri, P. Antimicrobial potential of leaf extracts of ten mangrove species from Indian sundarban. Int. J. Pharm. Biol. Sci. 2014, 5, 294–304. [Google Scholar]
- Reddy, A.R.K.; Grace, J.R. Anticancer activity of methanolic extracts of selected mangrove plants. Int. J. Pharm. Sci. Res. 2016, 38, 3852–3856. [Google Scholar]
- Janmanchi, H.; Raju, A.; Degani, M.; Ray, M.; Rajan, M. Antituberculosis, antibacterial and antioxidant activities of Aegiceras corniculatum, a mangrove plant and effect of various extraction processes on its phytoconstituents and bioactivity. S. Afr. J. Bot. 2017, 113, 421–427. [Google Scholar] [CrossRef]
- Rout, P.; Basak, U.C. Antioxidant properties in leaf and root extracts of some medicinally important mangrove species of Odisha coast. Am. J. Pharm. Tech. Res. 2012, 4, 1–13. [Google Scholar]
- Shafie, M.M.; Forghani, A.H.; Moshtaghiyan, J. Anti-inflammatory effects of hydro-alcoholic extracts of mangrove (Avicennia marina) and vitamin c on arthritis rats. Bull. Environ. Pharm Life Sci. 2013, 2, 32–37. [Google Scholar]
- Karami, L.; Majd, A.; Mehrabian, S.; Nabiuni, M.; Salehi, M.; Irian, S. Antimutagenic and anticancer effects of Avicennia marina leaf extract on Salmonella typhimurium ta100 bacterium and human promyelocytic leukaemia hl-60 cells. Sci. Asia 2012, 38, 349–355. [Google Scholar] [CrossRef]
- Sharief, M.N.; Umamaheswararao, V. Antibacterial activity of stem and root extracts of Avicennia officinalis l. Int. J. Appl. Pharm. 2011, 2, 231–236. [Google Scholar]
- Al Maqtari Maher, A. Screening of salt-stress, antioxidant enzyme, and antimicrobial activity of leave extracts of mangroves Avicennia marina l. From hodaidah, Yemen. J. Stress Physiol. Biochem. 2014, 10, 190–199. [Google Scholar]
- Ramanathan, T. Phytochemical characterization and antimicrobial efficiency of mangrove plants Avicennia marina and Avicennia officinalis. Int. J. Pharm. Biol. Arch. 2012, 3, 348–351. [Google Scholar]
- Sura, S.; Anbu, J.; Sultan, M.; Uma, B. Antiulcer effect of ethanolic leaf extract of Avicennia officinalis. Pharmacologyonline 2011, 3, 12–19. [Google Scholar]
- Hossain, M.H.; Howlader, M.S.I.; Dey, S.K.; Hira, A.; Ahmed, A. Evaluation of diuretic and neuropharmacological properties of the methanolic extract of Avicennia officinalis l. Leaves from bangladesh. Int. J. Pharm. Phytopharmacol. Res. 2012, 2, 2–6. [Google Scholar]
- A Rege, A.; Y Ambaye, R.; A Deshmukh, R. In-vitro testing of anti-hiv activity of some medicinal plants. Int. J. Prod. Res. 2010, 1, 193–199. [Google Scholar]
- Hossain, M.L. Medicinal activity of Avicennia officinalis: Evaluation of phytochemical and pharmacological properties. Saudi J. Med. Pharm. Sci. 2016, 2, 250–255. [Google Scholar]
- Lakshmi, V.; Sonkar, R.; Khanna, A.K. Antihyperlipidemic and antioxidant activities of Bruguiera cylindrinca (l). Chron Young Sci. 2012, 3, 236. [Google Scholar] [CrossRef]
- Gawali, P.; Jadhav, B. Antioxidant activity and antioxidant phytochemical analysis of mangrove species Sonneratia alba and Bruguiera cylindrica. Asian J. Microbiol. Biotechnol. Environ. Sci. 2011, 13, 257–261. [Google Scholar]
- Barik, R.; Sarkar, R.; Biswas, P.; Bera, R.; Sharma, S.; Nath, S.; Karmakar, S.; Sen, T. 5,7-dihydroxy-2-(3-hydroxy-4,5-dimethoxy-phenyl)-chromen-4-one-a flavone from Bruguiera gymnorrhiza displaying anti-inflammatory properties. Indian J. Pharmacol. 2016, 48, 304. [Google Scholar] [PubMed]
- Uddin, S.J.; Grice, I.D.; Tiralongo, E. Cytotoxic effects of Bangladeshi medicinal plant extracts. Evid. Based Complement. Altern. Med. 2011, 2011, 1–7. [Google Scholar] [CrossRef]
- Bunyapraphatsara, N.; Jutiviboonsuk, A.; Sornlek, P.; Therathanathorn, W.; Aksornkaew, S.; Fong, H.H.; Pezzuto, J.M.; Kosmeder, J. Pharmacological studies of plants in the mangrove forest. Thai J. Phytopharm. 2003, 10, 1–12, 2546. [Google Scholar]
- Abeysinghe, P.D. Antibacterial activity of some medicinal mangroves against antibiotic resistant pathogenic bacteria. Indian J. Pharm. Sci. 2010, 72, 167–172. [Google Scholar] [CrossRef]
- Hossain, H.; Moniruzzaman, S.; Nimmi, I.; Kawsar, H.; Hossain, A.; Islam, A.; Jahan, I.A. Anti-inflammatory and antioxidant activities of the ethanolic extract of Ceriops decandra (griff.) ding hou bark. Orient. Pharm. Exp. Med. 2011, 11, 215–220. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, H.; Tam, N.F.Y. Polyphenols, tannins and antioxidant activities of eight true mangrove plant species in south China. Plant Soil 2014, 374, 549–563. [Google Scholar] [CrossRef]
- Paul, T.; Ramasubbu, S. The antioxidant, anticancer and anticoagulant activities of Acanthus ilicifolius l. Roots and Lumnitzera racemosa willd. Leaves, from southeast coast of India. J. Appl. Pharm. Sci. 2017, 7, 081–087. [Google Scholar]
- Vadlapudi, V.; Naidu, K.C. In vitro bioefficiency of marine mangrove plant activity of Rhizophora conjugata. Int. J. Pharma. Tech. Res. 2009, 1, 1598–1600. [Google Scholar]
- SÁnchez Perera, L.M.; Batista, N.Y.; Rodríguez, A.; Farrada, F.; Bulnes, C. Gastric and duodenal antiulcer effects of Rhizophora mangle. Pharm. Biol. 2004, 42, 225–229. [Google Scholar] [CrossRef]
- Sánchez, J.C.; García, R.F.; Cors, M.T.M. 1, 1-diphenyl-2-picrylhydrazyl radical and superoxide anion scavenging activity of Rhizophora mangle (l.) bark. Pharmacog. Res. 2010, 2, 279. [Google Scholar] [CrossRef]
- Zhang, L.-L.; Lin, Y.-M.; Zhou, H.-C.; Wei, S.-D.; Chen, J.-H. Condensed tannins from mangrove species Kandelia candel and Rhizophora mangle and their antioxidant activity. Molecules 2010, 15, 420–431. [Google Scholar] [CrossRef]
- Gao, M.; Xiao, H. Activity-guided isolation of antioxidant compounds from Rhizophora apiculata. Molecules 2012, 17, 10675–10682. [Google Scholar] [CrossRef]
- Lim, S.; Darah, I.; Jain, K. Antimicrobial activities of tannins extracted from Rhizophora apiculata barks. J. Trop. For. Sci. 2006, 18, 59–65. [Google Scholar]
- Loo, A.; Jain, K.; Darah, I. Antioxidant activity of compounds isolated from the pyroligneous acid, Rhizophora apiculata. Food Chem. 2008, 107, 1151–1160. [Google Scholar] [CrossRef]
- Rahim, A.A.; Rocca, E.; Steinmetz, J.; Kassim, M.J.; Ibrahim, M.S.; Osman, H. Antioxidant activities of mangrove Rhizophora apiculata bark extracts. Food Chem. 2008, 107, 200–207. [Google Scholar] [CrossRef]
- Sulaiman, S.; Ibrahim, D.; Kassim, J.; Sheh-Hong, L. Antimicrobial and antioxidant activities of condensed tannin from Rhizophora apiculata barks. J. Chem. Pharm. Res. 2011, 3, 436–444. [Google Scholar]
- Chakraborty, K.; Raola, V.K. Two rare antioxidant and anti-inflammatory oleanenes from loop root Asiatic mangrove Rhizophora mucronata. Phytochemistry 2017, 135, 160–168. [Google Scholar] [CrossRef]
- Joel, E.L.; Bhimba, V. Isolation and characterization of secondary metabolites from the mangrove plant Rhizophora mucronata. Asian Pac. J. Trop. Biomed. 2010, 3, 602–604. [Google Scholar] [CrossRef]
- Gurudeeban, S.; Ramanathan, T.; Satyavani, K. Antimicrobial and radical scavenging effects of alkaloid extracts from Rhizophora mucronata. Pharm. Chem. J. 2015, 49, 34–37. [Google Scholar] [CrossRef]
- Ramanathan, T.A.H. In Studies on Analgesic Activity of a Mangrove Species—Rhizophora mucronata Poir. In Proceedings of the Annual International Conference on Advances in Biotechnology (BIOTECH 2011), Bozen, Italy, 12–13 January 2011. [Google Scholar]
- Jadhav, R.; Jadhav, B. Evaluation of antimicrobial principles of rhizophora species along mumbai coast. J. Adv. Sci. Res. 2012, 3, 30–33. [Google Scholar]
- Wahyuni, W.T.; Darusman, L.K.; Surya, N.K. Potency of rhizopora spp. Extracts as antioxidant and inhibitor of acetylcholinesterase. Procedia Chem. 2015, 16, 681–686. [Google Scholar] [CrossRef]
- Manilal, A.; Merdekios, B.; Idhayadhulla, A.; Muthukumar, C.; Melkie, M. An in vitro antagonistic efficacy validation of Rhizophora mucronata. Asian Pac. J. Trop. Dis. 2015, 5, 28–32. [Google Scholar] [CrossRef]
- Hardoko, E.S.; Puspitasari, Y.; Amalia, R. Study of ripe Rhizophora mucronata fruit flour as functional food for antidiabetic. Int. Food Res. J. 2015, 22, 953–959. [Google Scholar]
- Alikunhi, N.M.; Kandasamy, K.; Manoharan, C.; Subramanian, M. Insulin-like antigen of mangrove leaves and its anti-diabetic activity in alloxan-induced diabetic rats. Nat. Prod. Res. 2012, 26, 1161–1166. [Google Scholar] [CrossRef]
- Pandey, A.K.; Gupta, P.P.; Lal, V.K. Hypoglycemic effect of Rhizophora mucronata in streptozotocin induced diabetic rats. J. Complement. Integr. Med. 2014, 11, 179–183. [Google Scholar] [CrossRef]
- Simlai, A.; Roy, A. Biological activities and chemical constituents of some mangrove species from sundarban estuary: An overview. Pharmacog. Rev. 2013, 7, 170. [Google Scholar]
- Eldeen, I.M.; Effendy, M.A. Antimicrobial agents from mangrove plants and their endophytes. In Méndez-Vilas 9ed. Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education; Formatex Research Center: Badajoz, Spain, 2013; pp. 872–882. [Google Scholar]
- Kathiresan, K.; Boopathy, N.S.; Kavitha, S. Coastal vegetation—An underexplored source of anticancer drugs. Indian J. Nat. Prod. Resour. 2006, 5, 115–119. [Google Scholar]
- Sithranga Boopathy, N.; Kathiresan, K. Anticancer drugs from marine flora: An overview. J. Oncol. 2010, 214186, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Sudirman, S.; Jacoeb, A. Proximate compositions, bioactive compounds and antioxidant activity from large-leafed mangrove (Bruguiera gymnorrhiza) fruit. Int. Food Res. J. 2014, 21, 2387–2391. [Google Scholar]
- Rahman, M.; Ahmed, A.; Shahid, I. Phytochemical and pharmacological properties of Bruguiera gymnorrhiza roots extract. Int. J. Pharm. Res. 2011, 3, 63–67. [Google Scholar]
- Sreedhar, S.; Christy, P.H. Phytochemical analysis of Rhizophora mucronata. Scrut. Int. Res. Agric. Plant Biotechnol. Biol. Prod. 2015, 2, 39–43. [Google Scholar]
- Rohini, R.; Das, A. Antidiarrheal and anti inflammatory activities of lupeol, quercetin, β-sitosterol, adene-5-en-3-ol and caffeic acid isolated from Rhizophora mucronata bark. Der Pharm. Lett. 2010, 2, 95–101. [Google Scholar]
- Fabiyi, O.A. Toxicity of Acanthus ilicifolius (L) fractions against Pratylechus spp. on maize (Zea mays). Alban. J. Agric. Sci. 2015, 14, 149–156. [Google Scholar]
- Ganesh, S.; Jannet Vennila, J. Phytochemical analysis of acanthus ilicifolius and Avicennia officinalis by gc-ms. Res. J. Phytochem. 2011, 5, 60–65. [Google Scholar] [CrossRef]
- Lin, N.; Yi, B.; Li, J.; Zhang, W.; Zhang, X. A new sugar ester from the roots of Acanthus ilicifolius. Rec. Nat. Prod. 2017, 11, 74–76. [Google Scholar]
- Singh, D.; Aeri, V. Phytochemical and pharmacological potential of Acanthus ilicifolius. J. Pharm. Bioallied Sci. 2013, 5, 17. [Google Scholar] [PubMed]
- Tan, D.; Jiang, C.; Tao, Y. Chemical constituents of Acanthus ilicifolius. Chem. Nat. Comp. 2016, 52, 951–952. [Google Scholar] [CrossRef]
- Ghosh, D.; Mondal, S.; Ramakrishna, K. Pharmacobotanical, physicochemical and phytochemical characterisation of a rare salt-secreting mangrove Aegialitis rotundifolia roxb.,(plumbaginaceae) leaves: A comprehensive pharmacognostical study. S. Afr. J. Bot. 2017, 113, 212–229. [Google Scholar] [CrossRef]
- Fauvel, M.-T.; Bousquet-Melou, A.; Moulis, C.; Gleye, J.; Jensen, S.R. Iridoid glucosides from Avicennia germinans. Phytochemistry 1995, 38, 893–894. [Google Scholar] [CrossRef]
- Fauvel, M.-T.; Moulis, C.; Bon, M.; Fourasté, I. A new iridoid glucoside from African Avicennia germinans. Nat. Prod. Lett. 1997, 10, 139–142. [Google Scholar] [CrossRef]
- Laphookhieo, S.; Karalai, C.; Ponglimanont, C.; Chantrapromma, K. Pentacyclic triterpenoid esters from the fruits of Bruguiera cylindrica. J. Nat. Prod. 2004, 67, 886–888. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.M.; Kabir, M.Z.; Paul, P.K.; Islam, M.R.; Rahman, S.; Jahan, R.; Rahmatullah, M. A review on a mangrove species from the sunderbans, bangladesh: Bruguiera gymnorrhiza (l.) lam. (Rhizophoraceae). Am.-Eurasian J. Sustain. Agric. 2013, 7, 340–355. [Google Scholar]
- Sun, Y.-Q.; Guo, Y.-W. Gymnorrhizol, an unusual macrocyclic polydisulfide from the Chinese mangrove Bruguiera gymnorrhiza. Tetrahedron Lett. 2004, 45, 5533–5535. [Google Scholar] [CrossRef]
- Thirunavukkarasu, P.; Asha, S.; Reddy, R.; Priya, D.; Hari, R.; Sudhakar, N. Phytochemical analysis of medicinal mangrove plant species Ceriops decandra. Glob. J. Pharmacol. 2018, 12, 24–30. [Google Scholar]
- Anjaneyulu, A.S.; Rao, V.L. Ceriopsins f and g, diterpenoids from Ceriops decandra. Phytochemistry 2003, 62, 1207–1211. [Google Scholar] [CrossRef]
- Poompozhil, S.; Kumarasamy, D. Studies on phytochemical constituents of some selected mangroves. J. Acad. Ind. Res. 2014, 2, 590–592. [Google Scholar]
- Nagababu, P.; Umamaheswara, R. Phytochemical, antibacterial and antioxidant evaluation of Ceriops decandra (griff). Ding hou leaf extract. J. Chem. Pharm. Res. 2014, 6, 428. [Google Scholar]
- Chen, J.-D.; Yi, R.-Z.; Lin, Y.-M.; Feng, D.-Q.; Zhou, H.-C.; Wang, Z.-C. Characterization of terpenoids from the root of Ceriops tagal with antifouling activity. Int. J. Mol. Sci. 2011, 12, 6517–6528. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, W.-J.; Wu, J. Two new dolabranes from the Chinese mangrove Ceriops tagal. J. Asian Nat. Prod. Res. 2016, 18, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Du, S.S.; Wang, C.F.; Li, J.; Zhang, H.M.; Liu, Q.Z.; Liu, Z.L.; Deng, Z.W. Antifeedant diterpenoids against tribolium castaneum from the stems and twigs of Ceriops tagal (Rhizophoraceae). Molecules 2011, 16, 6060–6067. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.-M.; Li, M.-Y.; Li, J.; Xiao, Q.; Feng, G.; Wu, J. Dolabranes from the Chinese mangrove, Ceriops tagal. J. Nat. Prod. 2010, 73, 1701–1705. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, J.; Yu, S.; Proksch, P.; Gu, J.; Lin, W. Tnf-α inhibitory diterpenoids from the Chinese mangrove plant Excoecaria agallocha l. Phytochemistry 2010, 71, 2124–2131. [Google Scholar] [CrossRef] [PubMed]
- Ponnapalli, M.G.; Ankireddy, M.; Annam, S.C.V.R.; Ravirala, S.; Sukki, S.; Tuniki, V.R. Unusual ent-isopimarane-type diterpenoids from the wood of Excoecaria agallocha. Tetrahedron Lett. 2013, 54, 2942–2945. [Google Scholar] [CrossRef]
- Konishi, T.; Konoshima, T.; Fujiwara, Y.; Kiyosawa, S. Excoecarins d, e, and k, from Excoecaria agallocha. J. Nat. Prod. 2000, 63, 344–346. [Google Scholar] [CrossRef] [PubMed]
- DeSouza, L.; Wahidullah, S. Antibacterial phenolics from the mangrove Lumnitzera racemosa. Indian J. Mar. Sci. 2010, 39, 294–298. [Google Scholar]
- Jasna, T.; Chandra, P.R.; Khaleel, K. Preliminary phytochemical screening and gc ms analysis of chloroform extract of Kandelia candel (l.) druce. Int. J. Pharm. Sci. Res. 2017, 8, 3530–3533. [Google Scholar]
- Prasad, N.; Yang, B.; Kong, K.W.; Khoo, H.E.; Sun, J.; Azlan, A.; Ismail, A.; Romli, Z.B. Phytochemicals and antioxidant capacity from Nypa fruticans Wurmb. Fruit. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Ebana, R.; Etok, C.; Edet, U. Phytochemical screening and antimicrobial activity of Nypa fruticans harvested from Oporo River in the niger delta region of nigeria. Int. J. Innov. Appl. Stud. 2015, 10, 1120. [Google Scholar]
- Balasubramanian, V.; Rajesh, P.; Rajaram, R.; Kannan, V.R. A review on Rhizophora genus: Therapeutically important perspective phytochemical constituents. In Bioactive Phytochemicals: Perspectives for Modern Medicine; Gupta, V.K., Ed.; Daya Publishing House: New Delhi, India, 2015; Volume 3. [Google Scholar]
- Kandil, F.; Grace, M.; Seigler, D.; Cheeseman, J. Polyphenolics in Rhizophora mangle l. Leaves and their changes during leaf development and senescence. Trees 2004, 18, 518–528. [Google Scholar] [CrossRef]
- Andrade-Cetto, A.; Escandón-Rivera, S.M.; Torres-Valle, G.M.; Quijano, L. Phytochemical composition and chronic hypoglycemic effect of Rhizophora mangle cortex on stz-na-induced diabetic rats. Rev. Bras. Farmacogn. 2017, 27, 744–750. [Google Scholar] [CrossRef]
- Martins, J.N.; Figueiredo, F.S.; Martins, G.R.; Leitão, G.G.; Costa, F.N. Diterpenes and a new benzaldehyde from the mangrove plant Rhizophora mangle. Rev. Bras. Farmacogn. 2017, 27, 175–178. [Google Scholar] [CrossRef]
- Wu, Y.B.; Liu, D.; Liu, P.Y.; Yang, X.M.; Liao, M.; Lu, N.N.; Sauriol, F.; Gu, Y.C.; Shi, Q.W.; Kiyota, H. New limonoids from the seeds of Xylocarpus granatum. Helv. Chim. Acta 2015, 98, 691–698. [Google Scholar] [CrossRef]
- Krauss, K.W.; Allen, J.A. Influences of salinity and shade on seedling photosynthesis and growth of two mangrove species, Rhizophora mangle and Bruguiera sexangula, introduced to Hawaii. Aquat. Bot. 2003, 77, 311–324. [Google Scholar] [CrossRef]
- Harvey, P. Australian Mangrove and Saltmarsh Resource: Bruguiera sexangula. Available online: https://coastalresearch.csiro.au/?q=node/119 (accessed on 16 April 2019).
- Use, P. Rhizophora apiculata (prosea). 2016. Available online: https://uses.plantnet-project.org/en/Rhizophora_apiculata_(PROSEA) (accessed on 20 February 2018).
- Kathiresan, K. A review of studies on pichavaram mangrove, southeast India. Hydrobiologia 2000, 430, 185–205. [Google Scholar] [CrossRef]
- Patra, J.K.; Mohanta, Y.K. Antimicrobial compounds from mangrove plants: A pharmaceutical prospective. Chin. J. Integr. Med. 2014, 20, 311–320. [Google Scholar] [CrossRef] [PubMed]


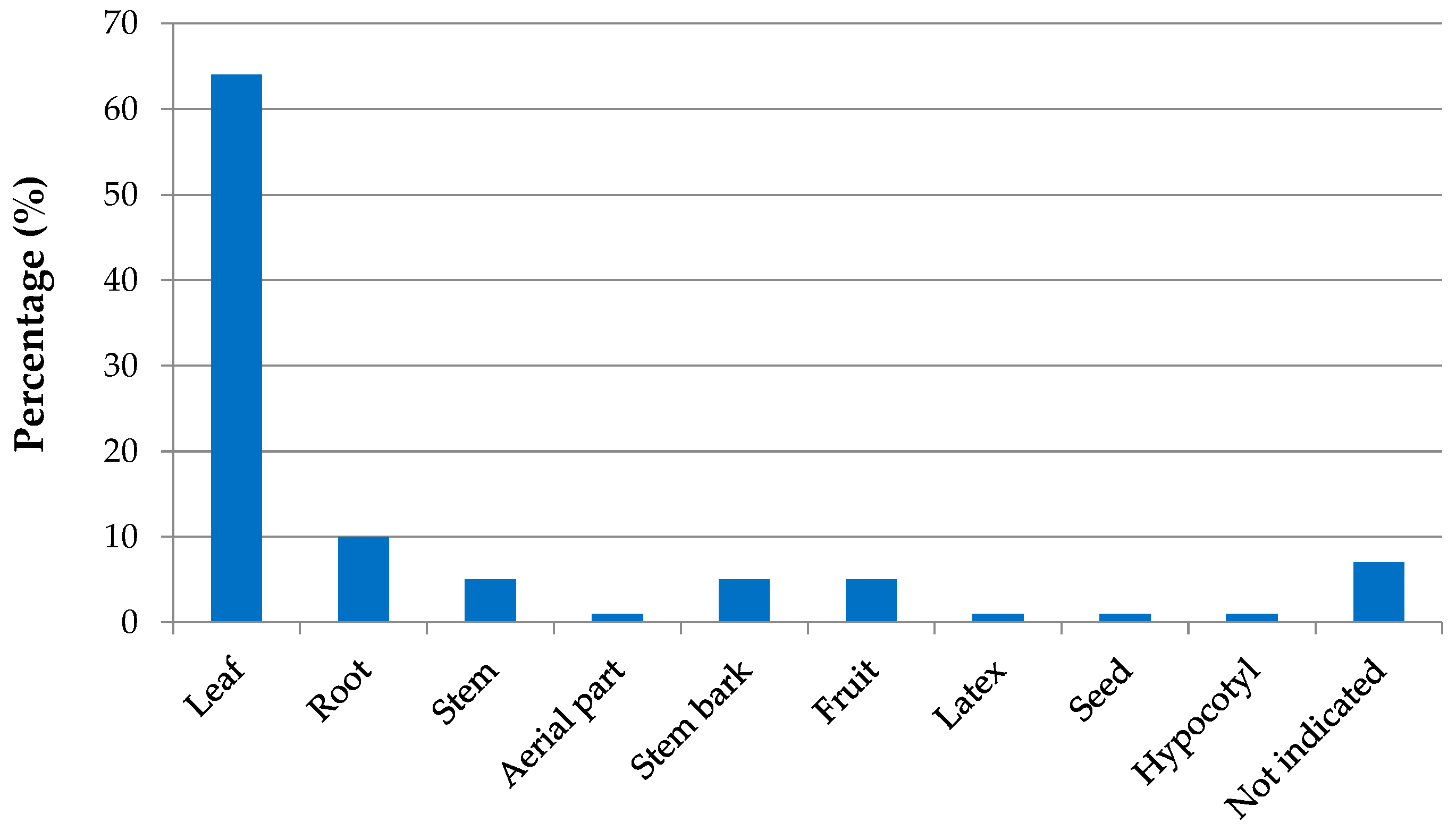
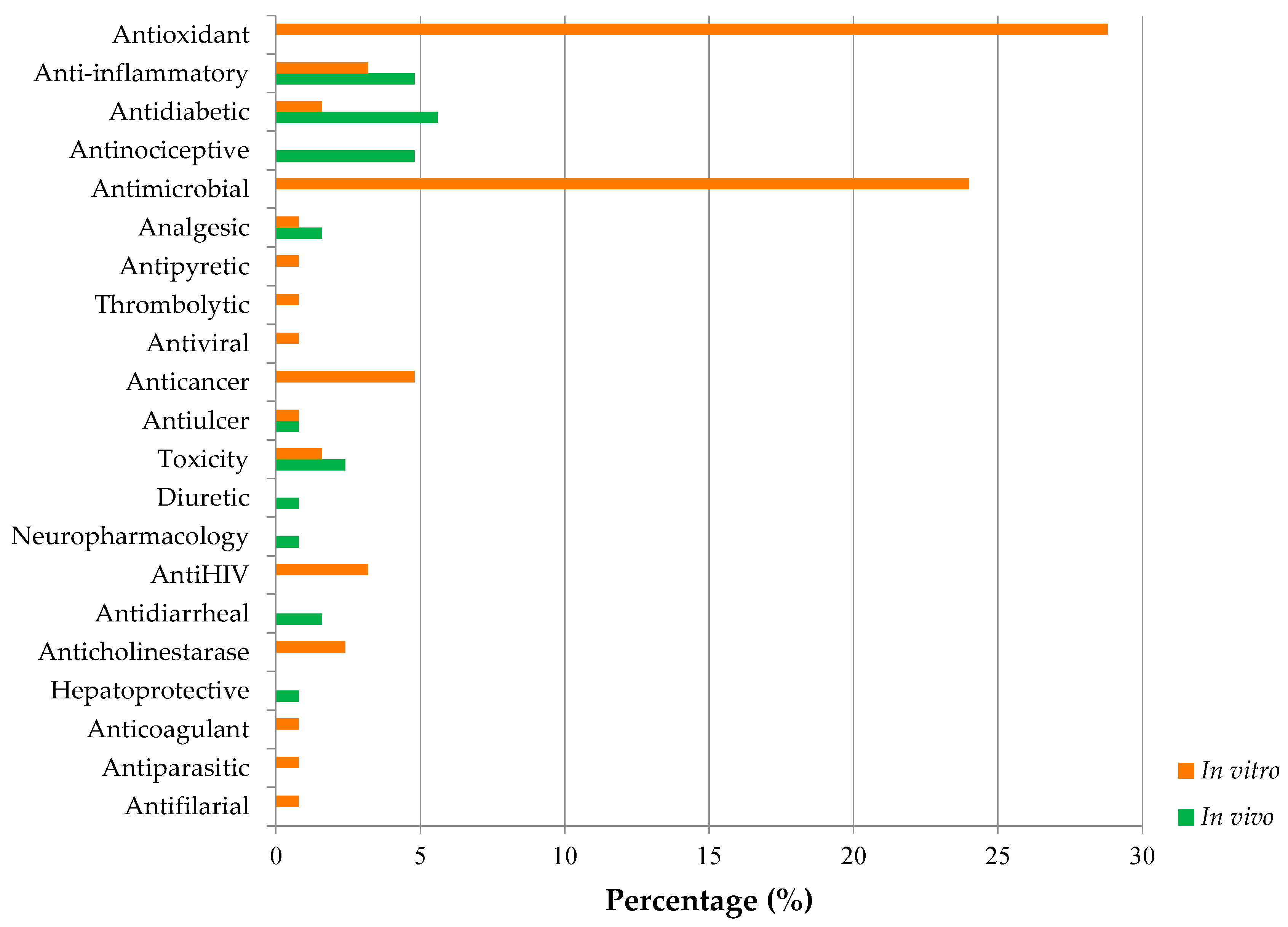
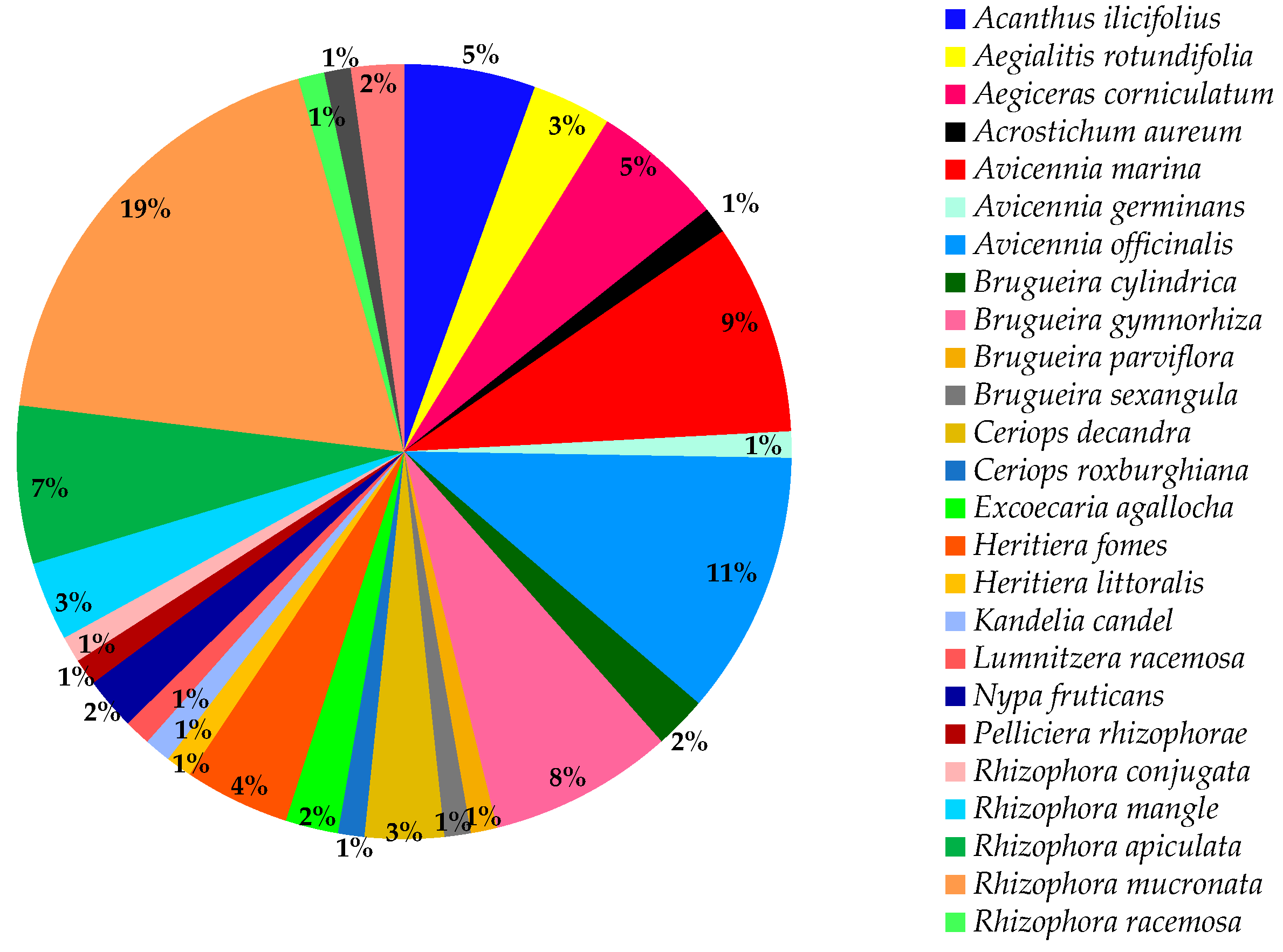
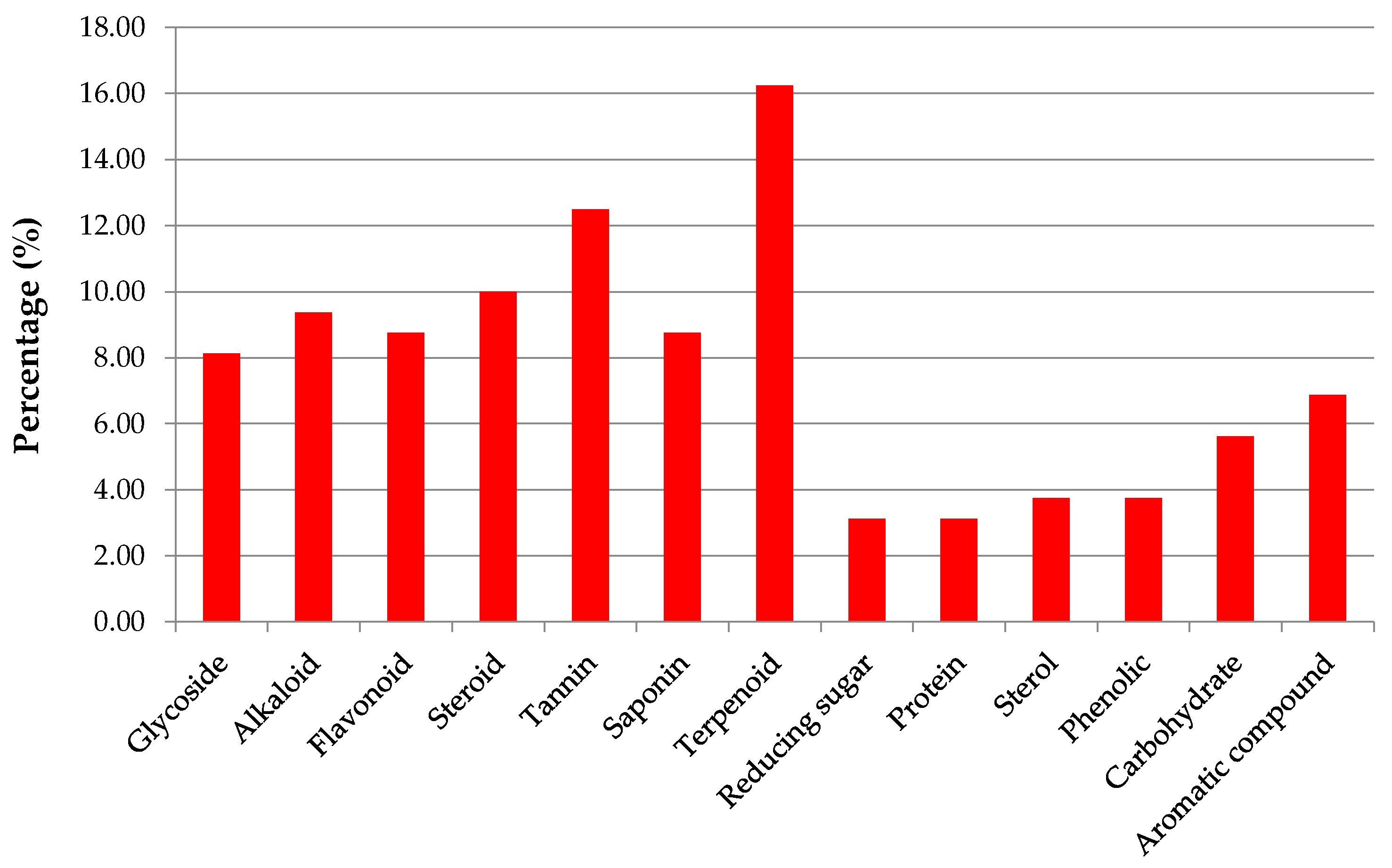
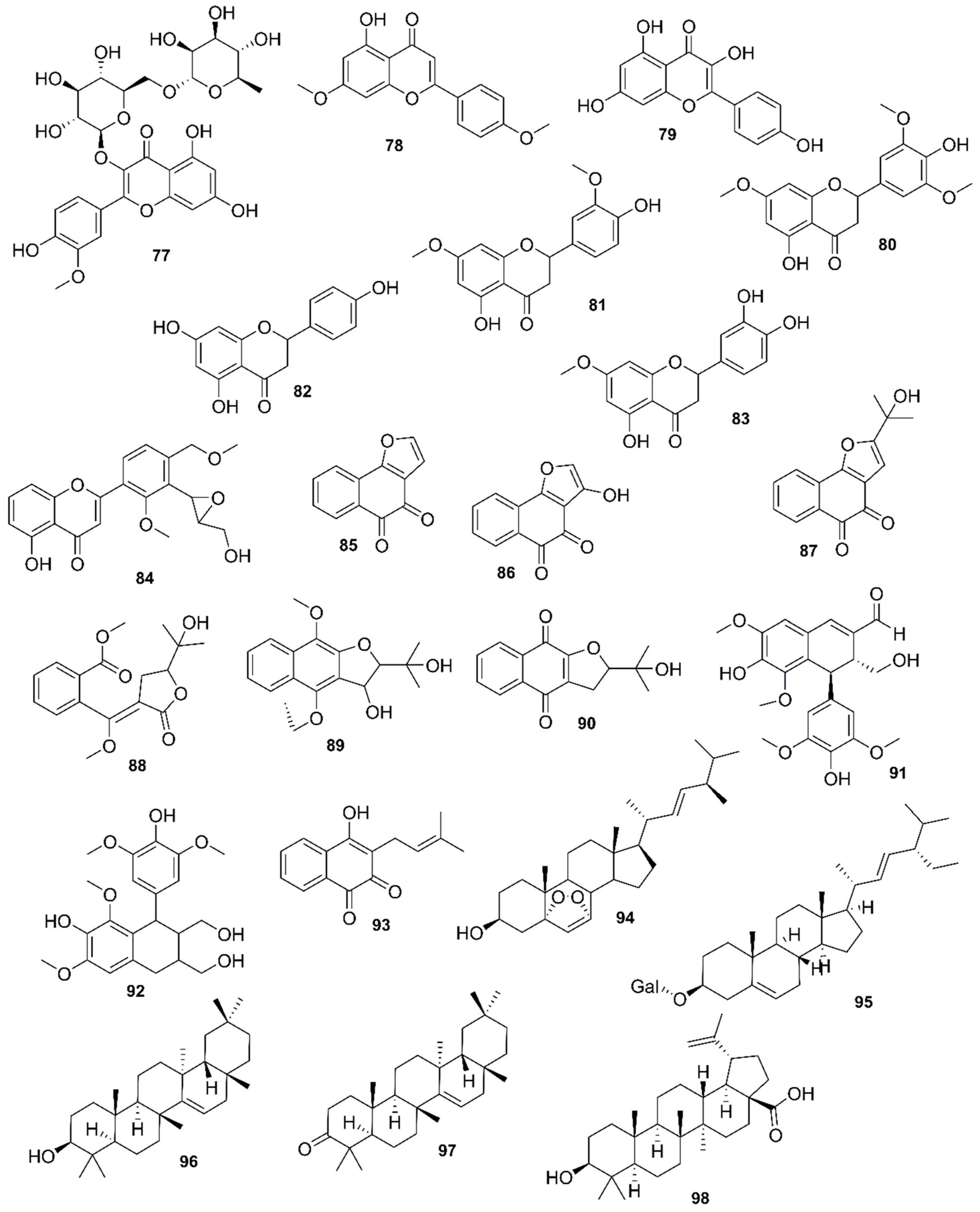
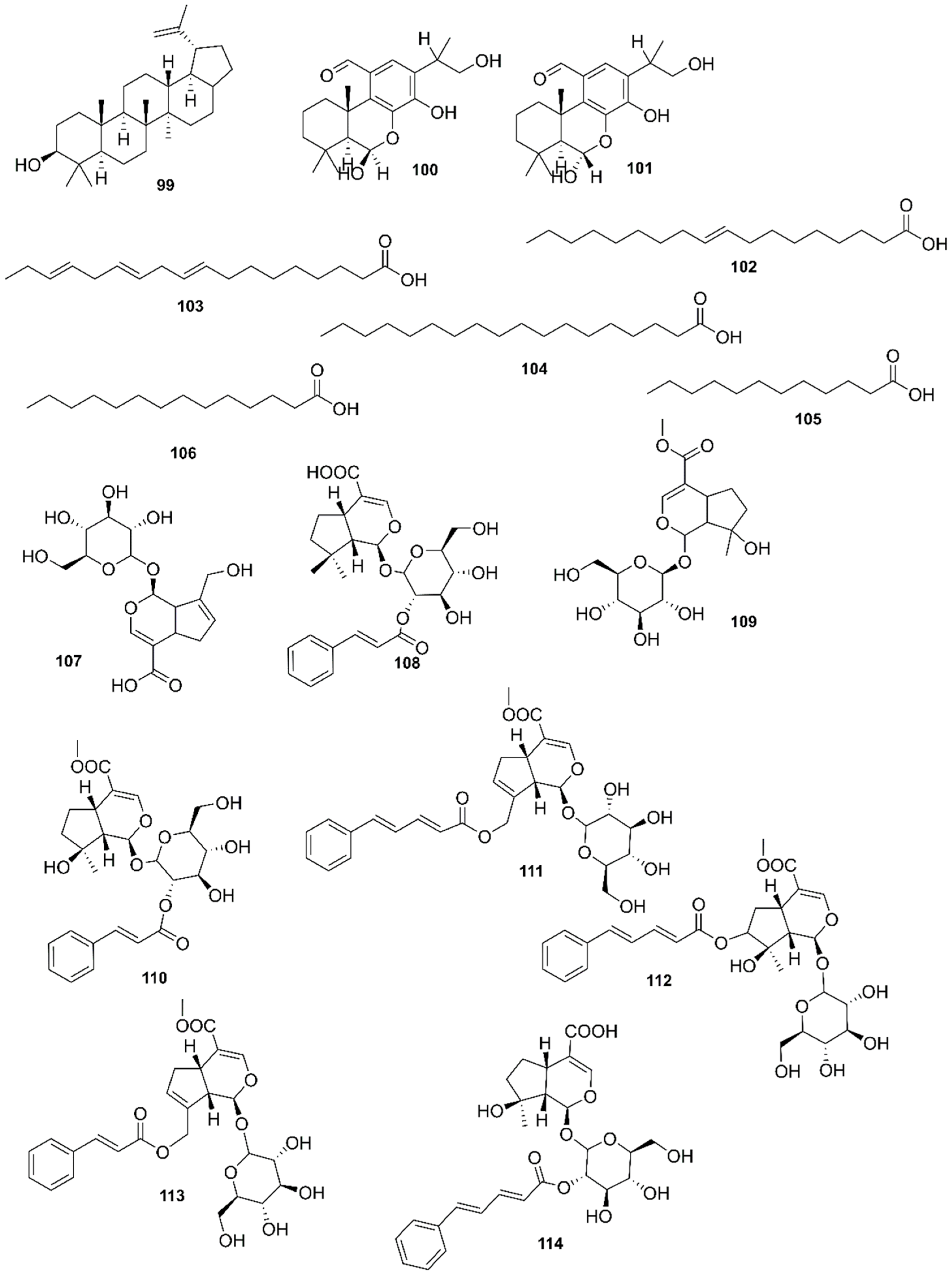
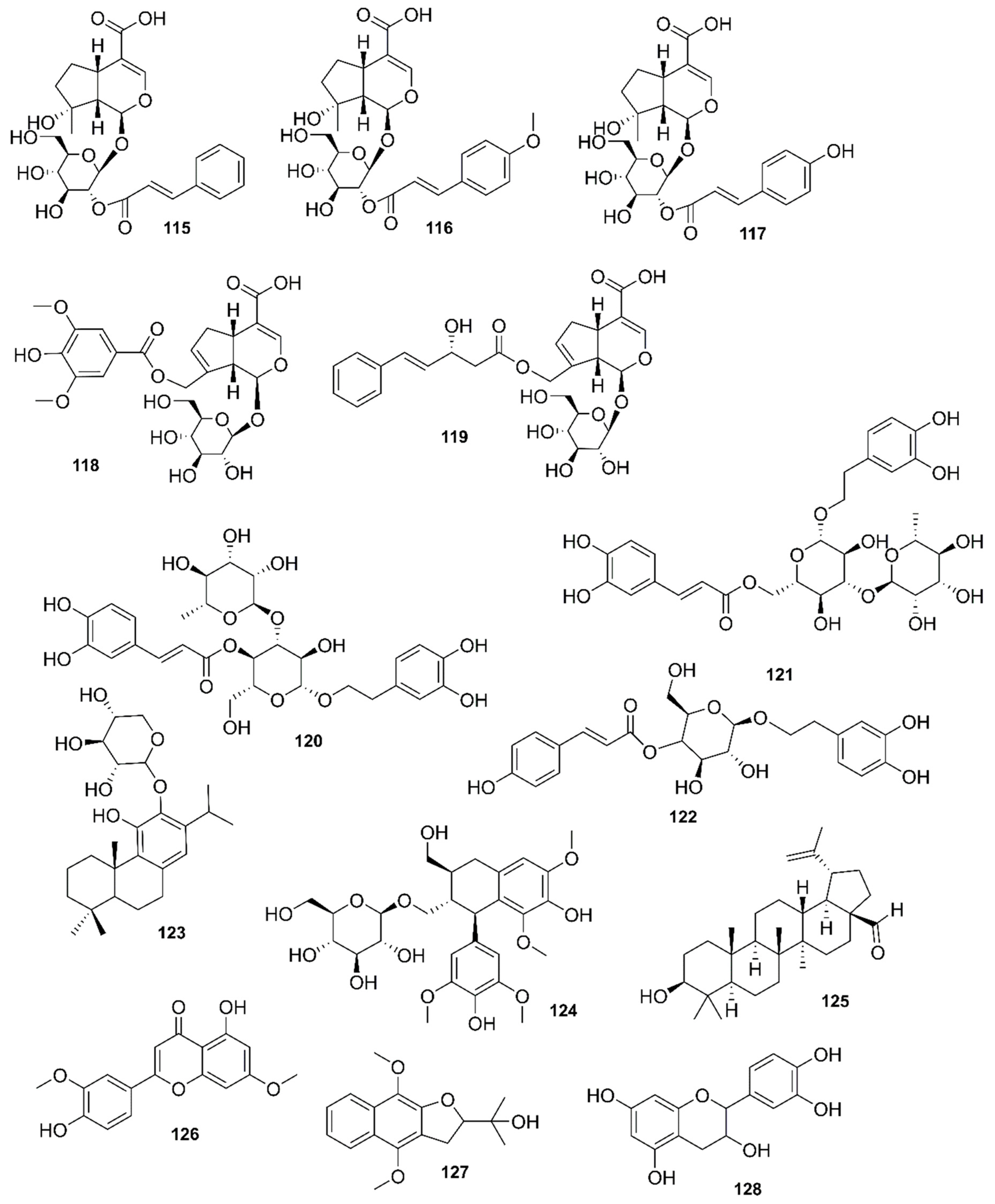
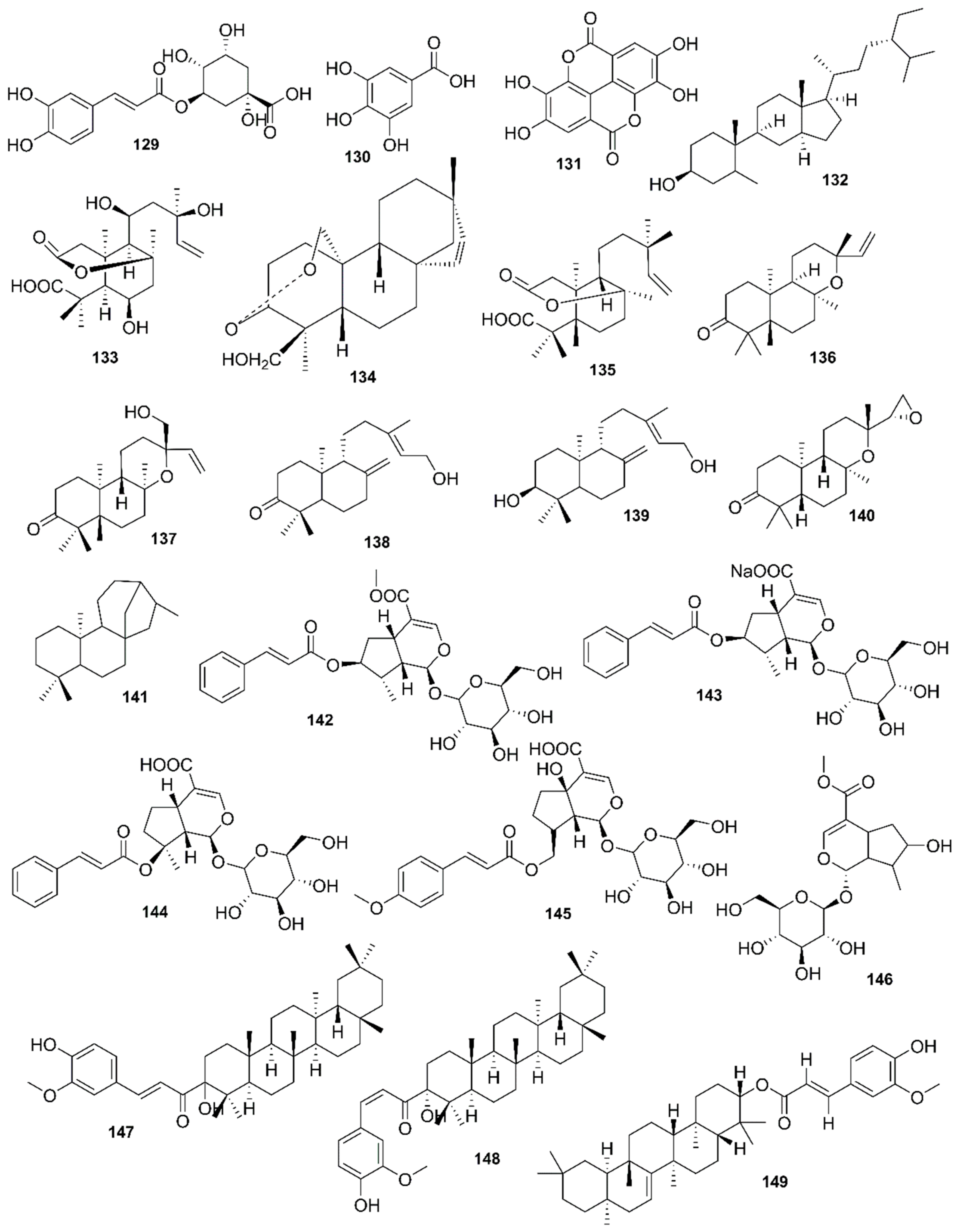
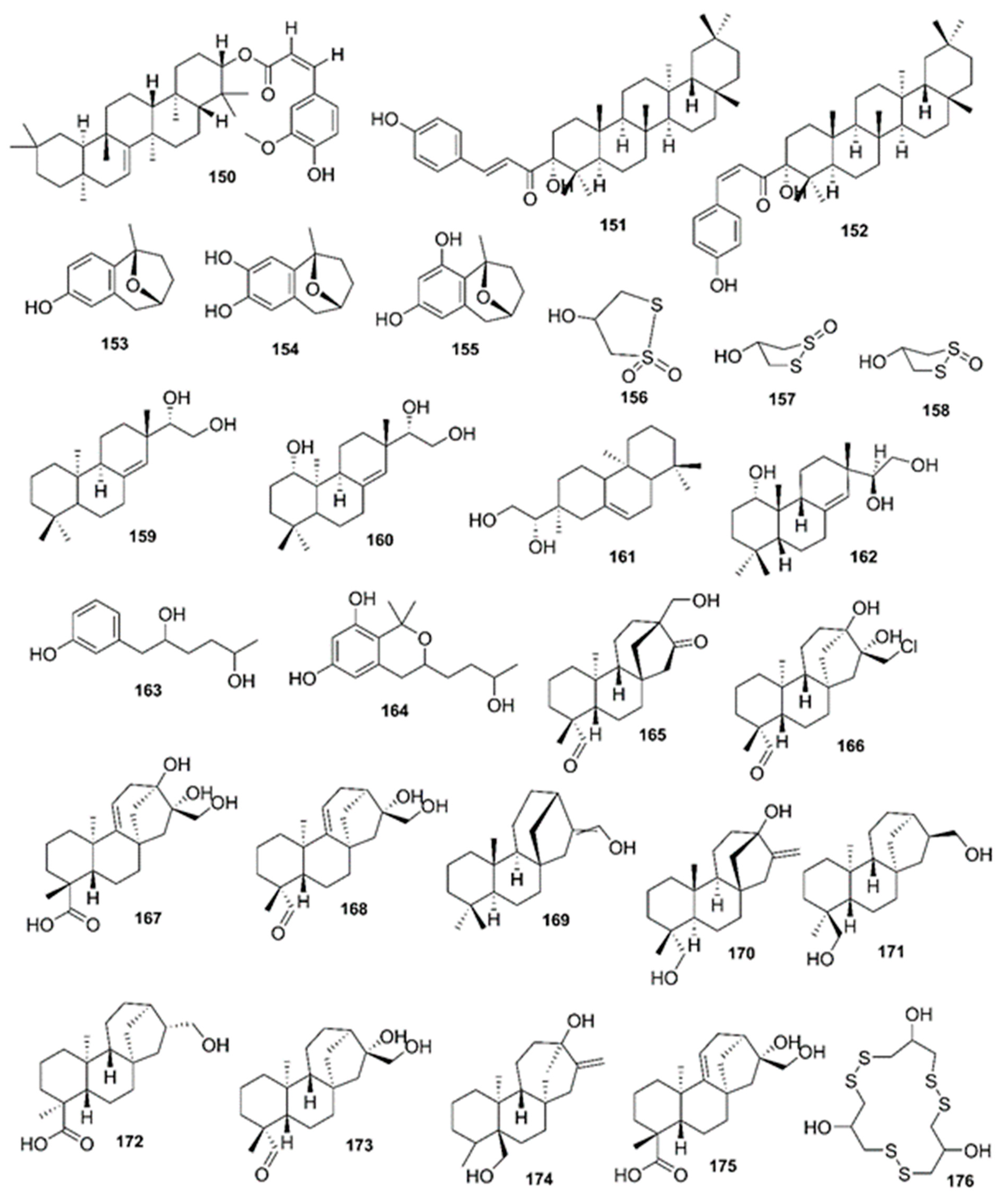
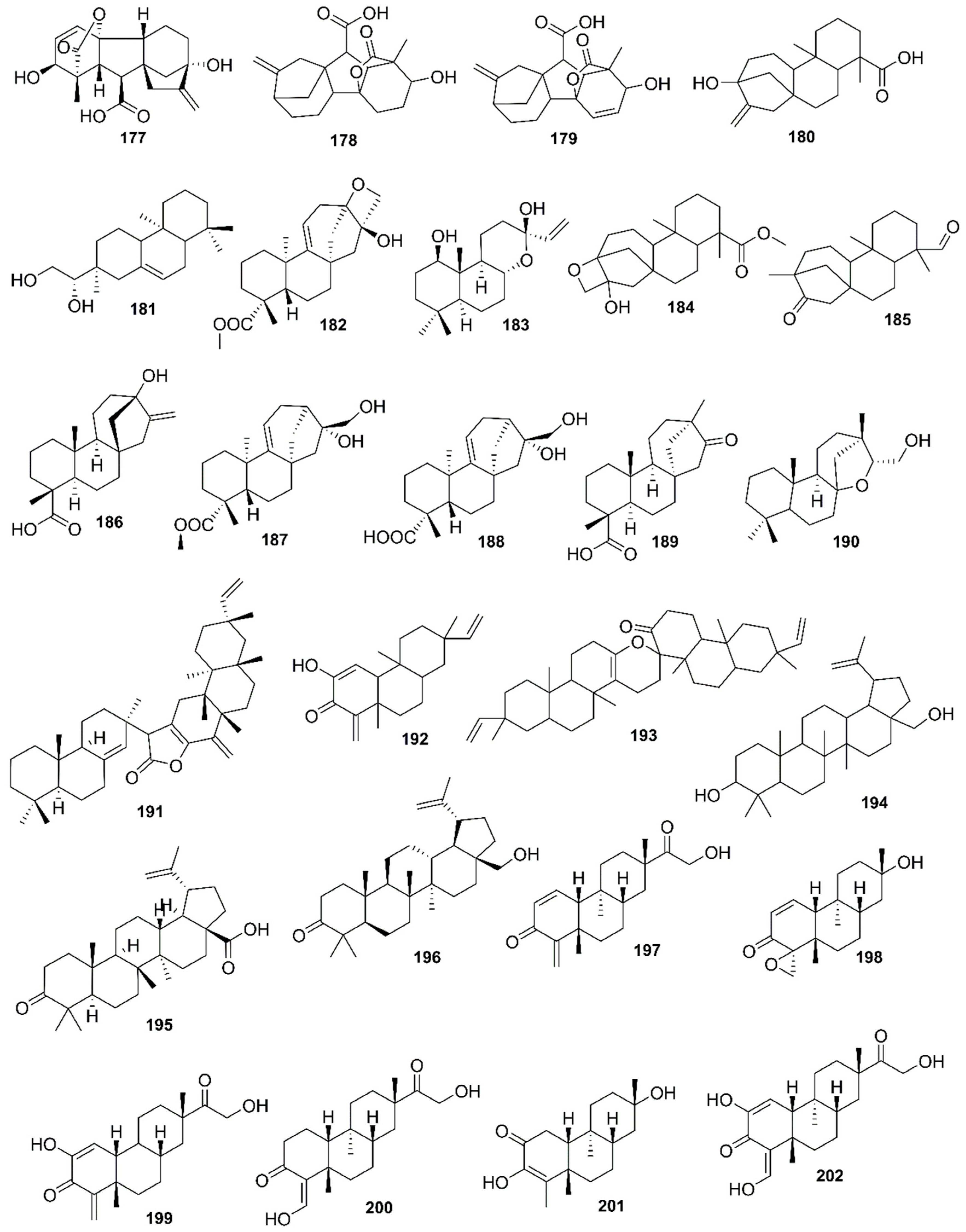
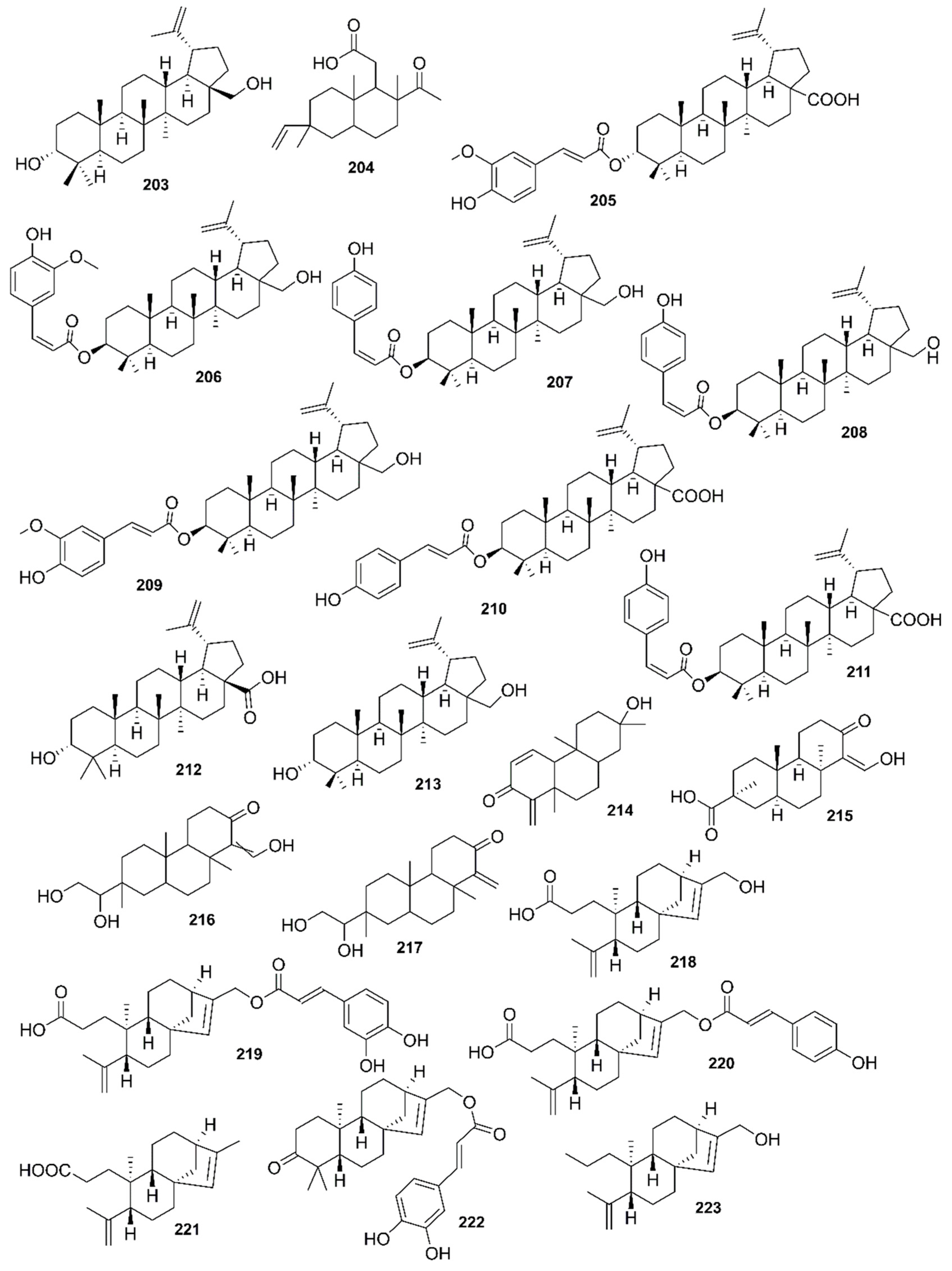
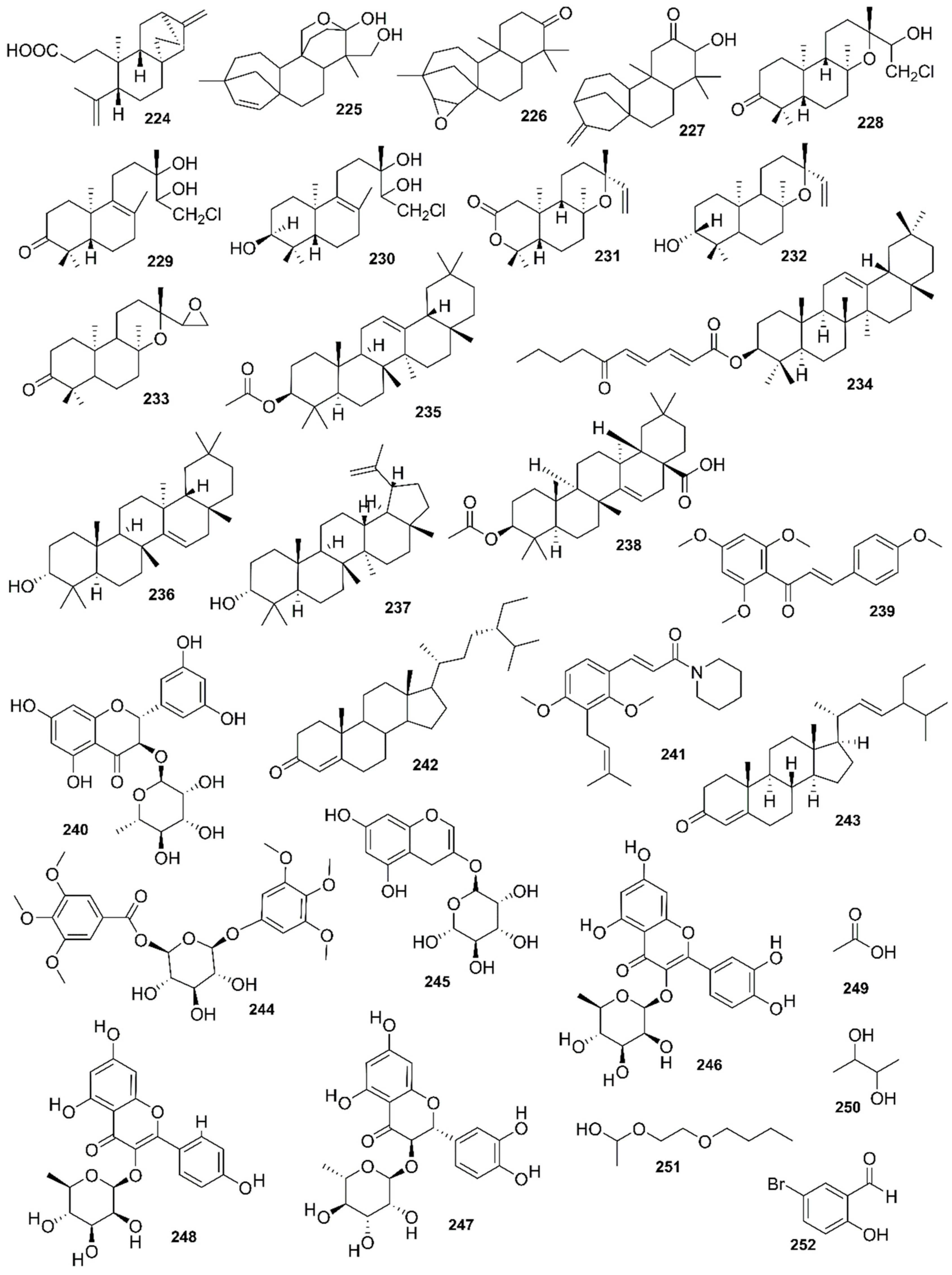
| Country | Local Names | Reference |
|---|---|---|
| Netherlands | Vloedbosschen (mangrove community), mangrove (individual species) | [6,12,13] |
| United Kingdom | Mangrove | |
| France | Manglier, Paletuvier | |
| Germany | Mangrove | |
| Madagascar | Honkalahy, Voandrano | |
| Malaysia | Manggi-manggi | |
| Mauritius Rodrigues Comoros | Manglier, Paletuvier, Mangrove | |
| Spain | Manglar | |
| Surinam | Mangro |
| Red | Black | White | ||
|---|---|---|---|---|
| Characteristics | Leaves | Very shiny, very pointy green on both sides, green on both sides | Less shiny, pointy, grey in color in bottom surface | Shiny on both sides, round |
| Roots | Rhizophores or arc-shaped prop roots, roots come out of the stem and grow downwards to end in the soil | Pneumatophores or pencil-like roots, roots grow against gravity from the soil surface | - | |
| Fruits | Cigar-shaped | Teardrop-shaped | Smallest in size | |
| Examples | R. mucronata, R. mangle | A. germinans, B. gymnorhiza | L.racemosa |
| Rank | Country | km2 | % Global Total |
|---|---|---|---|
| 1 | Indonesia | 42,278 | 25.79 |
| 2 | Brazil | 17,287 | 10.55 |
| 3 | Malaysia | 7616 | 4.65 |
| 4 | Venezuela | 7516 | 4.59 |
| 5 | Nigeria | 6908 | 4.21 |
| 6 | Papua New Guinea | 6236 | 3.80 |
| 7 | Colombia | 6236 | 3.80 |
| 8 | Mexico | 6036 | 3.68 |
| 9 | Thailand | 3936 | 2.40 |
| 10 | Gabon | 3864 | 2.36 |
| 11 | Myanmar | 3783 | 2.31 |
| 12 | Australia | 3314 | 2.02 |
| 13 | Panama | 2673 | 1.63 |
| 14 | Mozambique | 2658 | 1.62 |
| 15 | Cuba | 2407 | 1.47 |
| 16 | Bangladesh | 2314 | 1.41 |
| 17 | Philippines | 2084 | 1.27 |
| 18 | Ecuador | 1906 | 1.16 |
| 19 | United States | 1554 | 0.95 |
| 20 | Cameroon | 1323 | 0.81 |
| Species | Family | Height | Aerial Roots | Bark | Leaves | Fruits | Flowers | Reference |
|---|---|---|---|---|---|---|---|---|
| Acanthus ilicifolius L. | Acanthaceae | Up to 2 m | Stilt | - | Spiny edges | Kidney shaped | Large light-violet petals | [42] |
| Aegiceras corniculatum (L.) Blanco | Primulaceae | Up to 6 m | - | Smooth, greyish | Alternate, obovate, 3–10 cm long, 1.5–5 cm wide | Light green to pink, curved cylinder, 2–7.5 cm long | Fragrant, white, clusters of 10–30 | [43] |
| Aegralitis rotundifolia | Plumbaginaceae | 2–3 m | - | - | Broad ovate, obtuse apex, 5–8.8 cm long, 4.5–8.5 cm wide | - | - | [44,45] |
| Avicennia germinans (L.) L. | Acanthaceae | Up to 30–50 m | Pneumatophores | Rough with irregular flattened scales, dark brown or black | Opposite, elliptical, thick with glands on upper surface, green on upper surface, grey on bottom surface, 3–15 cm long | Dark green, flat, velvety pericarp beneath, 2–3 cm in diameter | White, auxiliary clusters, 1–2 cm in diameter | [7] |
| Avicennia integra N.C.Duke | Acanthaceae | 2–7 m | Pneumatophores | Smooth, brown to reddish | Opposite, simple, elliptical, shiny green on upper surface, pale and fine on bottom surface, 5–14 cm long | Pale green, furry, ovoid pods, 21–23 mm long, 12–15 mm wide | Golden yellow or orange, zygomorphic | [7] |
| Avicennia bicolour Standl. | Acanthaceae | 8–20 m | - | - | - | - | White corolla with yellow throat, hairy petals, zygomorphic, 5–6 mm in diameter | [7] |
| Avicennia marina (Forssk.) Vierh. | Acanthaceae | Up to 14 m | Pneumatophores | Smooth light grey made up of thin, stiff, brittle flakes | Thick, bright, and glossy on upper leaf, grey or silvery-white on bottom leaf, 5–8 cm long | Green, oval, 20–25 mm in diameter | White or golden yellow, clusters of 3–5 | [7,46] |
| Avicennia officinalis L. | Acanthaceae | Up to 30 m | Pneumatophores | Smooth, dirty green to dark grey. Slightly fissured and does not flake | Shiny green with round apex, golden brown on upper leaf, 10 cm long, 5 cm wide | Green or brown, heart-shaped | Orange yellow to lemon yellow, 6–10 mm in diameter | [47] |
| Avicennia schauerina Stapf & Leechm. ex Moldenke | Acanthaceae | - | - | - | - | Pale sap green with purple tinge, flat | Slightly hairy corolla | [7] |
| Bruguiera cylindrica (L.) Blume | Rhizophoraceae | Up to 20 m | Pneumatophores | Smooth and grey, with corky raised patches containing lenticels (pores) | Glossy, elliptical with pointed apex | Curved cylinder, 15 cm long | Greenish white, clusters of 2–5 | [48] |
| Bruguiera sexangula (Lour.) Poir. | Rhizophoraceae | Up to 15 m | Pneumatophores | Smooth, grey-brown | Smooth, glossy green with pointed apex, 9.5–20 cm long, 3–7 cm wide | Green, cigar shaped, 5–12 cm long, 1–2 cm wide | Pale yellow-green to pinkish orange sepals | [43] |
| Bruguiera gymnorhiza (L.) Lam | Rhizophoraceae | 5–8 m | Pneumatophores | Rough, reddish-brown | Large, dark green, shiny, elliptical in shape with reddish petiole, 3–4.5 cm long | Green, cigar shaped, 2 cm long | Creamy white to brown | [13] |
| Ceriops tagal (Perr.) C. B. Robb. | Rhizophoraceae | Up to 25 m | Buttress | Smooth, lenticels, silvery-grey to orangeish-brown | Opposite in pairs, obovate, yellowish-green on bottom surface, 6 cm long, 3 cm wide | Ovoid, 3 cm long, brown | - | [49] |
| Excoecaria agallocha L. | Euphorbiaceae | Up to 15 m | Elbow-shaped pegs | - | Alternate, elliptical, apex shortly acuminate, narrow base, 3–8 cm long, 1.5–3 cm wide | 3-lobed, 8 mm in diameter | Yellow, Unisexual | [50] |
| Heritiera fomes Buch.-Ham | Sterculiaceae | 15-25 m | Pneumatophores | - | Elliptical | - | Pink or orange, bell-shaped, 5 mm across | [51] |
| Heritiera littoralis Aiton | Sterculiaceae | Up to 25 m | Pneumatophores | - | Dark green, short petioles of 1 cm, elliptical, acute apex, 10–23 cm long, 4–10 cm wide | Light green to brown | Unisexual | [51] |
| Kandelia candel (L.) Druce | Rhizophoraceae | Up to 10 m | - | Flaky, reddish brown with lenticels | - | Oval, 25 cm long | White | [49] |
| Nypa fruticans Wurmb | Arecaceae | Up to 9 m | - | - | Palm-like | Woody nut | Catkin-like, red or yellow | [52] |
| Pelliciera rhizophorae Planch. & Triana | Tetramerisataceae | Up to 20 m | Buttress | Brown | Dark green, leather-like, smooth on both upper and bottom surface, small hairs on edges, elongated, pointed, 20 cm long, 5 cm wide | Brown, spherical with a pointed end | 5-rayed symmetric red or white petals | [53] |
| Rhizophora apiculata Blume | Rhizophoraceae | Up to 30 m | Stilt | Grey, almost smooth, 50 cm diameter | Decussate, rosette-like at end of twigs, acute apex, reddish petiole, 1.5–3 cm long | Brown, ovoid or inversely pear-shaped berry, rough, 2–3.5 cm long | Yellow, bisexual, 4-lobed calyx | [54,55] |
| Rhizophora mangle L. | Rhizophoraceae | Up to 24 m | Stilt | Grey or grey-brown, smooth, thin | Opposite, elliptical, acute apex, thick, shiny green on upper surface, yellow-green, black dots on bottom surface, 6–12 cm long, 2.5–6 cm wide | - | Pale pink | [34,56] |
| Rhizophora mucronata Lam. | Rhizophoraceae | 20–25 m | Stilt roots buttressing the trunk | - | Thick, dark green, distinct mucronate tip, covered with minute black spots on inferior surface | Green, cigar-shaped | Creamy-white | [13] |
| Rhizophora racemosa G. Mey | Rhizophoraceae | Up to 30 m | Stilt | - | Opposite, elliptical, hairless blades | - | - | [26]) |
| Rhizophora stylosa Griff. | Rhizophoraceae | Up to 15 m | - | Dark brown to black | - | Ovoid to pear-shaped, 4 cm long | - | [57] |
| Xylocarpus granatum J. Koenig | Meliaceae | 3–8 m | Buttress long | Light brown, yellowish or greenish, smooth, flaky | Bright light green to dark green, round apex, pinnate | - | White, 8 mm across | [24] |
| Species | Region/Country | Plant Part(s) | Use(s) in Traditional Medicine | References |
|---|---|---|---|---|
| Acanthus ilicifolius L. | Bangladesh | WP | Aphrodisiac, rheumatism, relief for asthma, diabetes, diuretic, dyspepsia, leprosy, hepatitis, blood purifier, cure for cold, gangrenous wounds, skin allergies, snake bites | [33,65] |
| West Bengal | NI | Analgesic, wound healing effect | [66] | |
| NI | L | Pain reliever | [67] | |
| Sundarbans, India | L | Rheumatism, neuralgia, snake bite, paralysis, asthma | [68] | |
| NI | WP | Aphrodisiac, astringent, rheumatic pain, leucorrhea | [69] | |
| Pichavaram, India | F | Snake bites | [58] | |
| WP | Detoxification, kidney stone, small pox, skin diseases, ulcer | [70] | ||
| South Thailand | NI | Rheumatism, asthma, paralysis, psoriasis, leucorrhea | [71] | |
| Thailand | L | Blood purifier, dressing against snake bites, rheumatism | ||
| Aegialitis rotundifolia Roxb. | NI | L | Pain reliever, inflammation treatment, anti-ache agent | [72] |
| Bangladesh | L | Antidote for insect bites, pyrexia | [73] | |
| Aegiceras corniculatum (L.) Blanco | Sindh, Pakistan | St | Rheumatism, painful arthritis, inflammation | [74] |
| Sindh, Pakistan | NI | Inflammatory diseases | [75] | |
| Acrostichum aureum L. | Kerala, India | WP | Astringent in hemorrhage, worm remedy | [76] |
| Avicennia germinans (L.) L | NI | B, L, F | Astringent, malaria, hemorrhoids, treatment for hemorrhage, rheumatism, swellings, throat ailments | [7] |
| NI | NI | Diarrhea, hemorrhage, rheumatism, hemorrhoids, tumors, swellings | [77] | |
| Avicennia marina (Forssk.) Vierh. | NI | B, L | Small pox, skin diseases, treatment for ulcers, throat pains | [65] |
| Iran | L | Ulcers, rheumatism, burns | [78] | |
| Avicennia officinalis L. | Tamil Nadu, India | F | Tumor, boil | [79] |
| S | Inflammation, ulcer | |||
| R | Aphrodisiac | |||
| B | Skin disease (scabies), contraceptive, astringent, hepatitis | |||
| Re | Snake bite, wound healing, contraceptive | |||
| Tamil Nadu, India | L | Asthma, paralysis, dyspepsia, rheumatism, ulcer, snake bite, skin disease, small pox sores, tumor | [80] | |
| Pichavaram, India | L | Asthma, bronchial, detoxification, joints pain, stomach disorders, urinary disorders | [58] | |
| Bruguiera cylindrica (L.) Blume | NI | B | Hemorrhage, ulcers | [63] |
| Bruguiera gymnorhiza (L.) Lam | Sundarbans, India | B, L | Diarrhea, fever | [81] |
| India | B, R | Diabetes, viral fever | [82] | |
| Selangor, Malaysia | St | Burns, intestinal worms, liver disorders | [83] | |
| Guangxi Province, China | L | Diarrhea | [54,65] | |
| China | F | Shingles, eye disease, malaria | [84] | |
| Indonesia | F | Angina, hemorrhage, hematuria | [85] | |
| South Andaman Island | L, R | Eye diseases, shingles | [62] | |
| F | Diarrhea, malaria, burns | |||
| B, R, L | Diabetes, hemorrhage, hypertension, stings of toxic lagoon fish | [13] | ||
| Comoros, Mauritius | R | Eye disease | [86] | |
| Pichavaram forest, India | L | Constipation | [58] | |
| Pichavaram, India | WP | Diarrhea, fever, burns, intestinal worms | [64] | |
| NI | B, L, F | |||
| Bruguiera parviflora (Roxb.) Wight & Arn. ex Griff. | NI | B | Diabetes | [61] |
| Ceriops decandra (Griff.) W. Theob. | Tamil Nadu, India | B, F, L | Hepatitis, ulcers | [87] |
| Ceriops roxburghiana Arn. | NI | WP | Diabetes, ulcers | [88] |
| Ceriops tagal (Perr.) C. B. Rob. | NI | B | Hemorrhage | [61] |
| Excoecaria agallocha L. | NI | NI | Epilepsy, ulcers, leprosy, rheumatism, paralysis | [50] |
| Pichavaram, India | La | Toothache | [58] | |
| Heritiera fomes Buch.-Ham. | Bhitarkanika, India | L, R, S | Cardiovascular diseases, gastrointestinal disorders, skin diseases, hepatic disorders, gastrointestinal disorders (diarrhea, dysentery, constipation, stomach ache, dyspepsia), skin diseases (rash, eczema, boils, itch, sores, scabies), infections, jaundice, hepatitis, wound healing, diabetes, goiter (hyperthyroidism) | [7,89,90] |
| Sundarbans, India | WP | [51] | ||
| Sundarbans, India | WP | Heart disease, bloating, stomach ache, diabetes, pain, diarrhea, skin disease, hepatic disorders, goiter | [91] | |
| NI | T | Toothache, oral infection | [69] | |
| Heritiera littoralis Aiton | Philippines | Sa | Fish, arrowhead, and spearhead poisoning | [92] |
| S | Diarrhea, dysentery, hematuria | |||
| Kandelia candel (L.) Druce. | NI | NI | Cardiovascular disease, cancer, neurodegenerative disorders | [93] |
| Kandelia rheedii Wight & Arn. | India | NI | Tuberculosis | [94] |
| Lumnitzera racemosa Willd. | Orissa, India | NI | Snake bites, rheumatism, skin allergies, blood purifier, asthma, diabetes, anti-fertility | [95] |
| Nypa fruticans Wurmb | Malaysia | NI | Diabetes | [96] |
| Philippines | F, L | Diabetes, snake bite | [61] | |
| Rhizophora apiculata Blume | Tamil Nadu, India | WP | Prevent colitis, inflammatory bowel disease (IBD) | [97,98] |
| Pichavaram, India | B | Amoebiasis, diarrhea, nausea, vomiting | [58] | |
| Rhizophora mucronata Lam. | India | WP | Angina, dysentery, hematuria, hepatitis, ulcers, diabetes, hemorrhage | [98] |
| Tamil Nadu, India | B | Diarrhea, nausea, vomiting, amoebiasis, antiseptic, stop bleeding | [58] | |
| Mauritius | L, R | Astringent, antidote against toxic fish stings, diabetes, fever, hypertension | [13] | |
| Porong, Indonesia | WP | Elephantiasis, hematoma, hepatitis, ulcer, febrifuge | [60,61] | |
| India | L, R | Angina, blood in urine, diabetes, diarrhea, dysentery, fever | [99] | |
| Malaysia | L, R | Childbirth, hemorrhage | [100] | |
| China | B | Diarrhea | ||
| Japan | B | Diarrhea | ||
| NI | L | Astringent, antiseptic | ||
| NI | WP | Diarrhea, elephantiasis, hematuria | [33] | |
| New Guinea | St | Constipation, cure fertility, menstruation disorders | [59] | |
| Pichavaram, India | B | Diarrhea, nausea, vomiting | [58] | |
| Thailand | B | Diarrhea, dysentery, leprosy | [71] | |
| Rhizophora conjugata L. | India | B | Diabetes | [101] |
| Rhizophora mangle L. | India | B, L | Diabetes | [88,101] |
| Rhizophora racemosa G. Mey. | Nigeria | L | Toothache, dysmenorrhea | [102] |
| NI | NI | Malaria | [103] | |
| Xylocarpus granatum J.Koenig | NI | NI | Cholera, diarrhea, elephantiasis, inflammation, pain, swelling of breasts | [104] |
| East Africa | B | Cholera, diarrhea, fever, malaria | ||
| South East Asia | L | Diarrhea | ||
| Indian coastal region | F | Diarrhea, dyslipidemia, hyperglycemia | ||
| Pichavaram, India | B | Cholera, diarrhea, dysentery | [58] | |
| Thailand | B | Cholera | [71] |
| Species | Traditionally Used | Pharmacologically Validated |
|---|---|---|
| Acanthus ilicifolius | ✓ | ✓ |
| Aegiceras rotundifolia | ✓ | ✓ |
| Aegiceras corniculatum | ✓ | ✓ |
| Acrostichum aureum | ✓ | ✓ |
| Avicennia germinans | ✓ | ✓ |
| Avicennia marina | ✓ | ✓ |
| Avicennia officinalis | ✓ | ✓ |
| Bruguiera cylindrica | ✓ | ✓ |
| Bruguiera gymnorhiza | ✓ | ✓ |
| Bruguiera parviflora | ✓ | ✓ |
| Bruguiera sexangula | ✓ | ✓ |
| Ceriops decandra | ✓ | ✓ |
| Ceriops roxburghiana | ✓ | ✓ |
| Ceriops tagal | ✓ | ✕ |
| Excoecaria agallocha | ✓ | ✓ |
| Heritiera fomes | ✓ | ✓ |
| Heritiera littoralis | ✓ | ✓ |
| Kandelia candel | ✓ | ✓ |
| Kandelia rheedii | ✓ | ✕ |
| Lumnitzera racemosa | ✓ | ✓ |
| Nypa fruticans | ✓ | ✓ |
| Pelliciera rhizophorae | ✕ | ✓ |
| Rhizophora apiculata | ✓ | ✓ |
| Rhizophora mucronata | ✓ | ✓ |
| Rhizophora stylosa | ✕ | ✓ |
| Rhizophora conjugata | ✓ | ✓ |
| Rhizophora mangle | ✓ | ✓ |
| Rhizophora racemosa | ✓ | ✓ |
| Xylocarpus granatum | ✓ | ✓ |
| Total number of species | 27 | 27 |
| Species | Plant Part(s) | Extract | Study/Assays | Activity | Reference |
|---|---|---|---|---|---|
| Acanthus ilicifolius L. | L, R | Me | Antioxidant-DPPH (In vitro) | IC50 (mg/mL): L = 2501.53 ± 182.62, R = 1319.66 ± 150.76 | [105] |
| L, R | Me | Antioxidant-FRAP (In vitro) | AAE (mg/g): L = 1.10 ± 0.03, R = 1.62 ± 0.03 | ||
| L | Me | Antinociceptive- Acetic acid-induced writhing test (In vivo) | Control (10 mL/kg) number of writhings = 51.5 ± 4.1, at 250 and 500 mg/kg (extract), %inhibition = 33.0% and 51.1% respectively | [68] | |
| L | Me | Antinociceptive-Formalin test (In vivo) | At 250 and 500 mg/kg, %inhibition = 37.54 and 50.18 respectively for 5 min and 45.5% and 67.24% respectively for 30 min | ||
| L | Me | Anti-inflammatory- Carrageenan-induced paw edema (In vivo) | ED50 (mg/kg) = 146.2, 95% Cl = 69.38–286.2 both at early and late phases. After 2 h, ED50 (mg/kg) = 194, 95% Cl = 135.8–301.4. With BW755C (COX-LOX inhibitor) the paw edema decreased significantly. No significant inhibitory activity was shown with indomethacin | [66] | |
| L | Me | Anti-inflammatory- Acetic acid-induced peritoneal inflammation (In vivo) | At 200 and 400 mg/kg, %inhibition = 48 and 77, respectively | ||
| L | Me | Antioxidant-DPPH (In vitro) | IC50 (g/mL): extract = 8.40 ± 0.06, Quercetin = 5.28 ± 0.08, Vitamin C = 6.62 ± 0.05 | ||
| L | Me | Antioxidant- ABTS (In vitro) | IC50 (g/mL): extract = 10.34 ± 0.02, Quercetin = 3.60 ± 0.03, Vitamin C = 4.86 ± 0.03 | ||
| L | Me | Antioxidant- SO (In vitro) | IC50 (g/mL): extract = 78.12 ± 2.51, Quercetin = 30.19 ± 1.32, Vitamin C = 52.18 ± 3.14 | ||
| L | Me | Antioxidant- HO (In vitro) | IC50 (g/mL): extract = 24.60 ± 1.10, Quercetin = 14.32 ± 0.52, Vitamin C = 21.08 ± 0.34 | ||
| L | A | Antimicrobial (In vitro) | Zone of inhibition (mm) against BS = 20, SA = 18, PA = 18, CA = 22 | [67] | |
| L | Bu | Zone of inhibition (mm) against BS = 16, SA = 8, PA = 10, CA = 15 | |||
| L | C | Zone of inhibition (mm) against BS = 22, SA = 21, PA = 20, CA = 26 | |||
| L | A | Antimicrobial-Disc diffusion assay (In vitro) | Active against EC, AGT, STM, SA, AF, and TR. Zone of inhibition (mm) = 7.5 ± 0.4, 8 ± 0.5, 7 ± 0.1, 8.2 ± 0.3, 8.0 ± 0.7 and 7.9 ± 0.3, respectively. Me and EA extracts are inactive against TR | [106] | |
| Aegialitis rotundifolia Roxb. | L | Aq | Anti-inflammatory- Cotton pellet-induced granuloma (In vitro) | At 400 mg/kg, %inhibition = 29.1, while %inhibition of standard drug = 63.22% | [73] |
| L | Aq | Anti-inflammatory- Carrageenan induced hind paw edema (In vitro) | At 400 mg/kg, %inhibition = 26.75%, while %inhibition of indomethacin = 40.13% | ||
| L | Aq | Analgesic- Acetic acid induced writhing test (In vitro) | At 200 and 400 mg/kg, %inhibition = 47.86% and 57.1% respectively | ||
| L | Aq | Antipyretic (In vitro) | At 400 mg/kg, a moderate antipyretic activity is reported by decreasing the temperature at 36.61 °C | ||
| L | Aq | Cytotoxicity using micro culture tetrazolium assay (MTT assay) (In vitro) | Active; IC50 at 200 µg/mL = 97.77 | [107] | |
| L | Me | Thrombolytic activity (In vitro) | At dosage 2, 4, 6, 8, and 10 mg/mL, %of clot lysis = 9.57 ± 1.06%, 13.35 ± 1.67%, 19.35 ± 1.84%, 28.23 ± 1.97%, and 32.76 ± 1.22%, respectively | [45] | |
| L | Me | Membrane stabilizing activity―Hypotonic solution-induced hemolysis (In vitro) | At dosage 2, 4, 6, 8, and 10 mg/mL, %inhibition of hemolysis = 22.80 ± 0.49%, 30.80 ± 0.6%, 35.30 ± 0.74%, 40.80 ± 0.89%, and 45.80 ± 0.77%, respectively | ||
| L | Me | Antibacterial―Disc diffusion (In vitro) | Active against 100 µL of ST and EC. Inactive against SA and PA | ||
| Aegiceras corniculatum (L.) Blanco | St | H | Toxicity (In vivo) | Non-toxic at 1 g/kg | [74] |
| St | EA | LD50(mg/kg) = 850 | |||
| St | Me | Toxic above 200 mg/kg | |||
| St | EA | Antinociceptive- Acetic acid-induced writhings in mice (In vivo) | At 10 and 50 mg/kg, %inhibition = 29 ± 2.5% and 53 ± 3.0%, respectively, IC50 (mg/kg) at 50 mg/kg = 52 ± 4.2 | ||
| St | H | At 25, 50, and 100 mg/kg, %inhibition = 12 ± 0.7%, 28 ± 2.5%, and 37 ± 3.5%, respectively | |||
| St | Me | At 1, 5, and 10 mg/kg, %inhibition = 33.4 ± 3.3%, 55.6 ± 6.2%, and 82.4 ± 7.3%, respectively. Me extract at 5 mg/kg is more potent with IC50 value of 4.2 ± 0.99 | |||
| AP | H | Anti-inflammatory- Carrageenan induced paw edema in rats (In vivo) | At 10, 25, and 50 mg/kg, % inhibition = 15.8 ± 2.0%, 39.2 ± 3.9%, and 65.0 ± 4.0%, respectively | [75] | |
| AP | EA | At 1, 5, and 10 mg/kg, % inhibition = 28.4 ± 4.7%, 40.6 ± 2.1%, and 51.4 ± 2.7%, respectively | |||
| AP | Me | At 100 mg/kg, % inhibition = 10.8 ± 3.4% | |||
| L | CE | Antibacterial using REMA assay (In vitro) | Active against BS (gram-positive) and EC (gram-negative) at 5 mg/mL | [108] | |
| L, Sb, R | Me | Antioxidant-FRAP (In vitro) | AAE (mg/g) for the 3 methanolic extracts of each plant parts = 5.31 ± 0.11, 8.18 ± 0.14, and 5.03 ± 0.73, respectively | [105] | |
| L, Sb, R | Me | Antioxidant-DPPH (In vitro) | IC50 (mg/mL) for the 3 methanolic extracts of each plant parts = 129.95 ± 3.29, 96.74 ± 2.52, and 233.53 ± 56.25, respectively | ||
| L | EA | Antimicrobial-Disc diffusion assay (In vitro) | Zone of inhibition (mm) against EC, AGT, STM, and SA = 6.9 ± 0.4, 8.25 ± 0.3, 6.5 ± 0.5, and 8.0 ± 0.4, respectively, Inactive against AF and TR | [106] | |
| Acrostichum aureum L. | L | Me | Antibacterial-Disc diffusion (In vitro) | Zone of inhibition (mm) against EC = 10 ± 0.12, SM = 7.6 ± 0.58 | [76] |
| L | Ac | Zone of inhibition (mm) against PA, SA, EC and SM = 12.3 ± 0.23, 9.7 ± 0.48, 10.6 ± 0.14, and 7 ± 0.32, respectively | |||
| L | PE | No activity observed | |||
| L | W | No activity observed | |||
| Avicennia marina (Forssk.) Vierh | L | A | Antimicrobial- Agar well diffusion (In vitro) | Active against BC, EF, SA, SM, and AT | [7] |
| L | E | Anti-inflammatory- Rat model of rheumatoid arthritis (In vivo) | Inflammatory markers were observed to be reduced and joint lesions were improved | [109,110] | |
| L | E | Antiviral (In vitro) | Active against HIV, SFV, EMVC, and HBV | [98] | |
| L | E | Antimutagenic- MTT assay (In vitro) | Strong effect with inhibition rates of 68% and 71% with and without metabolic activation S9 | [111] | |
| L | E | Anticancer- MTT assay (In vitro) | Significant cytotoxic effect on HL-60 cells and induced apoptosis in HL-60 cell line | ||
| NI | Me | Antioxidant- ABTS (In vitro) | Strong activity | [112] | |
| L | NI | Antimicrobial (In vitro) | Zone of inhibition (mm) against EC, SA, BS, CA, and AN = 12, 6, 7, 9, and 10, respectively for 30 µl of extract | [113] | |
| L | Ac | Antimicrobial- Disc diffusion assay (In vitro) | Zone of inhibition (mm) against AGT, STM, SA, and TR are 6.8 ± 0.9, 7.5 ± 0.5, 9.1 ± 0.3, and 6.5 ± 0.35, respectively. Inactive against EC and TR | [106] | |
| L | CE | Antimicrobial- Disc diffusion assay (In vitro) | Zone of inhibition (mm) against SA, KP, PA, BS, EC, ENA, PS, SP, and CS = 18, 24, 26, 16, 27, 8, 12, 5, and 1, respectively | [114] | |
| L | CE | Antioxidant- DPPH (In vitro) | %radical scavenging = 88.93% | ||
| Avicennia germinans (L.) L. | L | Me | Antibacterial- Disc diffusion assay (In vitro) | At 100 mg, zone of inhibition (mm) against EC, KS, PS, and SA = 16, 22, 12, and 18 | [77] |
| Avicennia officinalis L | L | E | Antioxidant- DPPH (In vitro) | IC50 (control) = 65.12 ± 54, IC50 (extract) at 0.1 mg/mL = 40.77 ± 3.43 | [80] |
| L | E | Antioxidant- HO (In vitro) | IC50 (control) = 64.35 ± 1.34, IC50 (extract) = 38.23 ± 3.84 | ||
| L | E | Antioxidant- NO (In vitro) | At 0.1 mg/mL, IC50: control = 62.97 ± 8.64, extract = 39.87 ± 4.78 | ||
| L | E | Antioxidant- ABTS (In vitro) | At 0.1 mg/mL, IC50: control = 61.84 ± 1.33, extract = 38.78 ± 9.62 | ||
| L | EA | Antimicrobial- Disc diffusion assay (In vitro) | Zone of inhibition (mm) against EC, STM, and SA = 7.8 ± 0.7, 7 ± 0.1, and 7.7 ± 0.5, respectively, inactive against AF and TR | [106] | |
| R | A, E, Me | Antimicrobial- Agar well diffusion (In vitro) | For the three extracts activity observed with EC, SA, ENA, KP, PA, BS, LD, and SP | [112] | |
| NI | E | Antiulcer- Indomethacine-induced gastric ulcer (In vitro) | Gastric ulcers observed to decrease when glutathione is reduced in the gastric mucosa | [115] | |
| L | Me | Anti-inflammatory- Carrageenan induced paw edema (In vivo) | Inhibition of prostaglandin effect more potent in chronic model than in acute model | [79] | |
| L | Me | Diuretic- Lipschitz dirutic model (In vivo) | At dosage 200 and 400 mg/kg, volume of urine = 3.06 ± 0.18 and 3.89 ± 0.13 mL, respectively | [116] | |
| L | Me | Neuropharmacological- Pentobarbital induced hypnosis test (In vivo) | At dosage 250 and 500 mg/kg, total sleeping time = 6.74 ± 2.83 and 82.07 ± 3.57 min, respectively while with control (0.1% Tween 80), time = 32.06 ± 1.20 min | ||
| L | Me | Neuropharmacological- Open field test (In vivo) | At dosage 250 mg/kg, number of movements before and after drug administration after 90 min = 110.50 ± 2.12 and 41.85 ± 3.35, respectively | ||
| At dosage 500 mg/kg, number of movements before and after drug administration after 90 min = 107.99 ± 2.70 and 30.06 ± 2.64, respectively | |||||
| L | Me | Neuropharmacological- Hole cross test (In vivo) | At dosage 250 mg/kg, number of holes crossed before and after drug administration after 90 min = 7.57 ± 0.18 and 5.30 ± 0.69, respectively | ||
| At dosage 500 mg/kg, number of movements before and after drug administration after 90 min = 6.61 ± 0.72 and 4.90 ± 0.67, respectively | |||||
| L | PE | Anti-HIV- Reverse transcriptase (RT) inhibition assay (In vitro) | %inhibition: control = 71.04 ± 1.94, extract = 74.79 ± 3.47 | [117] | |
| L | E | %inhibition: control (AZT) = 71.04 ± 1.94, extract = 82.00 ± 0.26 | |||
| Fr | E | Antioxidant- ABTS (In vitro) | Activity highest with ABTS compared to DPPH and FRAP | [112] | |
| L | E | Toxicity (In vivo) | No significant change observed in the majority of the mice. Mortality rate was zero | [115] | |
| L | E | Antioxidant- DPPH (In vitro) | At dosage 10 and 100 µg/mL, %inhibition = 16.34% and 63.64%, respectively | [118] | |
| L | E | Cytotoxic (In vitro) | LC50 (µg/mL) = 131.2 | ||
| L | E | Antibacterial- Disc diffusion (In vitro) | Active against EC and ST, MIC (µg/mL) against EC = 62.5, ST = 125 | ||
| Bruguiera cylindrica (L.) Blume | St | Bu, C, E, H, Aq | Antioxidant- Oxygen free radical generation (In vitro) | %inhibition for all extracts ranged from 18–77 for superoxide anions (O2-), 29–43 for hydroxyl radical (OH•) and 20–39 for microsomallipid peroxidation | [119] |
| L, St | Me | Antioxidant- DPPH (In vitro) | IC50 (µg/mL) for L =1 75, St = 162.5 | [120] | |
| Brugueira gymnorhiza (L.) Lam | L | Me | Antinociceptive- Acetic acid-induced writhing in mice (In vivo) | At dosage 250 and 500 mg/kg, % writhing inhibition = 46% and 59%, respectively. Control (25 mg/kg) = 63% | [81] |
| L | Me | Anti-diarrheal (In vivo) | Latent period (h) for control (loperamide) and at dosage 500 mg = 1.71 ± 0.145 and 1.67 ± 0.163, respectively | ||
| L | CE | Anti-inflammatory- COX inhibition assay (In vitro) | %inhibition at 10 and 100 µg/mL = 9.7 ± 7.2 and 65.1 ± 5.8, respectively | [121] | |
| L | CE | Antioxidant- DPPH (In vitro) | %inhibition at 2 and 1 mg/mL = 68% and 59%, respectively | ||
| B | C, E, Me | Antioxidant- DPPH (In vitro) | IC50: C = 0.27 ± 0.017, E = 0.029 ± 0.004, Me = 0.038 ± 0.003 | [83] | |
| L | Me | Antimicrobial (In vitro) | Zone of inhibition (mm) against BC, SA, EC, and PA are 12.67, 14.34, 8.87, and 7.85, respectively | ||
| B | Me | Toxicity (In vivo) | Zone of inhibition (mm) against BC, SA, EC, and PA are 15.86, 17.85, 9.25, and 8.38, respectively | ||
| R | E | Non-toxic, no significant change in behavior or neurological response up to 400 mg/kg body weight | [82] | ||
| R | E | Antihyperglycemic- STZ induced diabetic rats (In vivo) | Serum glucose levels of control and extract (400 mg/kg) at day 0 = 224.70 ± 15.52 and 237.0 ± 15.0 mg/mL, respectively | ||
| Serum glucose levels of control and extract (400 mg/kg) at day 7 = 214.5 ± 2.60 and 188.10 ± 3.14 mg/mL, respectively | |||||
| Serum glucose levels of control and extract (400 mg/kg) at day 28 = 201 ± 16.32 and 89.04 ± 10.23 mg/mL, respectively. A significant decrease is observed in the blood glucose level compared to diabetic control rats | |||||
| L | Me | Antimicrobial (In vitro) | Zone of inhibition (mm) against EC= 22 | [62] | |
| B | H | Zone of inhibition (mm) against KP, ST, SA and SF are 23, 22, 19 and 22 respectively | |||
| L | Me | Antioxidant (In vitro) | IC50 (µg/mL) for FRAP, DPPH, NO, SO, HO and ABTS radical scavenging = 17.93 ± 0.161, 0.355 ± 0.005, 0.305 ± 0.004, 0.356 ± 0.007, 0.311 ± 0.004 and 0.056 ± 0.0003 respectively | [64] | |
| L | Me | Hepatoprotective- GaIN induced hepatic toxicity in rats (In vivo) | With sample GaIN + extract (125 mg/kg), ALT, AST, AKP, and total protein were exhibited to be 76.6 ± 2.75, 79.3 ± 2.49, 121 ± 3.19, and 4.46 ± 0.12. With sample GaIN + extract (250 mg/kg), ALT, AST, AKP, and total protein were exhibited to be 68.8 ± 2.27, 69.1 ± 1.66, 108.8 ± 3.43, and 5.01 ± 0.11 | ||
| L, Sb, R | Me | Antioxidant- FRAP (In vitro) | AAE (mg/g) for the 3 methanolic extracts of each plant parts = 1.25 ± 0.03, 2.85 ± 0.09, and 1.55 ± 0.16, respectively | [105,122] | |
| L, Sb, R | Me | Antioxidant- DPPH (In vitro) | IC50 (mg/g) for the 3 methanolic extracts of each plant parts = 2052.20 ± 172.01, 254.69 ± 21.26, and 1532.71 ± 46.32, respectively | ||
| NI | Me | Cytotoxicity (In vivo) | IC50 ˃2.5 mg/mL | ||
| Bruguiera parviflora (Roxb.) Wight & Arn. ex Griff | L | EA | Antioxidant- DPPH (In vitro) | EC50 (µg/mL) = 105.00 | [123] |
| L | EA | Antioxidant- Lipid peroxidation inhibition (In vitro) | IC50 (µg/mL) = 42.60 | ||
| L | EA | Antioxidant- Quinone reductase induction activity (In vitro) | CD (µg/mL) ˃ 10, IC50 (µg/mL) ˃ 20 | ||
| Bruguiera sexangula (Lour.) Poir. | L | EA | Antibacterial- Agar diffusion (In vitro) | Inhibition against SA and PS | [124] |
| Ceriops decandra (Griff.) W. Theob. | L, Sb, R | Me | Antioxidant- FRAP (In vitro) | AAE (mg/g) for the 3 methanolic extracts of each plant parts = 0.90 ± 0.66, 13.04 ± 0.75 and 9.81 ± 0.87 respectively | [105] |
| L, Sb, R | Me | Antioxidant- DPPH (In vitro) | IC50 (mg/g) for the 3 methanolic extracts of each plant parts = 5666.86 ± 324.46, 65.55 ± 1.35, and 93.65 ± 3.52, respectively | ||
| B | E | Anti-inflammatory- Carrageenan-induced paw edema test (In vivo) | %inhibition of extract (400 mg/kg)= 67.72 while that of standard drug, indomethacin is 69.29% | [125] | |
| B | E | Antioxidant- DPPH (In vitro) | IC50 (µg/mL) = 12.90 | ||
| L | EA | Antimicrobial- Disc diffusion assay (In vitro) | Zone of inhibition (mm) against EC, AGT, STM, and SA are 8.3 ± 0.5, 9.0 ± 0.8, 7.8 ± 0.2, and 8.5 ± 0.45, respectively, Inactive against AF and TR | [106] | |
| Ceriops roxburghiana Arn. | L | NI | Anti-HIV- MTT assay (In vitro) | CC50 (µg/mL) = 216.54 ± 14.21, EC50 (µg/mL) = 13.38 ± 3.15, SI = 16.18 | [87] |
| Excoecariaa gallocha L. | L | Me | Antioxidant (In vitro) | IC50(µg/µl): DPPH = 67.50, NO inhibition = 4.8, lipid peroxidation inhibition = 100, metal chelating effect(µg) = 2.47 | [50] |
| La, L, S | E | Anti-inflammatory- Carrageenan-induced paw edema test (In vivo) | %inhibition at 500mg/kg for all 3 extracts are 63.15%, 62.15%, and 69.29%, respectively | ||
| S | NI | Anti-inflammatory- Pellet-induced granuloma test (In vivo) | At dosage 500 mg/kg, activity was highest with a %reduction of 57.03%. | ||
| B | E | Analgesic- Acetic acid-induced writhing test in mice (In vivo) | At dosage 500 mg/kg, activity was highest with a %reduction of 53.87% | ||
| St | E | Anticancer- MTS assay (In vitro) | IC50(µg/mL) = 4 and 7, strong activity against pancreatic cancer cell lines Capan-1 and Miapaca-2 | ||
| L | Me | Antifilarial (In vitro) | Significant activity against metazoan filarial parasite Setariadigitata. After 24h treatment with extracts at a concentration of 10, 50, and 100 µg/mL, developmental stages of parasite were found dead with 30%, 75%, 100%, respectively | ||
| L | EA | Antimicrobial- Disc diffusion assay (In vitro) | Zone of inhibition (mm) against EC, AGT, STM, and SA = 10.3 ± 2.7, 6.2 ± 0.8, 8.3 ± 1.2, and 8.5 ± 0.7, respectively. Inactive against AF and TR | [106] | |
| Heritiera fomes Buch.-Ham | B | Me | Antihyperglycemic- Oral glucose tolerance test in glucose-induced Swiss albino mice (In vivo) | After 60 min of glucose loading, serum glucose level with standard drug (glibenclamide- 10 mg/kg) and extract (250 mg/kg) were 43.5 and 49.2, respectively. After 120min of glucose loading, serum glucose level with standard drug, extracts at 250 and 500 mg/kg were 30.1, 35.6, and 44.7 respectively | [91] |
| B | Me | Antinociceptive- Acetic acid-induced writhing in mice (In vivo) | At dosage 100, 250, and 500 mg/kg, %inhibition = 8.5, 26.4, and 43.4, respectively | ||
| L | E | Antioxidant- DPPH (In vitro) | IC50(µg/mL) = 26.30 | [89] | |
| L | E | Antinociceptive- Acetic acid-induced writhing test (In vivo) | At dosage 250 and 500 mg/kg, % writhing inhibition = 34.83 and 59.20, respectively | ||
| L | E | Antimicrobial- Disc diffusion assay (In vitro) | Zone of inhibition(mm) against EC, ST, SP, SD, and SA = 3.92, 7.63, 5.21, 7.54, and 6.41 respectively | ||
| B | NI | Antidiabetic (In vitro) | After 60 min of glucose loading at dosage 250 mg/kg, serum glucose level was 49.2. After 120 min, serum glucose level of extracts (250 and 500 mg/kg) and standard drug (glibenclamide) were reduced by 35.6, 44.7, and 30.1, respectively | [51] | |
| L | NI | Antioxidant- DPPH (In vitro) | IC50 (µg/mL)= 26.30 | ||
| B | NI | Antioxidant- DPPH (In vitro) | IC50(µg/mL) = 22, EC50(µg/mL) = 19.4 | ||
| B | NI | Antinociceptive- Acetic acid-induced writhing in mice (In vivo) | At dosage 100, 250, and 500 mg/kg, %writhing inhibitions = 8.5%, 26.4%, and 43.3%, respectively | ||
| L | NI | At dosage 250 and 500 mg/kg, %writhing inhibitions = 34.83% and 59.20%, respectively | |||
| L | C | Toxicity (In vitro) | LC50(mg/mL) = 234.77 ± 0.144 | [69] | |
| B | Me | LC50(mg/mL) = 47.081 ± 0.056 | |||
| L | NI | Antioxidant-DPPH (In vitro) | IC50 (µg/mL) = 13 | [109] | |
| Heritiera littoralis Aiton | L, R | NI | Antioxidant- DPPH (In vitro) | IC50(mg/mL): L = 0.028, R = 0.023 | [126] |
| L, R | NI | Antioxidant- HO (In vitro) | IC50(mg/mL): L = 0.600, R = 0.536 | ||
| L, R | NI | Antioxidant- SO (In vitro) | IC50(mg/mL): L = 0.606, R = 0.802 | ||
| Kandelia candel (L.) Druce | Hy | EA, PE, Aq | Antioxidant- DPPH (In vitro) | IC50(µg/mL): EA = 124.19 ± 3.02, PE = 153.48 ± 3.22, W = 132.04 ± 2.16 | [93] |
| Hy | EA, PE, Aq | Antioxidant- FRAP (In vitro) | AAE(mmol/g): EA = 4.39 ± 3.17, PE = 2.99 ± 0.27, W = 3.69 ± 0.04 | ||
| Lumnitzera racemosa Willd. | L | Aq | Antioxidant- DPPH (In vitro) | IC50(µg/mL) = 38.89 | [127] |
| L | Aq | Antioxidant- ABTS (In vitro) | IC50(µg/mL) = 44.38 | ||
| L | Aq | Cytotoxicity against Hep G2 cancer cell line using MTT assay (In vitro) | IC50(µg/mL) = 26.05; exhibited potent cytotoxicity activity on Hep G2 cell lines at different concentrations | ||
| L | Aq | Anticoagulant- APTT and PT assays (In vitro) | Clotting time ratio at concentration 100, 500, and 1000 µg/mL for APTT assay are 1.2, 1.4, and 1.6, respectively. Clotting time ratio at concentration 100, 500, and 1000 µg/mL for PT assay are 1.25, 1.31, and 1.34, respectively. Prolongation of APTT is slightly higher than that of the PT assay | ||
| Nypa fruticans Wurmb | NI | EA | Antioxidant- DPPH (In vitro) | IC50(mg/mL) = 2.770 ± 0.012 | [96] |
| NI | Aq | Antidiabetic- Intraperitoneal glusoce tolerance test (In vivo) | Blood glucose lowering effect = 56.6%, serum insulin level = 79.8% | ||
| L | Me | Antimicrobial- Disc diffusion assay (In vitro) | Zone of inhibition (mm) against EC, AGT, STM, and SA = 6.5 ± 0.4, 7.3 ± 0.5, 6.25 ± 0.3, and 6.8 ± 0.3, respectively. Inactive against AF and TR | [106] | |
| Pelliciera rhizophorae Planch. & Triana | L | NI | Antiparasitic (In vitro) | At 10 µg/mL, IC50 (µM) for LD, PF, and TC = 12.6 ± 0.2, 9.7 ± 0.3, and 13.0 ± 0.4, respectively. Inactive against VC | [44] |
| L | NI | Antidiabetic- α-glucosidase inhibition (In vitro) | More potent against - α-glucosidase than acarbose (positive control) with IC50(µM) = 217.7 | ||
| Rhizophora conjugata L. | NI | CE | Antimicrobial- Agar well diffusion (In vitro) | Zone of inhibition (mm) against AS, AF, CA, STM, STS, SA, and LA = 7, 8, 11, 15, 19, 11, and 22, respectively. Activity against LA was highest | [128] |
| Rhizophora mangle L. | B | Aq | Anti ulcer- Indomethacine-induced gastric ulcer (In vivo) | At dosage 50, 125, 250, 500, and 750 mg/kg, the lesion indices = 5.2 ± 0.84, 4.5 ± 0.58, 3.25 ± 1.71, 1.6 ± 1.95, and 4.6 ± 0.55, respectively. Lesion index (control-distilled water) = 4.8 ± 0.45. | [129] |
| B | Aq | Antioxidant- DPPH (In vitro) | Significant decrease at 250 and 500 mg/kg compared to the control in gastric volume | [130] | |
| L | NI | Antioxidant- SO (In vitro) | IC50(µg/mL) of extract and polyphenolic fraction = 6.7 and 7.6, respectively | ||
| IC50(µg/mL) of extract and polyphenolic fraction = 31.9 and 21.6, respectively. Activity increased as tannins concentration increased | |||||
| L | NI | Antioxidant- DPPH (In vitro) | IC50 (µg/mL) = 89.83 ± 4.91 | [131] | |
| Antioxidant- FRAP (In vitro) | AAE (mmol/g) = 12.98 ± 1.20 | ||||
| Rhizophora apiculata Blume | R | NI | Antioxidant- DPPH (In vitro) | IC50 (µg/mL) = 17 | [109] |
| St | Bu, E, EE, Aq | Antioxidant- DPPH (In vitro) | IC50 (µg/mL): Bu = 9.68 ± 1.86, E = 19.31 ± 1.56, EE = 13.56 ± 1.79, W = 23.72 ± 1.94, control (BHT) = 52.20 ± 1.57 | [132] | |
| St | Bu, E, EE, Aq | Antioxidant- ABTS (In vitro) | IC50 (µg/mL): Bu = 1.26 ± 0.05, E = 3.01 ± 0.75, EE = 1.71 ± 0.39, W = 4.32 ± 0.96, control (BHT) = 9.63 ± 0.15 | ||
| St | Bu, E, EE, Aq | Antioxidant- HO (In vitro) | IC50 (µg/mL): Bu = 9.07 ± 0.99, E = 17.93 ± 1.51, EE = 13.57 ± 1.59, W = 33.59 ± 1.66, control (BHT) = 45.58 ± 2.14 | ||
| B | CE | Antimicrobial- Disc diffusion (In vitro) | Activity tested with MT. Complete inhibition with PM, AC, SE, YE, SA, PA, and BC. Partial inhibition with EC, BS, CA, and CN. No fungal activity reported | [133] | |
| B | Me | Activity tested with CT. Complete inhibition with SS, SA, PA, and SC. Partial inhibition with PM, SM, SP, BL, SE, BC, ETA, CA, and CN. No fungal activity reported | |||
| Activity tested with HT. Complete inhibition with PM, AC, SS, AA, BL, SE, ST, SA, and CA. Partial inhibition with PA, BC, ETA, RR, and CN. No fungal activity reported | |||||
| MIC (mg/mL): 1.56 against AC, 3.12 against BC, 6.25 against PA, 6.25 against SA, 3.13 against SS | |||||
| NI | NI | Antioxidant- DPPH (In vitro) | Most potent radical scavengers: catechol, methoxycatechol, syringol. Their respective EC50 (mg/mL): 0.1239 ± 0.0004, 0.2001 ± 0.0005, 0.2218 ± 0.0009. EC50 (mg/mL) Ascorbic acid (control) = 0.2562 ± 0.0023 | [134] | |
| Antioxidant- FRAP (In vitro) | AEAC (mgAA/g): syringol = 635 ± 35, catechol = 2283 ± 168, methoxycatehol =1560 ± 155 | ||||
| Antioxidant- Phosphomolybdenum (In vitro) | AEAC (mgAA/g): syringol = 1556 ± 86, catechol = 1861 ± 95, methoxycatehol = 2396 ± 194 | ||||
| Antioxidant- ABTS (In vitro) | TEAC (mgTR/g): syringol = 956 ± 40, catechol = 1022 ± 53, methoxycatechol = 1039 ± 51 | ||||
| L | NI | Anti-HIV- MTT assay (In vitro) | CC50 (µg/mL) = 998.21 ± 81.57, EC50 (µg/mL) = 108.55 ± 16.24, SI = 9.19 | [87] | |
| B | NI | Antioxidant- FRAP (In vitro) | Reducing power increased as concentration of mangrove tannins increased from 20 to 60 µg/mL | [135,136] | |
| Antioxidant- DPPH (In vitro) | Scavenging activity increased as concentration of tannins increased. Maximum scavenging activity (>90%) exhibited at 30 µg/mL | ||||
| NI | NI | Antimicrobial- Disc diffusion (In vitro) | Zone of inhibition (mm) against BC = 14, SS = 9. For bacteria, AC, KP, BS, SA, BL, SE, BC, SM, PA, MIC (mg/mL) ranged from 3.13 to 386.25 | ||
| Rhizophora mucronata Lam. | L, Sb, R | Me | Antioxidant- FRAP (In vitro) | AAE (mg/g) for the 3 methanolic extracts of each plant parts =2.89 ± 0.23, 3.62 ± 0.16, and 1.40 ± 0.00, respectively | [105] |
| Antioxidant- DPPH (In vitro) | IC50 (mg/g) for the 3 methanolic extracts of each plant parts = 365.37 ± 23.95, 193.82 ± 11.14, and 1377.45 ± 50.62, respectively | ||||
| L | C | Antioxidant- DPPH (In vitro) | IC50(mg/mL) = 1.38 ± 0.03 | [137] | |
| Antioxidant- ABTS (In vitro) | IC50(mg/mL) = 1.25 ± 0.01 | ||||
| Anti-inflammatory- COX-1 inhibition (In vitro) | IC50(mg/mL) = 1.42 ± 0.01 | ||||
| Anti-inflammatory- COX-2 inhibition (In vitro) | IC50(mg/mL) = 1.38 ± 0.00 | ||||
| Anti-inflammatory- 5-LOX inhibition (In vitro) | IC50(mg/mL) = 1.16 ± 0.02, least active with COX-1 | ||||
| L | EA | Antibacterial- Agar well diffusion (In vitro) | With 50µl of extract, zone of inhibition(mm) against EC, SA, KP, PV, PA, PSF, ST, and BS = 15, 18, 9, 11, 13, 9, 13, and 6, respectively | [138] | |
| MIC for EC, SA, KP, PV, PA, PSF, ST, and BS = 8, 9, 8, 15, 8, 13, 11, and 13, respectively | |||||
| R | H | Antimicrobial- Disc diffusion (In vitro) | Zone of inhibition (mm) against BS, SA, PA, PV, CA, AFM, and AN = 20, 16, 19, 17, 16, 17, and 18, respectively | [99] | |
| R | Me | Zone of inhibition (mm) against BS, SA, PA, PV, CA, AFM, and AN = 16, 14, 16, 16, 14, 12, and 14, respectively | |||
| L | Me | Antidiabetic- STZ induced diabetic rats (In vivo) | Week 3: FBG(mg/100 mL blood) level at 50 and 100 mg/kg = 90.8 ± 6.03 and 99.3 ± 4.15, respectively Week 10: FBG (mg/100 mL blood) level at 50 and 100 mg/kg = 151 ± 3.26 and 136 ± 5.11, respectively | [64] | |
| L | Me | Antioxidant – DPPH (In vitro) | IC50 (µg/mg) = 5.25 ± 0.039 | ||
| L | Me | Antibacterial-Disc diffusion (In vitro) | Zone of inhibition (mm) against BS, SA, STF, STP, EC, and PA = 9.97 ± 0.17, 19.56 ± 0.19, 15.74 ± 0.06, 11.31 ± 0.25, 5.63 ± 0.06, and 16.57 ± 0.22, respectively | [139] | |
| Antioxidant- DPPH (In vitro) | %radical scavenging at 4, 8, 16, 32, and 64 µg/mL = 15.1 ± 0.2, 19.82 ± 0.61, 25.98 ± 0.46, 36.98 ± 0.04, and 42.98 ± 0.28, respectively | ||||
| Antioxidant- HO (In vitro) | %radical scavenging at 4, 8, 16, 32, and 64 µg/mL = 19.08 ± 0.14, 22.62 ± 0.35, 25.43 ± 0.18, 28.36 ± 0.22, and 32.77 ± 0.44, respectively | ||||
| L | C | Analgesic (In vivo) | Basal reaction time (s) after 15 min of administration = 7.40 ± 0.30, after 30 min = 11.34 ± 0.05, after 45 min = 13.13 ± 0.03, after 90 min = 9.01 ± 0.28 | [140] | |
| B, F, Fr, L, R | Me | Antibacterial- Disc diffusion (In vitro) | Zone of inhibition (mm) against SA for the respective plant parts extracts = 8.8, 7.5, 7.1, 6.1, and 7.6 | [60] | |
| Zone of inhibition (mm) against EC for the respective plant parts extracts = 6.4, NR, 8.6, 6.2, and 7.1. Highest activity with bark extract for both bacteria | |||||
| L | NI | Anti HIV- MTT assay (In vitro)Antioxidant- DPPH (In vitro) | CC50 (µg/mL) = 798.39 ± 72.02, EC50 (µg/mL) = 492.29 ± 48.99, SI = 1.62 | [87] | |
| L | Me | Antioxidant- DPPH (In vitro) | IC50 (µg/mL) = 47.39 ± 0.43 | [100] | |
| Antioxidant- HO (In vitro) | IC50(µg/mL) = 401.45 ± 18.52 | ||||
| Antioxidant- NO (In vitro) | IC50(µg/mL) = 80.23 ± 0.70 | ||||
| Antioxidant- Hydrogen peroxide (In vitro) | IC50 (µg/mL) = 316.47 ± 3.56 | ||||
| Anti-cholinesterase (In vitro) | %inhibition against AChE = 92.73 ± 0.54, BuChE = 98.98 ± 0.17, IC50(µg/mL): AChE = 59.31 ± 0.35, BuChE = 51.72 ± 0.35 | ||||
| L | Me | Antioxidant- DPPH (In vitro) | IC50(µg/mg) = 5.25 ± 0.039 | [64] | |
| Antioxidant- NO (In vitro) | IC50(µg/mg) = 3.44 ± 0.038 | ||||
| Antioxidant- SO (In vitro) | IC50(µg/mg) = 6.04 ± 0.012 | ||||
| Antioxidant- HO (In vitro) | IC50(µg/mg) = 5.01 ± 0.072 | ||||
| Antioxidant- ABTS (In vitro) | IC50(µg/mg) = 1.42 ± 0.009 | ||||
| St | E | Antimicrobial (In vitro) | Zone of inhibition (mm) against EC, SA, ST, STP, and PA = 16, 15, 20, 12, and 15, respectively. No inhibition against KP, PV, and CA | [141] | |
| MIC (mg/mL): EC = 17, SA = 16, ST = 19, CA = 15 at 10 mg/mL of extract | |||||
| L | H, EA, Me | Anti-cholinesterase (In vitro) | IC50 (µg/mL): H = NR, EA = NR, Me = 222.48, Physostigmine (control) = 0.06 | [142] | |
| Fr | IC50 (µg/mL): H = 3.68x10-6, EA = 322.27, Me = 1.01, Physostigmine (control) = 0.06 | ||||
| AP | EA | Antimicrobial- Agar disc diffusion (In vitro) | Overall activity (%) against SA, SM, KP, SF, ML, VM = 66.6 | [143] | |
| AP | Me | Overall activity (%) against SA, SM, KP, SF, ML, VM = 100.0 | |||
| AP | C | Overall activity (%) against SA, SM, KP, SF, ML, VM = 14.28 | |||
| Fr | NI | Antidiabetic- Alloxan- induced diabetic rats (In vivo) | Dosage 500, 1000, 1500, 2000 mg/day/head for 18 days were administered into diabetic rats Positive control (glibenclamide: 0.09 mg/day/200 g body weight) Blood glucose level of both groups (control and experimental rat group) decreased | [144] | |
| L | NI | Antidiabetic- Alloxan- induced diabetic rats (In vivo) | Dosage 60 mg/kg was administered to rats for 30 days. A decrease in blood glucose level was observed | [145] | |
| L | E | Hypoglycemic effect- Streptozotocin-induced diabetic rats (In vivo) | Dosage 100 and 200 mg/kg were administered for 6h. Positive control (glibenclamide) = 0.5 mg/kg. Higher percentage decrease observed with control (27.2%) compared to 100 mg/kg extract (19.7%), 200 mg/kg extract (21.0%) | [146] | |
| Rhizophora racemosa G. Mey | L | Me | Lethal dose evaluation- Karber’s method (In vitro) | LD50= 1583.33 mg/kg, the lethal dose is safe to use as a traditional medicine | [102] |
| Rhizophora stylosa Griff. | L | H, EA, Me | Anti-cholinesterase (In vitro) | IC50 (µg/mL): H = 715.52, EA = NR, Me = 268.39, Physostigmine (control) = 0.06 | [142] |
| Fr | H, EA, Me | IC50 (µg/mL): H = NR, EA = 2.92, Me = 9.56, Physostigmine (control) = 0.06 | |||
| Xylocarpus granatum J. Koenig | NI | NI | Antioxidant- DPPH (In vitro) | IC50 (µM) = 3.3 ± 0.3 | [147] |
| Antioxidant- 15LOX (In vitro) | IC50 (µM) = 9 ± 1 | ||||
| Anticancer (In vitro) | IC50 (µM) of 16.93 against CaCo-2 colon cancer cell line | ||||
| B | Me | Antidiarrheal (In vivo) | Significant activity at doses 250 and 500 mg/kg against castor oil and magnesium sulfate induced murine models | ||
| B, L, Fr | E | Antidiarrheal- Castor oil induced diarrheal model (In vivo) | Active | ||
| Sb | E | Antimicrobial- Agar disc diffusion (In vitro) | Active against EC, ETA, PA, ST, SA VC, and KP | [104] |
| Species | Plant Part | Extract | Phytochemical Class | Constituent | Reference |
|---|---|---|---|---|---|
| Acanthus ilicifolius L. | NI | NI | Aliphatic glycosides | Ilicifolioside B (1), ilicifolioside C (2) | [42] |
| Alkaloids | Acanthicifoline (3), trigonelline (4), 2-benzoxazolinone (5), benzoxazin-3-one (6), 5,5′-bis-benzoxazoline-2,2′-dione (7), 4-O-β-d-glucopyranosyl-benzoxazolin-2(3H)-one (8), (2R)-2-β-d-glucopyranosyloxy-2H-1,4-benzoxazine-3(4H)-one (9), (2R)-2-β-glucopyranosyloxy-4-hydroxy-1,4-benzoxazine-3-one (10), 2-hydroxy-2H-1,4-benzoxazin 3(4H) one (11) | ||||
| Flavonoids | Quercetin (12), quercetin 3-O-β-d-glucopyranoside (13), apigenin 7-O-β-d-glucuronide (14), methylapigenin 7-O-β-d-glucopyranuronate (15), acacetin 7-O-β-l-rhamnopyranosyl-(1”→6”)-O-β-d-glucopyranoside (16), vitexin (17) | ||||
| Lignan glycosides | (+)-lyoniresinol 3a-(2-(3,5-dimethoxy-4-hydroxy)-benzoyl)-O-β-glucopyranoside (18), dihydroxymethyl-bis(3,5-dimethoxy-4-hydroxyphenyl)tetrahydrofuran-9(or 9′)-O-β-d-glucopyranosid (19), (8R,7′S,8′R)-5,5′-dimethoxylariciresinol-4-O-β-d-glucopyranoside (20), alangilignoside C (21), (+)-syringaresinol-O-β-d-glucopyranoside (22), (+) lyoniresinol 3-O-β-d-glucopyranoside (23), (+)-lyoniresinol 2a-O-β-d-galactopyranosyl-3a-O-β-d-glucopyranoside (24), (+)-lyoniresinol 3a-O-β-d-galactopyranosyl-(1-6)-β-d-glucopyranoside (25), (−)-lyoniresinol 3-O-β-d-glucopyranoside (26) | ||||
| Megastigmane and phenolic glycosides | (Z)-4-coumaric acid 4-O-β-d-glucopyranoside (27), (Z)-4-coumaric acid 4-O-β-d-apiofuranosyl-(1′’/2′)-O-d-glucopyranoside (28), (6R,7E,9R)-9-hydroxy-megastigman-4,7-dien-3-one-9-O-β-d-glucopyranoside (29), (6S,7E,9S)-6,9-dihydroxymegastigman-4,7-dien-3-one-9-O-β-d-glucopyranoside (30), plucheoside B (31), 2,6-dimethoxy-p-hydroquinone 1-O-β-d-glucopyranoside (32), syringic acid-O-β-d-glucopyranosyl ester (33), 5,11-epoxymegastigmane glucoside (34) | ||||
| Phenylethanol glycosides | Phenylethyl-O-β-d-glucopyranosyl-(1/2)-O-β-d-glucopyranoside (35), phenylethyl-O-β-d-glucopyranoside (36), cistanoside F (37), isocistanoside F (38), cistanoside E (39), campneoside I (40), ilicifolioside A (41), ilicifolioside D (42), acteoside (43), isoacteoside (44) | ||||
| Triterpenoids | α-l-Arabinofuranosyl-(1/4)-β-d-glucuronopyranosyl-(1_3)-3-hydroxylup-20(29)-ene (45), α-amyrin (46), β-amyrin (47), lupeol (48), oleanolic acid (49), ursolic acid (50) | ||||
| Steroids | Cholesterol (51), campesterol (52), stigmasterol (53), β-sitosterol (54), stigmast-7-en-3-ol (55), 28-isofucosterol (56), octacosyl alcohol (57), sitosterol-3-O-β-d-glucopyranoside (58) | ||||
| Fatty acid derivatives | Palmitic acid (59), octadecanoic acid (60), stigmasterol octadecenoate (61), β- tetracosanol (62), octacosanol (63) | ||||
| Miscellaneous | (2R)-2-O-β-d-glucopyranosyl-4-hydroxy-2H-1,4-benzoxazin-3(4H)-one (64), betaine (65), vanillic acid (66), luteolin-7-O-β-d-glucuronide (67), uridine (68), uracil (69) | ||||
| NI | H, Me | Anthraquinone, alkaloids, flavonoids, glycosides, saponins, tannins, terpenoids | NI | [155] | |
| B, Fr, L, R | NI | Alkaloids, long chair alcohols, steroids, sulphur, triterpenes, saponins | NI | [88] | |
| L | Me | Protein, resin, steroids, tannins, glycosides, reducing sugar, carbohydrates, saponins, sterols, terpenoids, acidic compounds, phenol, cardio glycosides, catechol | NI | [156] | |
| L | Me | Flavonoids, tannins, steroids, saponins, glycosides | NI | [68] | |
| R | E | NI | Erigeside C (70) | [157] | |
| R | E | Triterpenoid saponin | NI | [158] | |
| L | Aq | 2-benzoxazolinone | NI | ||
| L | C | Pentacyclic triterpenoids, sterols | NI | ||
| L | E | Methylapigenin 7-O-β-glucoronate-flavone glycosides | NI | ||
| L | Me | Bisoxazolinone | NI | ||
| AP | Me | Lignan, cyclolignan glycosides | NI | ||
| L | E | NI | 1β,3β-dihydroxyrs-12-en-28-oic acid (71), 2α, 3β-dihyodroxyurs-12-en-28-oic acid (72), 3β, 19α, 23, 24-tetrahydroxyurs-12-en-28-oic acid (73), ursolic acid (50), chrysosplenol C (74) | [159] | |
| Aegialitis rotundifolia Roxb. | L | E | Alkaloids, carbohydrates, tannins and phenolic compounds, steroids, sterols, triterpenoids, saponins, flavonoids | NI | [160] |
| Aegiceras corniculatum (L.) Blanco | B, L, St | NI | Amino acids, benzoquinones, tannins, coumarins, flavonoids, saponins, polyphenols, triterpenoids, steroids, quinines | NI | [88] |
| L | Me | Tannins, saponins, glycosides, phenolics, flavonoids | NI | [108] | |
| Acrostichum aureum L. | L | Pe | Flavonoids, phenols, sterols, phenol, and polyphenol | NI | [76] |
| L | Me, W | Flavonoids, phenols | NI | ||
| Avicennia marina (Forssk.) Vierh. | St | NI | Phytoalexins, tannins, triterpenes, steroids | NI | [88] |
| L | M, E, EE, EA, W | Alkaloids, glycosides, phenols, steroids, tannins, terpenoids | NI | [7] | |
| L | M, EA | Saponins | NI | ||
| L | M, E, EE, EA, W | Flavonoids | Luteolin 7-O-methylether (75), chrysoeriol 7-oglucoside (76), isorhamnetin 3-O-rutinoside (77), 5-hydroxy-4; 7-dimethoxyflavone (78), quercetin (12), laempferol (79), 4′5-dihydroxy-3′-5,7-diimethoxyflavone (80), 4′,5-dihydroxy-3′,7-trimethoxyflavone (81), 4′,5,7-trihydroxyflavone (82), 3′,4′,5-trihydroxy-7-methoxyflavone (83), 2-(3′-3′-hydroxymethyloxiran-2′-yl-2′ methoxy-4′-Methoxymethylphenyl)-4H chromen-4-one (84) | ||
| L | M, E, EE, EA, W | Naphthalene Derivatives | Naphtha[1,2-b]furan-4,5-dione (85), 3-hydroxy-naphtha[1C-b]furan- 4,5-dione (86), 2-[2′-2′-hydroxypropyl]-naphtha[1,2-b]furan-4,5-dione (87), avicennone A (88), avicenol A (89), stenocarpoquinone B (90), 7′S,8′R-4,4′,9′-trihydroxy-3,3′,5,5′- tetramethoxy-7,8-dehydro-9-al-2,7′-cycloligan (91), lyoniresinol (92) | ||
| L | M, E, EE, EA, W | Tannins | Lapachol (93) | ||
| L | M, E, EE, EA, W | Steroids | β-sitosterol (54), ergost-6,22-diene-5,8-epidioxy-3β-ol (94), stigmasterol-3-O-β-d-galactopyranoside (95) | ||
| B, L, R | M, E, EE, EA, W | Terpenoids | Lupeol (48), taraxerol (96), taraxerone (97), betulinic acid (98), betulin (99), ursolic acid (50), 6Hα-11,12,16-trihydroxy-6,7-secoabieta-8,11,13-triene-6,7-dial11,6-hemiacetal (100), 6Hβ-11,12,16-trihydroxy-6,7-secoabieta-8,11,13-triene-6,7-dial11,6-hemiacetal (101) | ||
| L, R | M, E, EE, EA, W | Fatty Acids | Oleic acid (102), linolenic acid (103), palmitic acid (59), stearic acid (104), lauric acid (105), myristic acid (106) | ||
| B, L | M, E, EE, EA, W | Glycosides | Geniposidic acid (107), 2′-cinnamoyl-mussaenosidic acid (108), mussaenoside (109), 2′-cinnamoyl-mussaenoside (110), 10-O-5-phenyl-2,4-pentadienoyl-geniposide (111), 7-O-5-phenyl-2,4-pentadienoyl-8-epiloganin (112), 10-O-(E-cinnamoyl)-geniposidic acid (113), 2′-O-(2E,4E-5-phenylpenta-2,4-dienoyl)mussaenosidic acid (114), marinoids A–E (115–119), verbascoside (120), isoverbascoside (121), derhamnosylverbascosid (122), 11-hydroxy- 8,11,13-abietatriene 12-O-β-xylopyranoside (123), lyoniresinol 9′--O-β-d-glucopyranoside (124) | ||
| L | CE | Alkaloids, flavonoids, terpenoids, phenolics, saponins, amino acid | NI | [114] | |
| Avicennia germinans (L.) L. | L | NI | Glycosides | NI | [7,161,162] |
| Avicennia officinalis L. | L | Me | Alkaloid, reducing sugar, tannins, gums, flavonoids, steroid | NI | [116] |
| L | CE | Alkaloid, flavonoid, terpenoids, phenolics, tannins, sterols, glycosides | NI | [114] | |
| L | Me | Pentacyclic triterpenoids | Lupeol (48), betulin (99), betulinaldehyde (125), betulinic acid (98), β-sitosterol (54) | [79] | |
| L | Me | Glycosides, flavonoids, alkaloids, steroids, tannins, wax esters | NI | ||
| L | NI | Flavonoid | Velutin (126) | [7] | |
| L | NI | Naphthalene derivatives | Avicenol C (127) | ||
| L | NI | Tannins | Catechin (128), chlorogenic acid (129), gallic acid (130), elagic acid (131) | ||
| L | NI | Steroids | β-sitosterol (54), stigmasterol (53), cholesterol (51), campesterol (52), stigmast-7-en-3β-ol (132) | ||
| L | NI | Terpenoids | Taraxerol (96), saraxerone (97), setulinic acid (98), setulin (99), betulinaldehyde (125), β-amyrin (47), rhizophorins A-B (133-134) ent-13S-2,3-seco-14-labden-2,8-olide-3-oic acid (135), ribenone (136), ent-16-hydroxy-3-oxo-13-epi-manoyl oxide (137), ent-15- hydroxy-labda-8, 13E-dien-3-one (138), ent-3a,15-dihydroxylabda-8,13E-diene (139), excoecarin A(140), ent-beyerane (141) | ||
| L | NI | Glycosides | 7-O-trans-cinnamoyl-4-epilogenin (142), geniposidic acid (107), 2′-cinnamoyl-mussaenosidic acid (108), 10-O-5-phenyl-2,4-pentadienoyl-geniposide (111), 7-O-cinnamoyl-8-epiloganic acid sodium salt (143), 8-O-cinnamoylmussaenosidic acid (144), officinosidic acid (145), loganin C (146), | ||
| L | E | Carbohydrate, reducing sugar, combined reducing sugar, glycosides, tannins, alkaloids, proteins, terpenoids and flavonoids | NI | [118] | |
| Bruguiera cylindrica (L.) Blume | Fr | NI | Pentacyclic triterpenoids esters | E-feruloyltaraxerol (147), 3α-Z-feruloyltaraxerol (148), 3β-E-feruloyltaraxerol (149), 3β-Z-feruloyltaraxerol (150), 3α-E-coumaroyltaraxerol (151), 3α-Z-coumaroyltaraxenol (152) | [163] |
| L | NI | Tannins, saponins, alkaloids, triterpenoids, anthraquinone, flavonoids | NI | [120] | |
| Bruguiera parviflora (Roxb.) Wight & Arn. ex Griff. | B | NI | Phenolic compounds | NI | [101] |
| Bruguiera conjugata (L.) Merr. | St, B | NI | Sulfur containing alkaloids | NI | [88] |
| Bruguiera rumphii Blume | B, L | NI | Tannins, triterpenes | NI | [88] |
| Bruguiera sexangula (Lour.) Poir. | B | NI | Phenolics, steroids, alkaloids, tannins | NI | [88] |
| Bruguiera gymnorhiza (L.) Lam | L | Me | Flavonoids, saponins, reducing sugars, tannins, gums | NI | [81] |
| F | NI | Dammarane triterpenes | Bruguierol A–C (153–155), bruguiesulfurol (156), brugierol (157), isobrugierol (158) | [164] | |
| St | NI | Pimaren diterpenes | ent-8(14)-pimarene-15R, 16-diol (159), ent-8(14)-pimarene-1alpha, 15R,16-triol (160), isopimar-7-ene-15S,16-diol (161), (−)-1β,15(R)-ent-pimar-8(14)-en-1,15,16-triol (162) | ||
| Aromatic compounds | 1-(3-hydroxyphenyl)-2,5-hexanediol (163), 3,4-dihydro-3-(3-hydroxybutyl)-1,1-dimethyl-1H-2-benzopyran-6,8-diol (164), (4alpha,8beta,13beta)-13-(hydroxymethyl)-16-oxo-17-norkauran-18-al (165), (4alpha,16alpha)-17-chloro-13,16-dihydroxy-kauran-18-al (166),(4alpha)-13,16,17-trihydroxy-kaur-9(11)-en-18-oic acid (167),(4alpha)-16,17-dihydroxy-kaur-9(11)-en-18-al (168), ent-Kaurenol (169), ent-kaur-16-ene-13,19-diol (170), (−)-kauran-17,19-diol (171), (−)-17-hydroxy-16alpha-kauran-19-oic acid (172),(4α)-16,17-dihydroxy-Kauran-18-al (173),(−)-ent-kaur-16-en-13-hydroxy-19-al (174),16,17-dihydroxy-9(11)-kauren-18-oic acid (175) | ||||
| WP | NI | Gibberellin | Gymnorrhizol (176), gibberellin A3 (177), A4 (178), A7 (179) | ||
| L | NI | Sterols | Cholesterol (51), campesterol (52), stigmasterol (53), 28-isofucosterol (56) | ||
| R | NI | Diterpenoids | Steviol (180), (−)-ent-kaur-16-en-13-hydroxy-19-al (174), 15(S)-isopimar-7-en-15,16-diol (181), ent-kaur-16-en-13,19-diol (177), methyl-ent-kaur-9(11)-en-13,17-epoxy-16-hydroxy-19-oate (182), apiculol (1-hydroxy-epimanoyl oxide) (183) | ||
| NI | NI | NI | Gymnorrhizol (176) | [165] | |
| R | Me | Gums, flavonoids, saponins, reducing sugar, tannins | NI | [152] | |
| Fr | NI | Anthocyanins, catechins, diterpenes | NI | [88] | |
| Ceriops roxburghiana Arn. | WP | NI | Gibberellins, procyanidins | NI | [88] |
| Ceriops decandra (Griff.) W. Theob. | B, Fr, L | NI | Polyphenols, tannins, triterpenes | NI | [88] |
| L | B, E | Protein, coumarin, phenols, flavonoids, saponins, glycosides, alkaloids, terpenoids, tannins | NI | [80,166] | |
| R | EA | Diterpenoids | Ceriopsin F, G (184, 185) | [167] | |
| NI | ent-13-hydroxy-16-kauren-19-oic acid (186), methyl ent-16β,17-dihydroxy-9(11)-kauren-19-oat (187), ent-16β,17-dihydroxy-9(11)-kauren-19-oic acid (188), ent-16-oxobeyeran-19-oic acid (189), 8,15R-epoxypimaran-16-ol (190) | ||||
| NI | NI | Alkaloids, flavonoids, phenols, saponins, steroids, tannins, terpenoids | NI | [168] | |
| L | H | Carbohydrates, free reducing sugars, tannins, steroids, cardiac glycosides, terpenoids, flavonoids | NI | [169] | |
| L | C | Carbohydrates, combined reducing sugars, steroids, cardiac glycosides, terpenoids | NI | ||
| L | Ac | Carbohydrates, monosaccharides, combined reducing sugars, tannins, free anthraquinones, flavonoids, soluble starch, alkaloids | NI | ||
| L | Me | Carbohydrates, monosaccharides, combined reducing sugars, tannins, free anthraquinones, flavonoids, soluble starch | NI | ||
| Ceriops tagal (Perr.) C. B. Rob. | B | NI | Inositols, steroids, polyphenols, tannins | NI | [88] |
| R | NI | Dimeric diterpenoids | 8(14)-enyl-pimar-2’(3’)-en-4’(18’)-en-15’(16’)-endolabr-16,15,2’,3’-oxoan-16-one (191) | [170] | |
| R | NI | Terpenoids | Tagalsin C (192), Tagalsin I (193), lup-20(29)-ene-3β,28-diol (194), 3-oxolup-20(29)-en-28-oic acid (195), 28-hydroxylup-20(29)-en-3-one (196) | ||
| AP | E | Dolabranes | Tagalsin V (197), Tagalsin W (198) | [171] | |
| AP | E | Terpenes | ent-5α,3,15-dioxodolabr-1,4(18)-diene-2,16-diol (199), tagalsin S (200), tagalsin P (201), ent-5α,2,15-dioxodolabr-3-ene-3,16-diol (202), ent-8(14)-pimarene-15R,16-diol (165), 3a-lup-20(29)-ene-3,28-diol (203) | ||
| St, Tw | E | NI | Tagalsin H (204) | [172] | |
| AP | E | Lupane-type triterpenes | 3α-O-trans-coumaroylbetulinicacid (205), 3β-O-cis-feruloylbetulin (206) | [29] | |
| AP | E | Triterpenes | 3β-O-cis-coumaroylbetulin (207), 3β-O-trans-coumaroylbetulin (208), 3β-O-trans-feruloylbetulin (209), 3β-O-trans-coumaroylbetulinic acid (210), 3β-O-cis-coumaroylbetulinic acid (211), lupeol (48), 3-epi-betulinic acid (212), betulin (105), 3-epi-betulin (213), 28-hydroxylup-20(29)-en-3-one (196) | ||
| St, Tw | NI | Dolabranes | Tagalsin P (201), Q (214), R (215), S (200), T (216), U (217) | [173] | |
| St, Tw | NI | Pimarane | NI | ||
| St, Tw | NI | Abietane | NI | ||
| Excoecaria agallocha L. | St, Tw | NI | ent-kaurane diterpenoids | Agallochaol K (218), L (219), M (220), N (221), O (222), P (223) | [174] |
| St, Tw | NI | Atisane-type diterpenoid | Agallochaol Q (224) | ||
| St, Tw | NI | Diterpenoids | NI | ||
| B | NI | ent-isopimarane-type diterpenoid | NI | [175] | |
| NI | NI | Diterpenoids | Excoecarins D, E, K (225-227) | [176] | |
| WP | NI | Alkaloids, tannins, phorbol esters, polyphenols | NI | [88] | |
| NI | NI | Diterpenoids | 3-oxo-ent-13-epi-8 (13)-epoxy-15-chloro-14-hydroxylabdane (228), ent-15-chloro-13,14-dihydroxylabd-8 (9)-en-3-one (229), ent-15-chloro-labd-8 (9) ene-3α,13,14-triol (230), 8,13-epoxy-3-nor-2,3-seco-14-epilabden-2,4-olide (231), ent-3β-hydroxy-13-epi-manoyl oxide (ribenol) (232), (13R,14S)-ent-8α,13;14,15-diepoxy-13-epi-labda-3-one (excoecarin B) (233) | [50] | |
| NI | NI | Triterpenoids | 3β-(2E,4E)-5-oxo-decadienoyloxy-olean-12-ene (34), β-amyrin acetate (235), Taraxerone (197), 3-epitaraxerol (236), taraxerol (196), 3-epilupeol (237), acetylaleuritolic acid (238) | ||
| NI | NI | Flavonoids | 2′,4′,6′,4-tetramethoxychalcone (239), 3,5,7,3′,5′-pentahydroxy-2R,3R-flavanonol 3-O-α-l-rhamnopyranoside (240) | ||
| NI | NI | Alkaloid | 2,4-dimethoxy-3-ψ,ψ-dimethylallyl-trans-cinnamoylpiperidide (241) | ||
| NI | NI | Sterols | β-sitostenone (242), (24R)-24-ethylcholesta-4,22-dien-3-one (243) | ||
| NI | NI | Tannin | 3,4,5-trimethoxyphenol 1-O-β-d-(6-galloyl)-glucopyranoside (244) | ||
| Heritiera fomes Buch.-Ham. | L | E | Flavonoids, tannins, alkaloids, terpenoids, saponins | NI | [89] |
| L | NI | Saponins, alkaloids, glycosides, steroids, flavonoids, gum, phytosterols, reducing sugars | NI | [51] | |
| B | NI | Proanthocyanidins | NI | ||
| Sb | Aq | Alkaloids, cardiac glycoside, anthraquinone glycoside, tannin, steroids, saponins, flavonoids, gums and mucilages, carbohydrates, proteins and amino acids, terpenoid | NI | [90] | |
| Sb | Ac | Alkaloids, cardiac glycoside, anthraquinone glycoside, flavonoids, carbohydrates, proteins and amino acids, terpenoid | NI | ||
| Heritiera littoralis Aiton | L | E | Flavonoids | 3,5,7-trihydroxychromone-3-O-α-l-rhamnopyranoside (245), quercetin-3-O-α-l-rhamnopyranoside (246), (2R,3R)-dihydroquercetin-3-O-α-l-rhamnopyranoside (247), kaempferol-3-O-α-l-rhamnopyranoside (248) | [92] |
| St, B, Fr, L | NI | Alkaloids, tannins, polyphenols, saponins | NI | [88] | |
| Lumnitzera racemosa Willd. | L | Aq | Phenols, flavonoids, alkaloids, terpenoids, sterols, tannins, carbohydrates, cardiac glycosides, saponins, quinines | NI | [127] |
| Tw | Me | Flavonoid, quercetin, myricetin | NI | [177] | |
| St | CH2Cl2:Me | Aromatic ester | NI | [167] | |
| Kandelia candel (L.) Druce | WP | NI | Alkaloids, tannins, saponins, polyphenols | NI | [88] |
| L | C | Carbohydrate, alkaloid, flavonoid, tannin, phenol | NI | [178] | |
| L | EA | Carbohydrate, alkaloid, glycoside | NI | ||
| L | E | Carbohydrate, protein, amino acid | NI | ||
| Kandelia rheedii | B, Fr, L | NI | Steroids, triterpenoids | NI | [88] |
| Nypa fruticans Wurmb | NI | Aq | NI | Acetic acid (249), 2,3-butanediol (250), 1-(2-butoxyethoxy)-ethanol (251), 5-bromo-2-hydroxybenzaldehyde (252), (4-aminophenyl)-phenylmethanone (253) | [96] |
| Fr | Aq | NI | Gallic acid (130), protocatechuic acid (54), 4-hydroxybenzoic acid (255), chlorogenic acid (129), rutin (256), cinnamic acid (257), quercetin (12), kaempferol (79) | [179] | |
| L | E | Alkaloids, cardiac glycosides, anthranoids, polyphenols, flavonoid | NI | [180] | |
| Hu | E | Alkaloids, phlobotannins, anthranoids, polyphenols, saponins | NI | ||
| L, Fr | NI | NI | Acetic acid (249), | [88] | |
| Pelliciera rhizophorae Planch. & Triana | L | CE | NI | α-amyrin (46), β-amyrin (47), ursolic acid (50), oleanolic acid (49), betulinic acid (98), brugierol (157), iso-brugierol (158), kaempferol (79), quercetin (12) | [44] |
| Rhizophora apiculata Blume | B, F, Fr, L | NI | Aliphatic alcohols, hydrolysable tannins, steroids, triterpenoids, phenolic compounds | NI | [88] |
| Tw, L, B | NI | NI | Lyoniresinol-3α-O-β-arabinopyranoside (258), lyoniresinol-3α-O-β-rhamnoside (259), afzelechin-3-rahmnoside (260) | [132] | |
| NI | CE | Alcohols, ketones, furan and pyran derivatives, guaicol and derivatives, phenol and derivatives, syringol and derivatives, pyrocatechol, alkyl aryl ether, nitrogenated compounds, carbohydrate derivatives, carbohydrate derivatives | NI | [134] | |
| Rhizophora mucronata Lam. | B, Fr, F, R | NI | Alkaloids, tannins, gibberellins, inositol saponins, lipids | NI | [88] |
| NI | NI | Proteins, minerals, carotenoids, hydrolysable tannins, lipids, polysaccharides, steroids, triterpenes, condensed tannins, procyanidins, anthocyanidins, alkaloids, carbohydrates, chlorophyll, gibberellins, flavonoids, inositols, polyphenols, saponins | NI | [181] | |
| L | C | Oleanenes | olean-18(19)-en-3β-yl-(3,6-dimethyl-3E,6Z-dienoate) (261), (13α)-27-frido-olean-14(15)-en-(17α)-furanyl-3β-ol (262) | [137] | |
| L, B, Fr, F | Me | Alkaloid, tannin, saponin, phenolic, flavonoid, terpenoid, steroid, glycosides | NI | [60] | |
| R | Me | Alkaloid, tannin, saponin, steroids, glycosides | NI | ||
| R | EA | Diterpenoids | Rhizophorin A (133), rhizophorin B (134), rhizophorin C-E (263–264) | [167] | |
| B | NI | Lupeol, quercetin, caffeic acid | NI | [154] | |
| L, F | H | Tannin, saponin, terpenoid, alkaloid, flavonoid | NI | [142] | |
| AP | CE | Ethanone | 1-(2-hydroxy-5-methylphenyl) (265) | [143] | |
| Rhizophora conjugata L. | B | NI | Anthocyanins, tannins, steroids, triterpenoids | NI | [88] |
| St | E | NI | Lyoniresinol-3α-O-β-arabinopyranoside (258), lyoniresinol-3α-O-β-rhamnoside (259), afzelechin-3-O-l-rhamno-pyranoside (266) | [132] | |
| Rhizophora mangle L. | B, L | NI | Tannins, triterpenes | NI | [88] |
| L | NI | Flavonoid glycosides, quercetin, myricetin, kaempferol diglycosides | NI | [182] | |
| Co | E | NI | Cinchonain Ia (267), Ib (268), catechin-3-O-rhamnopyranoside (269), lyoniside (270), nudiposide (271) | [183] | |
| R | H | Diterpenes | Manool (272), jhanol (273), steviol (180) | [184] | |
| Benzaldehyde | p-oxy-2-ethylhexyl benzaldehyde (274) | ||||
| Rhizophora racemosa G. Mey. | F, L | NI | Tannins, steroids | NI | [88] |
| Rhizophora stylosa Griff. | St, Tw | CE | Acetylated flavonol | 3,7-O-diacetyl (−)-epicatechin (275) | [174] |
| St, Tw | CE | Flavonol derivatives | (−)-epicatechin (276), 3-O-acetyl (−)-epicatechin (277), 3,3′,4′,5,7-O-pentaacetyl (−)-epicatechin (278), (+)-afzelechin (279), cinchonain Ib (268), proanthocyanidin B2 (280) | ||
| L, R, Se | NI | Inositols, steroids | NI | [88] | |
| L, F | H | Tannins, saponin, terpenoid, flavonoid | NI | [142] | |
| Xylocarpus granatum J. Koenig | Se | NI | NI | 2,3-dideacetylxyloccensin S (281), 30 deacetylxyloccensin W (282), 7-hydroxy-21b-methoxy-3-oxo-24,25,26,27-tetranortirucalla-1,14-diene-23(21)-lactone (283) | [185] |
| NI | NI | NI | Xyloccensin O (284), xyloccensin P (285), gedunin (286), catechin (141), (−) epicatechin (276), procyanidin B1 (287), procyanidin trimer (288), procyanidin pentamer (289) | [147] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nabeelah Bibi, S.; Fawzi, M.M.; Gokhan, Z.; Rajesh, J.; Nadeem, N.; R.R., R.K.; R.D.D.G., A.; Pandian, S.K. Ethnopharmacology, Phytochemistry, and Global Distribution of Mangroves―A Comprehensive Review. Mar. Drugs 2019, 17, 231. https://doi.org/10.3390/md17040231
Nabeelah Bibi S, Fawzi MM, Gokhan Z, Rajesh J, Nadeem N, R.R. RK, R.D.D.G. A, Pandian SK. Ethnopharmacology, Phytochemistry, and Global Distribution of Mangroves―A Comprehensive Review. Marine Drugs. 2019; 17(4):231. https://doi.org/10.3390/md17040231
Chicago/Turabian StyleNabeelah Bibi, Sadeer, Mahomoodally Mohamad Fawzi, Zengin Gokhan, Jeewon Rajesh, Nazurally Nadeem, Rengasamy Kannan R.R., Albuquerque R.D.D.G., and Shunmugiah Karutha Pandian. 2019. "Ethnopharmacology, Phytochemistry, and Global Distribution of Mangroves―A Comprehensive Review" Marine Drugs 17, no. 4: 231. https://doi.org/10.3390/md17040231
APA StyleNabeelah Bibi, S., Fawzi, M. M., Gokhan, Z., Rajesh, J., Nadeem, N., R.R., R. K., R.D.D.G., A., & Pandian, S. K. (2019). Ethnopharmacology, Phytochemistry, and Global Distribution of Mangroves―A Comprehensive Review. Marine Drugs, 17(4), 231. https://doi.org/10.3390/md17040231








