Divergolides T–W with Apoptosis-Inducing Activity from the Mangrove-Derived Actinomycete Streptomyces sp. KFD18
Abstract
1. Introduction
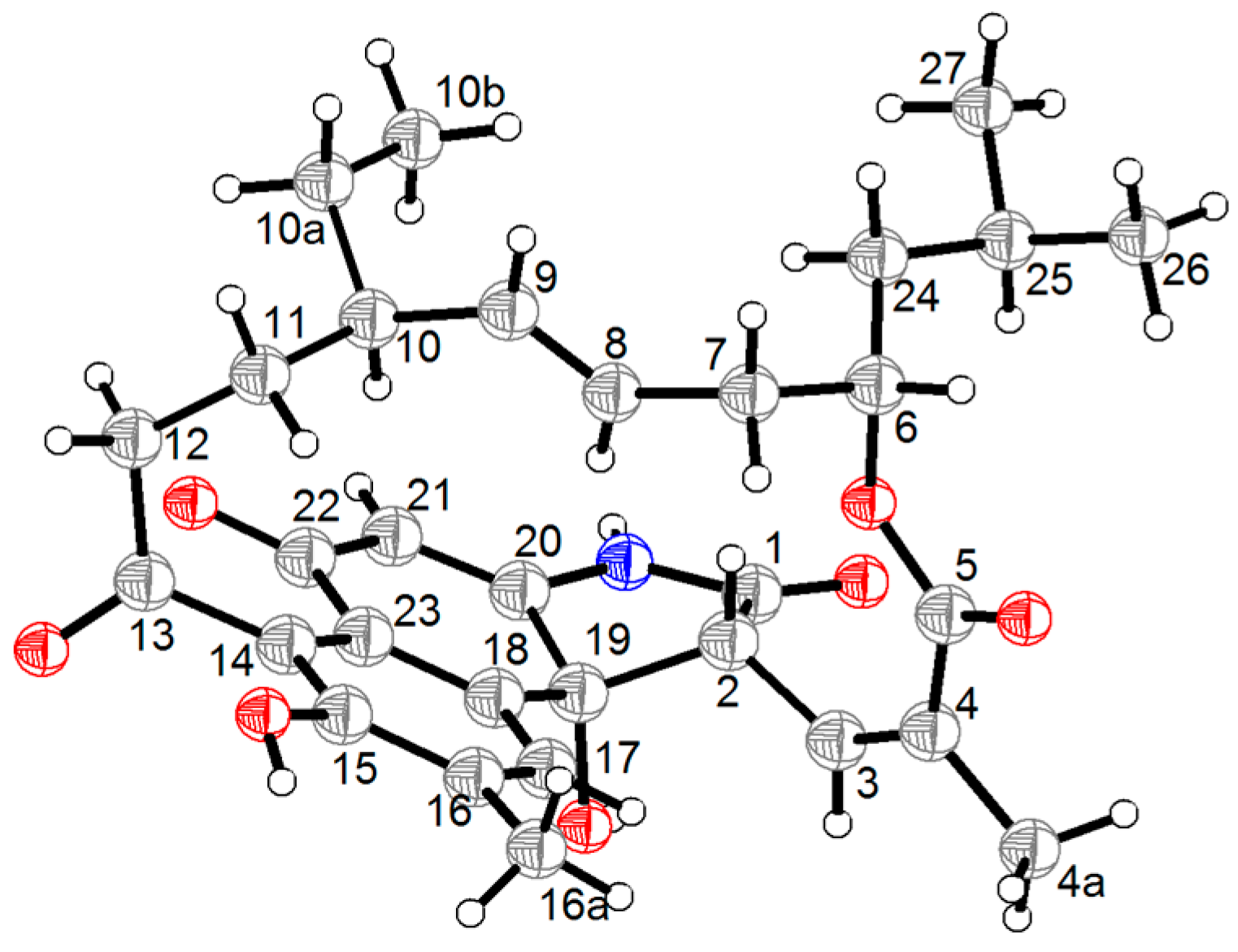
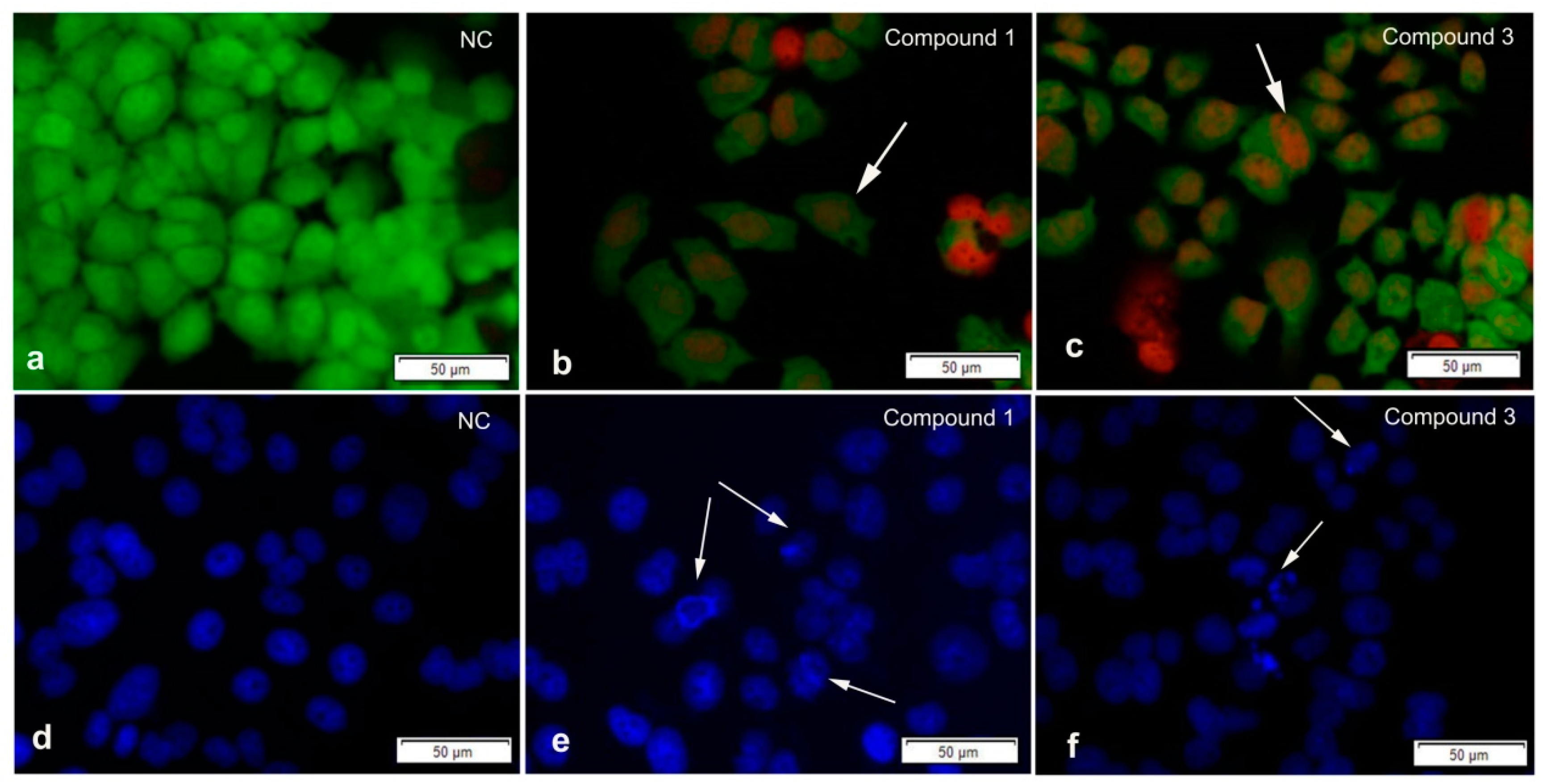
2. Results and Discussion
3. Experimental Section
3.1. General Experimental Procedure
3.2. Strain and Fermentation
3.3. Extraction and Isolation
3.4. Bioassays for Cytotoxic and Apoptosis-Inducing Activity
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Fukuyo, Y.; Hunt, C.R.; Horikoshi, N. Geldanamycin and its anti-cancer activities. Cancer Lett. 2010, 290, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Floss, H.G.; Yu, T.W. Rifamycin mode of action, resistance, and biosynthesis. Chem. Rev. 2005, 105, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Cassady, J.M.; Chan, K.K.; Floss, H.G.; Leistner, E. Recent developments in the maytansinoid antitumor agents. Chem. Pharm. Bull. 2004, 52, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Higashide, E.; Asai, M.; Ootsu, K.; Tanida, S.; Kozai, Y.; Hasegawa, T.; Kishi, T.; Sugino, Y.; Yoneda, M. Ansamitocin, a group of novel maytansinoid antibiotics with antitumour properties from Nocardia. Nature 1977, 270, 721–722. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Maier, A.; Fiebig, H.H.; Görls, H.; Lin, W.H.; Peschel, G.; Hertweck, C. Divergolides A–D from a mangrove endophyte reveal an unparalleled plasticity in ansa-macrolide biosynthesis. Angew. Chem. 2011, 123, 1668–1672. [Google Scholar] [CrossRef]
- Ding, L.; Franke, J.; Hertweck, C. Divergolide congeners illuminate alternative reaction channels for ansamycin diversification. Org. Biomol. Chem. 2015, 13, 1618–1623. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Baunach, M.; Ding, L.; Peng, H.; Franke, J.; Hertweck, C. Biosynthetic code for divergolide assembly in a bacterial mangrove endophyte. ChemBioChem 2014, 15, 1274–1279. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Li, S.; Guo, Z.; Sun, M.; Lu, C. Overexpression of div 8 increases the production and diversity of divergolides in Streptomyces sp. W112. RSC Adv. 2015, 5, 98209–98214. [Google Scholar] [CrossRef]
- Kong, F.D.; Ma, Q.Y.; Huang, S.Z.; Wang, P.; Wang, J.F.; Zhou, L.M.; Yuan, J.Z.; Dai, H.F.; Zhao, Y.X. Chrodrimanins K-N and related meroterpenoids from the fungus Penicillium sp. SCS-KFD09 isolated from a marine worm, Sipunculus nudus. J. Nat. Prod. 2017, 80, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.D.; Zhang, R.S.; Ma, Q.Y.; Xie, Q.Y.; Wang, P.; Chen, P.W.; Zhou, L.M.; Dai, H.F.; Luo, D.Q.; Zhao, Y.X. Chrodrimanins O–S from the fungus Penicillium sp. SCS-KFD09 isolated from a marine worm, Sipunculusnudus. Fitoterapia 2017, 122, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.D.; Huang, X.L.; Ma, Q.Y.; Xie, Q.Y.; Wang, P.; Chen, P.W.; Zhou, L.M.; Yuan, J.Z.; Dai, H.F.; Luo, D.Q. Helvolic acid derivatives with antibacterial activities against Streptococcus agalactiae from the marine-derived fungus Aspergillus fumigatus HNMF0047. J. Nat. Prod. 2018, 81, 1869–1876. [Google Scholar] [CrossRef] [PubMed]
- An, C.L.; Kong, F.D.; Ma, Q.Y.; Xie, Q.Y.; Yuan, J.Z.; Zhou, L.M.; Dai, H.F.; Yu, Z.F.; Zhao, Y.X. Chemical Constituents of the Marine-Derived Fungus Aspergillus sp. SCS-KFD66. Mar. Drugs 2018, 16, 468. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Li, Y.; Deng, J.; Li, S.; Shen, Y.; Wang, H.; Shen, Y. Hygrocins C–G, cytotoxic naphthoquinone ansamycins from gdmAI-disrupted Streptomyces sp. LZ35. J. Nat. Prod. 2013, 76, 2175–2179. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.S.; Novak, R.; Murray, P.J.; Eischen, C.M.; Susin, S.A.; Kroemer, G.; Halle, A.; Weber, J.R.; Tuomanen, E.I.; Cleveland, J.L. Apoptosis-inducing factor mediates microglial and neuronal apoptosis caused by pneumococcus. J. Infect. Dis. 2001, 184, 1300–1309. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.F.; Tang, Y.M.; Xu, X.J.; Li, S.S.; Zhang, J.Y. 10-Hydroxycamptothecin induces apoptosis in human neuroblastoma SMS-KCNR cells through p53, cytochrome c and caspase 3 pathways. Neoplasma 2016, 63, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Liang, F.; Chen, B.; Sun, Z.Y.; Xue, T.D.; Yang, R.L.; Luo, D.Q. Identification of demethylincisterol A3 as a selective inhibitor of protein tyrosine phosphatase Shp2. Eur. J. Pharmacol. 2017, 795, 124–133. [Google Scholar] [CrossRef] [PubMed]
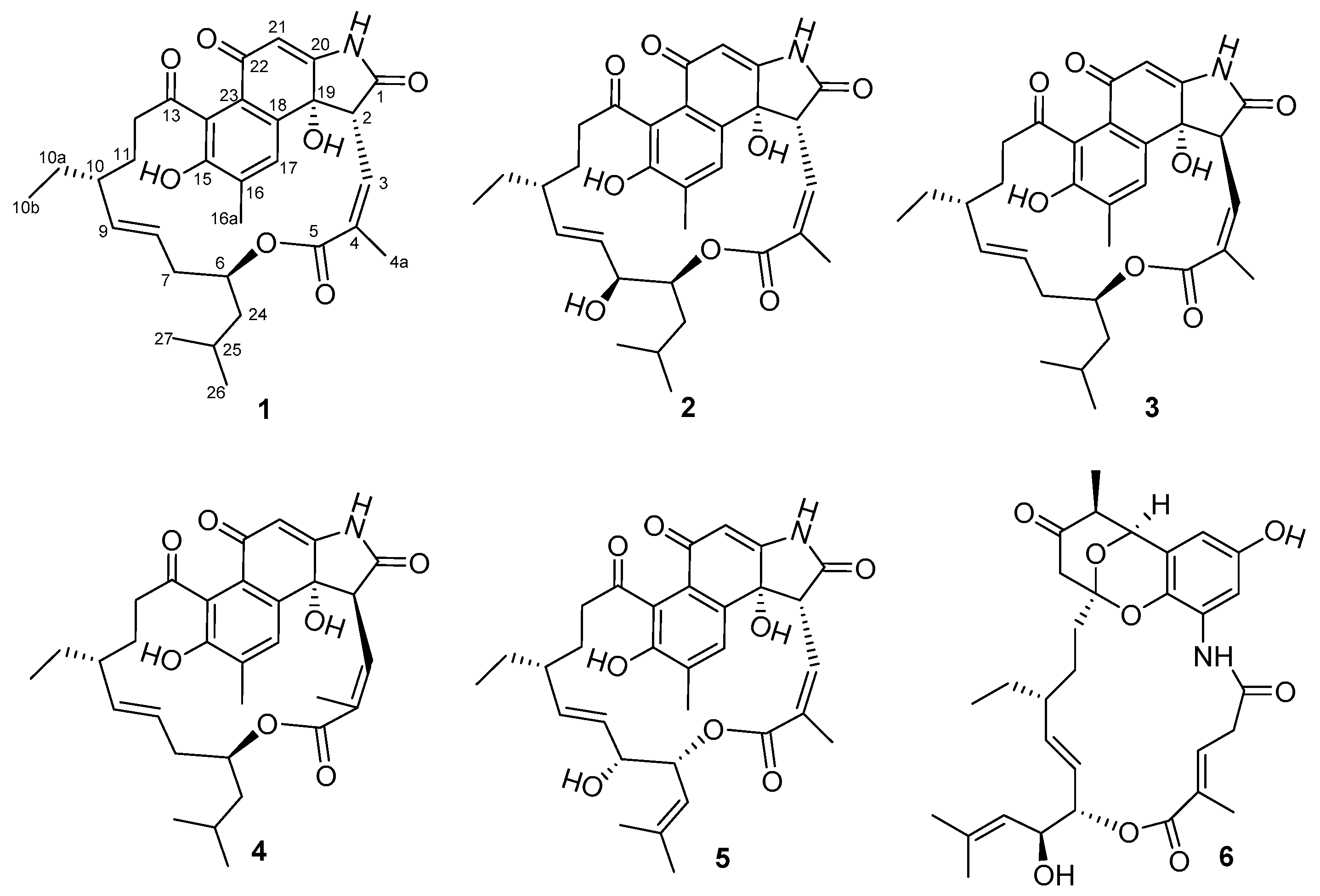
 ) and HMBC (→) correlations of 1–4.
) and HMBC (→) correlations of 1–4.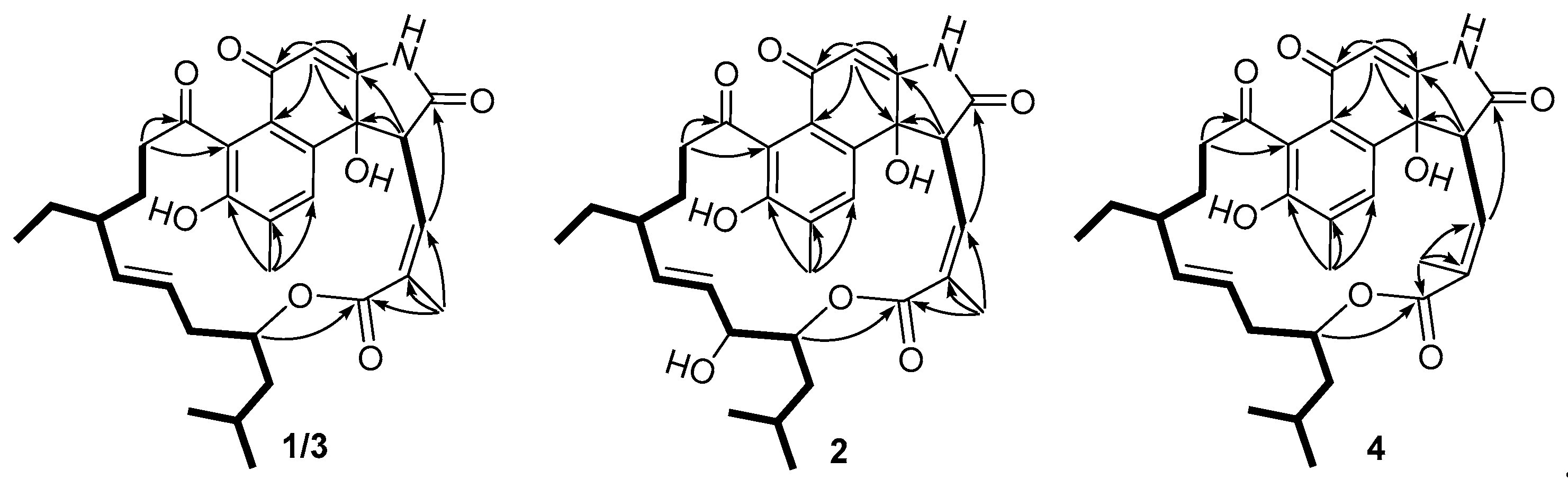

| Position | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| δC | δC | δC | δC | |
| 1 | 177.2, C | 177.1, C | 177.2, C | 176.8, C |
| 2 | 55.2, CH | 55.3, CH | 55.8, CH | 55.3, CH |
| 3 | 131.7, CH | 132.9, CH | 126.7, CH | 132.5 CH |
| 4 | 136.8, C | 136.1, C | 138.4, C | 135.0, C |
| 4a | 22.0, CH3 | 22.1, CH3 | 21.4, CH3 | 13.5, CH3 |
| 5 | 168.0, C | 167.7, C | 169.5, C | 167.9, C |
| 6 | 74.6, CH | 76.7, CH | 74.5, CH | 74.0, CH |
| 7 | 36.1, CH2 | 70.5, CH | 36.6, CH2 | 36.3, CH2 |
| 8 | 125.1, CH | 128.2, CH | 126.3, CH | 125.3, CH |
| 9 | 139.6, CH | 136.1, CH | 138.8, CH | 138.6, CH |
| 10 | 46.0, CH | 45.9, CH | 44.2, CH | 43.2, CH |
| 10a | 26.9, CH2 | 27.0, CH2 | 29.3, CH2 | 25.6, CH2 |
| 10b | 13.2, CH3 | 13.2, CH3 | 12.7, CH3 | 11.1, CH3 |
| 11 | 31.7, CH2 | 31.5, CH2 | 34.8, CH2 | 31.6, CH2 |
| 12 | 40.5, CH2 | 40.5, CH2 | 42.5, CH2 | 42.0, CH2 |
| 13 | 212.4, C | 212.4, C | 212.1, C | 212.1, C |
| 14 | 130.1, C | 130.2, C | 127.8, C | 130.6, C |
| 15 | 153.5, C | 153.5, C | 153.4, C | 152.9, C |
| 16 | 132.6, C | 132.9, C | 133.1, C | 133.4, C |
| 16a | 17.0, CH3 | 17.0, CH3 | 17.0, CH3 | 16.9, CH3 |
| 17 | 130.8, CH | 130.8, CH | 132.8, CH | 131.9, CH |
| 18 | 134.3, C | 136.1, C | 134.3, C | 135.0, C |
| 19 | 73.7, C | 73.8, C | 73.6, C | 75.2, C |
| 20 | 164.8, C | 164.7, C | 164.6, C | 164.6, C |
| 21 | 103.9, CH | 104.0, CH | 103.4, CH | 104.3, CH |
| 22 | 185.4, C | 185.4, C | 185.7, C | 185.7, C |
| 23 | 129.8, C | 129.9, C | 130.4, C | 130.6, C |
| 24 | 42.6, CH2 | 38.4, CH2 | 41.5, CH2 | 41.9, CH2 |
| 25 | 25.5, CH | 25.7, CH | 25.3, CH | 25.6, CH |
| 26 | 22.7, CH3 | 22.2, CH3 | 22.1, CH3 | 22.4, CH3 |
| 27 | 23.0, CH3 | 23.8, CH3 | 23.7, CH3 | 23.6, CH3 |
| Position | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| δH (J in Hz) | δH (J in Hz) | δH (J in Hz) | δH (J in Hz) | |
| 2 | 4.74, d (10.9) | 4.84, d (10.6) | 4.09, d (10.9) | 4.06, d (8.4) |
| 3 | 6.60, dq (10.9, 1.6) | 6.67, dq (10.6, 1.6) | 6.36, dq (10.8, 1.6) | 5.89, dq (8.4, 1.6) |
| 4a | 2.20, d (1.6) | 2.21, d (1.6) | 2.17, d (1.6) | 2.08, d (1.0) |
| 6 | 5.05, m | 4.99, m | 5.04, m | 4.87, m |
| 7 | 1.96, m | 3.90, ddd (2.74, 2.6, 2.6) | 2.15, m | 2.25, m |
| 2.15, m | 2.15, m | 2.14, m | ||
| 8 | 3.93, ddd (15.3, 10.2, 3.6) | 4.06, dd (15.6, 2.8) | 3.78, ddd (15.6, 6.0, 6.0) | 4.77, ddd (15.6, 9.1, 4.9) |
| 9 | 5.01, dd (15.3, 9.3) | 5.24, dd (15.6, 9.3) | 4.87, dd (15.6, 9.4) | 5.24, dd (15.6, 7.7) |
| 10 | 1.32, overlap | 1.37, overlap | 1.46, overlap | 1.78, m |
| 10a | 0.89, m | 0.92, m | 1.02, m | 1.43, overlap |
| 0.89, m | 1.49, overlap | 1.34, overlap | 1.18, overlap | |
| 10b | 0.66, t (7.4) | 0.65, t (7.4) | 0.73, t (7.4) | 0.77, t (7.5) |
| 11 | 1.35, m | 1.37, overlap | 1.68, m | 1.29, overlap |
| 1.46, overlap | 1.49, overlap | 1.25, m | 1.57, m | |
| 12 | 2.61, m | 2.62, m | 2.64, ddd (14.0, 11.3, 2.8) | 2.46, m |
| 2.90, m | 2.99, m | 2.46, ddd (14.0, 7.4, 2.9) | 2.77, m | |
| 16a | 2.22, s | 2.21, s | 2.30, s | 2.31, s |
| 17 | 7.41, s | 7.38, s | 7.57, s | 7.28, s |
| 21 | 5.82, s | 5.82, s | 5.80, s | 5.85, s |
| 24 | 1.13, m | 1.14, m | 1.15, m | 1.20, overlap |
| 1.32, overlap | 1.31, overlap | 1.32, overlap | 1.30, overlap | |
| 25 | 1.46, overlap | 1.49, overlap | 1.47, overlap | 1.45, overlap |
| 26 | 0.81 d (6.6) | 0.83 d (6.6) | 0.88 d (6.5) | 0.88 d (6.6) |
| 27 | 0.81, d (6.6) | 0.79, d (6.6) | 0.82, d (6.5) | 0.83, d (6.6) |
| Compound | IC50 (μM) | |||
|---|---|---|---|---|
| SGC-7901 | K562 | Hela | A549 | |
| 1 | 2.8 | 6.6 | 9.6 | 14.9 |
| 2 | 9.8 | 9.0 | >50 | 24.7 |
| 3 | 4.7 | 7.6 | 14.1 | 20.9 |
| 4 | 20.9 | 16.3 | 29.5 | 33.2 |
| 5 | >50 | >50 | >50 | >50 |
| 6 | >50 | >50 | >50 | >50 |
| Imatinib | 86.8 | 0.2 | 18.8 | 45.6 |
| Adriamycin | 6.9 | 10.7 | 11.4 | 5.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, L.-M.; Kong, F.-D.; Xie, Q.-Y.; Ma, Q.-Y.; Hu, Z.; Zhao, Y.-X.; Luo, D.-Q. Divergolides T–W with Apoptosis-Inducing Activity from the Mangrove-Derived Actinomycete Streptomyces sp. KFD18. Mar. Drugs 2019, 17, 219. https://doi.org/10.3390/md17040219
Zhou L-M, Kong F-D, Xie Q-Y, Ma Q-Y, Hu Z, Zhao Y-X, Luo D-Q. Divergolides T–W with Apoptosis-Inducing Activity from the Mangrove-Derived Actinomycete Streptomyces sp. KFD18. Marine Drugs. 2019; 17(4):219. https://doi.org/10.3390/md17040219
Chicago/Turabian StyleZhou, Li-Man, Fan-Dong Kong, Qing-Yi Xie, Qing-Yun Ma, Zhong Hu, You-Xing Zhao, and Du-Qiang Luo. 2019. "Divergolides T–W with Apoptosis-Inducing Activity from the Mangrove-Derived Actinomycete Streptomyces sp. KFD18" Marine Drugs 17, no. 4: 219. https://doi.org/10.3390/md17040219
APA StyleZhou, L.-M., Kong, F.-D., Xie, Q.-Y., Ma, Q.-Y., Hu, Z., Zhao, Y.-X., & Luo, D.-Q. (2019). Divergolides T–W with Apoptosis-Inducing Activity from the Mangrove-Derived Actinomycete Streptomyces sp. KFD18. Marine Drugs, 17(4), 219. https://doi.org/10.3390/md17040219




