Brevenal, a Marine Natural Product, is Anti-Inflammatory and an Immunomodulator of Macrophage and Lung Epithelial Cells
Abstract
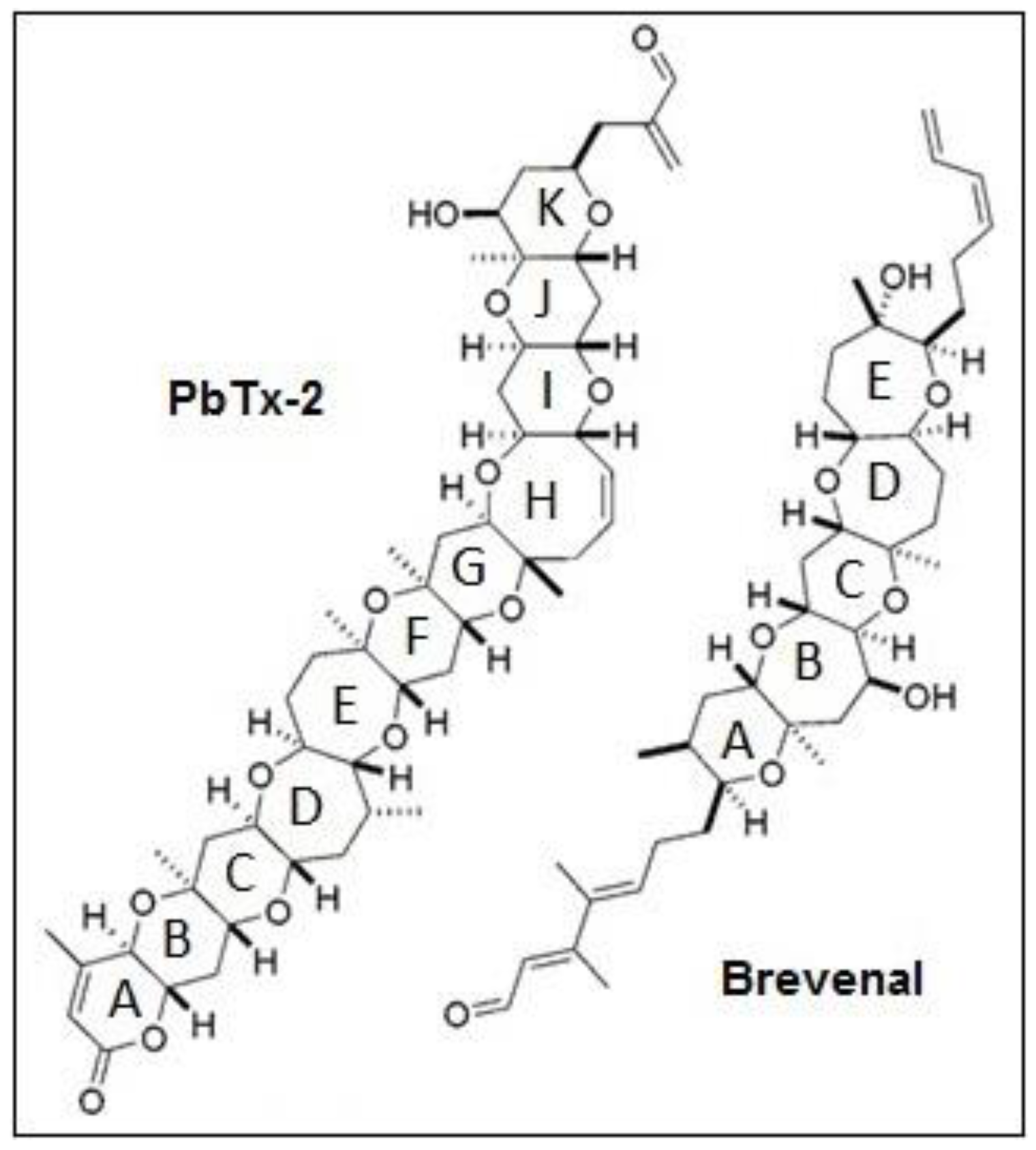
1. Introduction
2. Results
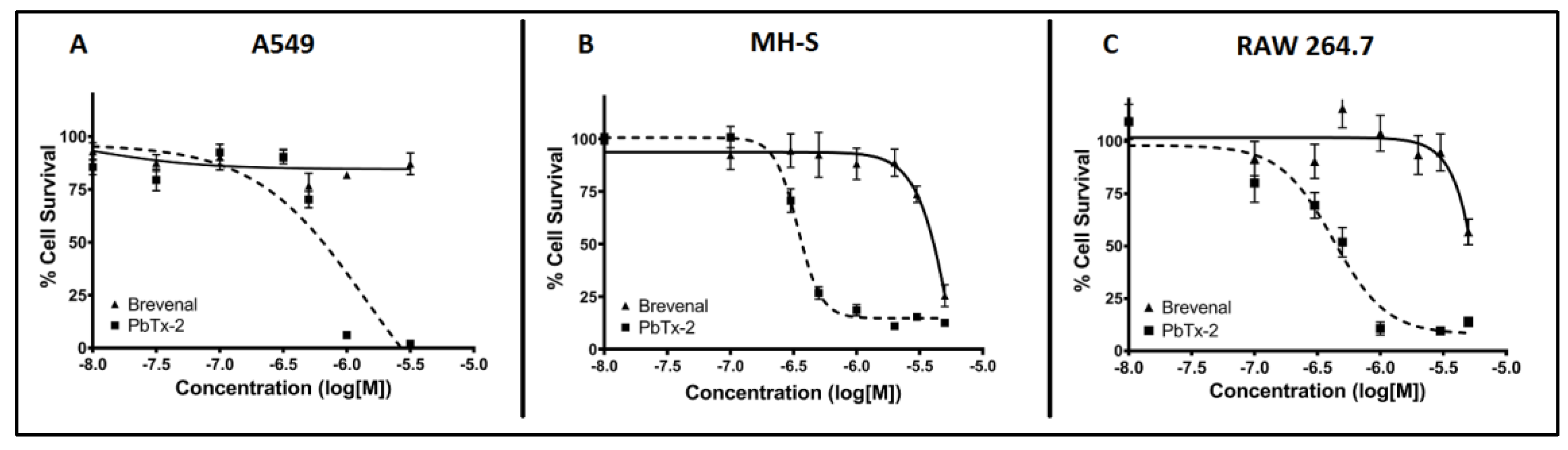
2.1. Brevenal is not Toxic for A549 Epithelial Lung Cells, MH-S Lung Macrophages, or RAW 264.7 Macrophages at Micromolar Concentrations
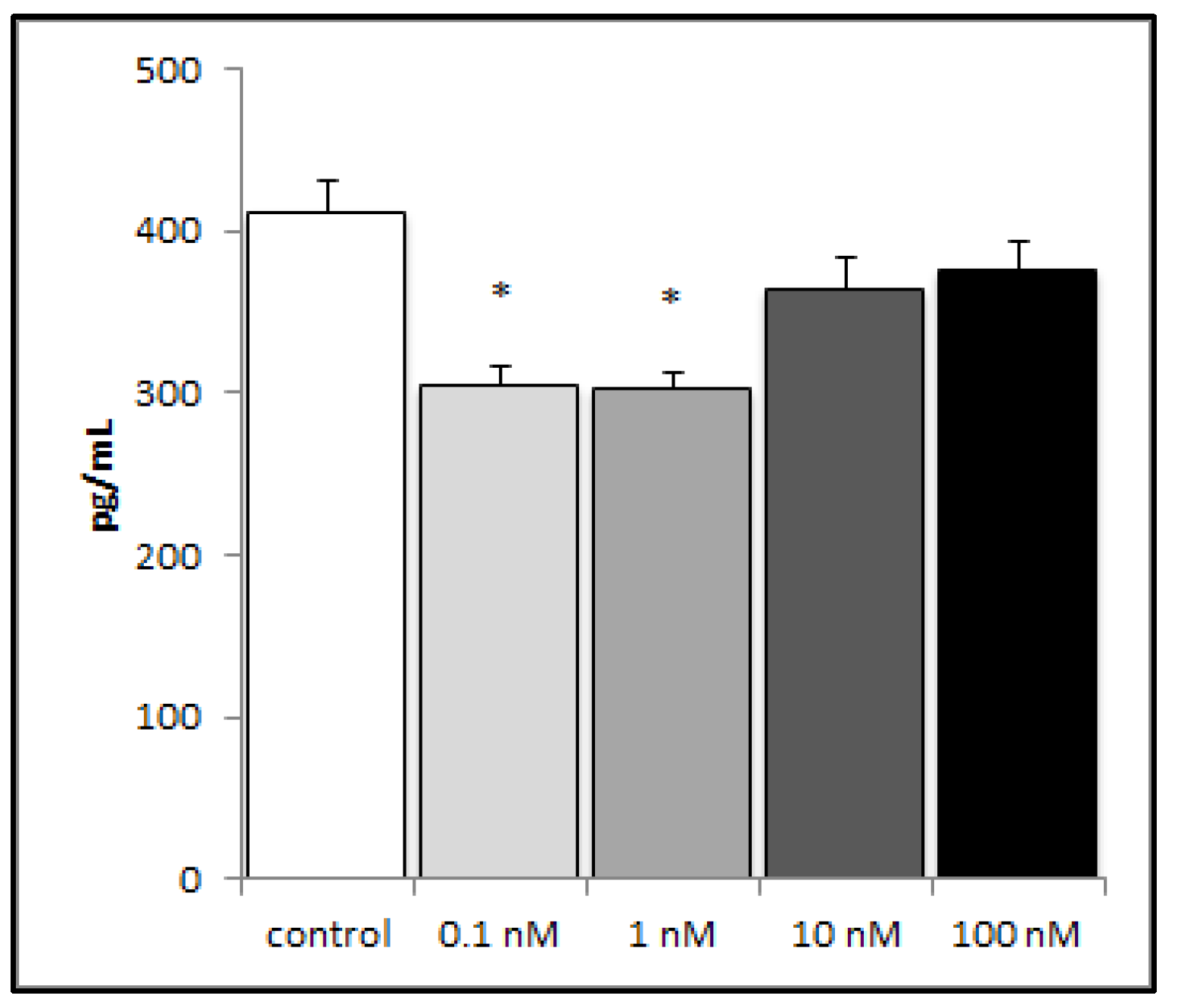
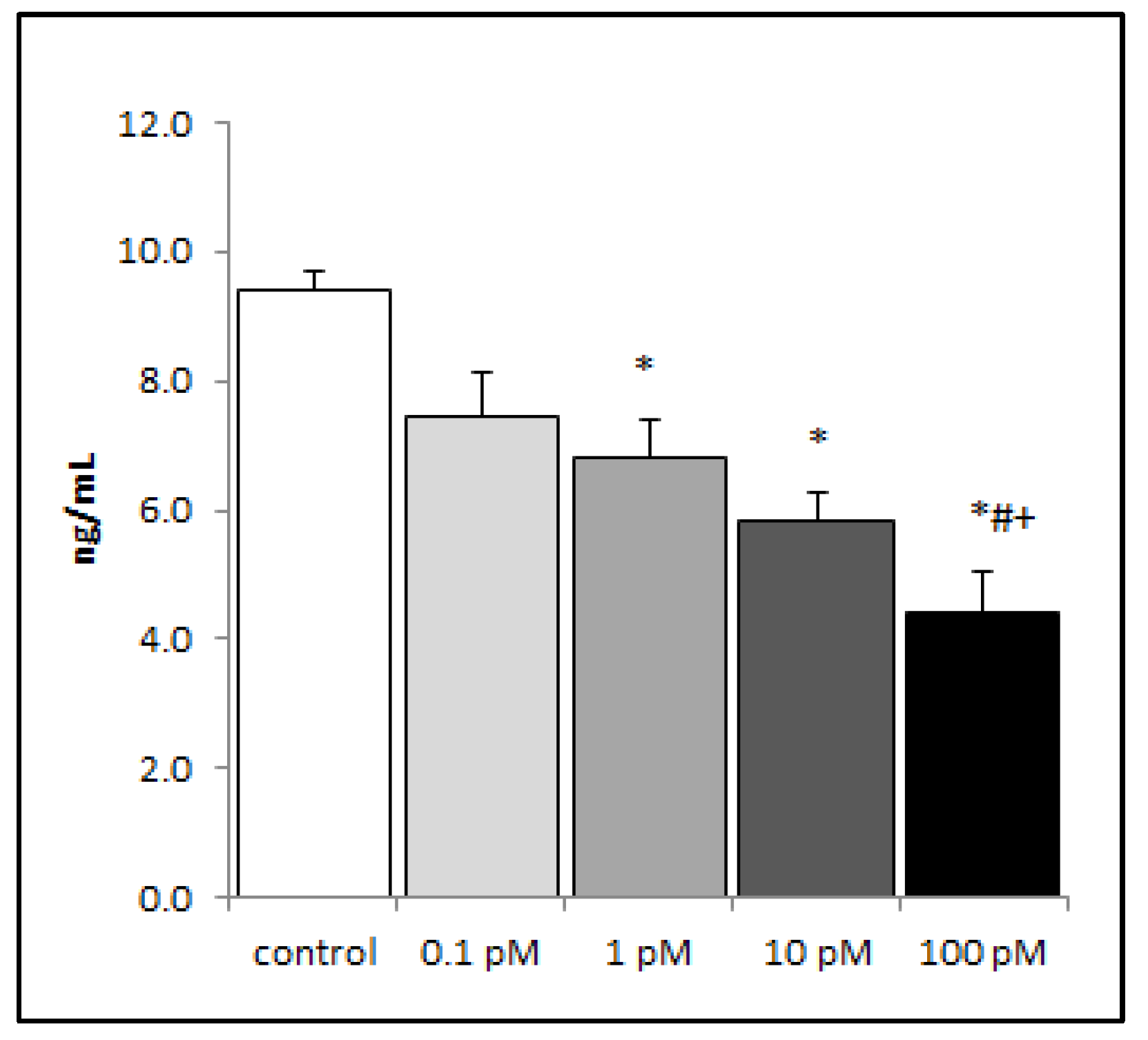
2.2. Brevenal Decreased LPS-Induced Production of the Proinflammatory Cytokine IL-8 from Human Lung Cells
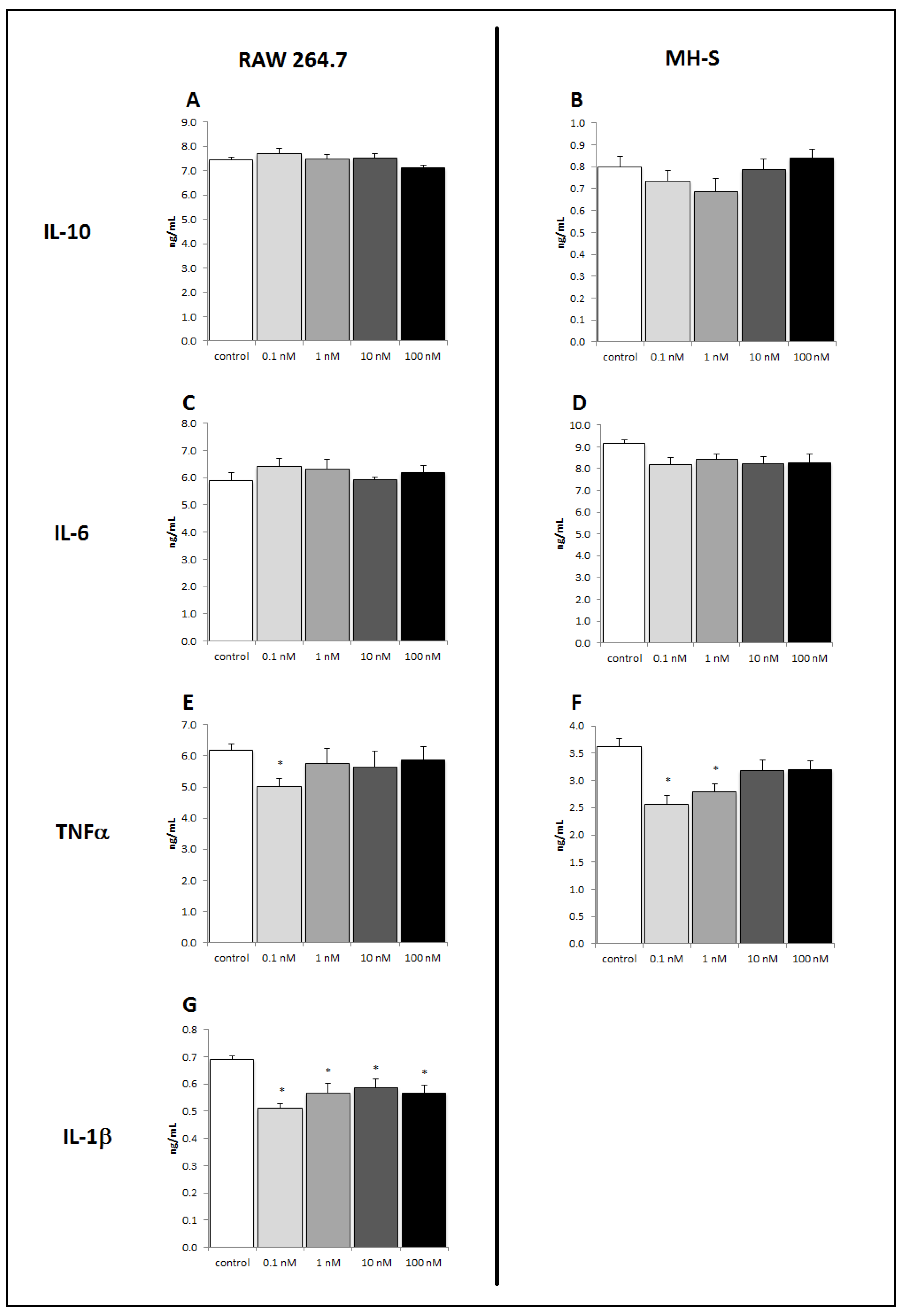
2.3. Brevenal Decreased LPS-Induced Production of Proinflammatory Cytokines from Murine Macrophages but Did Not Alter Pleiotropic or Anti-Inflammatory Cytokine Production
2.4. Treatment of RAW 264.7 Cells with Brevenal Did Not Change Cell-Surface Expression of TLR4, with or without LPS Co-Stimulation
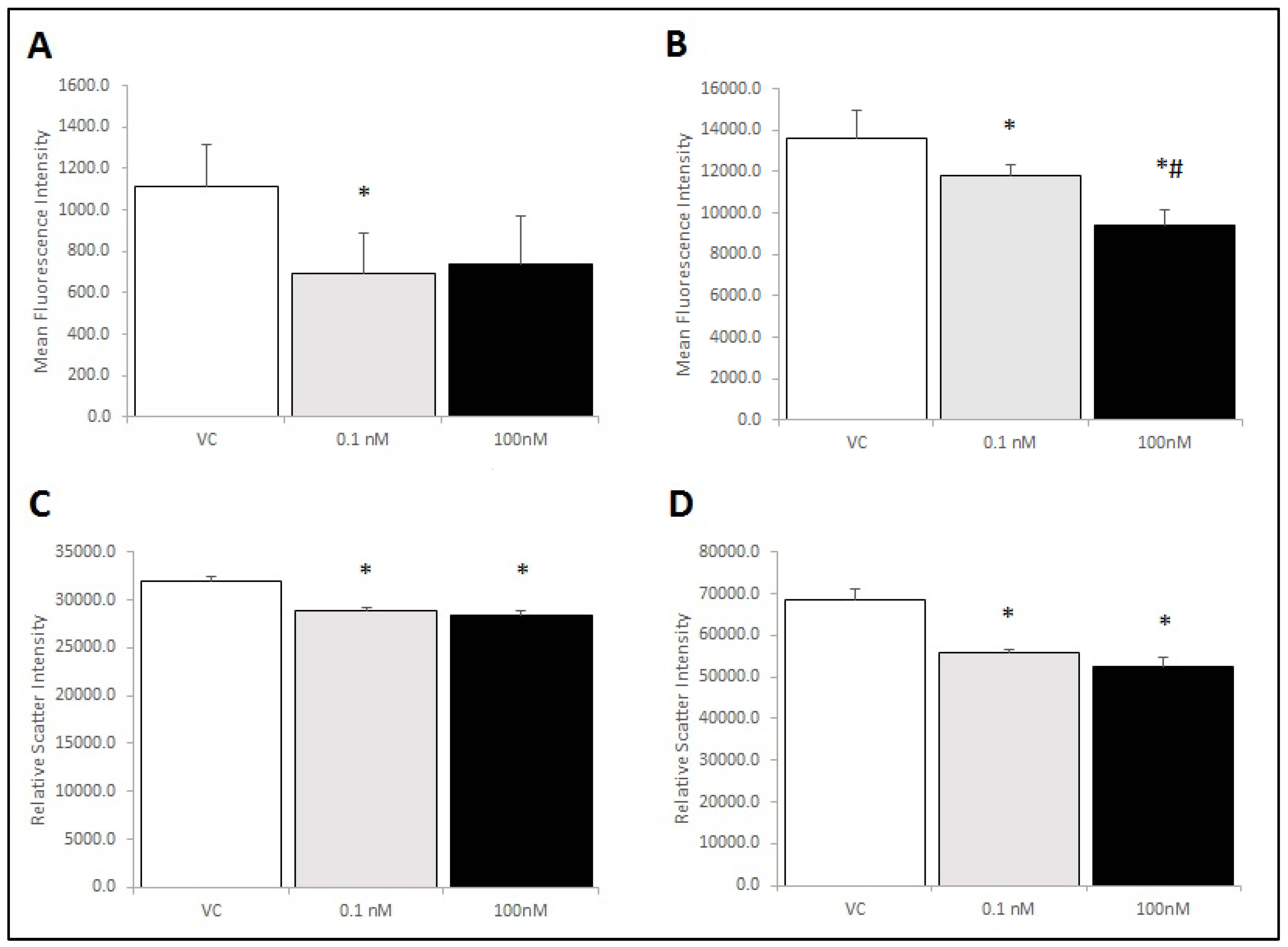
2.5. Brevenal Treatment Decreased Expression of Markers for Both M1 and M2 Phenotypes on RAW 264.7 Cells, Decreased Cell Size, and Decreased Cell Complexity
2.6. Brevenal Treatment Alters the Percentage of RAW 264.7 Cells that are Aggregated in Culture
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Brevenal Treatment
4.3. Cytotoxicity Assays
4.4. Quantification of Pro- and Anti-Inflammatory Cytokines
4.5. Flow Cytometry Assessment for Toll-Like Receptor 4 (TLR4), Mannose Receptor (CD206), and CD86 Receptor Expression
4.6. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Surveillance, Prevention and Control of Chronic Respiratory Diseases. A Comprehensive Approach; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Asaduzzaman, M.; Zhang, S.; Lavasani, S.; Wang, Y.S.; Thorlacius, H. LFA-1 and Mac-1 mediate pulmonary recruitment of neutrophils and tissue damage in abdominal sepsis. SHOCK 2008, 30, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, N.A.; Unsinger, J.; Hotchkiss, R.S.; Ayala, A. The new normal: Immunomodulatory agents against sepsis immune suppression. Trends Mol. Med. 2014, 20, 224–233. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The Global Burden of Disease: 2004 Update; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Burney, P.G.; Patel, J.; Newson, R.; Minelli, C.; Naghavi, M. Global and regional trends in COPD mortality, 1990–2010. Eur. Respir. J. 2015, 45, 1239–1247. [Google Scholar] [CrossRef]
- Stephenson, A.L.; Stanojevic, S.; Sykes, J.; Burgel, P.R. The changing epidemiology and demography of cystic fibrosis. Presse Med. 2017, 46, E87–E95. [Google Scholar] [CrossRef] [PubMed]
- Fitzsimmons, S.C. The changing epidemiology of cystic-fibrosis. J. Pediatr. 1993, 122, 1–9. [Google Scholar] [CrossRef]
- Stephenson, A.L.; Tom, M.; Berthiaume, Y.; Singer, L.G.; Aaron, S.D.; Whitmore, G.A.; Stanojevic, S. A contemporary survival analysis of individuals with cystic fibrosis: A cohort study. Eur. Respir. J. 2015, 45, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R.; Program, G. The global burden of asthma: Executive summary of the GINA Dissemination Committee Report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef]
- Baden, D.G.; Tomas, C.R. Variations in major toxin composition for six clones of Ptychodiscus brevis. Toxicon 1988, 26, 961–963. [Google Scholar] [CrossRef]
- Prasad, A.V.K.; Shimizu, Y.J. The structure of hemi-brevetoxin-B: A new type of toxin in the Gulf of Mexico red tide organism. J. Am. Chem. Soc. 1989, 111, 6476–6477. [Google Scholar] [CrossRef]
- Satake, M.; Campbell, A.; Van Wagoner, R.M.; Bourdelais, A.J.; Jacocks, H.; Baden, D.G.; Wright, J.L. Brevisin: An aberrant polycyclic ether structure from the dinoflagellate Karenia brevis and its implications for polyether assembly. Org. Chem. 2009, 74, 989–994. [Google Scholar] [CrossRef]
- Satake, M.; Bourdelais, A.J.; Van Wagoner, R.M.; Baden, D.G.; Wright, J.L. Brevisamide: An unprecedented monocyclic ether alkaloid from the dinoflagellate Karenia brevis that provides a potential model for ladder-frame initiation. Org. Lett. 2008, 10, 3465–3468. [Google Scholar] [CrossRef] [PubMed]
- Truxal, L.T.; Bourdelais, A.J.; Jacocks, H.M.; Abraham, W.M.; Baden, D.G. Characterization of tamulamides A and B: Polyethers isolated from the marine dinoflagellate Karenia brevis. J. Nat. Prod. 2010, 73, 536–540. [Google Scholar] [CrossRef]
- Bourdelais, A.J.; Jacocks, H.; Wright, J.L.; Bigwarfe, P.M.; Baden, D.G. A new polyether ladder compound produced by the dinoflagellate Karenia brevis. J. Nat. Prod. 2005, 68, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Bourdelais, A.J.; Campbell, S.; Jacocks, H.; Naar, J.; Wright, J.L.; Carsi, J.; Baden, D.G. Brevenal is a natural inhibitor of brevetoxin action in sodium channel receptor binding assays. Cell. Mol. Neurobiol. 2004, 24, 553–563. [Google Scholar] [CrossRef] [PubMed]
- LePage, K.T.; Rainier, J.D.; Johnson, W.B.; Baden, D.G.; Murray, T.F. Gambierol acts as a functional antagonist of neurotoxin site 5 on voltage-gated sodium channels in cerebellar granule neurons. J. Pharmacol. Exp. Ther. 2007, 323, 174–179. [Google Scholar] [CrossRef]
- Sayer, A.N.; Hu, Q.; Bourdelais, A.J.; Baden, D.G.; Gibson, J.E. The effect of brevenal on brevetoxin-induced DNA damage in human lymphocytes. Food. Chem. Toxicol. 2006, 44, 1082–1091. [Google Scholar] [CrossRef] [PubMed]
- Abraham, W.M.; Bourdelais, A.J.; Sebater, J.R.; Ahmed, A.; Lee, T.A.; Serebriakov, I.; Baden, D.G. Airway responses to aerosolized brevetoxins in an animal model of asthma. Am. J. Respir. Crit. Care Med. 2005, 171, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Abraham, W.M.; Baden, D.G. Novel pharmacological actions of natural antagonist derived from K. brevis. In Proceedings of the Society of Toxicology, Washington, DC, USA, 6–10 March 2011. [Google Scholar]
- Sabater, J.R.; Clarke, T.C.; Ahmed, N.; Bourdelais, A.J.; Baden, D.G.; Abraham, W.M. Comparative modulation of human neutrophil elastase-induced pathophysiological airway effects by clinical and experimental agents. In Proceedings of the American Thoracic Society 2012 International Conference, San Francisco, CA, USA, 18–23 May 2012. [Google Scholar]
- Abraham, W.M.; Sabater, J.R.; McClain, D.; Ball, R.W.; Beerman, M.; Baden, D.G.; Bourdelais, A.; Salathe, M.; Milla, C.E.; Cohen, I. The anti-inflammatory activity of the mucocilliary clearance agent brevenal enhances the efficacy of cystic fibrosis therapies. J. Cyst Fibros. 2017, 16, S38. [Google Scholar] [CrossRef]
- Holzman, D.C. A kinder side of red tides? Environ. Health Perspect. 2011, 119, a336. [Google Scholar] [CrossRef]
- Hilderbrand, S.C.; Murrell, R.N.; Gibson, J.E.; Brown, J.M. Marine brevetoxin induces IgE-independent mast cell activation. Arch. Toxicol. 2011, 85, 135–141. [Google Scholar] [CrossRef]
- Wilhelmi, V.; Fischer, U.; van Burlo, D.; Schulze-Osthoff, K.; Schins, R.P.F.; Albrecht, C. Evaluation of apoptosis induced by nanoparticles and fine particles on RAW 264.7 macrophages: Facts and artefacts. Toxicol. Vitro 2012, 26, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhang, P.; Hao, Y.; He, D.; Shen, Y.; Lu, R. Expression and significance of quantum dots in RAW 264.7 macrophages. Oncol. Lett. 2018, 16, 5997–6002. [Google Scholar] [CrossRef] [PubMed]
- Bickel, M. The role of interleukin-8 in inflammation and mechanisms of regulation. J. Periodontol. 1993, 64, 456–460. [Google Scholar]
- Ferraro, M.; Di Vincenzon, S.; Dino, P.; Bucchieri, S.; Cipollina, C.; Gjomarkaj, M.; Pace, E. Budesonide, aclidinium and formeoterol in combination limit inflammaging processes in bronchial epithelial cells exposed to cigarette smoke. Exp. Gerontol. 2019, 118, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.; Aberdein, J.; Jubrail, J.; Dockrell, D.H. Chapter Four–The Role of Macrophages in the Innate Immune Response to Streptococcus pneumoniiae and Staphylococcus aureus: Mechanisms and Contrasts. Adv. Micr. Phys. 2014, 65, 125–202. [Google Scholar]
- Parameswaran, N.; Patial, S. Tumor Necrosis Factor-α Signaling in Macrophages. Crit. Rev. Eukaryot. Gene Expr. 2010, 20, 87–103. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Biologic basis for interleukin-1 in disease. Blood 1996, 87, 2095–2147. [Google Scholar]
- Chaudhry, H.; Zhou, J.; Zhong, Y.; Ali, M.M.; McGuire, F.; Nagarkatti, P.S.; Nagarkatti, M. Role of Cytokines as a Double-edged Sword in Sepsis. In Vivo 2013, 27, 669–684. [Google Scholar] [PubMed]
- Sabat, R.; Grütz, G.; Warszawska, K.; Kirsch, S.; Witte, E.; Wolk, K.; Geginet, J. Biology of interleukin-10. Cytokine Growth Factor Rev. 2010, 21, 331–344. [Google Scholar] [CrossRef] [PubMed]
- Brattsand, R.; Linden, M. Cytokine modulation by glucocorticoids: Mechanisms and actions in cellular studies. Aliment. Pharmacol. Ther. 1996, 10, 81–90. [Google Scholar] [CrossRef]
- Debets, J.M.; Ruers, T.J.; van der Linden, M.P.; van der Linden, C.J.; Buurman, W.A. Inhibitory effect of corticosteroids on the secretion of tumour necrosis factor (TNF) by monocytes is dependent on the stimulus inducing TNF synthesis. Clin. Exp. Immunol. 1989, 78, 224–229. [Google Scholar] [PubMed]
- Wang, L.; Zhao, H.; Wang, D. Inflammatory cytokine expression in patients with sepsis at an intensive care unit. Exp. Ther. Med. 2018, 16, 2126–2131. [Google Scholar] [CrossRef]
- Sabry, O.M.M.; Andrews, S.; McPhail, K.L.; Goeger, D.E.; Yokochi, A.; LePage, K.T.; Murray, T.F.; Gerwick, W.H. Neurotoxic meroditerpenoids from the tropical marine brown alga Stypopodium flabelliforme. J. Nat. Prod. 2005, 68, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- McCall, J.R.; Elliot, E.A.; Bourdelais, A.J. A New Cytotoxicity Assay for Brevetoxins Using Fluorescence Microscopy. Mar. Drugs 2014, 12, 4868–4882. [Google Scholar] [CrossRef] [PubMed]
- Abraham, W.M.; Sabater, J.R.; Clarke, T.C.; Baden, D.G. Mechanisms of protease activated receptor-2 slowing of mucocilliary clearance in sheep. Am. J. Respir. Crit. Care Med. 2011, 183, A1221. [Google Scholar] [CrossRef]
- Lu, Y.C.; Yeh, W.C.; Ohashi, P.S. LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Shapouri-Moghaddam, A.; Mohammadian, S.; Vazini, H.; Taghadosi, M.; Esmaeili, S.; Mardani, M.; Seifi, B.; Mohammadi, A.; Afshari, J.T.; Sahebkar, A. Macrophage plasticity, polarization, and function in health and disease. J. Cell Physiol. 2018, 233, 6425–6440. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000 Prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef]
- Sica, A.; Mantovani, A. Macrophage plasticity and polarization: In vivo veritas. J. Clin. Investig. 2012, 122, 787–795. [Google Scholar] [CrossRef]
- Rettew, J.; McCall, S.; Marriott, I. GPR30/GPER-1 mediates rapid decreases in TLR4 expression on murine macrophages. Mol. Cell. Endocrinol. 2010, 328, 87–92. [Google Scholar] [CrossRef]
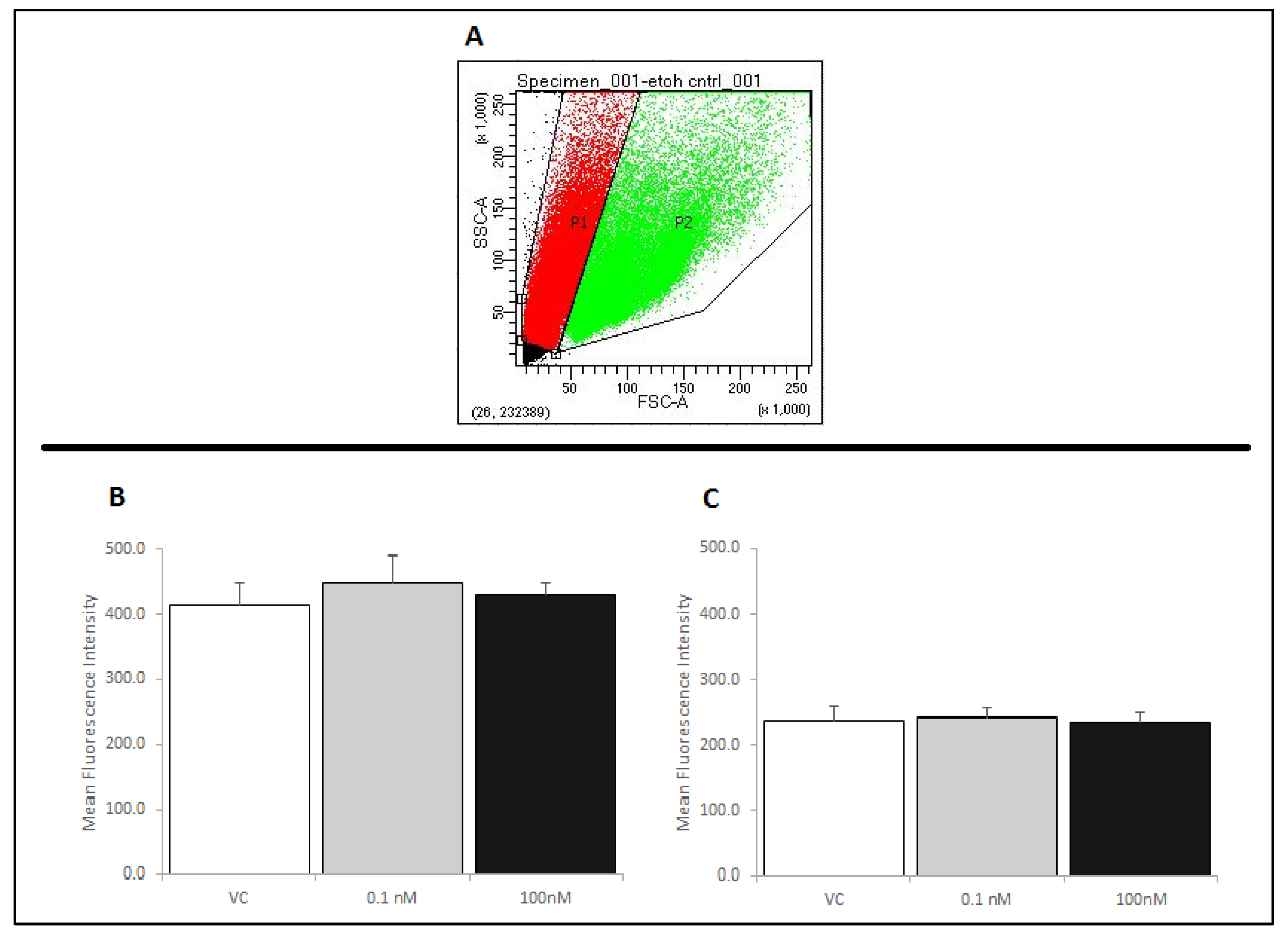
| Treatment | % P1 | % P2 |
|---|---|---|
| VC | 55.2% +/− 1.9% | 40.0% +/− 2.4% |
| 0.1 nM | 57.9% +/− 3.5% | 34.1% +/− 3.4% (*) |
| 100 nM | 55.9% +/− 1.7% | 34.9% +/− 2.7% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keeler, D.M.; Grandal, M.K.; McCall, J.R. Brevenal, a Marine Natural Product, is Anti-Inflammatory and an Immunomodulator of Macrophage and Lung Epithelial Cells. Mar. Drugs 2019, 17, 184. https://doi.org/10.3390/md17030184
Keeler DM, Grandal MK, McCall JR. Brevenal, a Marine Natural Product, is Anti-Inflammatory and an Immunomodulator of Macrophage and Lung Epithelial Cells. Marine Drugs. 2019; 17(3):184. https://doi.org/10.3390/md17030184
Chicago/Turabian StyleKeeler, Devon M., Meghan K. Grandal, and Jennifer R. McCall. 2019. "Brevenal, a Marine Natural Product, is Anti-Inflammatory and an Immunomodulator of Macrophage and Lung Epithelial Cells" Marine Drugs 17, no. 3: 184. https://doi.org/10.3390/md17030184
APA StyleKeeler, D. M., Grandal, M. K., & McCall, J. R. (2019). Brevenal, a Marine Natural Product, is Anti-Inflammatory and an Immunomodulator of Macrophage and Lung Epithelial Cells. Marine Drugs, 17(3), 184. https://doi.org/10.3390/md17030184




