Antioxidant and Neuroprotective Potential of the Brown Seaweed Bifurcaria bifurcata in an in vitro Parkinson’s Disease Model
Abstract
:1. Introduction
2. Results and Discussion
2.1. Antioxidant Activity of B. bifurcata
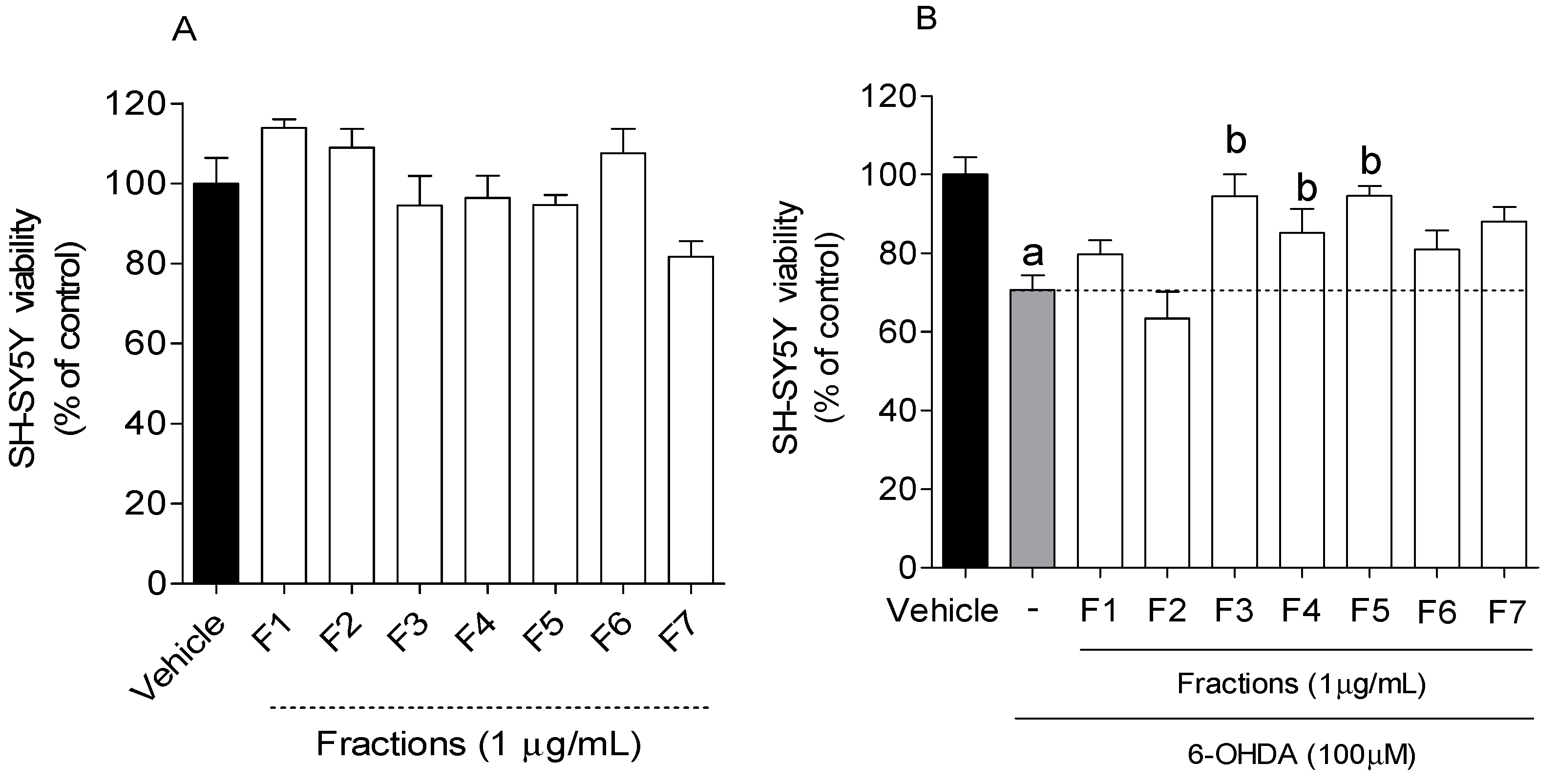
2.2. Neuroprotective Effect of B. bifurcata on SH-SY5Y Cells Exposed to 6-OHDA
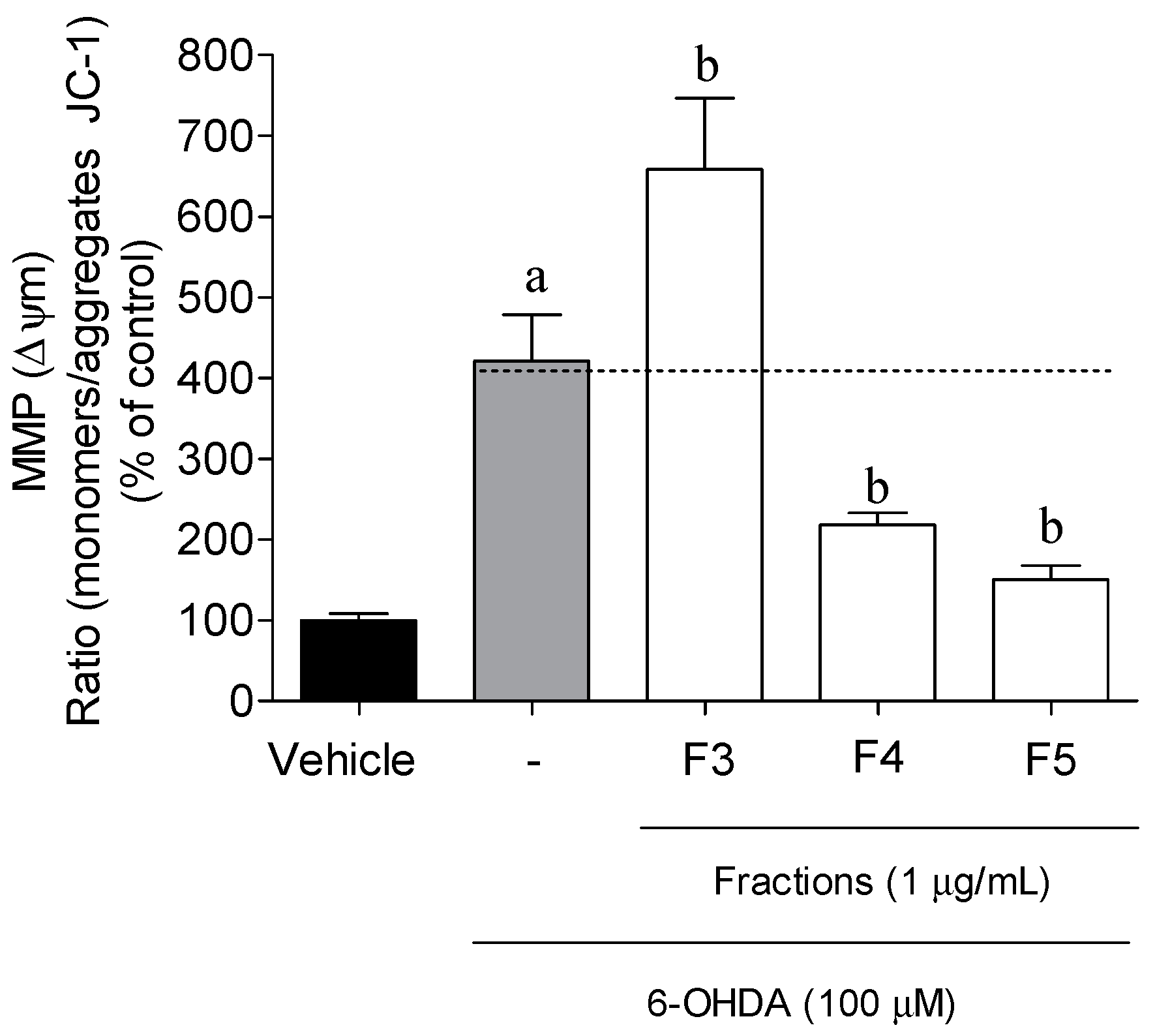
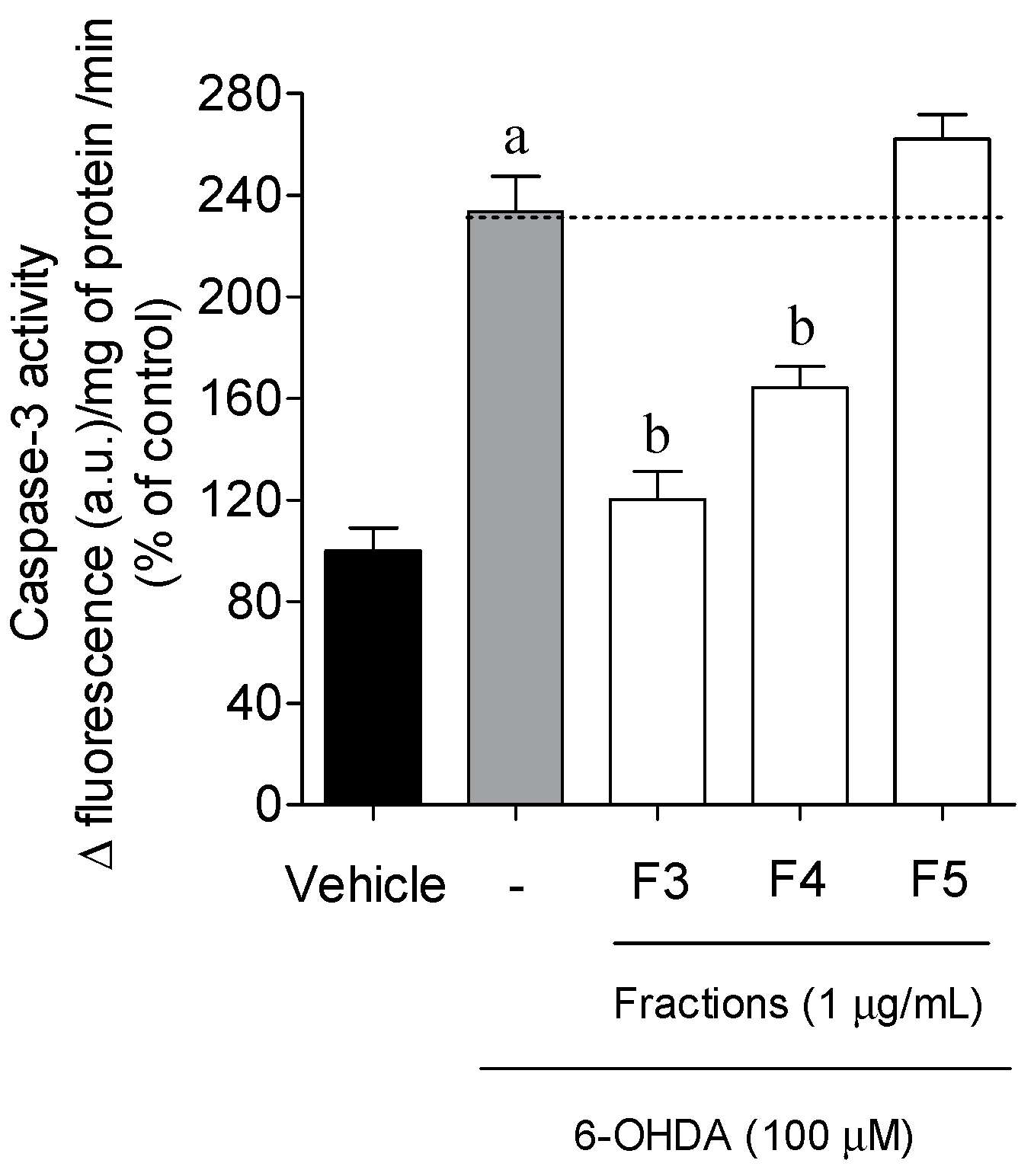
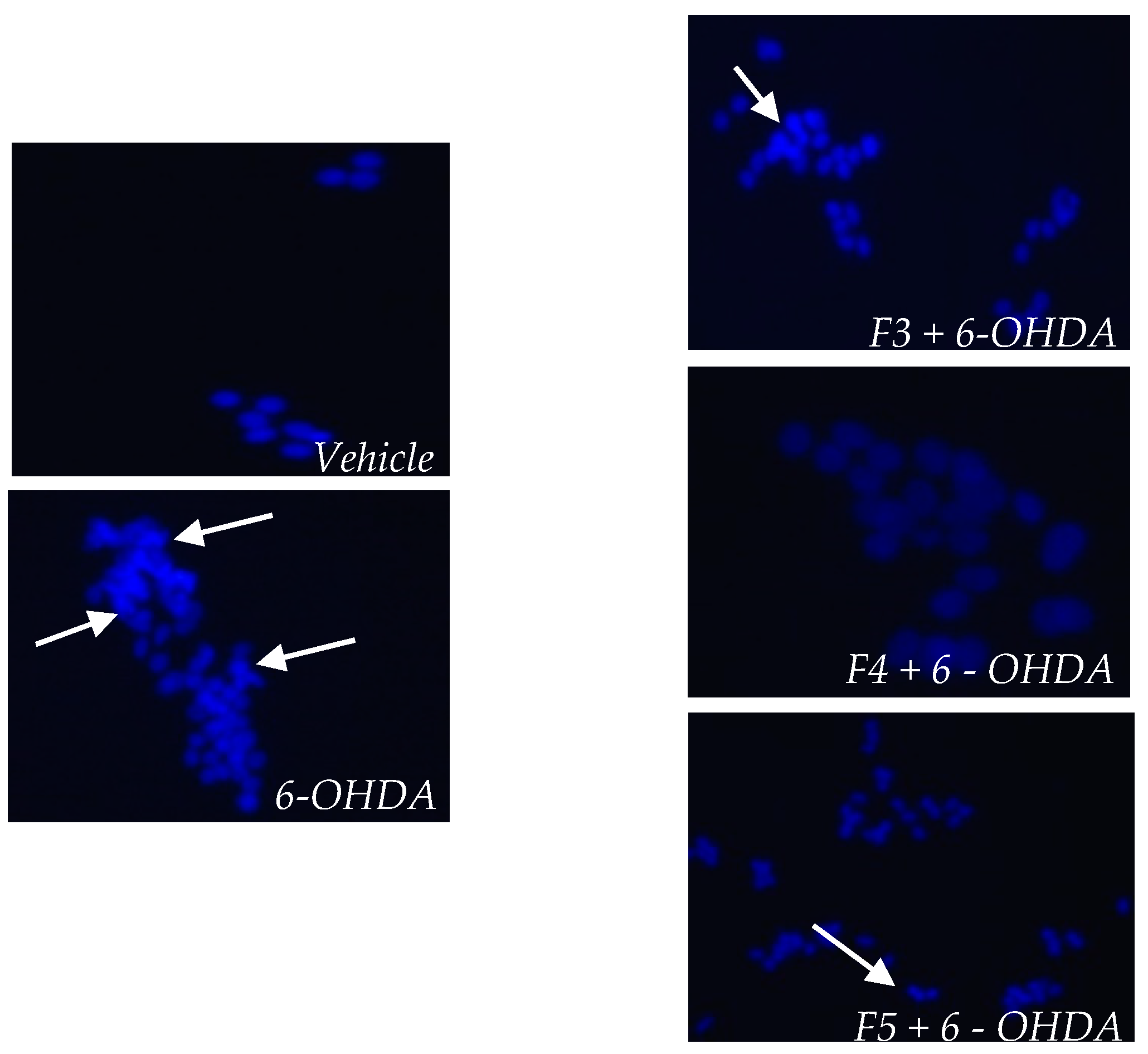
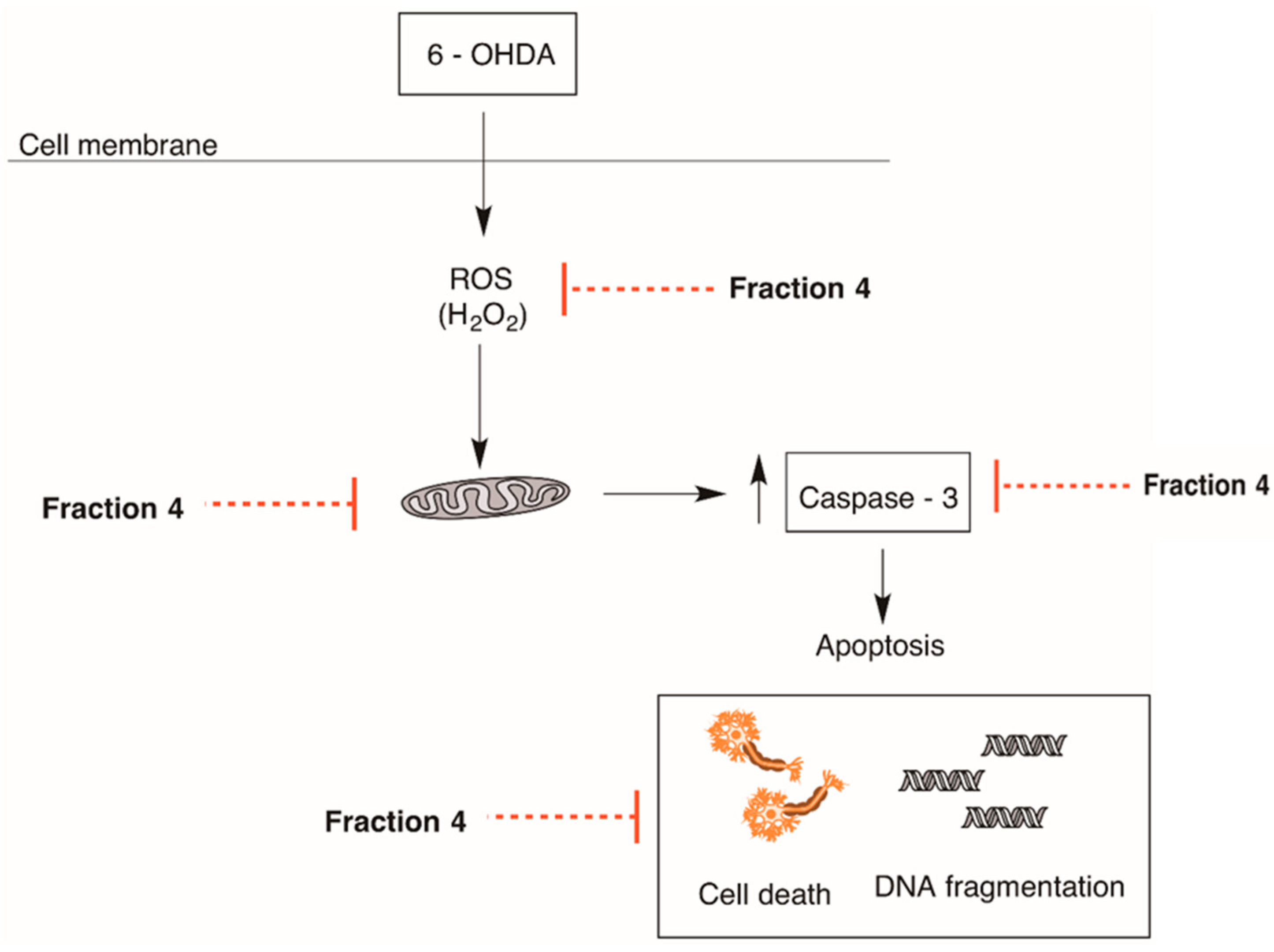
2.3. Cellular Mechanisms Associated with the Neuroprotective Effect of B. bifurcata
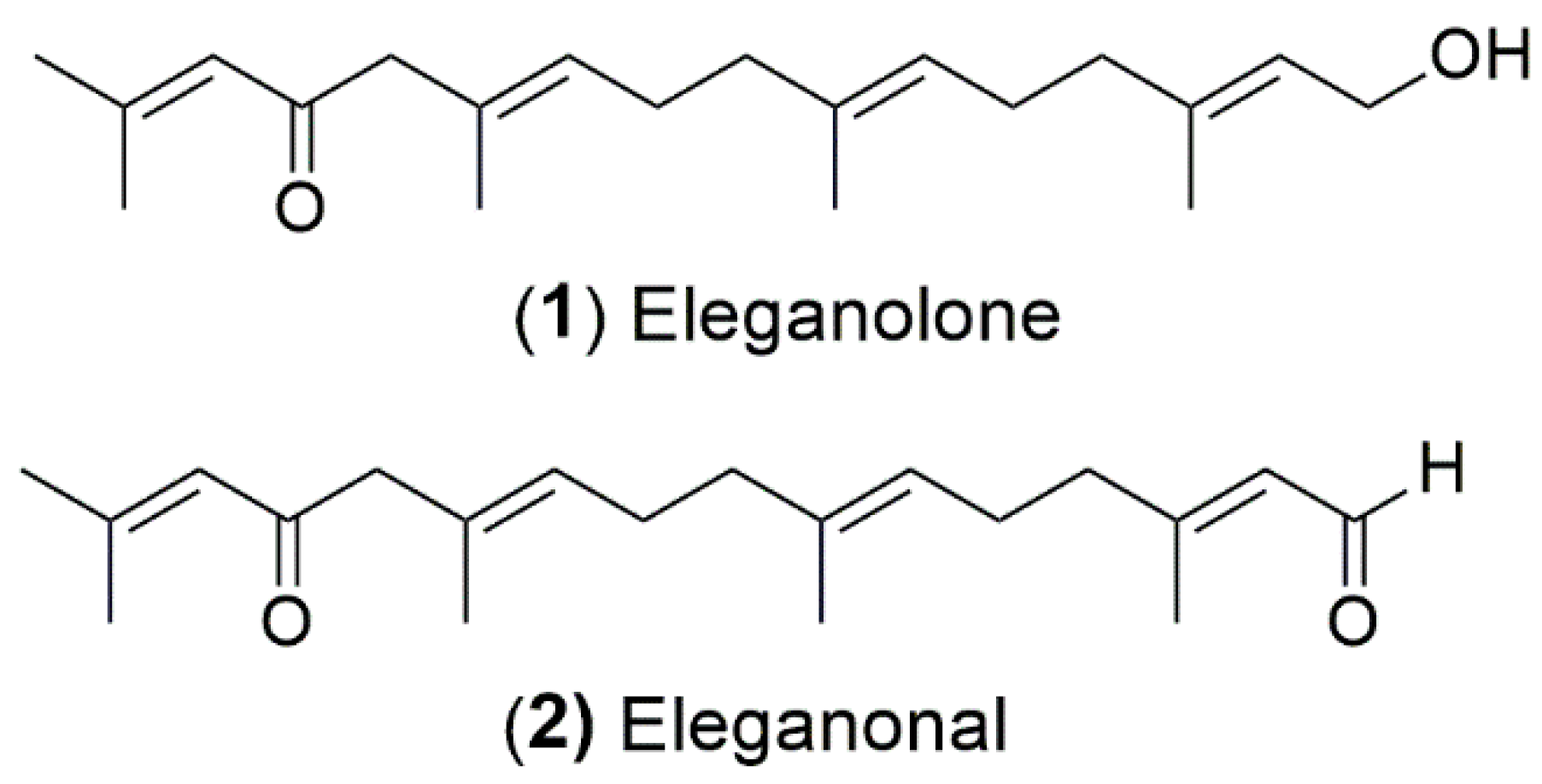
2.4. Isolation and Antioxidant Activity of B. bifurcata Compounds
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Seaweed Collection
3.3. Extraction and Fractionation of B. bifurcata
3.4. Isolation and Characterization of Bioactive Compounds
3.5. Antioxidant Activity
3.6. Mammalian Cell Strain and Culture Method
3.7. Neuroprotective Activity
3.8. Mechanisms Associated with Neuroprotective Potential
3.9. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ramkumar, M.; Rajasankar, S.; Gobi, V.V.; Dhanalakshmi, C.; Manivasagam, T.; Justin Thenmozhi, A.; Essa, M.M.; Kalandar, A.; Chidambaram, R. Neuroprotective effect of Demethoxycurcumin, a natural derivative of Curcumin on rotenone induced neurotoxicity in SH-SY5Y Neuroblastoma cells. BMC Complement. Altern. Med. 2017, 17, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.J.; Chen, W.F.; Guo, B.L.; Feng, C.W.; Hung, H.C.; Yang, W.Y.; Sung, C.S.; Tsui, K.H.; Chu, H.; Chen, N.F.; et al. The 1-tosylpentan-3-one protects against 6-hydroxydopamine-induced neurotoxicity. Int. J. Mol. Sci. 2017, 18, 1096. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Torres, V.; Encinar, J.A.; Herranz-López, M.; Pérez-Sánchez, A.; Galiano, V.; Barrajón-Catalán, E.; Micol, V. An updated review on marine anticancer compounds: The use of virtual screening for the discovery of small-molecule cancer drugs. Molecules 2017, 22, 1037. [Google Scholar] [CrossRef]
- Debbab, A.; Aly, A.H.; Lin, W.H.; Proksch, P. Bioactive Compounds from Marine Bacteria and Fungi. Microb. Biotechnol. 2010, 3, 544–563. [Google Scholar] [CrossRef] [Green Version]
- Pietra, F. Secondary metabolites from marine microorganisms: Bacteria, protozoa, algae and fungi. Achievements and prospects. Nat. Prod. Rep. 1997, 14, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Fischbach, M.A.; Walsh, C.T. Antibiotics for Emerging Pathogen. Science 2009, 325, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Song, F. Bioprospecting of Novel and Bioactive Compounds from Marine Actinomycetes Isolated from South China Sea Sediments. Curr. Microbiol. 2017, 75, 142–149. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Pinteus, S.; Mendes, S.; Pedrosa, R. Neuroprotective effects of seaweeds against 6-hydroxidopamine-induced cell death on an in vitro human neuroblastoma model. BMC Complement. Altern. Med. 2018, 18, 58. [Google Scholar] [CrossRef]
- Pinteus, S.; Silva, J.; Alves, C.; Horta, A.; Fino, N.; Rodrigues, A.I.; Mendes, S.; Pedrosa, R. Cytoprotective effect of seaweeds with high antioxidant activity from the Peniche coast (Portugal). Food Chem. 2017, 218, 591–599. [Google Scholar] [CrossRef]
- Rodrigues, D.; Alves, C.; Horta, A.; Pinteus, S.; Silva, J.; Culioli, G.; Thomas, O.P.; Pedrosa, R. Antitumor and antimicrobial potential of bromoditerpenes isolated from the Red Alga, Sphaerococcus coronopifolius. Mar. Drugs 2015, 13, 713–726. [Google Scholar] [CrossRef] [PubMed]
- El Gamal, A.A. Biological importance of marine algae. Saudi Pharm. J. 2010, 18, 1–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, S.A.O.; Trindade, S.S.; Oliveira, C.S.D.; Parreira, P.; Rosa, D.; Duarte, M.F.; Ferreira, I.; Cruz, M.T.; Rego, A.M.; Abreu, M.H.; et al. Lipophilic fraction of cultivated Bifurcaria bifurcata R. Ross: Detailed composition and in vitro prospection of current challenging bioactive properties. Mar. Drugs 2017, 15, 340. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine Natural Products. Nat. Prod. Rep. 2017, 34, 235–294. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L. Seaweed flora of the European North Atlantic and Mediterranean. In Springer Handbook of Marine Biotechnology; Springer: Berlin, Germany, 2015; pp. 65–178. ISBN 9783642539718. [Google Scholar]
- Smyrniotopoulos, V.; Merten, C.; Kaiser, M.; Tasdemir, D. Bifurcatriol, a new antiprotozoal acyclic diterpene from the brown alga Bifurcaria bifurcata. Mar. Drugs 2017, 15, 245. [Google Scholar] [CrossRef] [PubMed]
- Gallé, J.B.; Attioua, B.; Kaiser, M.; Rusig, A.M.; Lobstein, A.; Vonthron-Sénécheau, C. Eleganolone, a diterpene from the French marine alga Bifurcaria bifurcata inhibits growth of the human pathogens Trypanosoma brucei and Plasmodium falciparum. Mar. Drugs 2013, 11, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Göthel, Q.; Lichte, E.; Köck, M. Further eleganolone-derived diterpenes from the brown alga Bifurcaria bifurcata. Tetrahedron Lett. 2012, 53, 1873–1877. [Google Scholar] [CrossRef]
- Alves, C.; Pinteus, S.; Simões, T.; Horta, A.; Silva, J.; Tecelão, C.; Pedrosa, R. Bifurcaria bifurcata: A key macro-alga as a source of bioactive compounds and functional ingredients. Int. J. Food Sci. Technol. 2016, 51, 1638–1646. [Google Scholar] [CrossRef]
- Pinteus, S.; Silva, J.; Alves, C.; Horta, A.; Thomas, O.P.; Pedrosa, R. Antioxidant and cytoprotective activities of Fucus spiralis seaweed on a human cell in vitro model. Int. J. Mol. Sci. 2017, 18, 292. [Google Scholar] [CrossRef] [PubMed]
- Neto, J.; Uchôa, A.; Moura, P.; Filho, C.; Tenório, J.; Silva, A.; Ximenes, R.; Silva, M.; Correia, M. Phytochemical screening, total phenolic content and antioxidant activity of some plants from Brazilian flora. J. Med Plants Res. 2016, 10, 409–416. [Google Scholar]
- Farasat, M.; Khavari-Nejad, R.A.; Nabavi, S.M.B.; Namjooyan, F. Antioxidant activity, total phenolics and flavonoid contents of some edible green seaweeds from northern coasts of the Persian gulf. Iran. J. Pharm. Res. 2014. [Google Scholar] [CrossRef]
- Manochai, B.; Ingkasupart, P.; Lee, S.H.; Hong, J.H. Evaluation of antioxidant activities, total phenolic content (TPC), and total catechin content (TCC) of 10 sugar apple (Annona squamosa L.) cultivar peels grown in Thailand. Food Sci. Technol. 2018, 1–7. [Google Scholar] [CrossRef]
- Costa, L.S.; Fidelis, G.P.; Telles, C.B.S.; Dantas-Santos, N.; Camara, R.B.G.; Cordeiro, S.L.; Costa, M.S.S.P.; Almeida-Lima, J.; Melo-Silveira, R.F.; Oliveira, R.M.; et al. Antioxidant and antiproliferative activities of heterofucans from the seaweed Sargassum filipendula. Mar. Drugs 2011, 9, 952–966. [Google Scholar] [CrossRef] [PubMed]
- Kasote, D.M.; Katyare, S.S.; Hegde, M.V.; Bae, H. Significance of Antioxidant Potential of Plants and its Relevance to Therapeutic Applications. Int. J. Biol. Sci. 2015, 11, 982–991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, T.; Yue, W.; Li, Q. Comparison of the Phenolic Content and Antioxidant Activities of Apocynum venetum L. (Luo-Bu-Ma) and Two of Its Alternative Species. Int. J. Mol. Sci. 2010, 11, 4452–4464. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; O, W.; Li, W.; Jiang, Z.-G.; Ghanbari, H. Oxidative Stress and Neurodegenerative Disorders. Int. J. Mol. Sci. 2013, 14, 24438–24475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leyton, A.; Lienqueo, M.E. Purification of phlorotannins from Macrocystis pyrifera using macroporous resins. Food Chem. 2017, 237, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fu, X.; Duan, D.; Liu, X.; Xu, J.; Gao, X. Extraction and Identification of Phlorotannins from the Brown Alga, Sargassum fusiforme (Harvey) Setchell. Mar. Drugs 2017, 15, 49. [Google Scholar] [CrossRef]
- Choi, D.-Y.; Choi, H. Natural products from marine organisms with neuroprotective activity in the experimental models of Alzheimer’s disease, Parkinson’s disease and ischemic brain stroke: Their molecular targets and action mechanisms. Arch. Pharmacal Res. 2015, 38, 139–170. [Google Scholar] [CrossRef] [PubMed]
- Article, R.; Kovacic, P.; Weston, W. Chronicles of Pharmaceutical Science Treatment of Parkinson’s Disease with Phenolic Antioxidant Drugs: Oxidative Stress, Reactive Oxygen Species and Selectivity. Pharm. Sci. 2017, 1, 193–198. [Google Scholar]
- Brizi, C.; Santulli, C.; Micucci, M.; Budriesi, R.; Chiarini, A.; Aldinucci, C.; Frosini, M. Neuroprotective Effects of Castanea sativa Mill. Bark Extract in Human Neuroblastoma Cells Subjected to Oxidative Stress. J. Cell. Biochem. 2016, 117, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Alvariño, R.; Alonso, E.; Tribalat, M.; Gegunde, S.; Thomas, O.P.; Botana, L.M. Evaluation of the Protective Effects of Sarains on H2O2 -Induced Mitochondrial Dysfunction and Oxidative Stress in SH-SY5Y Neuroblastoma Cells. Neurotox. Res. 2017, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Souza, R.B.; Frota, A.F.; Sousa, R.S.; Cezario, N.A.; Santos, T.B.; Souza, L.M.F.; Coura, C.O.; Monteiro, V.S.; Cristino Filho, G.; Vasconcelos, S.M.M.; et al. Neuroprotective Effects of Sulphated Agaran from Marine Alga Gracilaria cornea in Rat 6-Hydroxydopamine Parkinson’s Disease Model: Behavioural, Neurochemical and Transcriptional Alterations. Basic Clin. Pharmacol. Toxicol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ba, Q.; Cui, C.; Wen, L.; Feng, S.; Zhou, J.; Yang, K. Schisandrin B shows neuroprotective effect in 6-OHDA-induced Parkinson’s disease via inhibiting the negative modulation of miR-34a on Nrf2 pathway. Biomed. Pharmacother. 2015, 75, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Lou, H.; Jing, X.; Wei, X.; Shi, H.; Ren, D.; Zhang, X. Naringenin protects against 6-OHDA-induced neurotoxicity via activation of the Nrf2/ARE signaling pathway. Neuropharmacology 2014, 79, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Perfeito, R.; Perfeito, R.; Rego, A.C. Papel da alfa-sinucleína e da disfunção mitocondrial associada à doença de Parkinson. Rev. Neurocienc. 2012, 20, 273–284. [Google Scholar]
- Rodriguez-Pallares, J.; Parga, J.A.; Muñoz, A.; Rey, P.; Guerra, M.J.; Labandeira-Garcia, J.L. Mechanism of 6-hydroxydopamine neurotoxicity: The role of NADPH oxidase and microglial activation in 6-hydroxydopamine-induced degeneration of dopaminergic neurons. J. Neurochem. 2007, 103, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Cui, N.; Wang, P.; Song, S.; Liang, H.; Ji, A. Neuroprotective effect od sulphated polysaccharide isolated from sea cucumber Stichopus japonicus on 6-OHDA-induced death in SH-SY5Y through inhibition of MAPK and NF-kB and activation of PI3K/Akt signaling pathways. Biochem. Biophys. Res. Commun. 2016, 470, 375–383. [Google Scholar] [CrossRef]
- Kich, D.M.; Bitencourt, S.; Alves, C.; Silva, J.; Pinteus, S.; Pedrosa, R.; Laufer, S.; de Souza, C.F.V.; Goettert, M.I. Neuromodulatory effects of Calyptranthes grandifolia extracts against 6-hydroxydopamine-induced neurotoxicity in SH-SY5Y cells. Biomed. Pharmacother. 2016, 84, 382–386. [Google Scholar] [CrossRef]
- Kaasik, A.; Vassiljev, V.; Poldoja, E.; Kalda, A. Do nuclear condensation or fragmentation and DNA fragmentation reflect the mode of neuronal death? NeuroReport 1999, 10, 1937–1942. [Google Scholar] [CrossRef]
- Venderova, K.; Park, D.S. Programmed cell death in Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2012. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.F.; Chakraborty, C.; Sung, C.S.; Feng, C.W.; Jean, Y.H.; Lin, Y.Y.; Hung, H.C.; Huang, T.Y.; Huang, S.Y.; Su, T.M.; et al. Neuroprotection by marine-derived compound, 11-dehydrosinulariolide, in an in vitro Parkinson’s model: A promising candidate for the treatment of Parkinson’s disease. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2012, 385, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili-Mahani, S.; Vazifekhah, S.; Pasban-Aliabadi, H.; Abbasnejad, M.; Sheibani, V. Protective effect of orexin-A on 6-hydroxydopamine-induced neurotoxicity in SH-SY5Y human dopaminergic neuroblastoma cells. Neurochem. Int. 2013, 63, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Daoudi, M.; Ortalo-Magne, A.; Valls, R.; Piovetti, L. (S)-12-Hydroxygeranylgeraniol-derived diterpenes from the brown alga Bifurcaria bifurcata. Phytochemistry 2001, 57, 529–535. [Google Scholar]
- Culioli, G.; Ortalo-Magné, A.; Daoudi, M.; Thomas-Guyon, H.; Valls, R.; Piovetti, L. Trihydroxylated linear diterpenes from the brown alga Bifurcaria bifurcata. Phytochemistry 2004, 65, 2063–2069. [Google Scholar] [CrossRef] [PubMed]
- Culioli, G.; Daoudi, M.; Mesguiche, V.; Valls, R.; Piovetti, L. Geranylgeraniol-derived diterpenoids from the brown alga Bifurcaria bifurcata. Phytochemistry 1999, 52, 1447–1454. [Google Scholar] [CrossRef]
- Ortalo-Magné, A.; Culioli, G.; Valls, R.; Pucci, B.; Piovetti, L. Polar acyclic diterpenoids from Bifurcaria bifurcata (Fucales, Phaeophyta). Phytochemistry 2005, 66, 2316–2323. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.; Zucca, F.; Duyn, J.; Crichton, R.; Zecca, L. The role of iron in brain ageing and neurodegenerative disorders. Lancet Neurol. 2014, 13, 1045–1060. [Google Scholar] [CrossRef] [Green Version]
- Berg, D.; Youdim, M.B.H. Role of iron in neurodegenerative disorders. Top. Magn. Reson. Imaging TMRI 2006, 17, 5–17. [Google Scholar] [CrossRef]
- Kurutas, E. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: Current state. Nutr. J. 2016, 15, 1–22. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A., Jr. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Dávalos, A.; Gómez-Cordovés, C.; Bartolomé, B. Extending Applicability of the Oxygen Radical Absorbance Capacity (ORAC-Fluorescein) Assay. J. Agric. Food Chem. 2004, 52, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996. [Google Scholar] [CrossRef] [PubMed]
- Rotter, B.A.; Oh, Y.N. MTS/PMS colorimetric assay is unsuitable for measuring mitogenic responses in porcine blood lymphocytes. J. Immunol. Methods 1996, 199, 205–209. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5). In Sect. Permut. Methods Microcomput. Power; Wageningen University & Research: Ithaca, NY, USA, 2002; Volume 10. [Google Scholar]
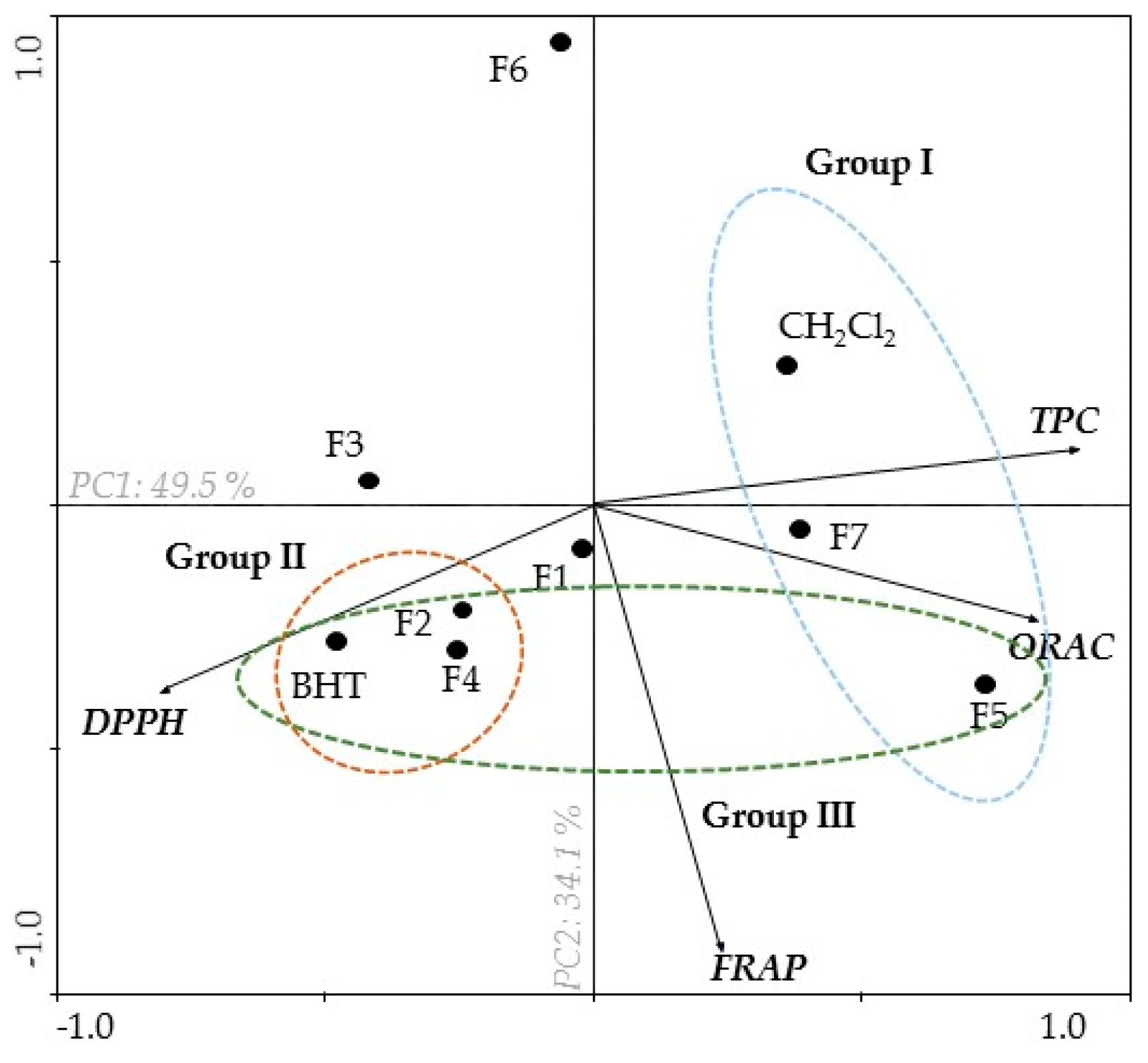

| Sample | TPC a | DPPH b,* | ORAC c | FRAP d |
|---|---|---|---|---|
| Crude Extract | 10.63 ± 1.40 | 43.34 (36.90–50.81) | 1886.55 ± 60.57 | 95.83 ± 4.48 |
| F1 | 4.4 ± 0.60 | 111.9 (80.40–155.70) | 353.23 ± 9.89 | 105.02 ± 1.88 |
| F2 | 5.3 ± 0.60 | >1000 | 768.01 ± 44.64 | 221.37 ± 11.77 |
| F3 | 3.08 ± 0.30 | >1000 | 412.55 ± 14.44 | 532.32 ± 3.00 |
| F4 | 2.06 ± 0.41 | >1000 | 1407.01 ± 44.56 | 548.19 ± 1.44 |
| F5 | 44.14 ± 1.30 | 49.73 (31.56–78.56) | 4469.14 ± 147.07 | 1128.20 ± 20.25 |
| F6 | 7.26 ± 0.50 | 64.28 (40.59–101.80) | 334.15 ± 49.65 | 7.64 ± 1.63 |
| F7 | 9.82 ± 0.80 | 114.60 (40.24–118.40) | 2861.25 ± 38.92 | 573.44± 33.79 |
| BHT | - | 205.00 (166.00–253.30) | 143.70 ± 23.36 | 2821.50 ± 63.03 |
| Compounds | DPPH a | ORAC b | FRAP c |
|---|---|---|---|
| Eleganolone (1) | >100 | 1663.83 ± 25.35 | 8341.18 ± 177.72 |
| Eleganonal (2) | >100 | 667.48 ± 10.96 | 8635.37 ± 389.54 |
| BHT | >100 | 143.70 ± 23.36 | 2821.50 ± 63.03 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, J.; Alves, C.; Freitas, R.; Martins, A.; Pinteus, S.; Ribeiro, J.; Gaspar, H.; Alfonso, A.; Pedrosa, R. Antioxidant and Neuroprotective Potential of the Brown Seaweed Bifurcaria bifurcata in an in vitro Parkinson’s Disease Model. Mar. Drugs 2019, 17, 85. https://doi.org/10.3390/md17020085
Silva J, Alves C, Freitas R, Martins A, Pinteus S, Ribeiro J, Gaspar H, Alfonso A, Pedrosa R. Antioxidant and Neuroprotective Potential of the Brown Seaweed Bifurcaria bifurcata in an in vitro Parkinson’s Disease Model. Marine Drugs. 2019; 17(2):85. https://doi.org/10.3390/md17020085
Chicago/Turabian StyleSilva, Joana, Celso Alves, Rafaela Freitas, Alice Martins, Susete Pinteus, Joana Ribeiro, Helena Gaspar, Amparo Alfonso, and Rui Pedrosa. 2019. "Antioxidant and Neuroprotective Potential of the Brown Seaweed Bifurcaria bifurcata in an in vitro Parkinson’s Disease Model" Marine Drugs 17, no. 2: 85. https://doi.org/10.3390/md17020085
APA StyleSilva, J., Alves, C., Freitas, R., Martins, A., Pinteus, S., Ribeiro, J., Gaspar, H., Alfonso, A., & Pedrosa, R. (2019). Antioxidant and Neuroprotective Potential of the Brown Seaweed Bifurcaria bifurcata in an in vitro Parkinson’s Disease Model. Marine Drugs, 17(2), 85. https://doi.org/10.3390/md17020085










