2-Alkyl-4-hydroxyquinolines from a Marine-Derived Streptomyces sp. Inhibit Hyphal Growth Induction in Candida albicans
Abstract
:1. Introduction
2. Results
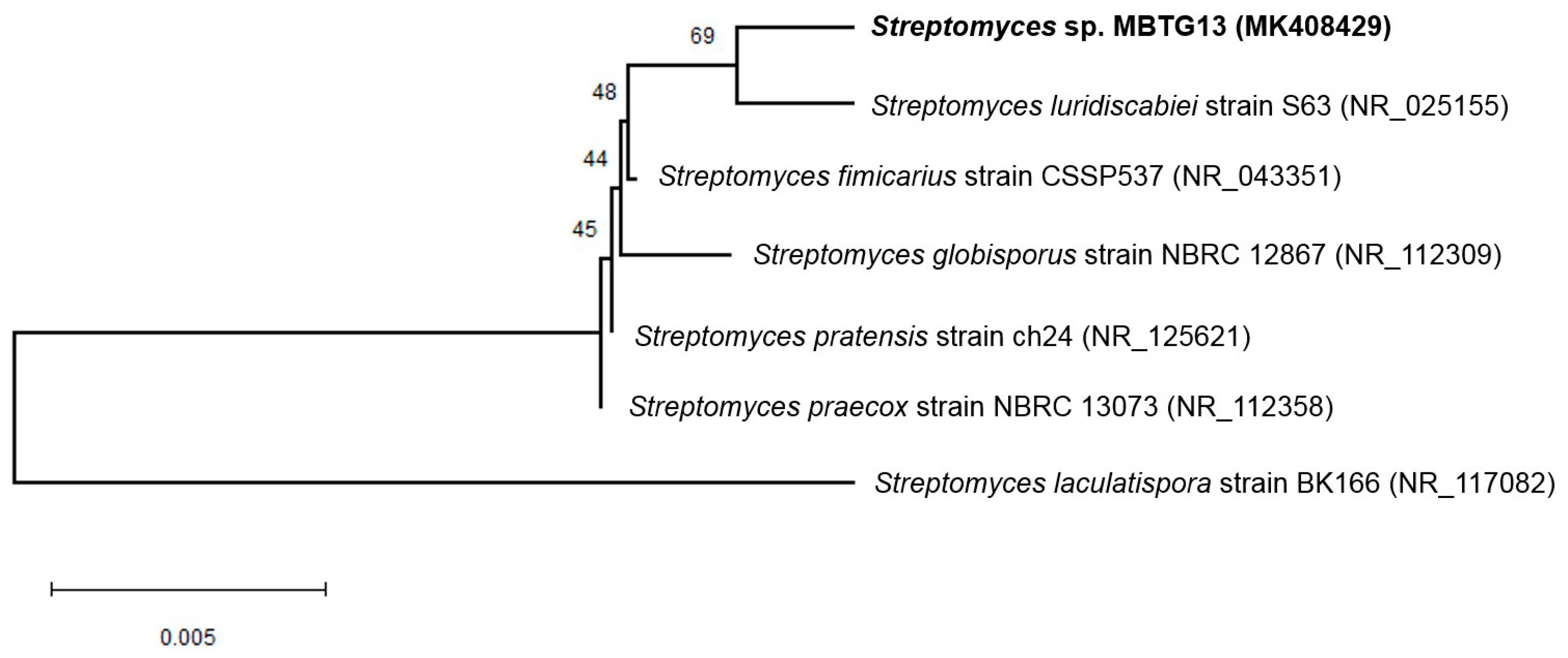
2.1. Taxonomy and Phylogenetic Analysis of MBTG13
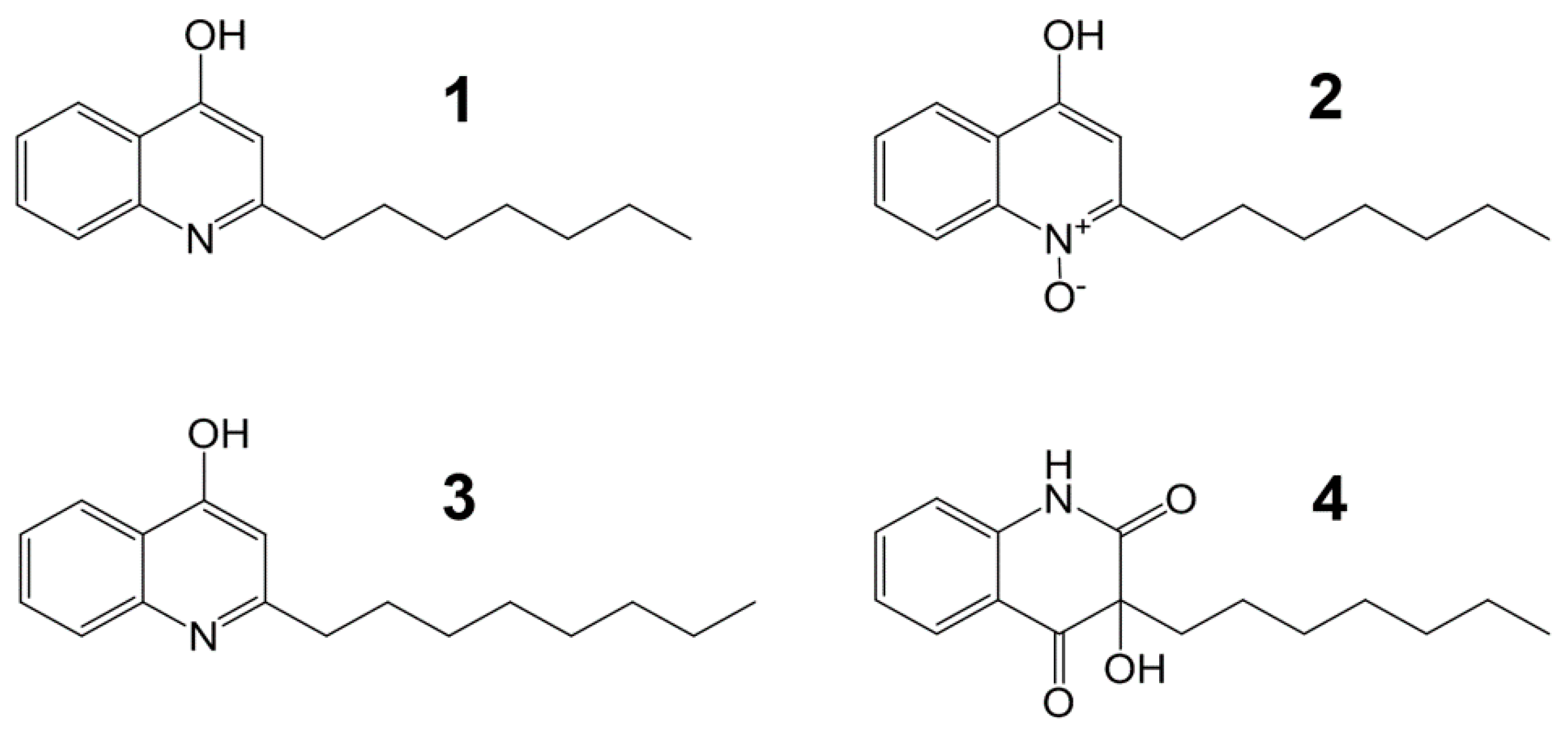
2.2. Isolation and Structural Elucidation of Compounds 1–4
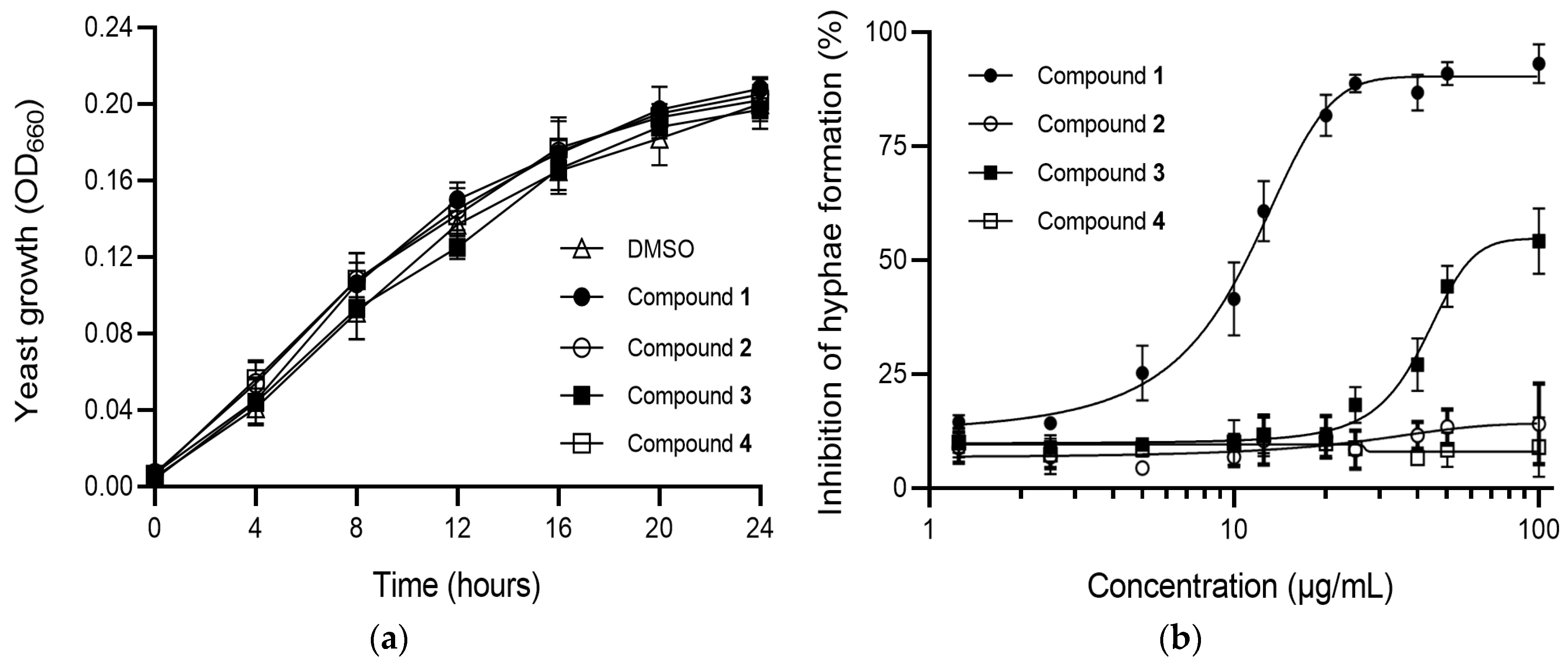
2.3. Antimicrobial Activity of Compounds 1–4
2.4. Effects of Compounds 1–4 on C. albicans Morphogenesis
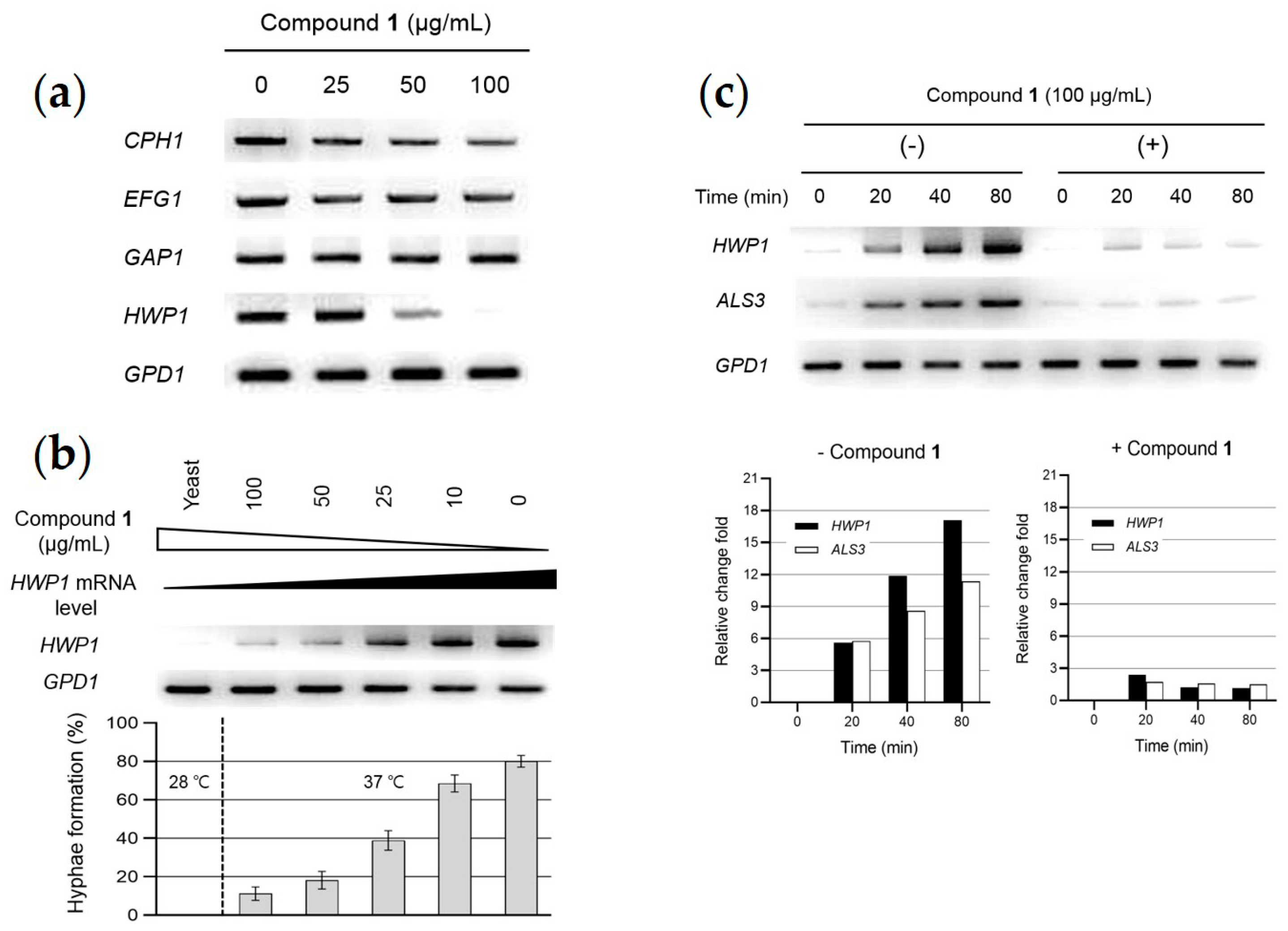
2.5. Analysis of Gene Expression Related to Hypha-Inducing Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Taxonomic Identification of the Producing Microorganism
4.3. Fermentation and Isolation
4.4. Antibacterial Activity Assay
4.5. Antifungal Activity Assay
4.6. Candida albicans Strain and Growth Medium
4.7. Candida albicans Growth and Morphological Transition Assay
4.8. Gene Expression Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Noble, S.M.; Gianetti, B.A.; Witchley, J.N. Candida albicans cell-type switching and functional plasticity in the mammalian host. Nat. Rev. Microbiol. 2017, 15, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Sudbery, P.E. Growth of Candida albicans hyphae. Nat. Rev. Microbiol. 2011, 9, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive candidiasis: A persistent public health problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Van Dijck, P.; Datta, A. Environmental sensing and signal transduction pathways regulating morphopathogenic determinants of Candida albicans. Microbiol. Mol. Biol. Rev. 2007, 71, 348–376. [Google Scholar] [CrossRef] [PubMed]
- Sayed, K.E.; Al-Said, M.S.; El-Feraly, F.S.; Ross, S.A. New quinoline alkaloids from Ruta chalepensis. J. Nat. Prod. 2000, 63, 995–997. [Google Scholar] [CrossRef]
- Zhao, N.; Li, Z.L.; Li, D.H.; Sun, Y.T.; Shan, D.T.; Bai, J.; Pei, Y.H.; Jing, Y.K.; Hua, H.M. Quinolone and indole alkaloids from the fruits of Euodia rutaecarpa and their cytotoxicity against two human cancer cell lines. Phytochemistry 2015, 109, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Cretton, S.; Breant, L.; Pourrez, L.; Ambuehl, C.; Marcourt, L.; Ebrahimi, S.N.; Hamburger, M.; Perozzo, R.; Karimou, S.; Kaiser, M.; et al. Antitrypanosomal quinoline alkaloids from the roots of Waltheria indica. J. Nat. Prod. 2014, 77, 2304–2311. [Google Scholar] [CrossRef] [PubMed]
- Kakinuma, N.; Iwai, H.; Takahashi, S.; Hamano, K.; Yanagisawa, T.; Nagai, K.; Tanaka, K.; Suzuki, K.; Kirikae, F.; Kirikae, T.; et al. Quinolactacins A, B and C: Novel quinolone compounds from Penicillium sp. EPF-6. J. Antibiot. 2000, 53, 1247–1251. [Google Scholar] [CrossRef]
- Teichert, A.; Schmidt, J.; Porzel, A.; Arnold, N.; Wessjohann, L. (Iso)-quinoline alkaloids from fungal fruiting bodies of Cortinarius subtortus. J. Nat. Prod. 2008, 71, 1092–1094. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.R.; Napier, E.J.; Fletton, R.A. G1499-2, a new quinoline compound isolated from the fermentation broth of Cytophaga johnsonii. J. Antibiot. 1978, 31, 952–958. [Google Scholar] [CrossRef]
- Kitamura, S.; Hashizume, K.; Iida, T.; Miyashita, E.; Shirahata, K.; Kase, H. Studies on lipoxygenase inhibitors. II. KF8940 (2-n-heptyl-4-hydroxyquinoline-N-oxide), a potent and selective inhibitor of 5-lipoxygenase, produced by Pseudomonas methanica. J. Antibiot. 1986, 39, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Royt, P.W.; Honeychuck, R.V.; Ravich, V.; Ponnaluri, P.; Pannell, L.K.; Buyer, J.S.; Chandhoke, V.; Stalick, W.M.; DeSesso, L.C.; Donohue, S.; et al. 4-Hydroxy-2-nonylquinoline: A novel iron chelator isolated from a bacterial cell membrane. Bioorg. Chem. 2001, 29, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Hassan, H.M.; Boonlarppradab, C.; Fenical, W. Actinoquinolines A and B, anti-inflammatory quinolone alkaloids from a marine-derived Streptomyces sp., strain CNP975. J. Antibiot. 2016, 69, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Hays, E.E.; Wells, I.C.; Katzman, P.A.; Cain, C.K.; Jacobs, F.A.; Thayer, S.A.; Doisy, E.A.; Gaby, W.L.; Roberts, E.C.; Muir, R.D.; et al. Antibiotic substances produced by Pseudomonas aeruginosa. J. Biol. Chem. 1945, 159, 725–750. [Google Scholar]
- Wells, I.C. Antibiotic substances produced by Pseudomonas aeruginosa; syntheses of pyo ib, pyo ic, and pyo III. J. Biol. Chem. 1952, 196, 331–340. [Google Scholar] [PubMed]
- Lépine, F.; Milot, S.; Déziel, E.; He, J.; Rahme, L.G. Electrospray/mass spectrometric identification and analysis of 4-hydroxy-2-alkylquinolines (HAQs) produced by Pseudomonas aeruginosa. J. Am. Soc. Mass Spectrom. 2004, 15, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Supong, K.; Thawai, C.; Supothina, S.; Auncharoen, P.; Pittayakhajonwut, P. Antimicrobial and anti-oxidant activities of quinoline alkaloids from Pseudomonas aeruginosa BCC76810. Phytochem. Lett. 2016, 17, 100–106. [Google Scholar] [CrossRef]
- Déziel, E.; Lépine, F.; Milot, S.; He, J.; Mindrinos, M.N.; Tompkins, R.G.; Rahme, L.G. Analysis of Pseudomonas aeruginosa 4-hydroxy-2-alkylquinolines (HAQs) reveals a role for 4-hydroxy-2-heptylquinoline in cell-to-cell communication. Proc. Natl. Acad. Sci. USA 2004, 101, 1339–1344. [Google Scholar] [CrossRef]
- Staab, J.F.; Bradway, S.D.; Fidel, P.L.; Sundstrom, P. Adhesive and mammalian transglutaminase substrate properties of Candida albicans Hwp1. Science 1999, 283, 1535–1538. [Google Scholar] [CrossRef]
- Sharkey, L.L.; Mcnemar, M.D.; Saporito-Irwin, S.M.; Sypherd, P.S.; Fonzi, W.A. HWP1 functions in the morphological development of Candida albicans downstream of EFG1, TUP1, and RBF1. J. Bacteriol. 1999, 181, 5273–5279. [Google Scholar]
- Kadosh, D.; Johnson, A.D. Induction of the Candida albicans filamentous growth program by relief of transcriptional repression: A genome-wide analysis. Mol. Biol. Cell 2005, 16, 2903–2912. [Google Scholar] [CrossRef] [PubMed]
- Argimón, S.; Wishart, J.A.; Leng, R.; Macaskill, S.; Mavor, A.; Alexandris, T.; Nicholls, S.; Knight, A.W.; Enjalbert, B.; Walmsley, R.; et al. Developmental regulation of an adhesion gene during cellular morphogenesis in the fungal pathogen Candida albicans. Eukaryot. Cell 2007, 6, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Oh, K.B.; Miyazawa, H.; Naito, T.; Matsuoka, H. Purification and characterization of an autoregulatory substance capable of regulating the morphological transition in Candida albicans. Proc. Natl. Acad. Sci. USA 2001, 98, 4664–4668. [Google Scholar] [CrossRef] [PubMed]
- Hornby, J.M.; Jensen, E.C.; Lisec, A.D.; Tasto, J.J.; Jahnke, B.; Shoemaker, R.; Dussault, P.; Nickerson, K.W. Quorum sensing in the dimorphic fungus Candida albicans is mediated by farnesol. Appl. Environ. Microbiol. 2001, 67, 2982–2992. [Google Scholar] [CrossRef] [PubMed]
- Bockmühl, D.P.; Ernst, J.F. A potential phosphorylation site for an A-type kinase in the Efg1 regulator protein contributes to hyphal morphogenesis of Candida albicans. Genetics 2001, 157, 1523–1530. [Google Scholar] [PubMed]
- Kebaara, B.W.; Langford, M.L.; Navarathna, D.H.; Dumitru, R.; Nickerson, K.W.; Atkin, A.L. Candida albicans Tup1 is involved in farnesol-mediated inhibition of filamentous growth induction. Eukaryot. Cell 2008, 7, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Langford, M.L.; Hargarten, J.C.; Patefield, K.D.; Marta, E.; Blankenship, J.R.; Fanning, S.; Nickerson, K.W.; Atkin, A.L. Candida albicans Czf1 and Efg1 coordinate the response to farnesol during quorum sensing, white-opaque thermal dimorphism, and cell death. Eukaryot. Cell 2013, 12, 1281–1292. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 11th ed.; M07; CLSI: Wayne, PA, USA, 2018. [Google Scholar]
- Clinical and Laboratory Standards Institute. Reference Methods for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi, 3rd ed.; M38; CLSI: Wayne, PA, USA, 2017. [Google Scholar]
- Davis-Hanna, A.; Piispanen, A.E.; Stateva, L.I.; Hogan, D.A. Farnesol and dodecanol effects on the Candida albicans Ras1-cAMP signaling pathway and the regulation of morphogenesis. Mol. Microbiol. 2008, 67, 47–62. [Google Scholar] [CrossRef]
| Compound | Minimum Inhibitory Concentration (MIC) (μg/mL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Gram(+) Bacteria | Gram(–) Bacteria | Fungi | ||||||||
| A | B | C | D | E | F | G | H | I | J | |
| 1 | 128 | 128 | >128 | >128 | >128 | 64 | >128 | >128 | >128 | >128 |
| 2 | 16 | 32 | 32 | 32 | >128 | >128 | >128 | >128 | >128 | >128 |
| 3 | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| 4 | 128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| Ampicillin | 0.07 | 0.13 | 0.13 | 0.13 | >128 | 16 | ||||
| Tetracycline | 0.5 | |||||||||
| Amphotericin B | 0.5 | 1 | 1 | 1 | ||||||
| Primer Name | Sequence |
|---|---|
| CPH1-For | 5′-GAAATGTGGCGCCGATGCAA-3′ |
| CPH1-Rev | 5′-ACCCGGCATTAGCAGTAGAT-3′ |
| EFG1-For | 5′-ACAGGCAATGCTAGCCAACA-3′ |
| EFG1-Rev | 5′-GCAGCAGTAGTAGTAGCAGC-3′ |
| GAP1-For | 5′-TTAAGTACTGGTGGACCAGC-3′ |
| GAP1-Rev | 5′-CAAACCCACTTTGAGCAAC-3′ |
| HWP1-For | 5′-GTGACAATCCTCTCAACCT-3′ |
| HWP1-Rev | 5′-GAGAGGTTTCACCGGCAGGA-3′ |
| ALS3-For | 5′-CCACTTCACAATCCCAT-3′ |
| ALS3-Rev | 5′-CAGCAGTAGTAGTAACAGTAGTAGTTTCATC-3′ |
| GPD1-For | 5′-AGTATGTGGAGCTTTACTGGGA-3′ |
| GPD1-Rev | 5′-CAGAAACACCAGCAACATCTTC-3′ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Hwang, J.-Y.; Chung, B.; Cho, E.; Bae, S.; Shin, J.; Oh, K.-B. 2-Alkyl-4-hydroxyquinolines from a Marine-Derived Streptomyces sp. Inhibit Hyphal Growth Induction in Candida albicans. Mar. Drugs 2019, 17, 133. https://doi.org/10.3390/md17020133
Kim H, Hwang J-Y, Chung B, Cho E, Bae S, Shin J, Oh K-B. 2-Alkyl-4-hydroxyquinolines from a Marine-Derived Streptomyces sp. Inhibit Hyphal Growth Induction in Candida albicans. Marine Drugs. 2019; 17(2):133. https://doi.org/10.3390/md17020133
Chicago/Turabian StyleKim, Heegyu, Ji-Yeon Hwang, Beomkoo Chung, Eunji Cho, Suhyun Bae, Jongheon Shin, and Ki-Bong Oh. 2019. "2-Alkyl-4-hydroxyquinolines from a Marine-Derived Streptomyces sp. Inhibit Hyphal Growth Induction in Candida albicans" Marine Drugs 17, no. 2: 133. https://doi.org/10.3390/md17020133
APA StyleKim, H., Hwang, J.-Y., Chung, B., Cho, E., Bae, S., Shin, J., & Oh, K.-B. (2019). 2-Alkyl-4-hydroxyquinolines from a Marine-Derived Streptomyces sp. Inhibit Hyphal Growth Induction in Candida albicans. Marine Drugs, 17(2), 133. https://doi.org/10.3390/md17020133





