Anti-Inflammatory Activity of β-thymosin Peptide Derived from Pacific Oyster (Crassostrea gigas) on NO and PGE2 Production by Down-Regulating NF-κB in LPS-Induced RAW264.7 Macrophage Cells
Abstract
1. Introduction
2. Results
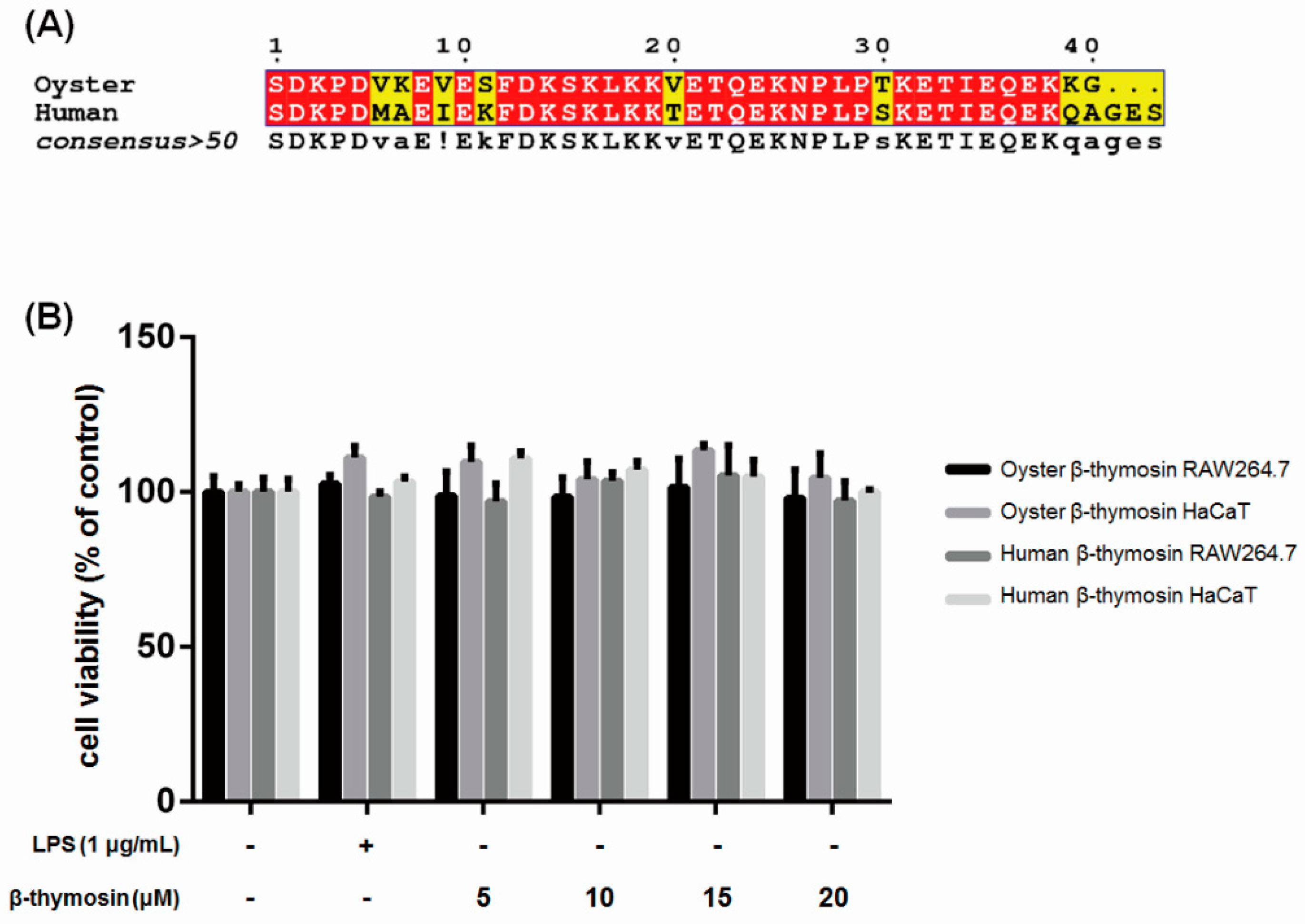
2.1. Sequence Alignment of Oyster and Human β-thymosin
2.2. Effects of β-thymosin on Cell Viability
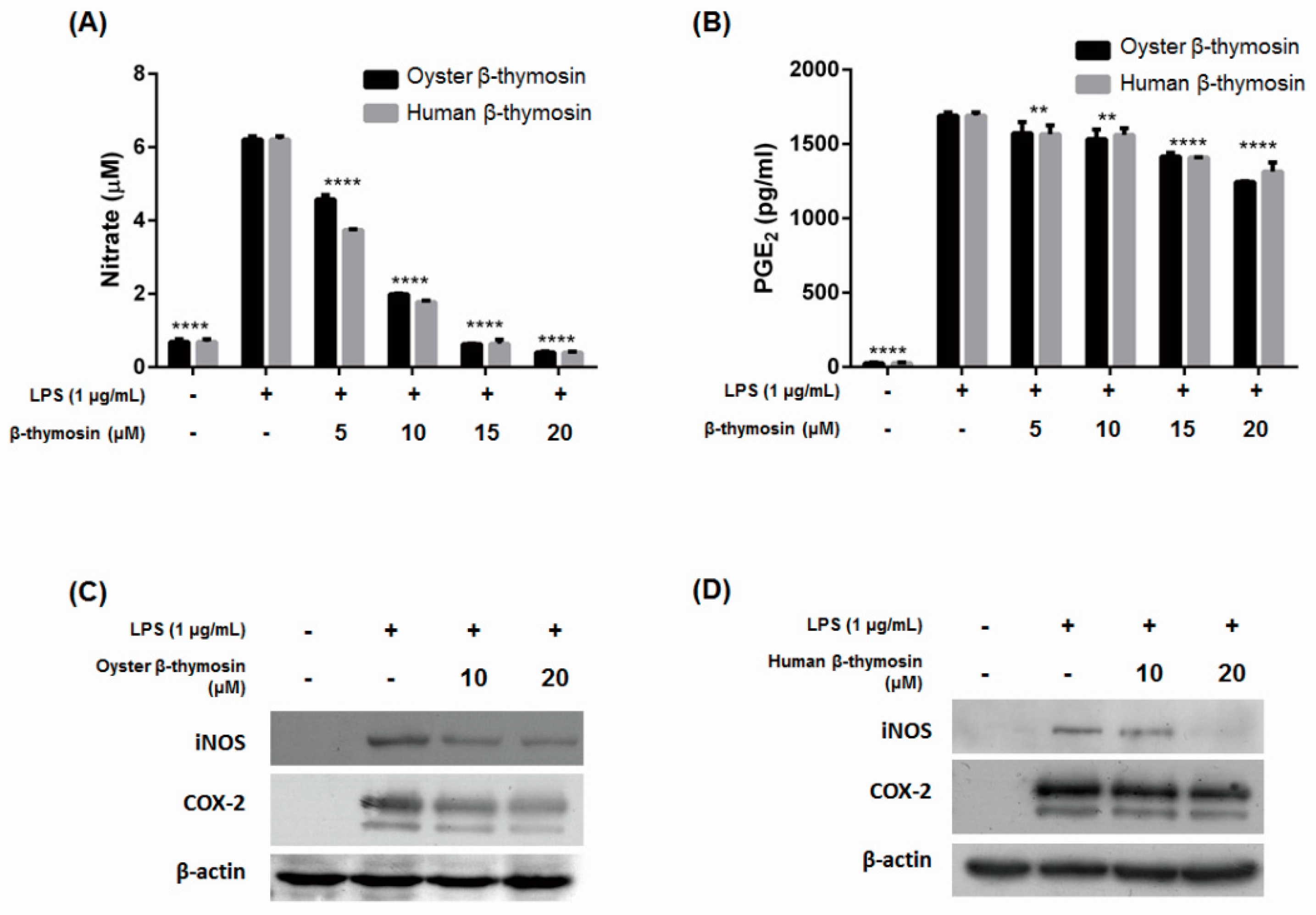
2.3. Inhibition of NO, PGE2, iNOS and COX-2 Expression by β-thymosin on LPS-Induced RAW264.7 Cells
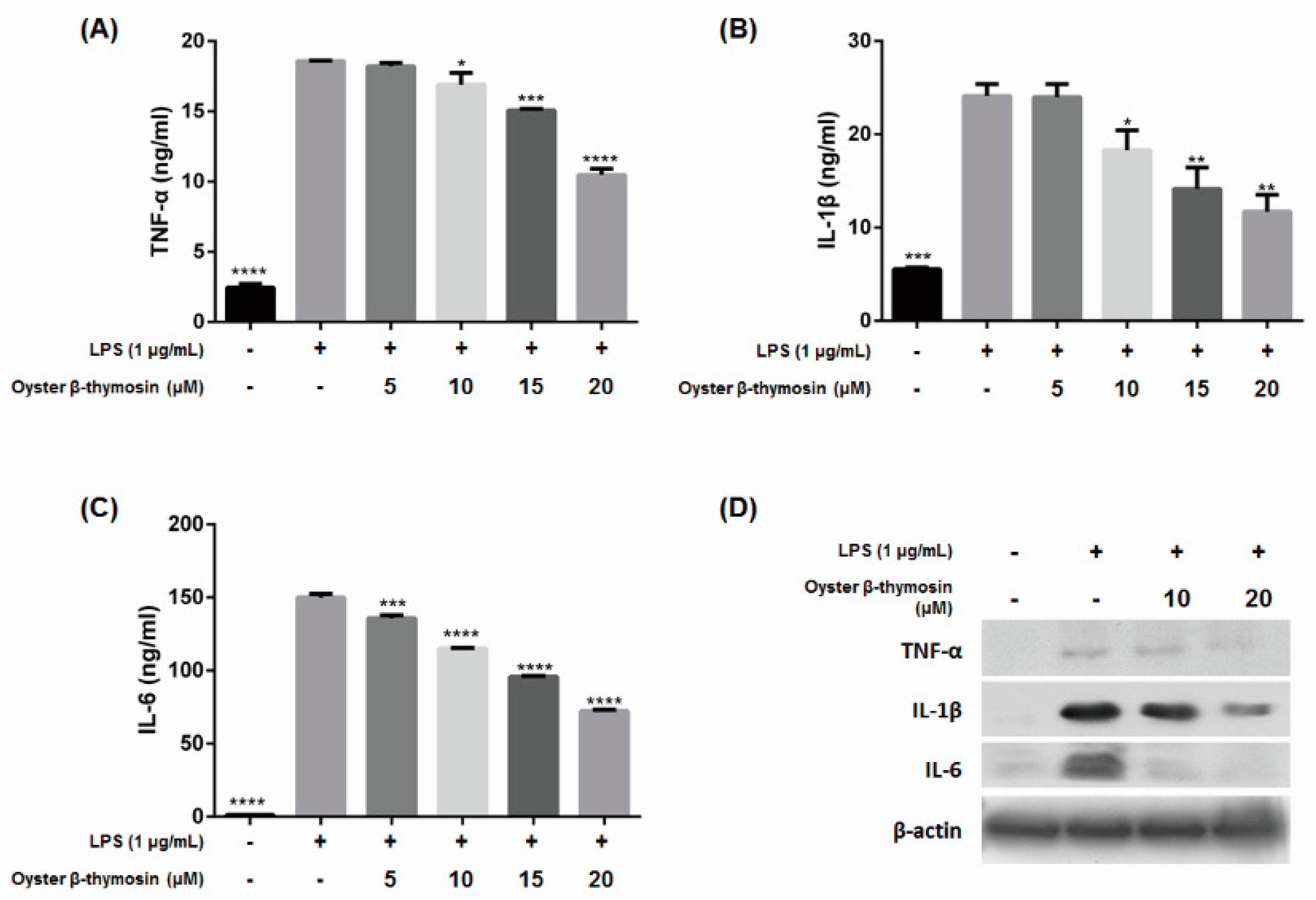
2.4. Inhibition of Cytokines Production by Oyster β-Thymosin on LPS-Stimulated RAW264.7 Cells
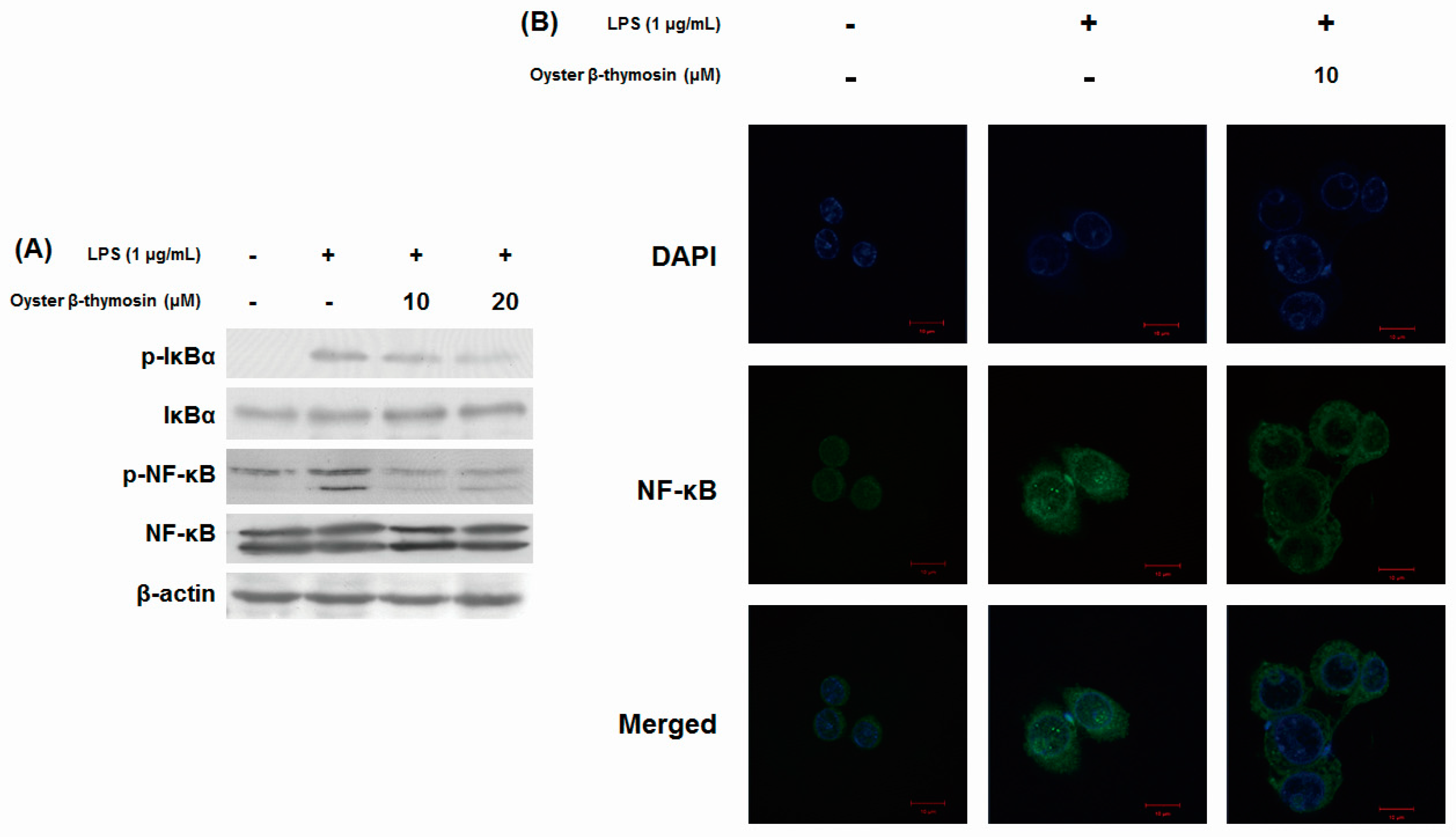
2.5. Inhibitory Effects of NF-κB Pathway by Oyster β-thymosin on LPS-stimulated RAW264.7 Cells
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Culture
4.3. Cell Viability Assay
4.4. NO Assay
4.5. PGE2 and Cytokine Production
4.6. Western Blot Analysis
4.7. Immunofluorescent Staining
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Abdulkhaleq, L.A.; Assi, M.A.; Abdullah, R.; Zamri-Saad, M.; Taufiq-Yap, Y.H.; Hezmee, M.N.M. The crucial roles of inflammatory mediators in inflammation: A review. Vet. World 2018, 11, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T.; Willoughby, D.A.; Gilroy, D.W. Anti-inflammatory lipid mediators and insights into the resolution of inflammation. Nat. Rev. Immunol. 2002, 2, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S. Alternative activation of macrophages. Nat. Rev. Immunol. 2003, 3, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Sweet, M.J.; Hume, D.A. Endotoxin signal transduction in macrophages. J. Leukoc. Biol. 1996, 60, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.S.; Mann, M.; DuBois, R.N. The role of cyclooxygenases in inflammation, cancer, and development. Oncogene 1999, 18, 7908–7916. [Google Scholar] [CrossRef] [PubMed]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef] [PubMed]
- Korhonen, R.; Lahti, A.; Kankaanranta, H.; Moilanen, E. Nitric oxide production and signaling in inflammation. Curr. Drug Targets-Inflamm. Allergy 2005, 4, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Higgs, E.A. Molecular mechanisms and therapeutic strategies related to nitric oxide. FASEB J. 1995, 9, 1319–1330. [Google Scholar] [CrossRef] [PubMed]
- Sprague, A.H.; Khalil, R.A. Inflammatory cytokines in vascular dysfunction and vascular disease. Biochem. Pharmacol. 2009, 78, 539–552. [Google Scholar] [CrossRef]
- Lee, S.I.; Yi, J.K.; Bae, W.J.; Lee, S.; Cha, H.J.; Kim, E.C. Thymosin beta-4 suppresses osteoclastic differentiation and inflammatory responses in human periodontal ligament cells. PLoS ONE 2016, 11, e0146708. [Google Scholar] [CrossRef]
- Sosne, G.; Szliter, E.A.; Barrett, R.; Kernacki, K.A.; Kleinman, H.; Hazlett, L.D. Thymosin beta 4 promotes corneal wound healing and decreases inflammation in vivo following alkali injury. Exp. Eye Res. 2002, 74, 293–299. [Google Scholar] [CrossRef]
- Lv, S.; Cheng, G.; Zhou, Y.; Xu, G. Thymosin beta4 induces angiogenesis through Notch signaling in endothelial cells. Mol. Cell. Biochem. 2013, 381, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Belsky, J.B.; Rivers, E.P.; Filbin, M.R.; Lee, P.J.; Morris, D.C. Thymosin beta 4 regulation of actin in sepsis. Expert Opin. Biol. Ther. 2018, 18, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Sosne, G.; Qiu, P.; Christopherson, P.L.; Wheater, M.K. Thymosin beta 4 suppression of corneal NFκB: A potential anti-inflammatory pathway. Exp. Eye Res. 2007, 84, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Gai, Y.; Zhao, J.; Song, L.; Wang, L.; Qiu, L.; Ning, X.; Zheng, X.; Zhang, Y.; Mu, C.; Zhang, Y.; et al. Two thymosin-repeated molecules with structural and functional diversity coexist in Chinese mitten crab Eriocheir sinensis. Dev. Comp. Immunol. 2009, 33, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wu, X. Molecular cloning and expression analysis of a β-Thymosin homologue from a gastropod abalone, Haliotis diversicolor supertexta. Fish Shellfish Immunol. 2009, 27, 379–382. [Google Scholar] [CrossRef]
- Nam, B.H.; Seo, J.K.; Lee, M.J.; Kim, Y.O.; Kim, D.G.; An, C.M.; Park, N.G. Functional analysis of Pacific oyster (Crassostrea gigas) β-thymosin: Focus on antimicrobial activity. Fish Shellfish Immunol. 2015, 45, 167–174. [Google Scholar] [CrossRef]
- Xue, Q.; Yan, Y.; Zhang, R.; Xiong, H. Regulation of iNOS on Immune Cells and Its Role in Diseases. Int. J. Mol. Sci. 2018, 19, 3805. [Google Scholar] [CrossRef]
- McAdam, B.F.; Catella-Lawson, F.; Mardini, I.A.; Kapoor, S.; Lawson, J.A.; FitzGerald, G.A. Systemic biosynthesis of prostacyclin by cyclooxygenase (COX)-2: The human pharmacology of a selective inhibitor of COX-2. Proc. Natl. Acad. Sci. USA 1999, 96, 272–277. [Google Scholar] [CrossRef]
- Tak, P.P.; Firestein, G.S. NF-κB: A key role in inflammatory diseases. J. Clin. Invest. 2001, 107, 7–11. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. Signaling to NF-κB by Toll-like receptors. Trends Mol. Med. 2007, 13, 460–469. [Google Scholar] [CrossRef] [PubMed]
- De Lorgeril, J.; Zenagui, R.; Rosa, R.D.; Piquemal, D.; Bachère, E. Whole transcriptome profiling of successful immune response to Vibrio infections in the oyster Crassostrea gigas by digital gene expression analysis. PLoS ONE 2011, 6, e23142. [Google Scholar] [CrossRef] [PubMed]
- Gueguen, Y.; Herpin, A.; Aumelas, A.; Garnier, J.; Fievet, J.; Escoubas, J.M.; Bulet, P.; Gonzalez, M.; Lelong, C.; Favrel, P.; et al. Characterization of a defensin from the oyster Crassostrea gigas Recombinant production, folding, solution structure, antimicrobial activities, and gene expression. J. Biol. Chem. 2006, 281, 313–323. [Google Scholar] [CrossRef]
- Groves, J.T.; Wang, C.C. Nitric oxide synthase: Models and mechanisms. Curr. Opin. Chem. Biol. 2000, 4, 687–695. [Google Scholar] [CrossRef]
- Clancy, R.M.; Abramson, S.B. Nitric oxide: A novel mediator of inflammation. Proc. Soc. Exp. Biol. Med. 1995, 210, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Muscará, M.N.; Wallace, J.L. Nitric Oxide. V. Therapeutic potential of nitric oxide donors and inhibitors. Am. J. Physiol. 1999, 276, G1313–G1316. [Google Scholar] [PubMed]
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and inflammation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 986–1000. [Google Scholar] [CrossRef]
- Sautebin, L. Prostaglandins and nitric oxide as molecular targets for anti-inflammatory therapy. Fitoterapia 2000, 71, S48–S57. [Google Scholar] [CrossRef]
- Shabab, T.; Khanabdali, R.; Moghadamtousi, S.Z.; Kadir, H.A.; Mohan, G. Neuroinflammation pathways: A general review. Int. J. Neurosci. 2017, 127, 624–633. [Google Scholar] [CrossRef]
- Medeiros, A.; Peres-Buzalaf, C.; Fortino Verdan, F.; Serezani, C.H. Prostaglandin E2 and the Suppression of Phagocyte Innate Immune Responses in Different Organs. Mediat. Inflamm. 2012, 2012, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Stenvinkel, P.; Ketteler, M.; Johnson, R.J.; Lindholm, B.; Pecoits-Filho, R.; Riella, M.; Heimbürger, O.; Cederholm, T.; Girndt, M. IL-10, IL-6, and TNF-α: Central factors in the altered cytokine network of uremia—the good, the bad, and the ugly. Kidney Int. 2005, 67, 1216–1233. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.M.; Ko, W.; Lee, S.J.; Kim, Y.C.; Son, J.Y.; Sohn, J.H.; Yim, J.H.; Oh, H. Anti-Inflammatory Effects of Curvularin-Type Metabolites from a Marine-Derived Fungal Strain Penicillium sp. SF-5859 in Lipopolysaccharide-Induced RAW264. 7 Macrophages. Mar. Drugs 2017, 15, 282. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.; Son, B.W.; Shin, P.G.; Choi, J.S.; Seo, Y.B.; Kim, G.D. Toluhydroquinone from Aspergillus sp. suppress inflammatory mediators via nuclear factor-κ B and mitogen-activated protein kinases pathways in lipopolysaccharide-induced RAW 264.7 cells. J. Pharm. Pharmacol. 2015, 67, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Robert, X.; Gouet, P. Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res. 2014, 42, W320–W324. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, D.; Kang, M.-j.; Jo, M.J.; Seo, Y.B.; Park, N.G.; Kim, G.-D. Anti-Inflammatory Activity of β-thymosin Peptide Derived from Pacific Oyster (Crassostrea gigas) on NO and PGE2 Production by Down-Regulating NF-κB in LPS-Induced RAW264.7 Macrophage Cells. Mar. Drugs 2019, 17, 129. https://doi.org/10.3390/md17020129
Hwang D, Kang M-j, Jo MJ, Seo YB, Park NG, Kim G-D. Anti-Inflammatory Activity of β-thymosin Peptide Derived from Pacific Oyster (Crassostrea gigas) on NO and PGE2 Production by Down-Regulating NF-κB in LPS-Induced RAW264.7 Macrophage Cells. Marine Drugs. 2019; 17(2):129. https://doi.org/10.3390/md17020129
Chicago/Turabian StyleHwang, Dukhyun, Min-jae Kang, Mi Jeong Jo, Yong Bae Seo, Nam Gyu Park, and Gun-Do Kim. 2019. "Anti-Inflammatory Activity of β-thymosin Peptide Derived from Pacific Oyster (Crassostrea gigas) on NO and PGE2 Production by Down-Regulating NF-κB in LPS-Induced RAW264.7 Macrophage Cells" Marine Drugs 17, no. 2: 129. https://doi.org/10.3390/md17020129
APA StyleHwang, D., Kang, M.-j., Jo, M. J., Seo, Y. B., Park, N. G., & Kim, G.-D. (2019). Anti-Inflammatory Activity of β-thymosin Peptide Derived from Pacific Oyster (Crassostrea gigas) on NO and PGE2 Production by Down-Regulating NF-κB in LPS-Induced RAW264.7 Macrophage Cells. Marine Drugs, 17(2), 129. https://doi.org/10.3390/md17020129




