Influence of Modified Fucoidan and Related Sulfated Oligosaccharides on Hematopoiesis in Cyclophosphamide-Induced Mice
Abstract
1. Introduction
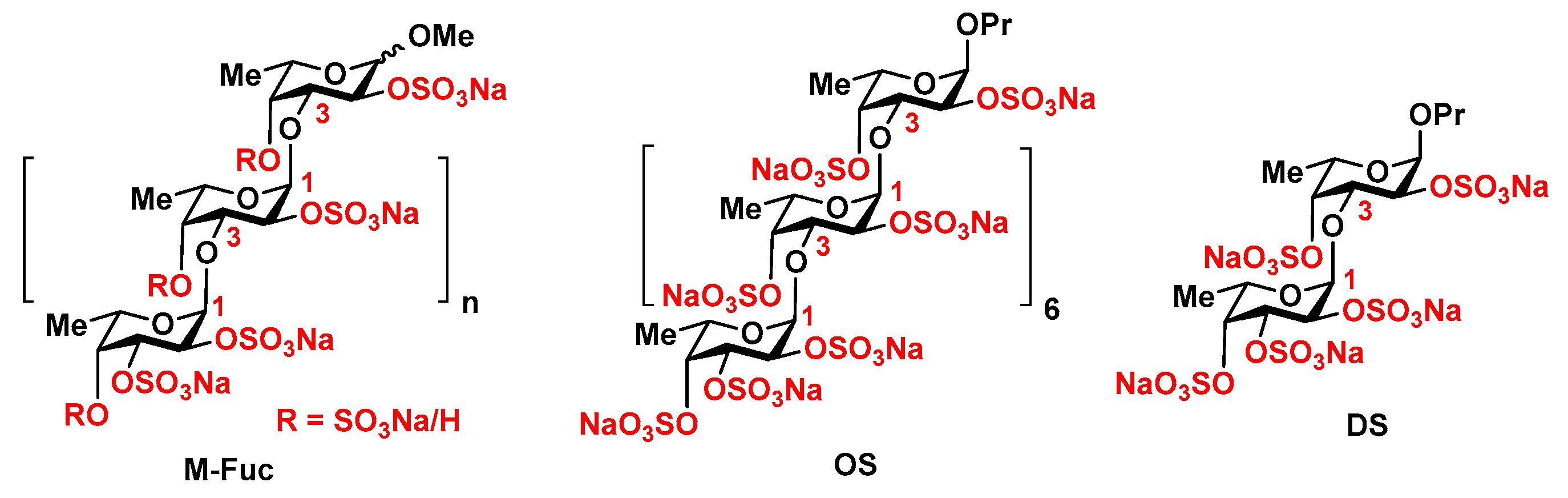
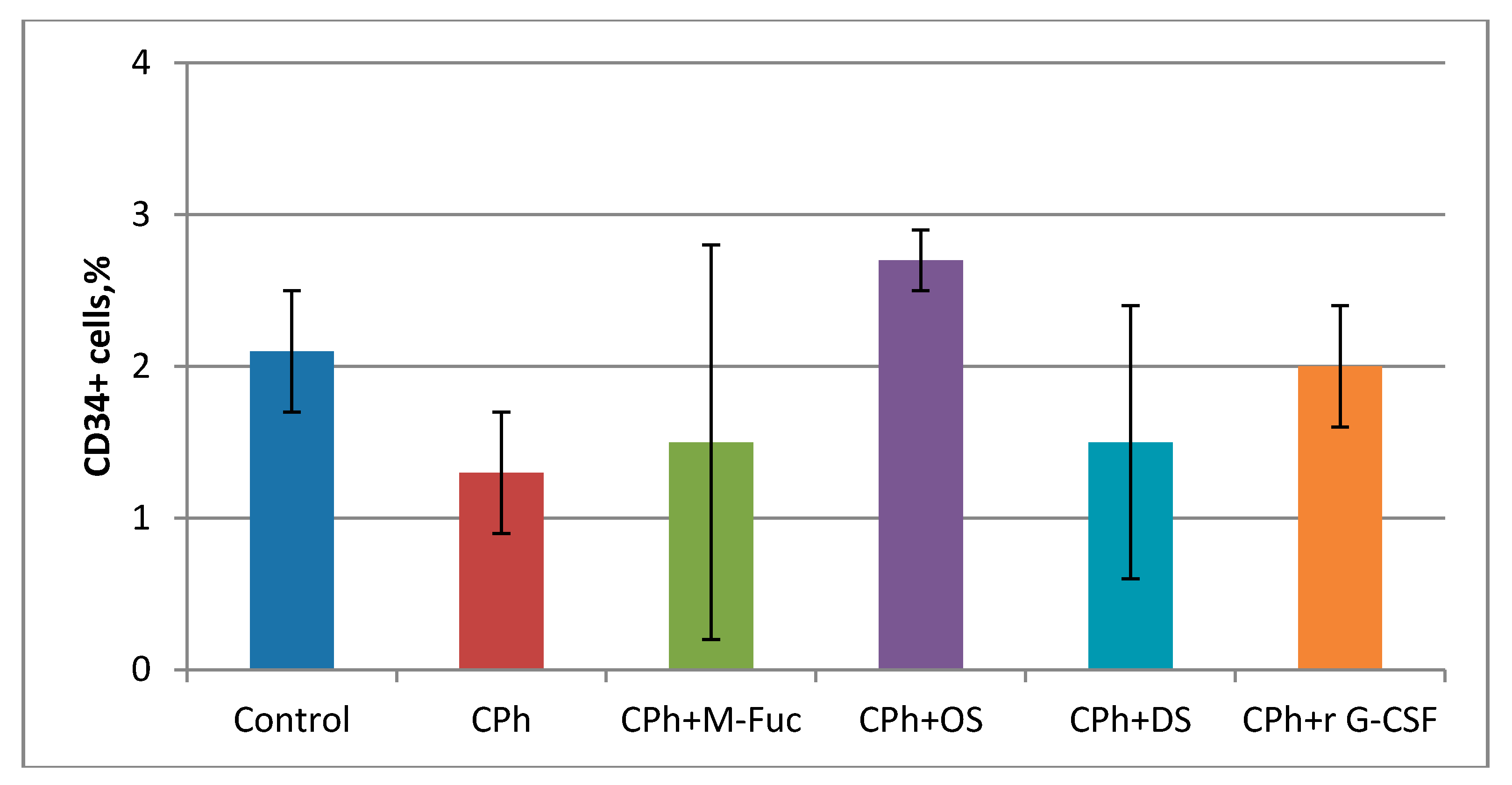
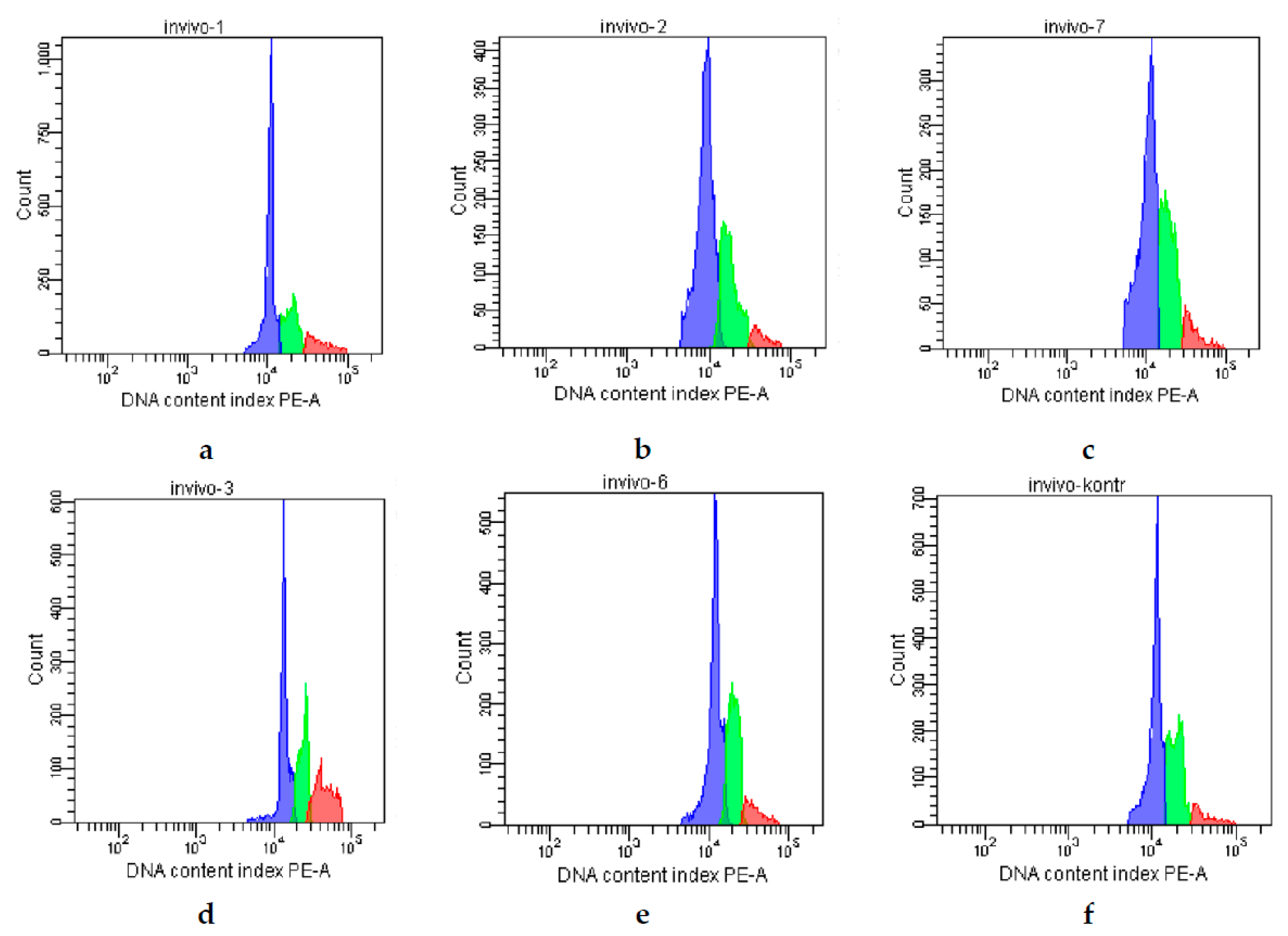
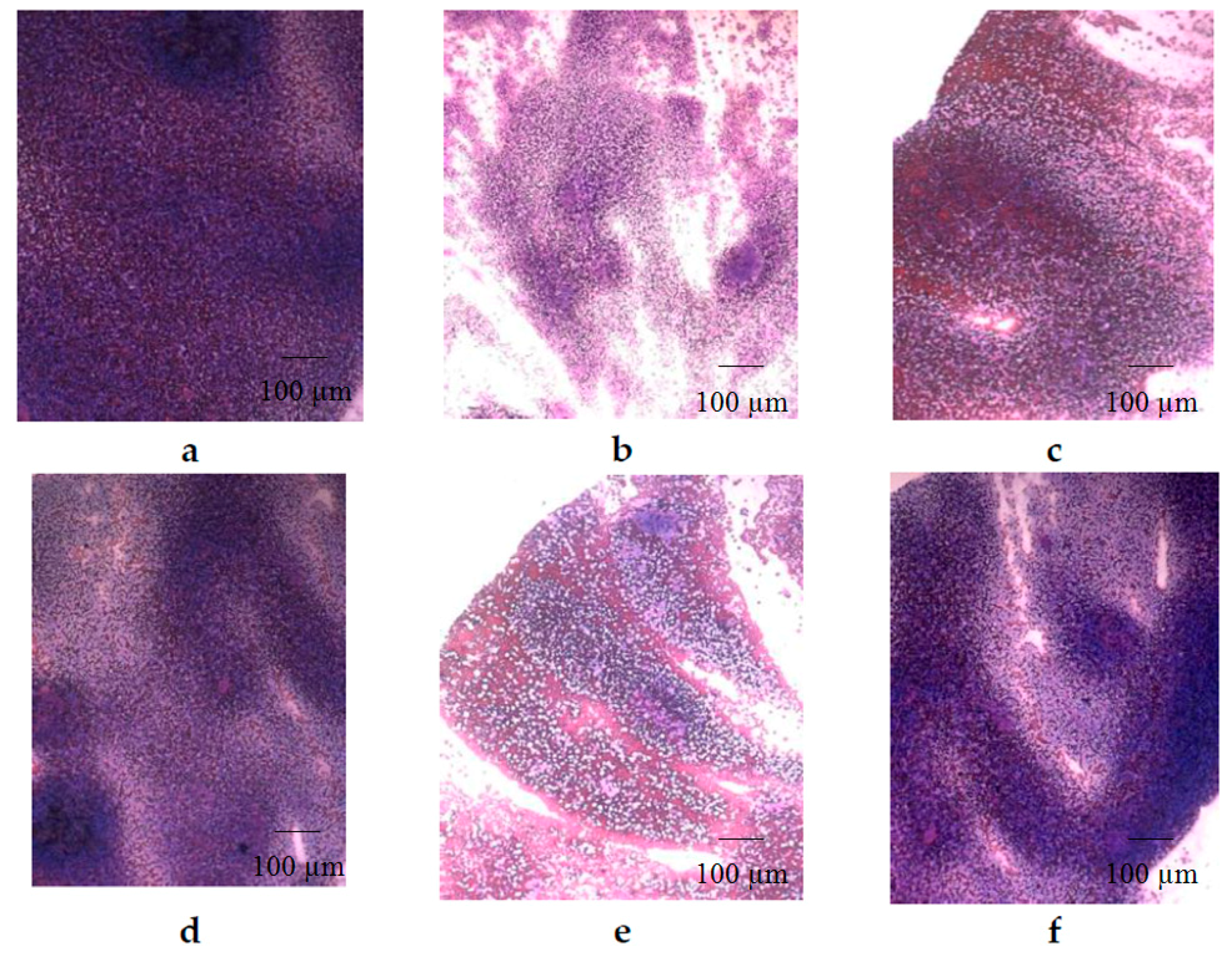
2. Results
3. Discussion
4. Materials and Methods
4.1. General Methods
4.2. Sulfated Polysaccharides
4.3. Animal Model
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nieto, Y. Pharmacodynamics of high-dose chemotherapy. Curr. Drug Metab. 2001, 2, 53–66. [Google Scholar] [PubMed]
- Schirmer, J.H.; Bremer, J.P.; Moosig, F.; Holle, J.U.; Lamprecht, P.; Wieczorek, S.; Haenisch, S.; Cascorbi, I. Cyclophosphamide treatment-induced leukopenia rates in ANCA-associated vasculitis are influenced by variant CYP450 2C9 genotypes. Pharmacogenomics 2016, 17, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Im, S.A.; Kim, K.H.; Kim, H.S.; Lee, K.H.; Shin, E.; Do, S.G.; Jo, T.H.; Park, Y.I.; Lee, C.K. Processed Aloe vera gel ameliorates cyclophosphamide-induced immunotoxicity. Int. J. Mol. Sci. 2014, 15, 19342–19354. [Google Scholar] [CrossRef] [PubMed]
- Kuter, D.J. Managing thrombocytopenia associated with cancer chemotherapy. Oncology 2015, 29, 282–294. [Google Scholar] [PubMed]
- Lo Celso, C.; Fleming, H.E.; Wu, J.W.; Zhao, C.X.; Miake-Lye, S.; Fujisaki, J.; Côté, D.; Rowe, D.W.; Lin, C.P.; Scadden, D.T. Live-animal tracking of individual haematopoietic stem/progenitor cells in their niche. Nature 2009, 457, 92–96. [Google Scholar] [CrossRef] [PubMed]
- De Grandis, M.; Lhoumeau, A.C.; Mancini, S.J.; Aurrand-Lions, M. Adhesion receptors involved in HSC and early-B cell interactions with bone marrow microenvironment. Cell. Mol. Life Sci. 2016, 73, 687–703. [Google Scholar] [CrossRef] [PubMed]
- AbuSamra, D.B.; Aleisa, F.A.; Al-Amoodi, A.S.; Jalal Ahmed, H.M.; Chin, C.J.; Abuelela, A.F.; Bergam, P.; Sougrat, R.; Merzaban, J.S. Not just a marker: CD34 on human hematopoietic stem/progenitor cells dominates vascular selectin binding along with CD44. Blood Adv. 2017, 1, 2799–2816. [Google Scholar] [CrossRef] [PubMed]
- Nabors, L.K.; Wang, L.D.; Wagers, A.J.; Kansas, G.S. Overlapping roles for endothelial selectins in murine hematopoietic stem/progenitor cell homing to bone marrow. Exp. Hematol. 2013, 41, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Kiel, M.J.; Yilmaz, O.H.; Iwashita, T.; Yilmaz, O.H.; Terhorst, C.; Morrison, S.J. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 2005, 121, 1109–1121. [Google Scholar] [CrossRef] [PubMed]
- Winkler, I.G.; Barbier, V.; Nowlan, B.; Jacobsen, R.N.; Forristal, C.E.; Patton, J.T.; Magnani, J.L.; Lévesque, J.P. Vascular niche E-selectin regulates hematopoietic stem cell dormancy, self renewal and chemoresistance. Nat. Med. 2012, 18, 1651–1657. [Google Scholar] [CrossRef] [PubMed]
- Crobu, D.; Spinetti, G.; Schrepfer, R.; Tonon, G.; Jotti, G.S.; Onali, P.; Dedoni, S.; Orsini, G.; Di Stefano, A. Preclinical and clinical phase I studies of a new recombinant Filgrastim (BK0023) in comparison with Neupogen®. BMC Pharmacol. Toxicol. 2014, 15, 7. [Google Scholar] [CrossRef] [PubMed]
- Anisimova, N.; Ustyuzhanina, N.; Donenko, F.; Bilan, M.; Usov, A.; Nifantiev, N.; Kiselevskiy, M. Fucoidan and fucosylated chondroitin sulfate stimulate hematopoiesis in cyclophosphamide-induced mice. Mar. Drugs 2017, 15, 301. [Google Scholar] [CrossRef] [PubMed]
- Bilan, M.I.; Vinogradova, E.V.; Tsvetkova, E.A.; Grachev, A.A.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. A sulfated glucuronofucan containing both fucofuranose and fucopyranose residues from the brown alga Chordaria flagelliformis. Carbohydr. Res. 2008, 343, 2605–2612. [Google Scholar] [CrossRef] [PubMed]
- Ustyuzhanina, N.E.; Bilan, M.I.; Dmitrenok, A.S.; Tsvetkova, E.A.; Nifantiev, N.E.; Usov, A.I. Modification of fucoidan from the seaweed Chordaria flagelliformis and study of anticoagulant and anti-inflammatory activity of the product. Carbohydr. Polym. under review.
- Ustuzhanina, N.E.; Krylov, V.B.; Grachev, A.A.; Gerbst, A.G.; Nifantiev, N.E. Synthesis, NMR and conformational studies of fucoidan fragments. VIII. Convergent block-wise synthesis of long chain linear and 2,3-branched oligosaccharides. Synthesis 2006, 23, 4017–4031. [Google Scholar]
- Krylov, V.B.; Kaskova, Z.M.; Vinnitskiy, D.Z.; Ustyuzhanina, N.E.; Grachev, A.A.; Chizhov, A.O.; Nifantiev, N.E. Acid-promoted total sulfation of fucoidan fragments. Carbohydr. Res. 2011, 346, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Bilan, M.I.; Grachev, A.A.; Ustuzhanina, N.E.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. A highly regular fraction of a fucoidan from the brown seaweed Fucus distichus L. Carbohydr. Res. 2004, 339, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Khatuntseva, E.A.; Ustuzhanina, N.E.; Zatonskii, G.V.; Shashkov, A.S.; Usov, A.I.; Nifant’ev, N.E. Synthesis, NMR and conformational studies of fucoidan fragments 1: Desulfated 2,3-and 3,4-branched trisaccharide fragments and constituting disaccharides. J. Carbohydr. Chem. 2000, 19, 1151–1173. [Google Scholar] [CrossRef]
- Anisimova, N.Yu.; Ustyuzhanina, N.E.; Donenko, F.V.; Bilan, M.I.; Ushakova, N.A.; Usov, A.I.; Nifantiev, N.E.; Kiselevskiy, M.V. Influence of fucoidans and their derivatives on antitumor and phagocytic activity of human blood leucocytes. Biochemistry 2015, 80, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Frenette, P.S.; Weiss, L. Sulfated glycans induce rapid hematopoietic progenitor cell mobilization: Evidence for selectin-dependent and independent mechanisms. Blood 2000, 96, 2460–2468. [Google Scholar] [PubMed]
- Sugiyama, T.; Kohara, H.; Noda, M.; Nagasawa, T. Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity 2006, 25, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, A.; Peired, A.J.; Weiss, L.A.; Katayama, Y.; Frenette, P.S. The integrin alphaMbeta2 anchors hematopoietic progenitors in the bone marrow during enforced mobilization. Blood 2004, 104, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Pomin, V.H. Fucanomics and galactanomics: Current status in drug discovery, mechanisms of action and role of the well-defined structures. Biochim. Biophys. Acta 2012, 1820, 1971–1999. [Google Scholar] [CrossRef] [PubMed]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.A.; et al. Comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Panagos, C.G.; Thomson, D.S.; Moss, C.; Hoghes, A.D.; Kelly, M.S.; Liu, Y.; Chai, W.; Venkatasamy, R.; Spina, D.; Page, C.P.; et al. Fucosylated chondroitin sulfates from the body wall of the sea cucumber Holothuria forskali. Conformation, selectin binding, and biological activity. J. Biol. Chem. 2014, 289, 28284–28298. [Google Scholar] [CrossRef] [PubMed]
- Hayes, A.J.; Melrose, J. Glycans and glycosaminoglycans in neurobiology: Key regulators of neuronal cell function and fate. Biochem. J. 2018, 475, 2511–2545. [Google Scholar] [CrossRef] [PubMed]
- Ananikov, V.P.; Eremin, D.B.; Yakukhnov, S.A.; Dilman, A.D.; Levin, V.V.; Egorov, M.P.; Karlov, S.S.; Kustov, L.M.; Tarasov, A.L.; Greish, A.A.; et al. Organic and hybrid systems: from science to practice. Mend. Commun. 2017, 27, 425–438. [Google Scholar] [CrossRef]
| Groups | WBC (×103/µL) | RBC (×106/µL) | Hemoglobin (g/dL) | Platelets (×103/µL) |
|---|---|---|---|---|
| Control | 6.7 ± 1.14 | 9.4 ± 0.45 | 16.6 ± 0.64 | 571.5 ± 54.71 |
| CPh | 2.4 ± 0.36 | 5.4 ± 0.07 | 11.3 ± 1.73 | 812.3 ± 46.35 |
| CPh + M-Fuc | 1.9 ± 1.20 | 5.4 ± 2.95 | 10.2 ± 5.52 | 589.0 ± 350.06 |
| CPh + OS | 5.3 ± 0.93 | 9.1 ± 1.11 | 16.7 ± 2.33 | 1036.3 ± 104.29 |
| CPh + DS | 2.3 ± 1.82 | 4.7 ± 2.89 | 8.7 ± 5.47 | 389.6 ± 207.74 |
| CPh + r G-CSF | 3.7 ± 0.47 | 7.0 ± 0.77 | 13.0 ± 1.30 | 541.0 ± 44.20 |
| Groups | Neutrophils (×103/µL) | Monocytes (×103/µL) | Lymphocytes (×103/µL) |
|---|---|---|---|
| Control | 1.9 ± 0.21 | 0.5 ± 0.01 | 4.4 ± 1.32 |
| CPh | 0.4 ± 0.25 | 0.2 ± 0.12 | 1.5 ± 0.38 |
| CPh + M-Fuc | 0.5 ± 0.39 | 0.2 ± 0.10 | 1.2 ± 0.71 |
| CPh + OS | 2.1 ± 0.88 | 0.5 ± 0.22 | 2.3 ± 0.02 |
| CPh + DS | 1.3 ± 1.22 | 0.1 ± 0.01 | 0.6 ± 0.32 |
| CPh + r G-CSF | 1.1 ± 0.74 | 0.4 ± 0.09 | 2.1 ± 0.47 |
| Groups | CD3+CD4+ (%) | CD3+CD8+ (%) | The Ratio CD4+/CD8+ | NK (%) |
|---|---|---|---|---|
| Control | 31 ± 0.7 | 16 ± 3.8 | 1.94 ± 0.28 | 1.3 ± 0.8 |
| CPh | 14 ± 3.4 | 16 ± 3.1 | 0.88 ± 0.10 | 2.1 ± 2.0 |
| CPh + M-Fuc | 16 ± 3.0 | 15 ± 2.2 | 1.07 ± 0.10 | 0.4 ± 1.2 |
| CPh + OS | 29 ± 2.4 | 15 ± 4.1 | 1.93 ± 0.19 | 2.3 ± 1.1 |
| CPh + DS | 24 ± 2.8 | 17 ± 1.4 | 1.41 ± 0.07 | 1.3 ± 0.2 |
| CPh + r G-CSF | 22 ± 0.8 | 16 ± 2.1 | 1.38 ± 0.50 | 3.2 ± 1.1 |
| Groups | CD11c+ | CD62p+ | ||
|---|---|---|---|---|
| % | p | % | p | |
| Control | 44 ± 5.4 | - | 48 ± 2.7 | - |
| CPh | 68 ± 8.1 | 0.057 | 70 ± 6.8 | 0.029 |
| CPh + M-Fuc | 27 ± 3.1 | 0.047 | 8 ± 2.4 | 0.001 |
| CPh + OS | 34 ± 6.2 | 0.278 | 38 ± 4.1 | 0.097 |
| CPh + DS | 22 ± 3.9 | 0.021 | 23 ± 2.5 | 0.002 |
| CPh + r G-CSF | 13 ± 7.5 | 0.02 | 14 ± 8.4 | 0.012 |
| Groups | E. coli+ Cells | ROS+ Cells | ||
|---|---|---|---|---|
| % | p | % | p | |
| Control | 32 ± 3.4 | - | 31 ± 1.6 | - |
| CPh | 54 ± 4.1 | 0.009 | 36 ± 0.1 | 0.026 |
| CPh + M-Fuc | 57 ± 3.1 | 0.003 | 35 ± 1.4 | 0.119 |
| CPh + OS | 43 ± 6.2 | 0.181 | 29 ± 1.1 | 0.350 |
| CPh + DS | 52 ± 3.9 | 0.012 | 47 ± 1.8 | 0.001 |
| CPh + r G-CSF | 29 ± 3.5 | 0.056 | 51 ± 0.8 | 0.001 |
| Groups | G0/G1 (%) | S (%) | G2/M (%) |
|---|---|---|---|
| Control | 63 | 23 | 14 |
| CPh | 65 | 29 | 4 |
| CPh + M-Fuc | 68 | 24 | 8 |
| CPh + OS | 47 | 28 | 25 |
| CPh + DS | 58 | 34 | 8 |
| CPh + r G-CSF | 60 | 32 | 8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anisimova, N.Y.; Ustyuzhanina, N.E.; Bilan, M.I.; Donenko, F.V.; Ushakova, N.A.; Usov, A.I.; Kiselevskiy, M.V.; Nifantiev, N.E. Influence of Modified Fucoidan and Related Sulfated Oligosaccharides on Hematopoiesis in Cyclophosphamide-Induced Mice. Mar. Drugs 2018, 16, 333. https://doi.org/10.3390/md16090333
Anisimova NY, Ustyuzhanina NE, Bilan MI, Donenko FV, Ushakova NA, Usov AI, Kiselevskiy MV, Nifantiev NE. Influence of Modified Fucoidan and Related Sulfated Oligosaccharides on Hematopoiesis in Cyclophosphamide-Induced Mice. Marine Drugs. 2018; 16(9):333. https://doi.org/10.3390/md16090333
Chicago/Turabian StyleAnisimova, Natalia Yu., Nadezhda E. Ustyuzhanina, Maria I. Bilan, Fedor V. Donenko, Natalia A. Ushakova, Anatolii I. Usov, Mikhail V. Kiselevskiy, and Nikolay E. Nifantiev. 2018. "Influence of Modified Fucoidan and Related Sulfated Oligosaccharides on Hematopoiesis in Cyclophosphamide-Induced Mice" Marine Drugs 16, no. 9: 333. https://doi.org/10.3390/md16090333
APA StyleAnisimova, N. Y., Ustyuzhanina, N. E., Bilan, M. I., Donenko, F. V., Ushakova, N. A., Usov, A. I., Kiselevskiy, M. V., & Nifantiev, N. E. (2018). Influence of Modified Fucoidan and Related Sulfated Oligosaccharides on Hematopoiesis in Cyclophosphamide-Induced Mice. Marine Drugs, 16(9), 333. https://doi.org/10.3390/md16090333







