Biochemical Characterization and Degradation Pattern of a Novel Endo-Type Bifunctional Alginate Lyase AlyA from Marine Bacterium Isoptericola halotolerans
Abstract
1. Introduction
2. Results and Discussions
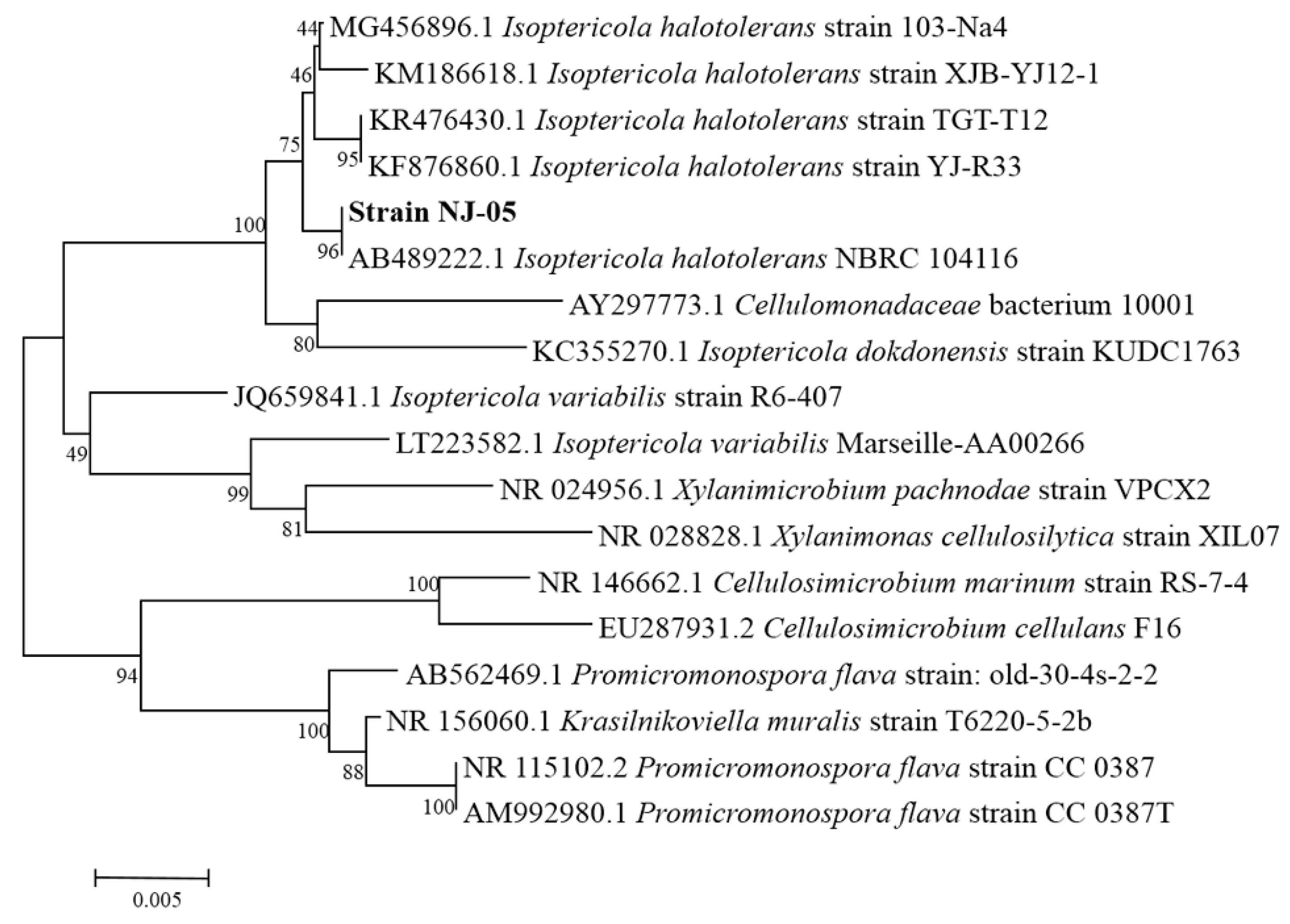
2.1. Screening and Identification of Strain NJ-05
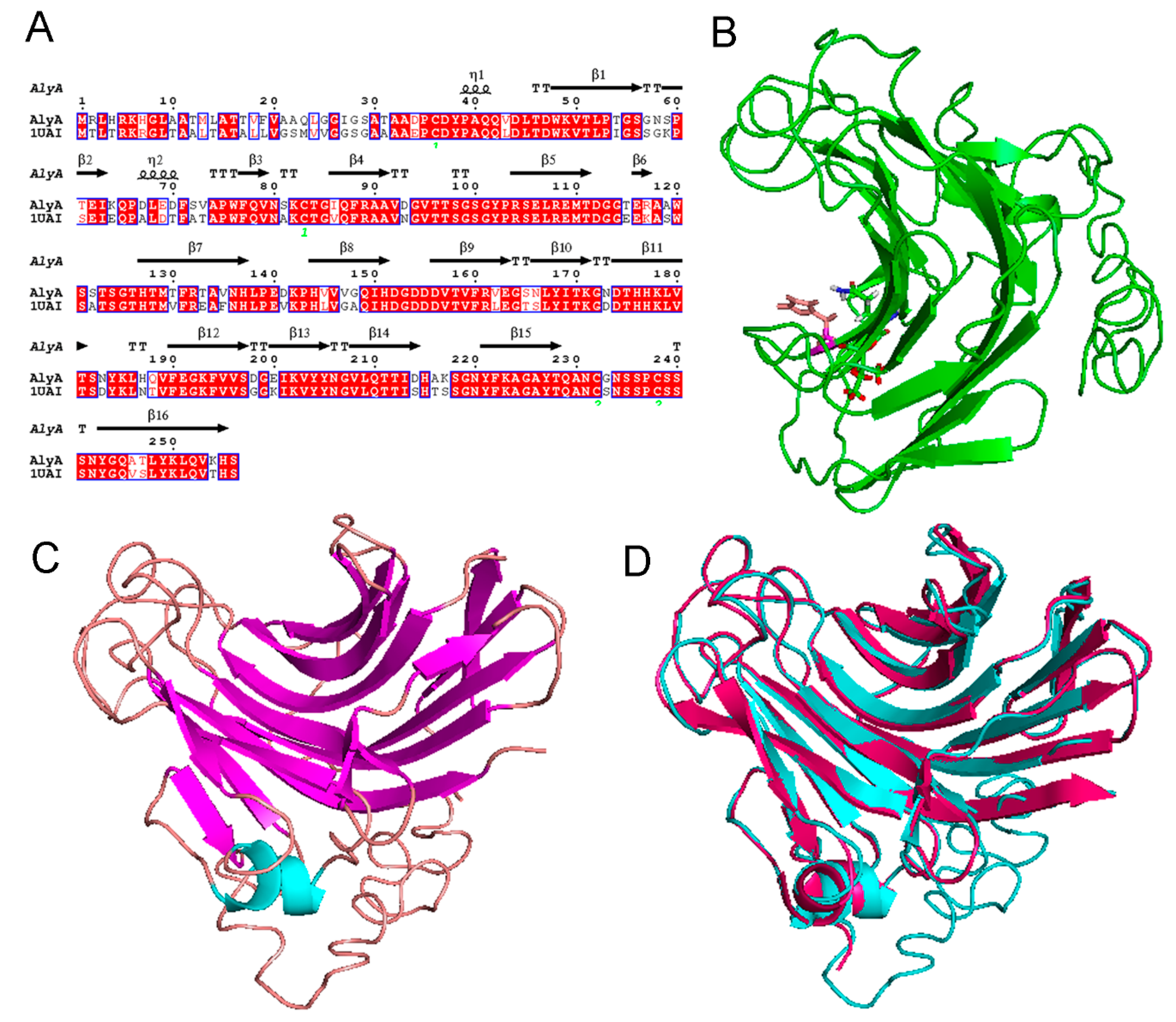
2.2. Sequence Analysis of Alginate Lyase
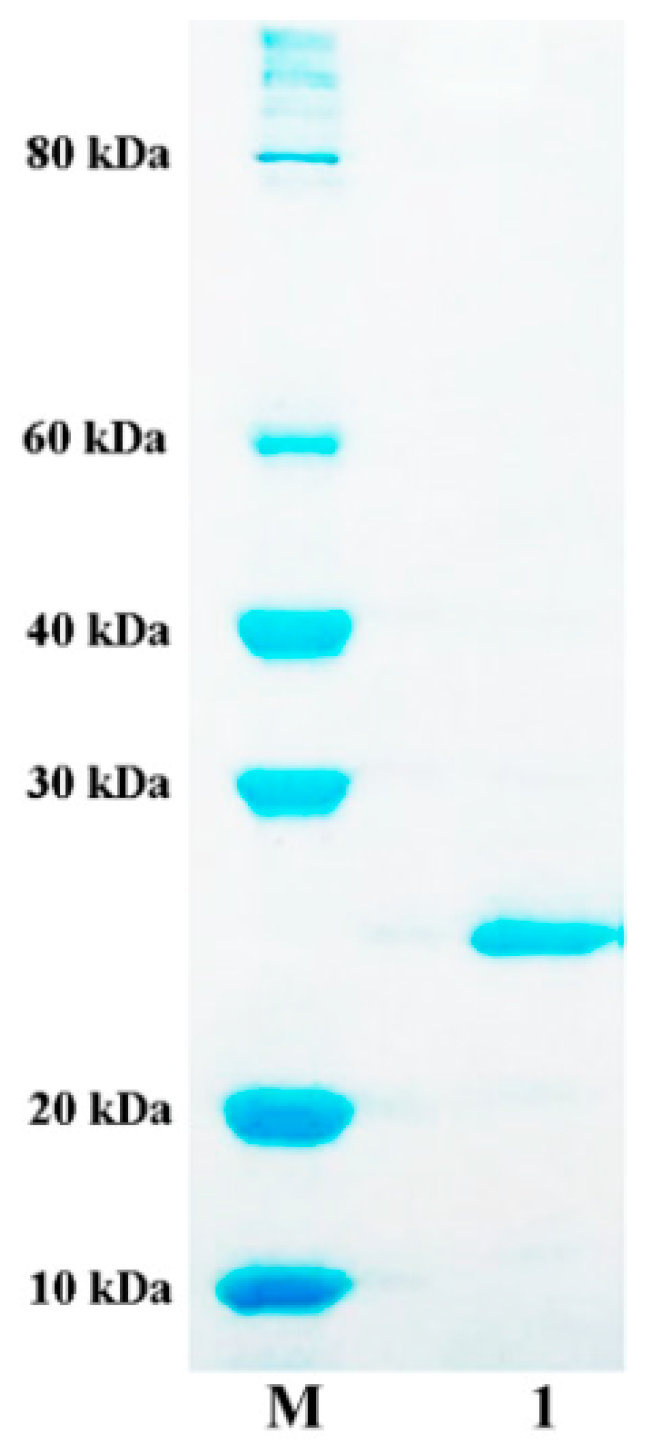
2.3. Expression and Purification of AlyA
2.4. Substrate Specifity and Enzymatic Kinetics of the Enzyme
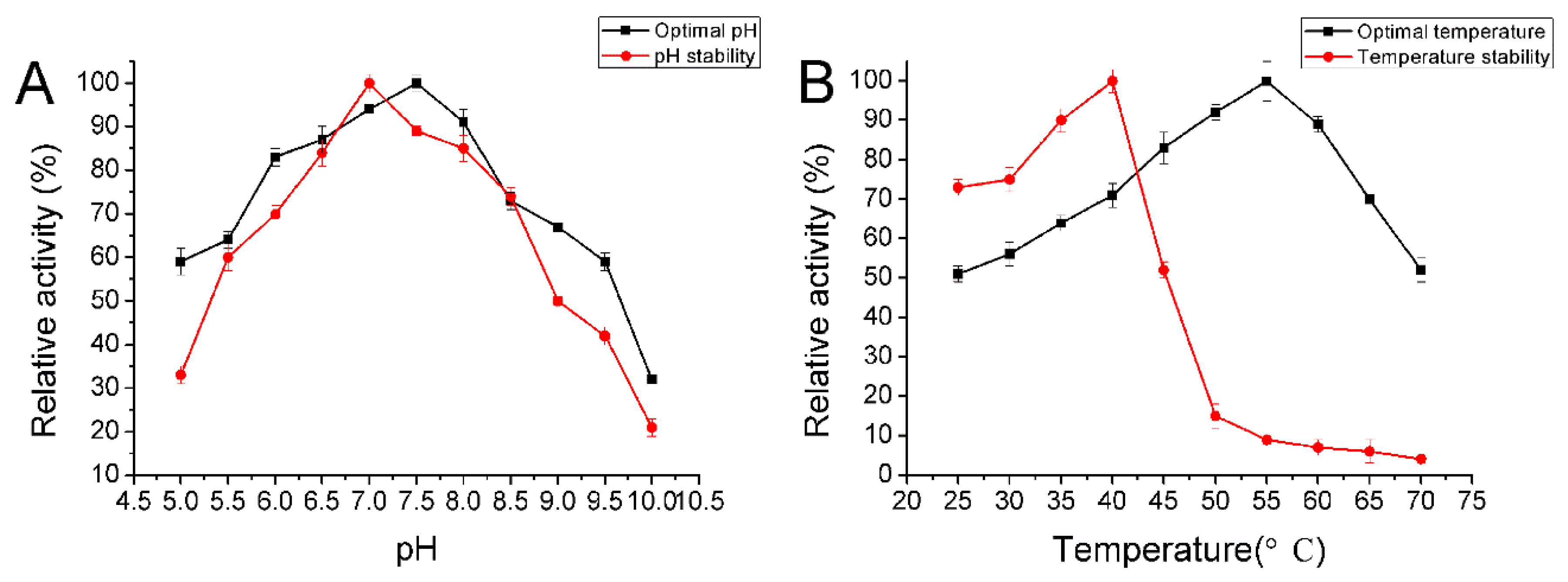
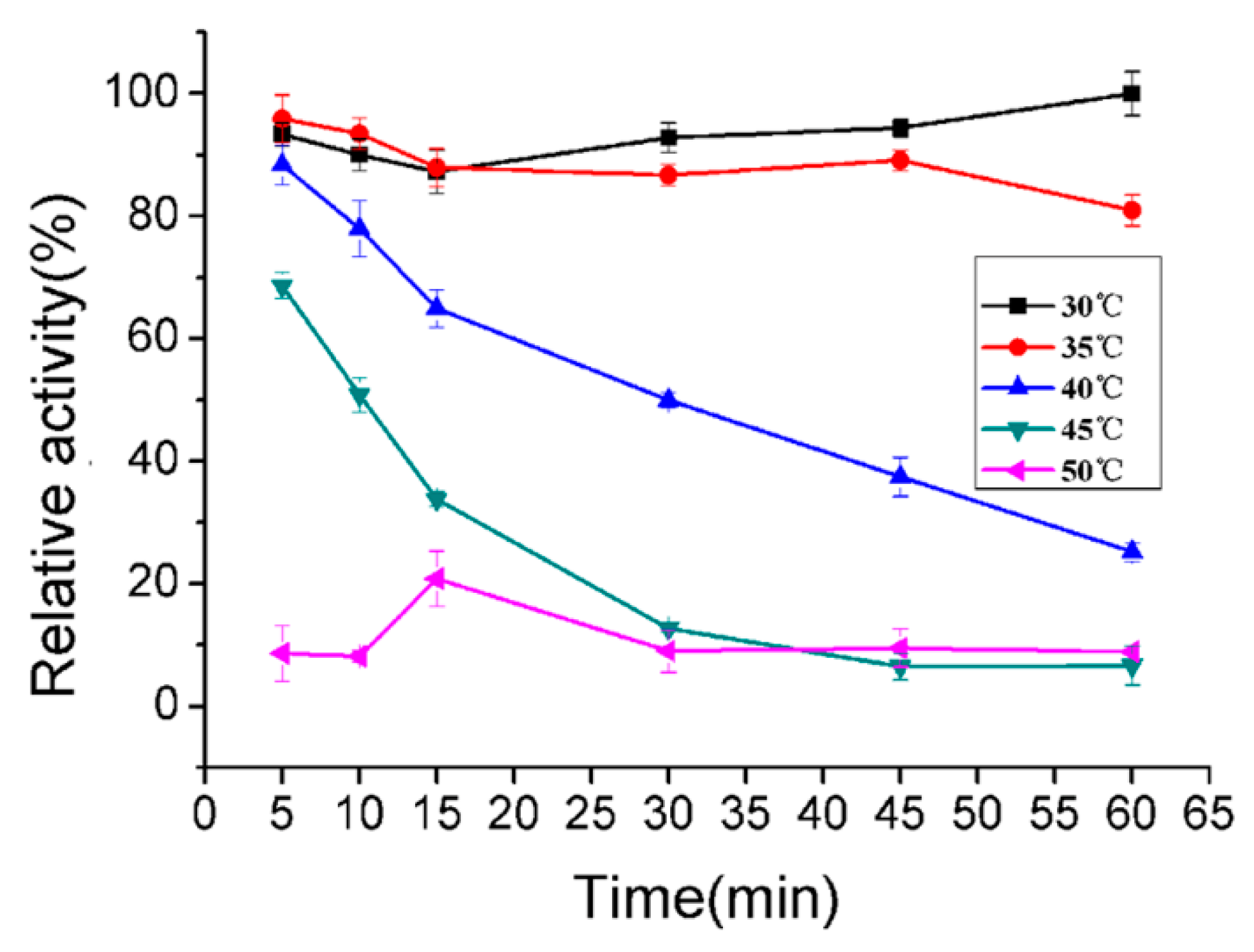
2.5. Biochemical Characterization of AlyA
3. Materials and Methods
3.1. Materials
3.2. Screening and Identification of Strain NJ-05
3.3. Cloning, Expression, and Purification of the Alginate Lyase
3.4. Enzyme Activity Assay
3.5. Substrate Specificity and Kinetic Measurement of Alginate Lyase
3.6. Biochemical Characterization of AlyA
3.7. Molecular Modeling and Structural Alignment
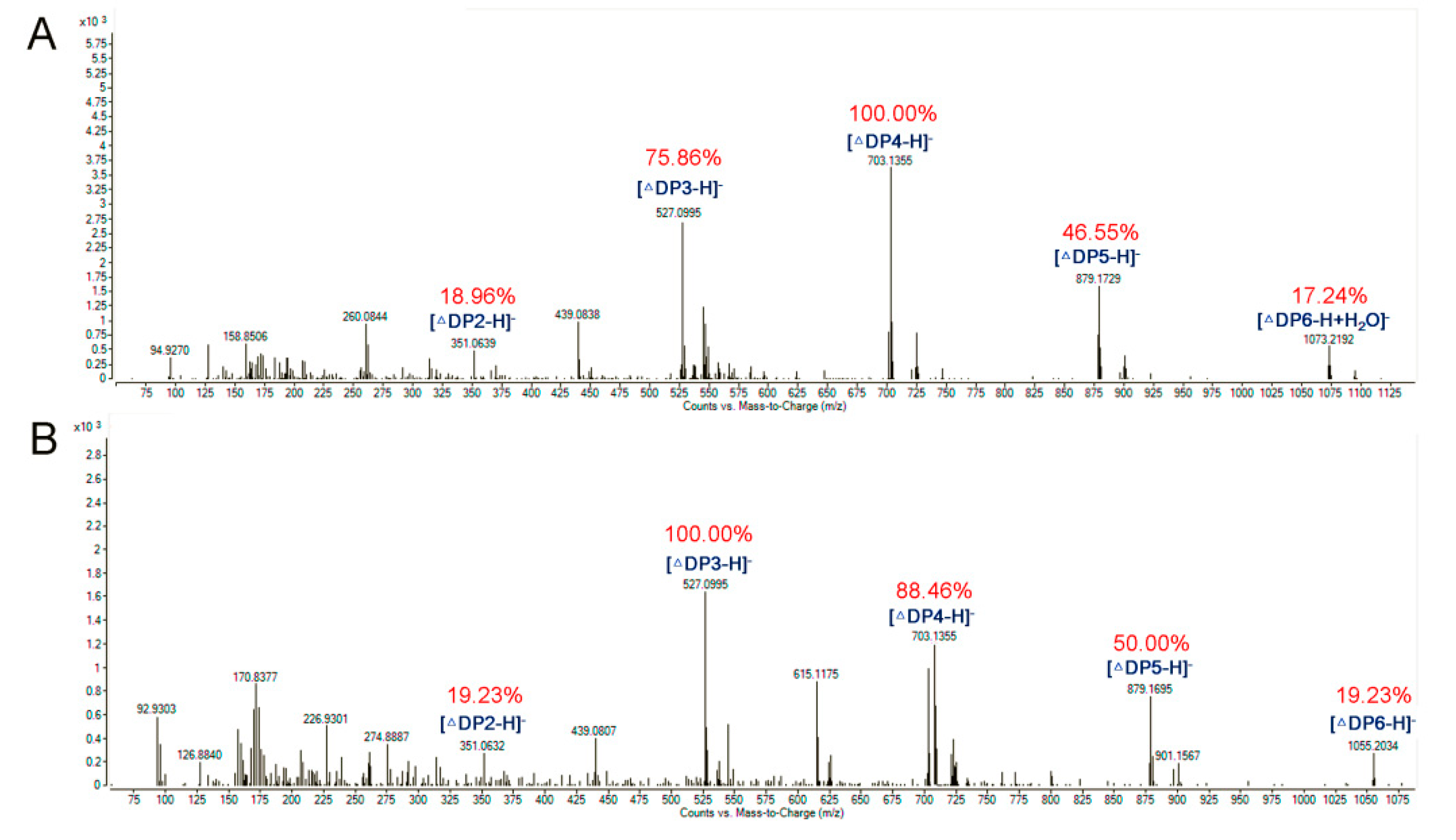
3.8. ESI-MS Analysis of the Degradation Products of AlyA
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gacesa, P. Enzymic degradation of alginates. Int. J. Biochem. 1992, 24, 545–552. [Google Scholar] [CrossRef]
- Pawar, S.N.; Edgar, K.J. Alginate derivatization: A review of chemistry, properties and applications. Biomaterials 2012, 33, 3279–3305. [Google Scholar] [CrossRef] [PubMed]
- Mabeau, S.; Kloareg, B. Isolation and analysis of the cell walls of brown algae: Fucus spiralis, F. ceranoides, F. vesiculosus, F. Serratus, Bifurcaria bifurcata and Laminaria digitata. J. Exp. Bot. 1987, 38, 1573–1580. [Google Scholar]
- Fujihara, M.; Nagumo, T. An influence of the structure of alginate on the chemotactic activity of macrophages and the antitumor activity. Carbohydr. Res. 1993, 243, 211–216. [Google Scholar] [CrossRef]
- Otterlei, M.; Ostgaard, K.; Skjak-Braek, G.; Smidsrod, O.; Soon-Shiong, P.; Espevik, T. Induction of cytokine production from human monocytes stimulated with alginate. J. Immunother. 1991, 10, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Bergero, M.F.; Liffourrena, A.S.; Opizzo, B.A.; Fochesatto, A.S.; Lucchesi, G.I. Immobilization of a microbial consortium on Ca-alginate enhances degradation of cationic surfactants in flasks and bioreactor. Int. Biodeter. Biodegr. 2017, 117, 39–44. [Google Scholar] [CrossRef]
- Yang, J.S.; Xie, Y.J.; He, W. Research progress on chemical modification of alginate: A review. Carbohydr. Polym. 2011, 84, 33–39. [Google Scholar] [CrossRef]
- Tai, H.B.; Tang, L.W.; Chen, D.D.; Irbis, C.; Bioconvertion, L.O. Progresses on preparation of alginate oligosaccharide. Life Sci. Res. 2015, 19, 75–79. [Google Scholar]
- Yang, J.H.; Bang, M.A.; Jang, C.H.; Jo, G.H.; Jung, S.K.; Ki, S.H. Alginate oligosaccharide enhances LDL uptake via regulation of LDLR and PCSK9 expression. J. Nutr. Biochem. 2015, 26, 1393–1400. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, M.; Kurachi, M.; Nakashima, T.; Kim, D.; Yamaguchi, K.; Oda, T.; Iwamoto, Y.; Muramatsu, T. Structure-activity relationship of alginate oligosaccharides in the induction of cytokine production from RAW264.7 cells. FEBS Lett. 2005, 579, 4423–4429. [Google Scholar] [PubMed]
- Yamamoto, Y.; Kurachi, M.; Yamaguchi, K.; Oda, T. Induction of multiple cytokine secretion from RAW264.7 cells by alginate oligosaccharides. Biosci. Biotechnol. Biochem. 2007, 71, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.Y.; Preston, L.A.; Schiller, N.L. Alginate lyase: Review of major sources and enzyme characteristics, structure-function analysis, biological roles, and applications. Annu. Rev. Microbiol. 2000, 54, 289–340. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.T.; Ko, H.J.; Kim, N.; Kim, D.; Lee, D.; Choi, I.G.; Woo, H.C.; Kim, M.D.; Kim, K.H. Characterization of a recombinant endo-type alginate lyase (Alg7D) from saccharophagus degradans. Biotechnol. Lett. 2012, 34, 1087–1092. [Google Scholar] [PubMed]
- Inoue, A.; Mashino, C.; Uji, T.; Saga, N.; Mikami, K.; Ojima, T. Characterization of an eukaryotic PL-7 alginate lyase in the marine red alga pyropia yezoensis. Curr. Biotechnol. 2015, 4, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Inoue, A.; Anraku, M.; Nakagawa, S.; Ojima, T. Discovery of a novel alginate lyase from Nitratiruptor sp. SB155-2 thriving at deep-sea hydrothermal vents and identification of the residues responsible for its heat stability. J. Biol. Chem. 2016, 291, 15551–15563. [Google Scholar] [CrossRef] [PubMed]
- Hata, M.; Kumagai, Y.; Rahman, M.M.; Chiba, S.; Tanaka, H.; Inoue, A.; Ojima, T. Comparative study on general properties of alginate lyases from some marine gastropod mollusks. Fisheries Sci. 2009, 75, 755. [Google Scholar] [CrossRef]
- Zhu, B.; Tan, H.; Qin, Y.; Xu, Q.; Du, Y.; Yin, H. Characterization of a new endo-type alginate lyase from vibrio sp. W13. Int. J. Biol. Macromol. 2015, 75, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Elyakova, L.A.; Favorov, V.V. Isolation and certain properties of alginate lyase VI from the mollusk Littorina sp. Biochim. Biophys. Acta 1974, 358, 341–354. [Google Scholar] [CrossRef]
- Lombard, V.; Bernard, T.; Rancurel, C.; Brumer, H.; Coutinho, P.M.; Henrissat, B. A hierarchical classification of polysaccharide lyases for glycogenomics. Biochem. J. 2010, 432, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Yin, H. Alginate lyase: Review of major sources and classification, properties, structure-function analysis and applications. Bioengineered 2015, 6, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Li, F.L.; Lu, M.; Ji, S.Q.; Wang, B. Biochemical and structural characterization of alginate lyases: an update. Curr. Biotechnol. 2015, 4, 223–239. [Google Scholar]
- Xu, F.; Wang, P.; Zhang, Y.Z.; Chen, X.L. Diversity of three-dimensional structures and catalytic mechanisms of alginate lyases. Appl. Environ. Microbiol. 2018, 84, e02040-17. [Google Scholar] [PubMed]
- Zhu, B.; Chen, M.; Yin, H.; Du, Y.; Ning, L. Enzymatic hydrolysis of alginate to produce oligosaccharides by a new purified endo-type alginate lyase. Mar. Drugs 2016, 14, 108. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, S.; Yu, W.; Gong, Q. Cloning, overexpression and characterization of a new oligoalginate lyase from a marine bacterium, shewanella sp. Biotechnol. Lett. 2015, 37, 665–671. [Google Scholar] [PubMed]
- Zhang, Z.; Yu, G.; Guan, H.; Zhao, X.; Du, Y.; Jiang, X. Preparation and structure elucidation of alginate oligosaccharides degraded by alginate lyase from Vibro sp. 510. Carbohydr. Res. 2004, 339, 1475–1481. [Google Scholar] [CrossRef] [PubMed]
- Inoue, A.; Kagaya, M.; Ojima, T. Preparation of protoplasts from Laminaria japonica using native and recombinant abalone alginate lyases. J. Appl. Phycol. 2008, 20, 633–640. [Google Scholar]
- Inoue, A.; Mashino, C.; Kodama, T.; Ojima, T. Protoplast preparation from Laminaria japonica with recombinant alginate lyase and cellulase. Mar. Biotechnol. 2011, 13, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Islan, G.A.; Bosio, V.E.; Castro, G.R. Alginate lyase and ciprofloxacin co-immobilization on biopolymeric microspheres for cystic fibrosis treatment. Macromol. Biosci. 2013, 13, 1238–1248. [Google Scholar] [CrossRef] [PubMed]
- Farrell, E.K.; Tipton, P.A. Functional characterization of AlgL, an alginate lyase from pseudomonas aeruginosa. Biochemistry 2012, 51, 10259–10266. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Hu, F.; Yuan, H.; Sun, Y.; Yao, Z. Biochemical characterization and degradation pattern of a unique pH-stable PolyM-specific alginate lyase from newly isolated Serratia marcescens NJ-07. Mar. Drugs 2018, 16, 129. [Google Scholar] [CrossRef] [PubMed]
- Kam, N.; Park, Y.J.; Lee, E.Y.; Kim, H.S. Molecular identification of a polym-specific alginate lyase from Pseudomonas sp. Strain ks-408 for degradation of glycosidic linkages between two mannuronates or mannuronate and guluronate in alginate. Can. J. Microbiol. 2011, 57, 1032–1041. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S. Cloning and expression of alginate lyase from a marine bacterium, Streptomyces sp. M3. J. Life Sci. 2009, 19, 1522–1528. [Google Scholar]
- Purification and Characterization of Extracellular Alginate Lyase from Corynebacterium sp. Aly-1 Strain. 1995. Available online: http://xueshu.baidu.com/s?wd=paperuri%3A%2841681fcc50d2b5e7270247da75b131af%29&filter=sc_long_sign&tn=SE_xueshusource_2kduw22v&sc_vurl=http%3A%2F%2Fci.nii.ac.jp%2Fnaid%2F110002947783%2Fen&ie=utf-8&sc_us=10692311932531899694 (accessed on 30 July 2018).
- Li, J.W.; Dong, S.; Song, J.; Li, C.B.; Chen, X.L.; Xie, B.B.; Zhang, Y.Z. Purification and characterization of a bifunctional alginate lyase from Pseudoalteromonas sp. SM0524. Mar. Drugs 2011, 9, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, Y.; Araki, R.; Iriyama, K.; Oda, T.; Fukuda, H.; Hayashida, S.; Muramatsu, T. Purification and characterization of bifunctional alginate lyase from Alteromonas sp. Strain no. 272 and its action on saturated oligomeric substrates. Biosci. Biotechnol. Biochem. 2001, 65, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Manns, D.; Nyffenegger, C.; Saake, B.; Meyer, A.S. Impact of different alginate lyases on combined cellulase–lyase saccharification of brown seaweed. Rsc Adv. 2016, 6, 45392–45401. [Google Scholar] [CrossRef]
- Mohapatra, B.R. Kinetic and thermodynamic properties of alginate lyase and cellulase co-produced by Exiguobacterium species Alg-S5. Int. J. Biol. Macromol. 2017, 98, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Swift, S.M.; Hudgens, J.W.; Heselpoth, R.D.; Bales, P.M.; Nelson, D.C. Characterization of AlgMsp, an alginate lyase from Microbulbifer sp. 6532A. PLoS ONE 2014, 9, e112939. [Google Scholar]
- Zhu, B.; Ni, F.; Ning, L.; Sun, Y.; Yao, Z. Cloning and characterization of a new pH-stable alginate lyase with high salt tolerance from marine Vibrio sp. Nj-04. Int. J. Biol. Macromol. 2018, 115, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Sun, Y.; Ni, F.; Ning, L.; Yao, Z. Characterization of a new endo-type alginate lyase from Vibrio sp. Nju-03. Int. J. Biol. Macromol. 2018, 108, 1140–1147. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Ni, F.; Sun, Y.; Yao, Z. Expression and characterization of a new heat-stable endo-type alginate lyase from deep-sea bacterium Flammeovirga sp. Nj-04. Extremophiles 2017, 21, 1027–1036. [Google Scholar] [PubMed]
- Thomas, F.; Lundqvist, L.C.E.; Jam, M.; Jeudy, A.; Barbeyron, T.; Sandström, C.; Michel, G.; Czjzek, M. Comparative characterization of two marine alginate lyases from Zobellia galactanivorans reveals distinct modes of action and exquisite adaptation to their natural substrate. J. Biol. Chem. 2013, 288, 23021. [Google Scholar] [CrossRef] [PubMed]
- Dou, W.; Wei, D.; Li, H.; Li, H.; Rahman, M.M.; Shi, J.; Xu, Z.; Ma, Y. Purification and characterisation of a bifunctional alginate lyase from novel Isoptericola halotolerans CGMCC 5336. Carbohydr. Polym. 2013, 98, 1476–1482. [Google Scholar] [CrossRef] [PubMed]
- Uchimura, K.; Miyazaki, M.; Nogi, Y.; Kobayashi, T.; Horikoshi, K. Cloning and sequencing of alginate lyase genes from deep-sea strains of Vibrio and Agarivorans and characterization of a new Vibrio enzyme. Mar. Biotechnol. 2010, 12, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Yagi, H.; Fujise, A.; Itabashi, N.; Ohshiro, T. Purification and characterization of a novel alginate lyase from the marine bacterium Cobetia sp. NAP1 isolated from brown algae. Biosci. Biotechnol. Biochem. 2016, 80, 2338–2346. [Google Scholar] [PubMed]
- Takeshita, S.; Sato, N.; Igarashi, M.; Muramatsu, T. A highly denaturant-durable alginate lyase from a marine bacterium: Purification and properties. Biosci. Biotechnol. Biochem. 1993, 57, 1125. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.M.; Li, S.Y.; Wu, Y.; Yu, W.G.; Han, F. Cloning and characterization of two thermo- and salt-tolerant oligoalginate lyases from marine bacterium Halomonas sp. Fems. Microbiol. Lett. 2016, 363, fnw079. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.W.; Huang, L.S.; Tan, H.D.; Qin, Y.Q.; Du, Y.G.; Yin, H. Characterization of a new endo-type polyM-specific alginate lyase from Pseudomonas sp. Biotechnol. Lett. 2015, 37, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, K.; Kodama, T. Purification and propertes of poly(β-d-mannuronate) lyase from Azotobacter chroococcum. Appl. Microbiol. Biot. 1996, 44, 576–581. [Google Scholar] [CrossRef]
- Maki, H.; Mori, A.; Fujiyama, K.; Kinoshita, S.; Yoshida, T. Cloning, sequence analysis and expression in escherichia coli of a gene encoding an alginate lyase from Pseudomonas sp. OS-ALG-9. J. Gen. Microbiol. 1993, 139, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Biochem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Osawa, T.; Matsubara, Y.; Muramatsu, T.; Kimura, M.; Kakuta, Y. Crystal structure of the alginate (poly α-l-guluronate) lyase from Corynebacterium sp. at 1.2 Å resolution. J. Mol. Biol. 2005, 345, 1111. [Google Scholar] [CrossRef] [PubMed]


| Parameters | Sodium Alginate | PolyG | PolyM |
|---|---|---|---|
| Specific activity (U/mg) | 7984.82 | 8643.29 | 7658.63 |
| Km (mM) | 5.6 | 3.2 | 6.7 |
| Vmax (nmol/s) | 3.22 | 1.74 | 1.89 |
| kcat (s−1) | 45.92 | 24.82 | 26.95 |
| kcat/Km (s−1·mM−1) | 8.20 | 7.56 | 4.02 |
| Enzyme | Specificity | Km, Vmax | Optimal Temperature and pH | Products (Dp) | Reference |
|---|---|---|---|---|---|
| AlyA | Bifunctional | 5.6 mM, 3.22 nmol/s | 55 °C, 7.5 | 2–5 | This study |
| Algb | polyG > polyM | 0.67 mg/mL, 473.93 U/mg | 30 °C, 8.0 | 2–5 | [17] |
| FsAlgA | polyG > polyM | 0.48 mM, 0.19 nmol/s | 50 °C, 7.0 | 2–5 | [41] |
| Alg7D | polyM > polyG | 3.0 mg/mL, 6.2 U/mg | 50 °C, 7.0 | 2–5 | [13] |
| AlgMsp | polyG > polyM | 3.4 mM, 57 pmol/s | 50 °C, 8.0 | 2–5 | [38] |
| AlgNJ-04 | polyG > polyM | 0.49 mM, 72 pmol/s | 40 °C, 7.0 | 2–5 | [41] |
| AlgNJU-03 | polyG > polyM | 8.5 mM, 1.67 nmol/s | 30 °C, 7.0 | 2–4 | [40] |
| AlgC-PL7 | Bifunctional | - | 55 °C, 8.0 | 1 | [45] |
| A9m | polyG > polyM | - | 30 °C, 7.5 | - | [44] |
| Reagent | Relative Activity (%) |
|---|---|
| Control | 100 ± 0.5 |
| Na+(100 mM) | 126 ± 2.2 |
| Na+(300 mM) | 180 ± 3.1 |
| Na+(500 mM) | 203 ± 4.6 |
| Na+(700 mM) | 136 ± 2.9 |
| Na+(900 mM) | 89 ± 7.9 |
| Zn2+ | 91 ± 2.3 |
| Cu2+ | 65 ± 3.2 |
| Mn2+ | 94 ± 2.1 |
| Co2+ | 75 ± 3.4 |
| Ca2+ | 174 ± 1.3 |
| Ca2+(10 mM) | 135 ± 5.7 |
| Fe3+ | 88 ± 2.1 |
| Mg2+ | 168 ± 2.7 |
| Mg2+(10 mM) | 119 ± 2.9 |
| Mg2+(50 mM) | 101 ± 3.2 |
| Ni2+ | 87 ± 1.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, B.; Ning, L.; Jiang, Y.; Ge, L. Biochemical Characterization and Degradation Pattern of a Novel Endo-Type Bifunctional Alginate Lyase AlyA from Marine Bacterium Isoptericola halotolerans. Mar. Drugs 2018, 16, 258. https://doi.org/10.3390/md16080258
Zhu B, Ning L, Jiang Y, Ge L. Biochemical Characterization and Degradation Pattern of a Novel Endo-Type Bifunctional Alginate Lyase AlyA from Marine Bacterium Isoptericola halotolerans. Marine Drugs. 2018; 16(8):258. https://doi.org/10.3390/md16080258
Chicago/Turabian StyleZhu, Benwei, Limin Ning, Yucui Jiang, and Lin Ge. 2018. "Biochemical Characterization and Degradation Pattern of a Novel Endo-Type Bifunctional Alginate Lyase AlyA from Marine Bacterium Isoptericola halotolerans" Marine Drugs 16, no. 8: 258. https://doi.org/10.3390/md16080258
APA StyleZhu, B., Ning, L., Jiang, Y., & Ge, L. (2018). Biochemical Characterization and Degradation Pattern of a Novel Endo-Type Bifunctional Alginate Lyase AlyA from Marine Bacterium Isoptericola halotolerans. Marine Drugs, 16(8), 258. https://doi.org/10.3390/md16080258






