Spirulina maxima Decreases Endothelial Damage and Oxidative Stress Indicators in Patients with Systemic Arterial Hypertension: Results from Exploratory Controlled Clinical Trial
Abstract
1. Introduction
2. Results
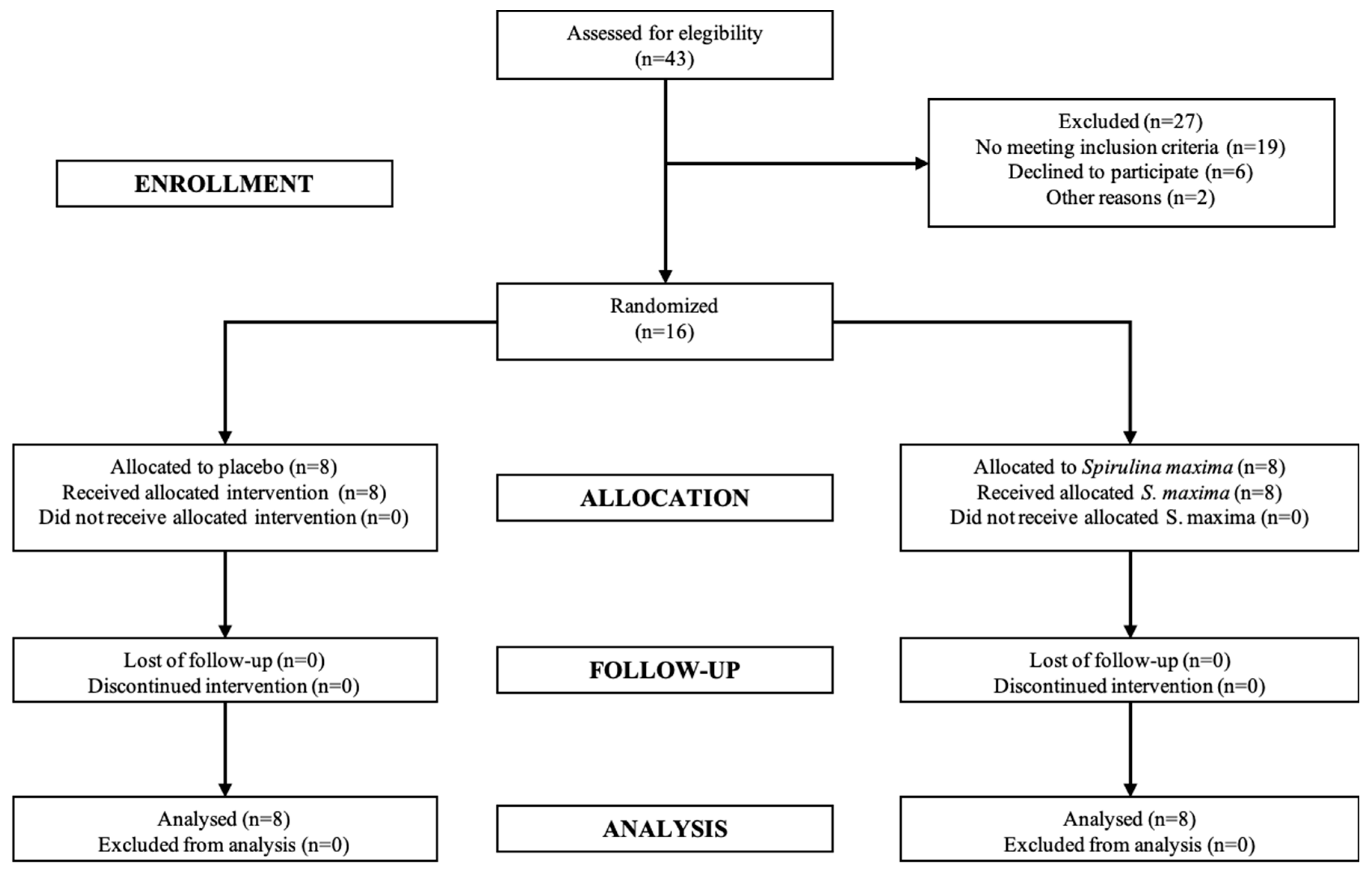
2.1. Pilot Clinical Trial
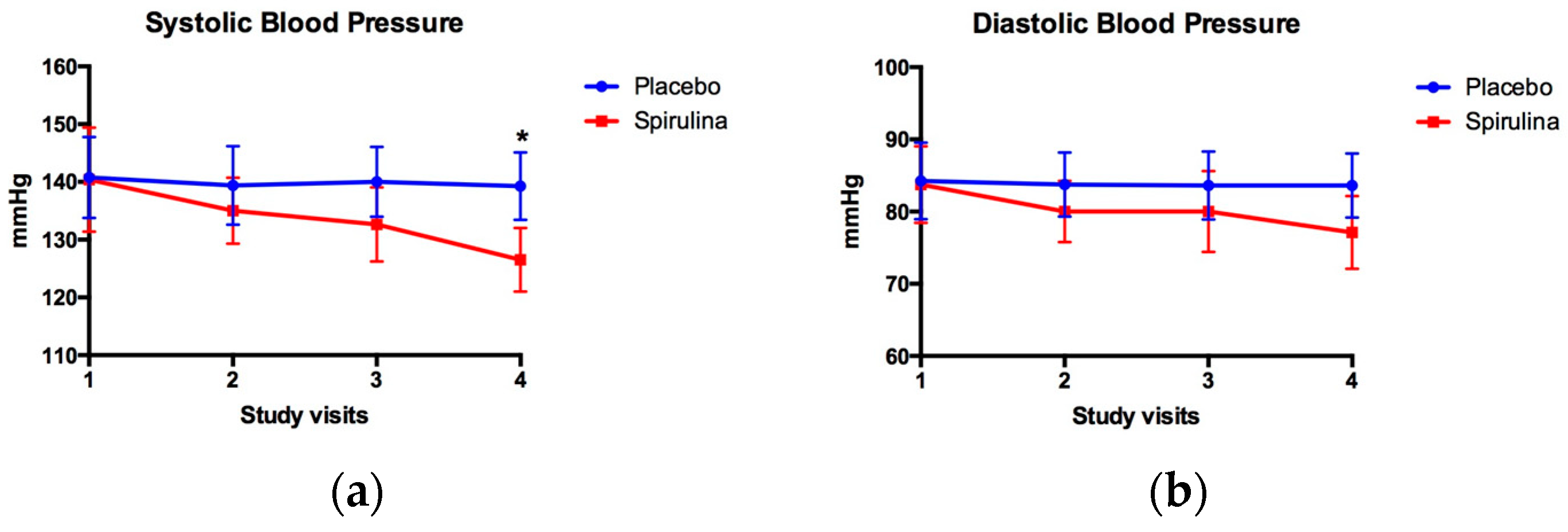
2.2. Blood Pressure Levels
2.3. Endothelial Damage Indicators
2.4. Antioxidant Status
2.5. Quality of Life
2.6. Safety
3. Discussion
3.1. Blood Pressure
3.2. Endothelial Damage Markers
3.3. Antioxidant Status
3.4. Safety
4. Materials and Methods
4.1. Pilot Clinical Trial
4.1.1. Outcome Measures
4.1.2. Safety
4.2. Spirulina Maxima and Placebo Treatment
4.3. Antioxidant Status Measures
4.3.1. Catalase
4.3.2. Superoxide Dismutase
4.3.3. Glutathione Reductase
4.3.4. Glutathione Peroxidase
4.3.5. Reduced and Oxidized Glutathione
4.3.6. Thiobarbituric Acid Reactive Substances
4.4. Endothelial Damage Indicators
4.5. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cifu, A.S.; Davis, A.M. Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. JAMA 2017, 318, 2132–2134. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.D.; Murray, C.C. The global burden of disease, 1990–2020. Nat. Med. 1998, 4, 1241–1243. [Google Scholar] [CrossRef] [PubMed]
- Campos-Nonato, I.; Hernandez-Barrera, L.; Pedroza-Tobias, A.; Medina, C.; Barquera, S. Hypertension in Mexican adults: Prevalence, diagnosis and type of treatment. Ensanut MC 2016. Salud Publica Mexico 2018, 60, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Wolf-Maier, K.; Cooper, R.S.; Banegas, J.R.; Giampaoli, S.; Hense, H.W.; Joffres, M.; Kastarinen, M.; Poulter, N.; Primatesta, P.; Rodriguez-Artalejo, F.; et al. Hypertension prevalence and blood pressure levels in 6 European countries, Canada, and the United States. JAMA 2003, 289, 2363–2369. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Q.J. The prevalence of prehypertension and hypertension among US adults according to the new joint national committee guidelines: New challenges of the old problem. Arch. Intern. Med. 2004, 164, 2126–2134. [Google Scholar] [CrossRef] [PubMed]
- Flaten, H.K.; Monte, A.A. The Pharmacogenomic and Metabolomic Predictors of ACE Inhibitor and Angiotensin II Receptor Blocker Effectiveness and Safety. Cardiovasc. Drugs Therapy 2017, 31, 471–482. [Google Scholar] [CrossRef] [PubMed]
- Roger, V.L.; Go, A.S.; Lloyd-Jones, D.M.; Adams, R.J.; Berry, J.D.; Brown, T.M.; Carnethon, M.R.; Dai, S.; de Simone, G.; Ford, E.S.; et al. Heart disease and stroke statistics--2011 update: A report from the American Heart Association. Circulation 2011, 123, e18–e209. [Google Scholar] [CrossRef]
- Guyenet, P.G. The sympathetic control of blood pressure. Nat. Rev. Neurosci. 2006, 7, 335–346. [Google Scholar] [CrossRef]
- Mann, S.J. Neurogenic hypertension: Pathophysiology, diagnosis and management. Clin. Auton. Res. 2018, 28, 363–374. [Google Scholar] [CrossRef]
- Nizar, J.M.; Bhalla, V. Molecular Mechanisms of Sodium-Sensitive Hypertension in the Metabolic Syndrome. Curr. Hypertens. Rep. 2017, 19. [Google Scholar] [CrossRef]
- Sanders, P.W. Vascular consequences of dietary salt intake. Am. J. Physiol. Renal. Physiol. 2009, 297, F237–F243. [Google Scholar] [CrossRef] [PubMed]
- Acelajado, M.C.; Oparil, S. Hypertension in the elderly. Clin. Geriatr. Med. 2009, 25, 391–412. [Google Scholar] [CrossRef] [PubMed]
- Te Riet, L.; van Esch, J.H.M.; Roks, A.J.M.; van den Meiracker, A.H.; Danser, A.H.J. Hypertension: Renin–Angiotensin–Aldosterone System Alterations. Circ. Res. 2015, 116, 960–975. [Google Scholar] [CrossRef] [PubMed]
- Tomaschitz, A.; Pilz, S.; Ritz, E.; Obermayer-Pietsch, B.; Pieber, T.R. Aldosterone and arterial hypertension. Nat. Rev. Endocrinol. 2010, 6, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.K. Linking endothelial dysfunction with endothelial cell activation. J. Clin. Investig. 2013, 123, 540–541. [Google Scholar] [CrossRef] [PubMed]
- Tycinska, A.M.; Mroczko, B.; Musial, W.J.; Sawicki, R.; Kaminski, K.; Borowska, H.; Sobkowicz, B.; Szmitkowski, M. Blood pressure in relation to neurogenic, inflammatory and endothelial dysfunction biomarkers in patients with treated essential arterial hypertension. Adv. Med. Sci. 2011, 56, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Austgulen, R.; Lien, E.; Vince, G.; Redman, C.W. Increased maternal plasma levels of soluble adhesion molecules (ICAM-1, VCAM-1, E-selectin) in preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 1997, 71, 53–58. [Google Scholar] [CrossRef]
- Cacciatore, F.; Bruzzese, G.; Vitale, D.F.; Liguori, A.; de Nigris, F.; Fiorito, C.; Infante, T.; Donatelli, F.; Minucci, P.B.; Ignarro, L.J.; et al. Effects of ACE inhibition on circulating endothelial progenitor cells, vascular damage, and oxidative stress in hypertensive patients. Eur. J. Clin. Pharmacol. 2011, 67, 877–883. [Google Scholar] [CrossRef]
- Napoli, C.; Sica, V.; de Nigris, F.; Pignalosa, O.; Condorelli, M.; Ignarro, L.J.; Liguori, A. Sulfhydryl angiotensin-converting enzyme inhibition induces sustained reduction of systemic oxidative stress and improves the nitric oxide pathway in patients with essential hypertension. Am. Heart J. 2004, 148, e5. [Google Scholar] [CrossRef]
- Kedziora-Kornatowska, K.; Czuczejko, J.; Szewczyk-Golec, K.; Motyl, J.; Szadujkis-Szadurski, L.; Kornatowski, T.; Pawluk, H.; Kedziora, J. Effects of perindopril and hydrochlorothiazide on selected indices of oxidative stress in the blood of elderly patients with essential hypertension. Clin. Exp. Pharmacol. Physiol. 2006, 33, 751–756. [Google Scholar] [CrossRef]
- Rubio-Guerra, A.F.; Vargas-Robles, H.; Vargas-Ayala, G.; Rodriguez-Lopez, L.; Escalante-Acosta, B.A. The effect of trandolapril and its fixed-dose combination with verapamil on circulating adhesion molecules levels in hypertensive patients with type 2 diabetes. Clin. Exp. Hypertens. 2008, 30, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Harrison, D.G. Basic science: Pathophysiology: Oxidative stress. J. Am. Soc. Hypertens. 2014, 8, 601–603. [Google Scholar] [CrossRef] [PubMed]
- Touyz, R.M. Reactive oxygen species and angiotensin II signaling in vascular cells—Implications in cardiovascular disease. Braz. J. Med. Biol. Res. 2004, 37, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Ngo-Matip, M.E.; Pieme, C.A.; Azabji-Kenfack, M.; Moukette, B.M.; Korosky, E.; Stefanini, P.; Ngogang, J.Y.; Mbofung, C.M. Impact of daily supplementation of Spirulina platensis on the immune system of naive HIV-1 patients in Cameroon: A 12-months single blind, randomized, multicenter trial. Nutr. J. 2015, 14, 70. [Google Scholar] [CrossRef] [PubMed]
- Winter, F.S.; Emakam, F.; Kfutwah, A.; Hermann, J.; Azabji-Kenfack, M.; Krawinkel, M.B. The effect of Arthrospira platensis capsules on CD4 T-cells and antioxidative capacity in a randomized pilot study of adult women infected with human immunodeficiency virus not under HAART in Yaounde, Cameroon. Nutrients 2014, 6, 2973–2986. [Google Scholar] [CrossRef] [PubMed]
- Mazokopakis, E.E.; Starakis, I.K.; Papadomanolaki, M.G.; Mavroeidi, N.G.; Ganotakis, E.S. The hypolipidaemic effects of Spirulina (Arthrospira platensis) supplementation in a Cretan population: A prospective study. J. Sci. Food Agric. 2014, 94, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Yakoot, M.; Salem, A. Spirulina platensis versus silymarin in the treatment of chronic hepatitis C virus infection. A pilot randomized, comparative clinical trial. BMC Gastroenterol. 2012, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Cingi, C.; Conk-Dalay, M.; Cakli, H.; Bal, C. The effects of spirulina on allergic rhinitis. Eur. Arch. Otorhinolaryngol. 2008, 265, 1219–1223. [Google Scholar] [CrossRef]
- Misbahuddin, M.; Islam, A.Z.; Khandker, S.; Ifthaker Al, M.; Islam, N.; Anjumanara. Efficacy of spirulina extract plus zinc in patients of chronic arsenic poisoning: A randomized placebo-controlled study. Clin. Toxicol. 2006, 44, 135–141. [Google Scholar] [CrossRef]
- Torres-Duran, P.V.; Ferreira-Hermosillo, A.; Juarez-Oropeza, M.A. Antihyperlipemic and antihypertensive effects of Spirulina maxima in an open sample of Mexican population: A preliminary report. Lipids Health Dis. 2007, 6, 33. [Google Scholar] [CrossRef]
- Suliburska, J.; Szulinska, M.; Tinkov, A.A.; Bogdanski, P. Effect of Spirulina maxima Supplementation on Calcium, Magnesium, Iron, and Zinc Status in Obese Patients with Treated Hypertension. Biol. Trace Elem. Res. 2016, 173, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Miczke, A.; Szulinska, M.; Hansdorfer-Korzon, R.; Kregielska-Narozna, M.; Suliburska, J.; Walkowiak, J.; Bogdanski, P. Effects of spirulina consumption on body weight, blood pressure, and endothelial function in overweight hypertensive Caucasians: A double-blind, placebo-controlled, randomized trial. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 150–156. [Google Scholar] [PubMed]
- Ichimura, M.; Kato, S.; Tsuneyama, K.; Matsutake, S.; Kamogawa, M.; Hirao, E.; Miyata, A.; Mori, S.; Yamaguchi, N.; Suruga, K.; et al. Phycocyanin prevents hypertension and low serum adiponectin level in a rat model of metabolic syndrome. Nutr. Res. 2013, 33, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Ren, D.F.; Xue, Y.L.; Sawano, Y.; Miyakawa, T.; Tanokura, M. Isolation of an antihypertensive peptide from alcalase digest of Spirulina platensis. J. Agric. Food Chem. 2010, 58, 7166–7171. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Sawano, Y.; Miyakawa, T.; Xue, Y.L.; Cai, M.Y.; Egashira, Y.; Ren, D.F.; Tanokura, M. One-week antihypertensive effect of Ile-Gln-Pro in spontaneously hypertensive rats. J. Agric. Food Chem. 2011, 59, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Shalia, K.K.; Mashru, M.R.; Vasvani, J.B.; Mokal, R.A.; Mithbawkar, S.M.; Thakur, P.K. Circulating levels of cell adhesion molecules in hypertension. Indian J. Clin. Biochem. 2009, 24, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Blann, A.D.; Tse, W.; Maxwell, S.J.; Waite, M.A. Increased levels of the soluble adhesion molecule E-selectin in essential hypertension. J. Hypertens. 1994, 12, 925–928. [Google Scholar] [CrossRef] [PubMed]
- Cottone, S.; Mule, G.; Amato, F.; Riccobene, R.; Vadala, A.; Lorito, M.C.; Raspanti, F.; Cerasola, G. Amplified biochemical activation of endothelial function in hypertension associated with moderate to severe renal failure. J. Nephrol. 2002, 15, 643–648. [Google Scholar]
- Cottone, S.; Mule, G.; Nardi, E.; Vadala, A.; Lorito, M.C.; Guarneri, M.; Arsena, R.; Palermo, A.; Cerasola, G. C-reactive protein and intercellular adhesion molecule-1 are stronger predictors of oxidant stress than blood pressure in established hypertension. J. Hypertens. 2007, 25, 423–428. [Google Scholar] [CrossRef]
- DeSouza, C.A.; Dengel, D.R.; Macko, R.F.; Cox, K.; Seals, D.R. Elevated levels of circulating cell adhesion molecules in uncomplicated essential hypertension. Am. J. Hypertens. 1997, 10, 1335–1341. [Google Scholar] [CrossRef]
- Hong, S.; Nelesen, R.A.; Krohn, P.L.; Mills, P.J.; Dimsdale, J.E. The association of social status and blood pressure with markers of vascular inflammation. Psychosom. Med. 2006, 68, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Madej, A.; Okopien, B.; Kowalski, J.; Haberka, M.; Herman, Z.S. Plasma concentrations of adhesion molecules and chemokines in patients with essential hypertension. Pharmacol. Rep. 2005, 57, 878–881. [Google Scholar] [PubMed]
- Palomo, I.; Marin, P.; Alarcon, M.; Gubelin, G.; Vinambre, X.; Mora, E.; Icaza, G. Patients with essential hypertension present higher levels of sE-selectin and sVCAM-1 than normotensive volunteers. Clin. Exp. Hypertens. 2003, 25, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Ali, E.A.; Barakat, B.M.; Hassan, R. Antioxidant and angiostatic effect of Spirulina platensis suspension in complete Freund’s adjuvant-induced arthritis in rats. PLoS ONE 2015, 10, e0121523. [Google Scholar] [CrossRef] [PubMed]
- Bashandy, S.A.; El Awdan, S.A.; Ebaid, H.; Alhazza, I.M. Antioxidant Potential of Spirulina platensis Mitigates Oxidative Stress and Reprotoxicity Induced by Sodium Arsenite in Male Rats. Oxid. Med. Cell. Longev. 2016, 2016, 7174351. [Google Scholar] [CrossRef] [PubMed]
- Chaiklahan, R.; Chirasuwan, N.; Triratana, P.; Loha, V.; Tia, S.; Bunnag, B. Polysaccharide extraction from Spirulina sp. and its antioxidant capacity. Int. J. Biol. Macromol. 2013, 58, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.L.; Lim, Y.W.; Radhakrishnan, A.K.; Lim, P.E. Protective effect of aqueous extract from Spirulina platensis against cell death induced by free radicals. BMC Complement. Altern. Med. 2010, 10, 53. [Google Scholar] [CrossRef]
- Gutierrez-Rebolledo, G.A.; Galar-Martinez, M.; Garcia-Rodriguez, R.V.; Chamorro-Cevallos, G.A.; Hernandez-Reyes, A.G.; Martinez-Galero, E. Antioxidant Effect of Spirulina (Arthrospira) maxima on Chronic Inflammation Induced by Freund’s Complete Adjuvant in Rats. J. Med. Food 2015, 18, 865–871. [Google Scholar] [CrossRef]
- Martin, S.J.; Sabina, E.P. Amelioration of anti-tuberculosis drug induced oxidative stress in kidneys by Spirulina fusiformis in a rat model. Ren. Fail. 2016, 38, 1115–1121. [Google Scholar] [CrossRef]
- Muga, M.A.; Chao, J.C. Effects of fish oil and spirulina on oxidative stress and inflammation in hypercholesterolemic hamsters. BMC Complement. Altern. Med. 2014, 14, 470. [Google Scholar] [CrossRef]
- Shabana, E.F.; Gabr, M.A.; Moussa, H.R.; El-Shaer, E.A.; Ismaiel, M.M.S. Biochemical composition and antioxidant activities of Arthrospira (Spirulina) platensis in response to gamma irradiation. Food Chem. 2017, 214, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Shetty, P.; Shenai, P.; Chatra, L.; Rao, P.K. Efficacy of spirulina as an antioxidant adjuvant to corticosteroid injection in management of oral submucous fibrosis. Indian J. Dent. Res. 2013, 24, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Tobon-Velasco, J.C.; Palafox-Sanchez, V.; Mendieta, L.; Garcia, E.; Santamaria, A.; Chamorro-Cevallos, G.; Limon, I.D. Antioxidant effect of Spirulina (Arthrospira) maxima in a neurotoxic model caused by 6-OHDA in the rat striatum. J. Neural Transm. 2013, 120, 1179–1189. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Velasco, M.; Gonzalez-Torres, L.; Lopez-Gasco, P.; Bastida, S.; Benedi, J.; Gonzalez-Munoz, M.J.; Sanchez-Muniz, F.J. Effects of glucomannan/spirulina-surimi on liver oxidation and inflammation in Zucker rats fed atherogenic diets. J. Physiol. Biochem. 2015, 71, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Inoguchi, T.; Sasaki, S.; Maeda, Y.; McCarty, M.F.; Fujii, M.; Ikeda, N.; Kobayashi, K.; Sonoda, N.; Takayanagi, R. Phycocyanin and phycocyanobilin from Spirulina platensis protect against diabetic nephropathy by inhibiting oxidative stress. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R110–R120. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Lee, Y.J.; Ryu, H.K.; Kim, M.H.; Chung, H.W.; Kim, W.Y. A randomized double-blind, placebo-controlled study to establish the effects of spirulina in elderly Koreans. Ann. Nutr. Metab. 2008, 52, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Kalafati, M.; Jamurtas, A.Z.; Nikolaidis, M.G.; Paschalis, V.; Theodorou, A.A.; Sakellariou, G.K.; Koutedakis, Y.; Kouretas, D. Ergogenic and antioxidant effects of spirulina supplementation in humans. Med. Sci. Sports Exerc. 2010, 42, 142–151. [Google Scholar] [CrossRef]
- Aquilano, K.; Baldelli, S.; Ciriolo, M.R. Glutathione: New roles in redox signaling for an old antioxidant. Front. Pharmacol. 2014, 5, 196. [Google Scholar] [CrossRef] [PubMed]
- Roca, C.A.; Dalfo, A.; Badia, X.; Aristegui, A.; Roset, M. Relation between clinical and therapeutic variables and quality of life in hypertension. J. Hypertens. 2001, 19, 1913–1919. [Google Scholar] [CrossRef]
- Gomez, M.; Garcia, C.; Gomez, V.; Mondragon, P. Calidad de vida en pacientes que viven con hipertensión arterial sistémica. Rev. Mex. Enf. Cardiol. 2011, 19, 7–12. [Google Scholar]
- Aebi, H.E. Detection and fixation of radiation-produced peroxide by enzymes. Radiat. Res. 1963, 3, 130–152. [Google Scholar] [CrossRef] [PubMed]
- Kono, Y. Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch. Biochem. Biophys. 1978, 186, 189–195. [Google Scholar] [CrossRef]
- Andersen, H.R.; Jeune, B.; Nybo, H.; Nielsen, J.B.; Andersen-Ranberg, K.; Grandjean, P. Low activity of superoxide dismutase and high activity of glutathione reductase in erythrocytes from centenarians. Age Ageing 1998, 27, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar] [PubMed]
- Torres-Duran, P.V.; Miranda-Zamora, R.; Paredes-Carbajal, M.C.; Mascher, D.; Ble-Castillo, J.; Diaz-Zagoya, J.C.; Juarez-Oropeza, M.A. Studies on the preventive effect of Spirulina maxima on fatty liver development induced by carbon tetrachloride, in the rat. J. Ethnopharmacol. 1999, 64, 141–147. [Google Scholar] [CrossRef]
| Baseline Parameter | Placebo Group (n = 8) | SM Group (n = 8) |
|---|---|---|
| Age (years) | 51.80 ± 9.44 | 57.00 ± 8.66 |
| Weight (kg) | 77.20 ± 18.89 | 79.36 ± 29.40 |
| Height (m) | 1.51 ± 0.10 | 1.56 ± 0.13 |
| BMI (kg/m2) | 34.19 ± 10.69 | 31.56 ± 7.11 |
| HR (bpm) | 71.20 ± 9.23 | 82.00 ± 7.34 |
| RR (bpm) | 19.40 ± 0.54 | 19.00 ± 1.00 |
| Temperature (°C) | 36.20 ± 0.27 | 36. 26 ± 0.35 |
| SBP (mm Hg) | 140.75 ± 7.03 | 140.38 ± 9.04 |
| DBP (mm Hg) | 84.25 ± 5.28 | 83.75 ± 5.31 |
| SAH evolution (years) | 4.20 ± 3.11 | 4.40 ± 3.05 |
| CHAL (points) | 25.20 ± 15.48 | 28.80 ± 11.56 |
| Indicator | Pre-Treatment | Post-Treatment | Intra Group Comparison (p) | ||||
|---|---|---|---|---|---|---|---|
| Placebo (n = 8) | Spirulina maxima (n = 8) | p | Placebo (n = 8) | Spirulina maxima (n = 8) | p | ||
| sVCAM-1 (ng/mL) | 508.90 ± 31.40 | 480.80 ± 10.30 * | * =0.033 | 501.60 ± 22.70 | 458.00 ± 19.60 **,+ | ** 0.0022 | + 0.0391 |
| sICAM-1 (ng/mL) | 375.10 ± 96.40 | 348.50 ± 85.20 | 0.64 | 308.50 ± 99.10 | 333.30 ± 99.10 | 0.88 | 0.64 |
| sE-Selectin (ng/mL) | 13.73 ± 4.16 | 12.12 ± 1.52 | 0.77 | 12.40 ± 1.24 | 9.71 ± 2.13 ** | ** 0.007 | + 0.0234 |
| Endothelin-1 (pg/mL) | 17.45 ± 6.11 | 14.42 ± 2.92 | 0.22 | 21.48 ± 2.17 | 11.90 ± 6.47 ** | ** 0.0002 | + 0.0391 |
| Oxidative Stress Indicator | Pre-Treatment | Post-Treatment | Intra Group Comparison (p) | ||||
|---|---|---|---|---|---|---|---|
| Placebo (n = 8) | Spirulina maxima (n = 8) | p | Placebo (n = 8) | Spirulina maxima (n = 8) | p | ||
| CAT (k/mL) | 3.35 ± 1.09 | 3.43 ± 1.15 | 0.800 | 3.35 ± 1.11 | 3.76 ± 1.23 ** | ** 0.016 | 0.054 |
| SOD (U/mL) | 77.63 ± 1.47 | 77.67 ± 1.87 | 0.980 | 76.63 ± 0.43 | 82.40 ± 1.32 ** | ** 0.0023 | Δ 0.009 |
| GR (µmol NADPH/min) | 61.45 ± 18.27 | 52.17 ± 28.32 | 0.480 | 41.49 ± 3.36 + | 51.93 ± 12.44 ** | 0.053 | + 0.0156 |
| GPx (µmol NADPH/min) | 354.70 ± 79.86 | 355.71 ± 42.26 | 0.490 | 322.43 ± 36.36 | 404.27 ± 25.89 **,Δ | ** 0.0002 | Δ 0.0234 |
| GSH (mg/mL) | 23.51 ± 5.08 | 23.35 ± 6.13 | 0.840 | 22.82 ± 2.32 | 26.94 ± 4.01 | 0.083 | 0.312 |
| GSSG (mg/mL) | 19.96 ± 3.32 | 26.76 ± 5.93 * | * 0.041 | 20.01 ± 1.58 | 37.88 ± 7.54 **,Δ | ** 0.0002 | Δ 0.0156 |
| TBARS (µg MDA/mL) | 5.74 ± 3.79 | 4.47 ± 2.68 | 0.570 | 3.89 ± 1.59 | 3.90 ± 1.16 | 0.999 | 0.740 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Sámano, J.; Torres-Montes de Oca, A.; Luqueño-Bocardo, O.I.; Torres-Durán, P.V.; Juárez-Oropeza, M.A. Spirulina maxima Decreases Endothelial Damage and Oxidative Stress Indicators in Patients with Systemic Arterial Hypertension: Results from Exploratory Controlled Clinical Trial. Mar. Drugs 2018, 16, 496. https://doi.org/10.3390/md16120496
Martínez-Sámano J, Torres-Montes de Oca A, Luqueño-Bocardo OI, Torres-Durán PV, Juárez-Oropeza MA. Spirulina maxima Decreases Endothelial Damage and Oxidative Stress Indicators in Patients with Systemic Arterial Hypertension: Results from Exploratory Controlled Clinical Trial. Marine Drugs. 2018; 16(12):496. https://doi.org/10.3390/md16120496
Chicago/Turabian StyleMartínez-Sámano, Jesús, Adriana Torres-Montes de Oca, Oscar Ivan Luqueño-Bocardo, Patricia V. Torres-Durán, and Marco A. Juárez-Oropeza. 2018. "Spirulina maxima Decreases Endothelial Damage and Oxidative Stress Indicators in Patients with Systemic Arterial Hypertension: Results from Exploratory Controlled Clinical Trial" Marine Drugs 16, no. 12: 496. https://doi.org/10.3390/md16120496
APA StyleMartínez-Sámano, J., Torres-Montes de Oca, A., Luqueño-Bocardo, O. I., Torres-Durán, P. V., & Juárez-Oropeza, M. A. (2018). Spirulina maxima Decreases Endothelial Damage and Oxidative Stress Indicators in Patients with Systemic Arterial Hypertension: Results from Exploratory Controlled Clinical Trial. Marine Drugs, 16(12), 496. https://doi.org/10.3390/md16120496





