Cloning, Expression and Characterization of a Novel Cold-adapted β-galactosidase from the Deep-sea Bacterium Alteromonas sp. ML52
Abstract
1. Introduction
2. Results
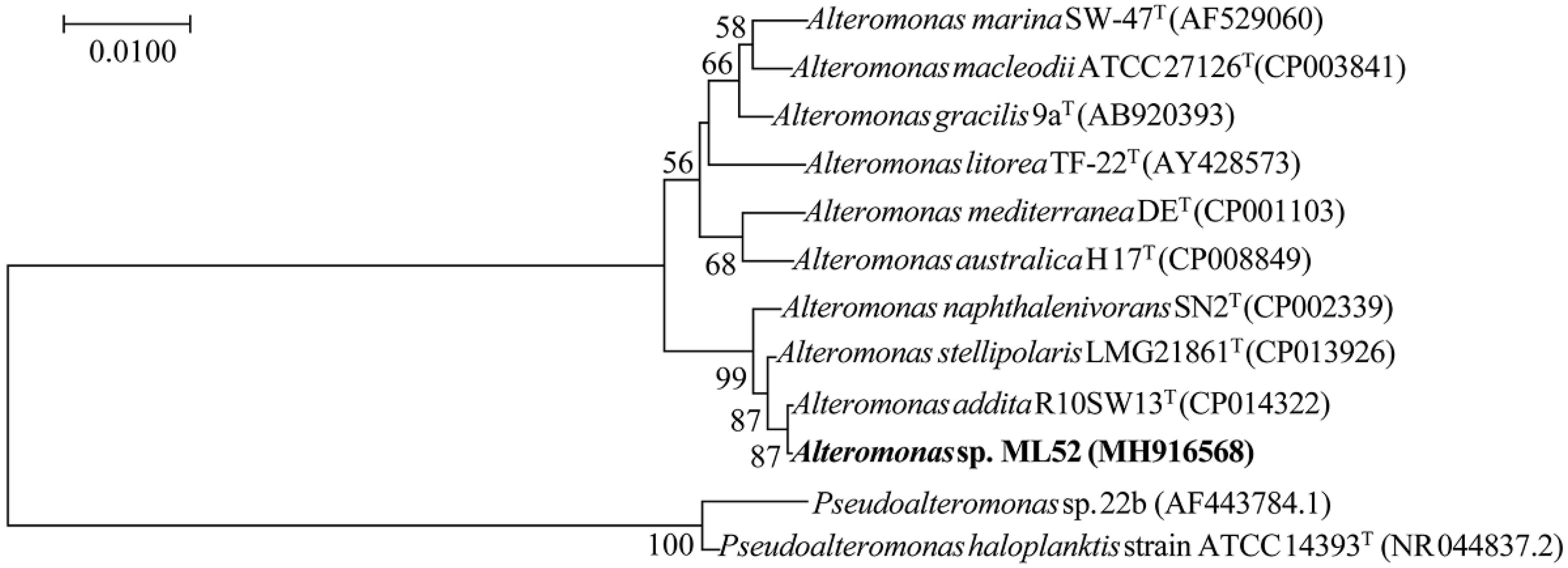
2.1. Characterization and Identification of Strain ML52
2.2. Molecular Cloning and Sequence Analysis
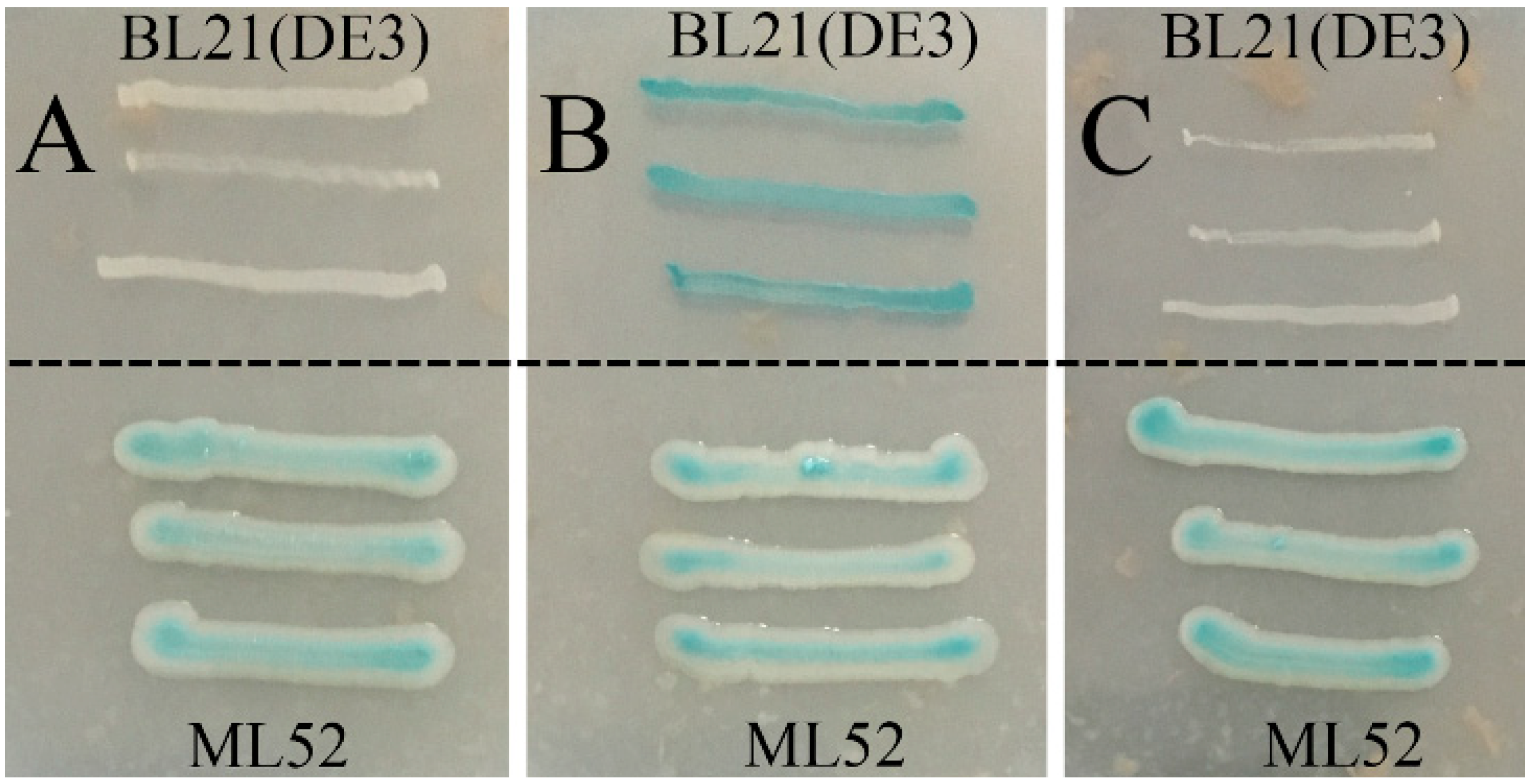
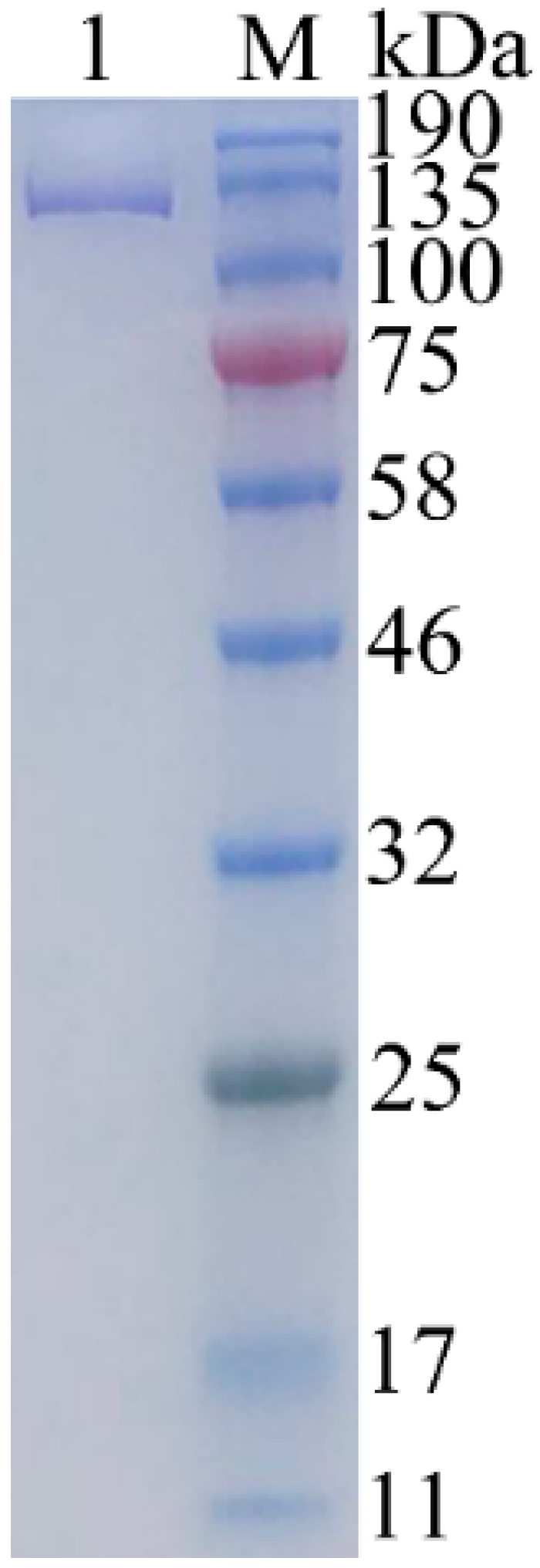
2.3. Expression and Purification of Recombinant Gal
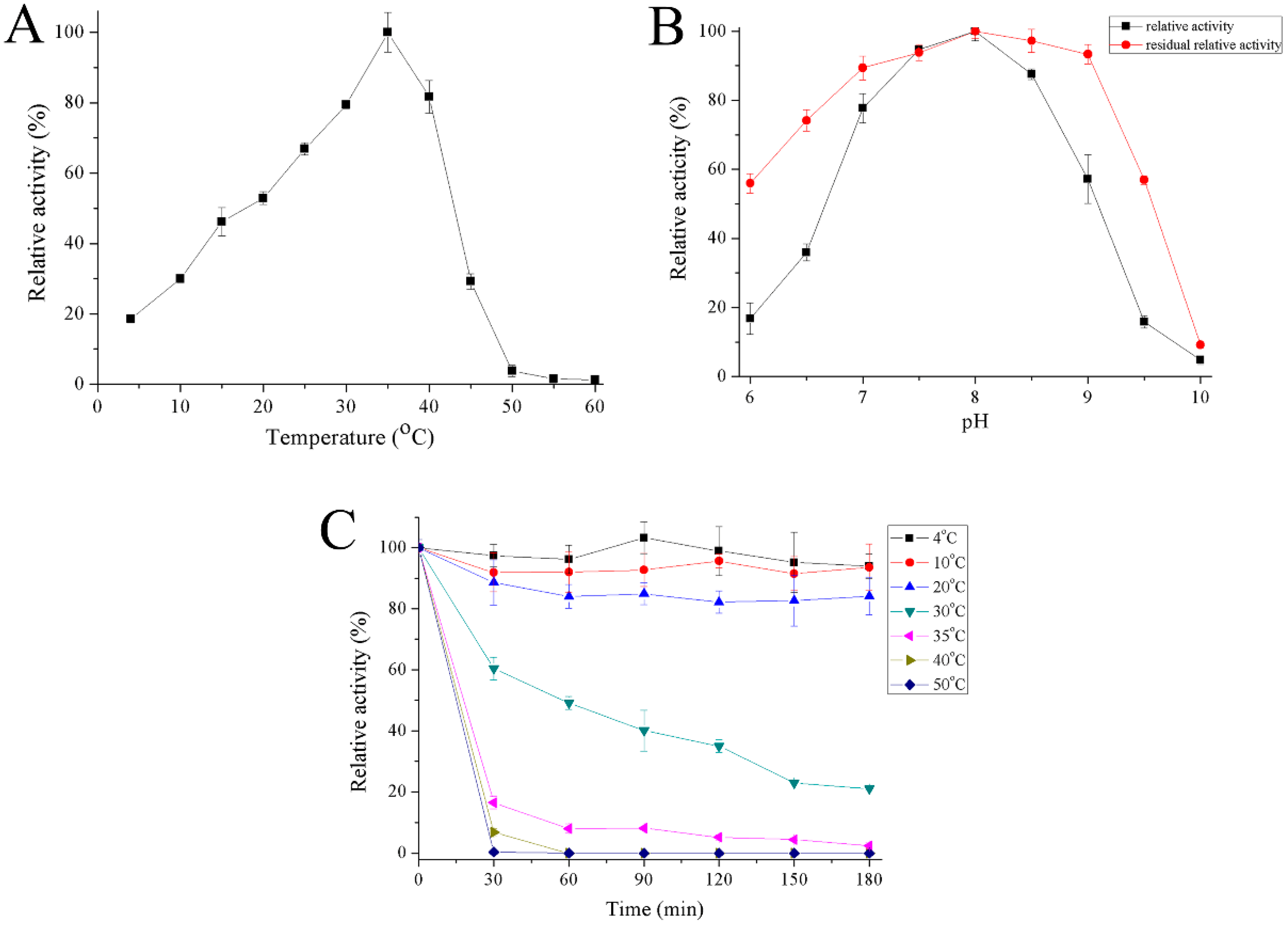
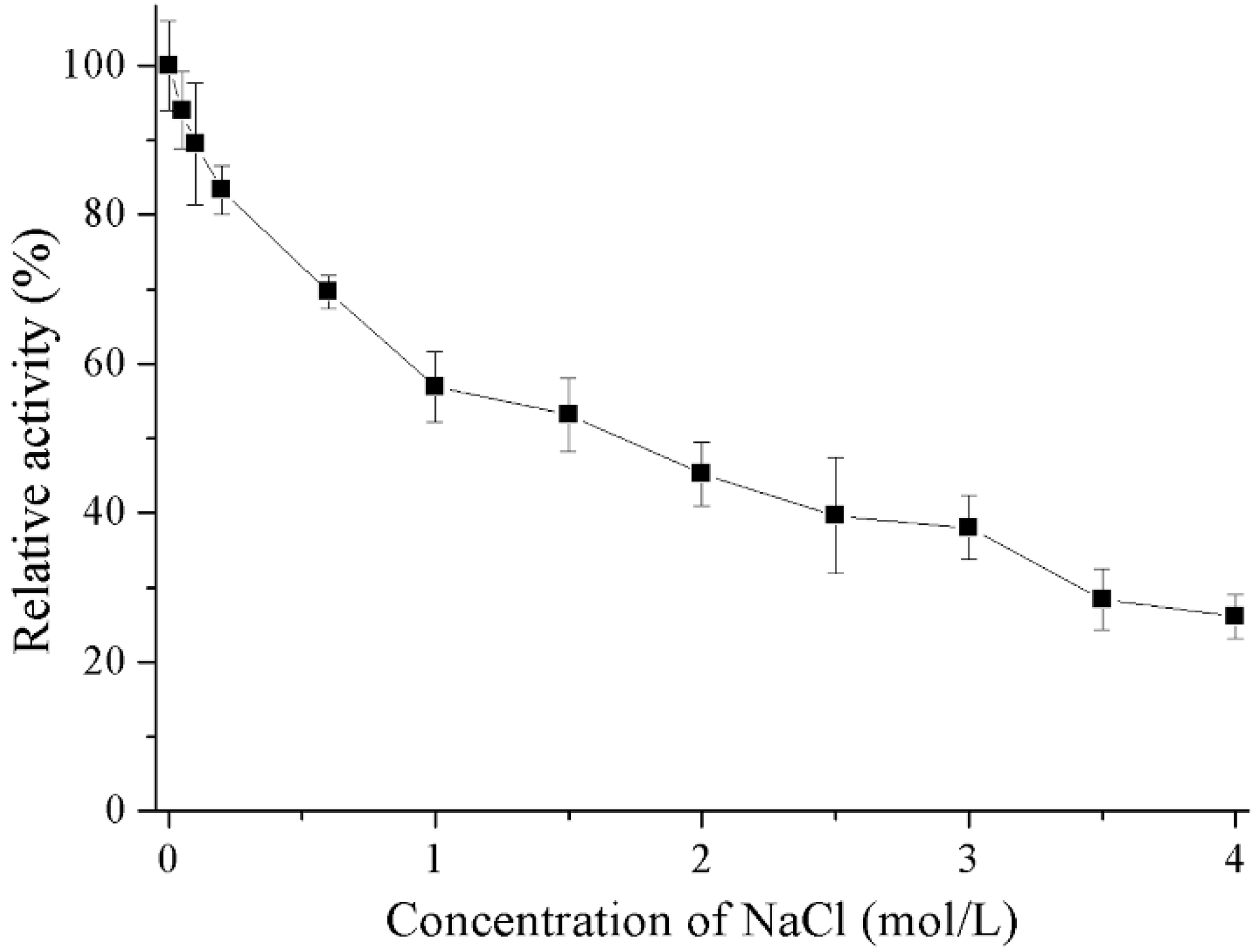
2.4. Properties of Recombinant Gal
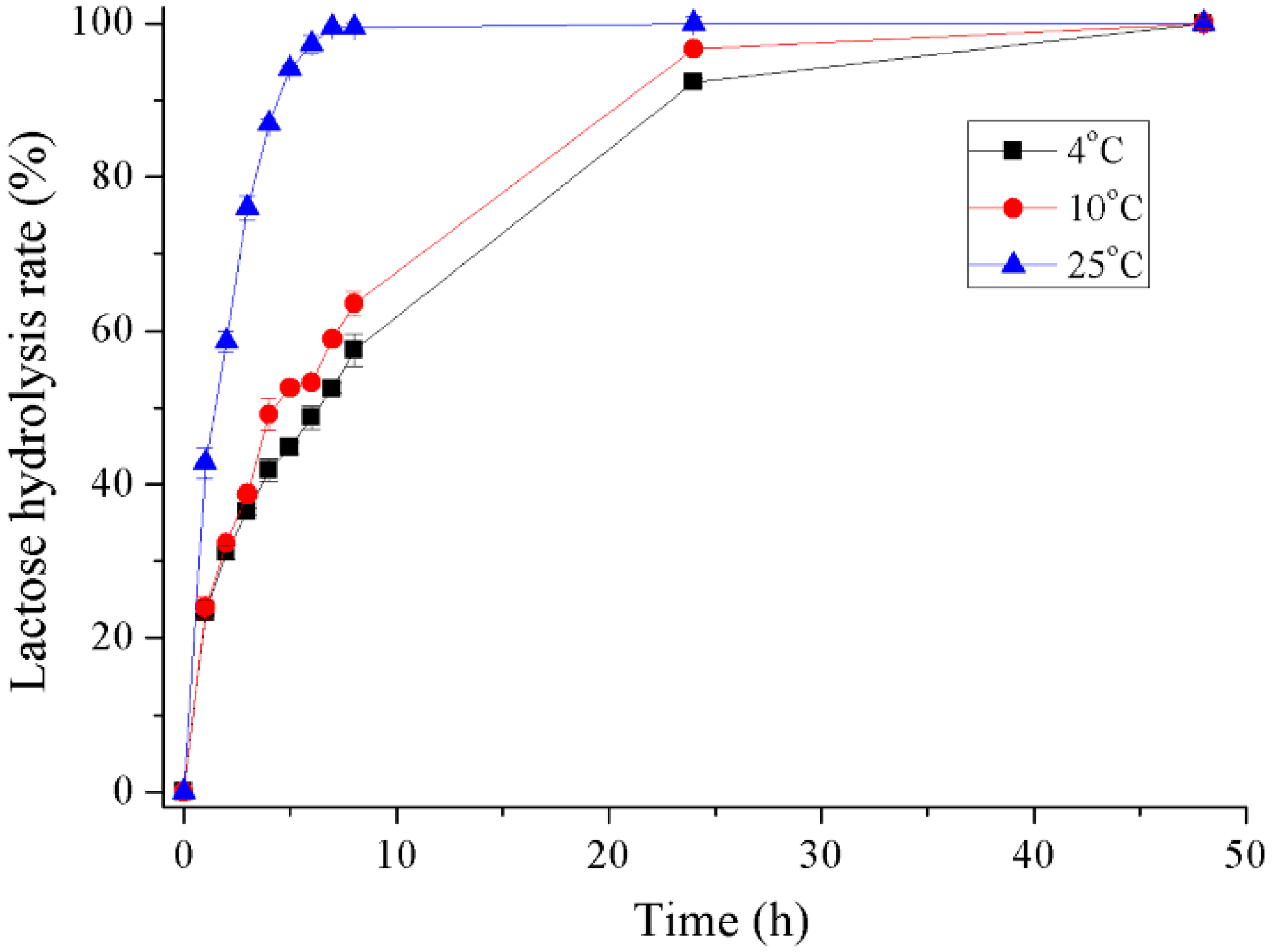
2.5. Hydrolysis of Lactose in Milk
3. Discussion
4. Materials and Methods
4.1. Isolation and Identification of Bacteria
4.2. Molecular Cloning and Sequence Analysis
4.3. Expression and Purification
4.4. Estimation of Molecular Weight
4.5. Beta-Galactosidase Activity Assay
4.6. Effect of Temperature and pH
4.7. Effect of Metal Ions and Chemicals
4.8. Determination of Substrate Specificity and Kinetic Parameters
4.9. Hydrolysis of Lactose in Milk
4.10. Nucleotide Sequence Accession Numbers
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Oliveira, C.; Guimarães, P.M.; Domingues, L. Recombinant microbial systems for improved β-galactosidase production and biotechnological applications. Biotechnol. Adv. 2011, 29, 600–609. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.A.; Satar, R. Recombinant β-galactosidases—Past, present and future: A mini review. J. Mol. Catal. B-Enzym. 2012, 81, 1–6. [Google Scholar] [CrossRef]
- Park, A.R.; Oh, D.K. Galacto-oligosaccharide production using microbial β-galactosidase: Current state and perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 1279–1286. [Google Scholar] [CrossRef] [PubMed]
- Husain, Q. Beta galactosidases and their potential applications: A review. Crit. Rev. Biotechnol. 2010, 30, 41–62. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekar, B.; van der Hoorn, R.A. Beta galactosidases in Arabidopsis and tomato—A mini review. Biochem. Soc. Trans. 2016, 44, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Vasiljevic, T.; Jelen, P. Lactose hydrolysis in milk as affected by neutralizers used for the preparation of crude β-galactosidase extracts from Lactobacillus bulgaricus 11842. Innov. Food Sci. Emerg. Technol. 2002, 3, 175–184. [Google Scholar] [CrossRef]
- Kim, C.S.; Ji, E.S.; Oh, D.K. A new kinetic model of recombinant beta-galactosidase from Kluyveromyces lactis for both hydrolysis and transgalactosylation reactions. Biochem. Biophys. Res. Commun. 2004, 316, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Panesar, P.S.; Panesar, R.; Singh, R.S.; Kennedy, J.F.; Kumar, H. Microbial production, immobilization and applications of beta-d-galactosidase. J. Chem. Technol. Biotechnol. 2006, 81, 530–543. [Google Scholar] [CrossRef]
- Horner, T.W.; Dunn, M.L.; Eggett, D.L.; Ogden, L.V. β-Galactosidase activity of commercial lactase samples in raw and pasteurized milk at refrigerated temperatures. J. Dairy Sci. 2011, 94, 3242–3249. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Colinas, B.; Fernandez-Arrojo, L.; Santos-Moriano, P.; Ballesteros, A.; Plou, F. Continuous Packed Bed Reactor with Immobilized β-Galactosidase for Production of Galactooligosaccharides (GOS). Catalysts 2016, 6, 189. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, L.; Lu, L.; Zhao, Y.; Kang, L.; Pang, X.; Liu, J.; Jiang, T.; Xiao, M.; Ma, B. Galactosylation of steroidal saponins by β-galactosidase from Lactobacillus bulgaricus L3. Glycoconj. J. 2016, 33, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Maischberger, T.; Leitner, E.; Nitisinprasert, S.; Juajun, O.; Yamabhai, M.; Nguyen, T.H.; Haltrich, D. Beta-galactosidase from Lactobacillus pentosus: Purification, characterization and formation of galacto-oligosaccharides. Biotechnol. J. 2010, 5, 838–847. [Google Scholar] [CrossRef] [PubMed]
- Feller, G.; Gerday, C. Psychrophilic enzymes: Hot topics in cold adaptation. Nat. Rev. Microbiol. 2003, 1, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Pakula, T.M.; Salonen, K.; Uusitalo, J.; Penttilä, M. The effect of specific growth rate on protein synthesis and secretion in the filamentous fungus Trichoderma reesei. Microbiology 2005, 151, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Stougaard, P. Identification, cloning and expression of a cold-active beta-galactosidase from a novel Arctic bacterium, Alkalilactibacillus ikkense. Environ. Technol. 2010, 31, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Alikkunju, A.P.; Sainjan, N.; Silvester, R.; Joseph, A.; Rahiman, M.; Antony, A.C.; Kumaran, R.C.; Hatha, M. Screening and Characterization of Cold-Active β-Galactosidase Producing Psychrotrophic Enterobacter ludwigii from the Sediments of Arctic Fjord. Appl. Biochem. Biotechnol. 2016, 180, 477–490. [Google Scholar] [CrossRef] [PubMed]
- Coker, J.A.; Sheridan, P.P.; Loveland-Curtze, J.; Gutshall, K.R.; Auman, A.J.; Brenchley, J.E. Biochemical characterization of a beta-galactosidase with a low temperature optimum obtained from an Antarctic Arthrobacter isolate. J. Bacteriol. 2003, 185, 5473–5482. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M.; Pulicherla, K.K.; Rekha, V.P.; Raja, P.K.; Sambasiva Rao, K.R. Cold active β-galactosidase from Thalassospira sp. 3SC-21 to use in milk lactose hydrolysis: A novel source from deep waters of Bay-of-Bengal. World J. Microbiol. Biotechnol. 2012, 28, 2859–2869. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Hua, X.; Zhang, Y.; Feng, Y.; Shen, Q.; Dong, J.; Zhao, W.; Zhang, W.; Jin, Z.; Yang, R. Cloning, expression and structural stability of a cold-adapted β-galactosidase from Rahnella sp. R3. Protein Expr. Purif. 2015, 115, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, T.; Ikehata, R.; Myoda, T.; Miyaji, T.; Tomizuka, N. Overexpression and functional analysis of cold-active beta-galactosidase from Arthrobacter psychrolactophilus strain F2. Protein Expr. Purif. 2007, 54, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, P.; Wanarska, M.; Kur, J. A new cold-adapted beta-d-galactosidase from the Antarctic Arthrobacter sp. 32c-gene cloning, overexpression, purification and properties. BMC Microbiol. 2009, 9, 151. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.X.; Gao, Y.; Hu, B.; Lu, X.L.; Liu, X.Y.; Jiao, B.H. A novel cold-adapted β-galactosidase isolated from Halomonas sp. S62: Gene cloning, purification and enzymatic characterization. World J. Microbiol. Biotechnol. 2013, 29, 1473–1480. [Google Scholar] [CrossRef] [PubMed]
- Wierzbicka-Woś, A.; Cieśliński, H.; Wanarska, M.; Kozłowska-Tylingo, K.; Hildebrandt, P.; Kur, J. A novel cold-active β-d-galactosidase from the Paracoccus sp. 32d—Gene cloning, purification and characterization. Microb. Cell Fact. 2011, 10, 108. [Google Scholar] [CrossRef]
- Hoyoux, A.; Jennes, I.; Dubois, P.; Genicot, S.; Dubail, F.; François, J.M.; Baise, E.; Feller, G.; Gerday, C. Cold-adapted beta-galactosidase from the Antarctic psychrophile Pseudoalteromonas haloplanktis. Appl. Environ. Microbiol. 2001, 67, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, T.; Ikehata, R.; Uchino, M.; Miyaji, T.; Takano, K.; Tomizuka, N. Cold-active acid beta-galactosidase activity of isolated psychrophilic-basidiomycetous yeast Guehomyces pullulans. Microbiol. Res. 2006, 161, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Karan, R.; Capes, M.D.; DasSarma, P.; DasSarma, S. Cloning, overexpression, purification, and characterization of a polyextremophilic β-galactosidase from the Antarctic haloarchaeon Halorubrum lacusprofundi. BMC Biotechnol. 2013, 13, 3. [Google Scholar] [CrossRef] [PubMed]
- Henrissat, B.; Davies, G. Structural and sequence-based classification of glycoside hydrolases. Curr. Opin. Struct. Biol. 1997, 7, 637–644. [Google Scholar] [CrossRef]
- Cieśliński, H.; Kur, J.; Białkowska, A.; Baran, I.; Makowski, K.; Turkiewicz, M. Cloning, expression, and purification of a recombinant cold-adapted beta-galactosidase from antarctic bacterium Pseudoalteromonas sp. 22b. Protein Expr. Purif. 2005, 39, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.M.; Li, H.; Cao, L.X.; Wu, P.C.; Zhang, C.T.; Sang, S.L.; Zhang, X.Y.; Chen, M.J.; Lu, J.Q.; Liu, Y.H. Molecular cloning and characterization of the gene encoding cold-active beta-galactosidase from a psychrotrophic and halotolerant Planococcus sp. L4. J. Agric. Food Chem. 2007, 55, 2217–2224. [Google Scholar] [CrossRef] [PubMed]
- Uchil, P.D.; Nagarajan, A.; Kumar, P. β-Galactosidase. Cold Spring Harb. Protoc. 2017, 774–779. [Google Scholar] [CrossRef] [PubMed]
- Skálová, T.; Dohnálek, J.; Spiwok, V.; Lipovová, P.; Vondrácková, E.; Petroková, H.; Dusková, J.; Strnad, H.; Králová, B.; Hasek, J. Cold-active beta-galactosidase from Arthrobacter sp. C2-2 forms compact 660 kDa hexamers: Crystal structure at 1.9A resolution. J. Mol. Biol. 2005, 353, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, K.; Mo, Z.; Liu, Y.; Hu, X. Crystallization and preliminary X-ray analysis of a cold-active β-galactosidase from the psychrotrophic and halotolerant Planococcus sp. L4. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2011, 67, 911–913. [Google Scholar] [CrossRef] [PubMed]
- Rutkiewicz-Krotewicz, M.; Pietrzyk-Brzezinska, A.J.; Sekula, B.; Cieśliński, H.; Wierzbicka-Woś, A.; Kur, J.; Bujacz, A. Structural studies of a cold-adapted dimeric β-D-galactosidase from Paracoccus sp. 32d. Acta Crystallogr. D Struct. Biol. 2016, 72, 1049–1061. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Arukha, A.P.; Mukhopadhyay, B.C.; Mitra, S.; Biswas, S.R. A constitutive unregulated expression of β-galactosidase in Lactobacillus fermentum M1. Curr. Microbiol. 2015, 70, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Białkowska, A.M.; Cieśliński, H.; Nowakowska, K.M.; Kur, J.; Turkiewicz, M. A new beta-galactosidase with a low temperature optimum isolated from the Antarctic Arthrobacter sp. 20B: Gene cloning, purification and characterization. Arch. Microbiol. 2009, 191, 825–835. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.Z.K.; Chen, X.D. Effects of temperature and pH on the catalytic activity of the immobilized β-galactosidase from Kluyveromyces lactis. Biochem. Eng. J. 2001, 9, 33–40. [Google Scholar] [CrossRef]
- Karasová-Lipovová, P.; Strnad, H.; Spiwok, V.; Maláa, Š.; Králová, B.; Russell, N.J. The cloning, purification and characterisation of a cold-active β-galactosidase from the psychrotolerant Antarctic bacterium Arthrobacter sp. C2-2. Enzym. Microb. Technol. 2003, 33, 836–844. [Google Scholar] [CrossRef]

| Ion/Reagent | Relative Activity (%) |
|---|---|
| None 1 | 100 ± 1.3 |
| K+ | 123 ± 0.6 |
| Ca2+ | 74.9 ± 5.4 |
| Mg2+ | 32.4 ± 1.4 |
| Mn2+ | 21.3 ± 0.9 |
| Ba2+ | 48.2 ± 2.1 |
| Zn2+ | 0.1 ± 0.04 |
| Ni2+ | 0.2 ± 0.03 |
| Cu2+ | 0 |
| EDTA | 0.2 ± 0.02 |
| Urea | 89.9 ± 0.3 |
| DTT | 116.9 ± 7 |
| 2-Mercaptoethanol | 108.9 ± 4 |
| SDS | 43.5 ± 1 |
| Substrate | Relative Activity (%) |
|---|---|
| 2-Nitrophenyl-β-d-galactopyranoside (ONPG) 1 | 100 ± 4.2 |
| 4-Nitrophenyl-β-d-galactopyranoside (PNPG) | 12.8 ± 0.5 |
| 4-Nitrophenyl-α-d-galactopyranoside | 0 |
| 2-Nitrophenyl-β-d-glucopyranoside | 0 |
| 4-Nitrophenyl-β-d-glucopyranoside | 0 |
| 4-Nitrophenyl-α-d-glucopyranoside | 0 |
| 4-Nitrophenyl-β-d-xylopyranoside | 0 |
| Substrate | Vmax (U/mg) | Km (mM) | kcat (S−1) | kcat/Km (S−1 mM−1) |
|---|---|---|---|---|
| ONPG | 464.7 | 0.14 | 3688.1 | 26343.6 |
| Lactose | 18.5 | 7.2 | 146.5 | 20.3 |
| Strain | Optimum | ONPG | Lactose | References | |||
|---|---|---|---|---|---|---|---|
| Temperature (°C) | pH | Km (mM) | kcat (S−1) | Km (mM) | kcat (S−1) | ||
| Alteromonas sp. ML52 | 35 | 8 | 0.14 | 3688.1 | 7.2 | 146.5 | This work |
| Arthrobacter psychrolactophilus strain F2 | 10 | 8 | 2.7 | 12.7 | 42.1 | 3.02 | [20] |
| Arthrobacter sp. 20B | 25 | 6–8 | - | - | - | - | [37] |
| Arthrobacter sp. C2-2 | 40 | 7.5 | - | - | 53.1 | 1106 | [39] |
| Arthrobacter sp. | 18 | 7 | 11.5 | 5.2 | - | - | [17] |
| Paracoccus sp. 32d | 40 | 7.5 | 1.17 | 71.81 | 2.94 | 43.23 | [23] |
| Pseudoalteromonas haloplanktis TAE 79 | - | 8.5 | - | 203 | 2.4 | 33 | [24] |
| Pseudoalteromonas sp. 22b | 40 | 6–8 | 0.28 | 312 | 3.3 | 157 | [28] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Yao, C.; Wang, W.; Zhuang, Z.; Liu, J.; Dai, F.; Hao, J. Cloning, Expression and Characterization of a Novel Cold-adapted β-galactosidase from the Deep-sea Bacterium Alteromonas sp. ML52. Mar. Drugs 2018, 16, 469. https://doi.org/10.3390/md16120469
Sun J, Yao C, Wang W, Zhuang Z, Liu J, Dai F, Hao J. Cloning, Expression and Characterization of a Novel Cold-adapted β-galactosidase from the Deep-sea Bacterium Alteromonas sp. ML52. Marine Drugs. 2018; 16(12):469. https://doi.org/10.3390/md16120469
Chicago/Turabian StyleSun, Jingjing, Congyu Yao, Wei Wang, Zhiwei Zhuang, Junzhong Liu, Fangqun Dai, and Jianhua Hao. 2018. "Cloning, Expression and Characterization of a Novel Cold-adapted β-galactosidase from the Deep-sea Bacterium Alteromonas sp. ML52" Marine Drugs 16, no. 12: 469. https://doi.org/10.3390/md16120469
APA StyleSun, J., Yao, C., Wang, W., Zhuang, Z., Liu, J., Dai, F., & Hao, J. (2018). Cloning, Expression and Characterization of a Novel Cold-adapted β-galactosidase from the Deep-sea Bacterium Alteromonas sp. ML52. Marine Drugs, 16(12), 469. https://doi.org/10.3390/md16120469




