Seaweed Secondary Metabolites In Vitro and In Vivo Anticancer Activity
Abstract
1. Introduction
2. Secondary Metabolites from Seaweeds with in Vitro Cytotoxic Activity
2.1. Mono-, Sesqui-, Diterpenes and Sterols
2.2. Carotenoid
2.3. Phenolic Compounds
2.3.1. Phlorotannins
2.3.2. Bromophenols
2.4. Alkaloids
3. Secondary Metabolites from Seaweeds with In Vivo Antitumor Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Akt | Serine/Threonine-Specific Protein Kinase |
| AMPK | Adenosine Monophosphate-Activated Protein Kinase |
| ATP | Adenosine Triphosphate |
| BALB | “Bagg Albino” |
| Bax | B-Cell Lymphoma 2-Associated X Protein |
| BCBL-1 | Body-Cavity-Based Lymphoma Cell Line |
| BCL-xL | B-Cell Lymphoma-Extra Large |
| BT549 | Human Carcinoma Breast Epithelial Cell Line |
| b.w. | Body Weight |
| CD44 | Cluster of Differentiation 44 |
| cdc2 | Cell Division Cycle Protein 2 |
| CDK4 | Cyclin-Dependent Kinase 4 |
| CpG | Cytosine Nucleotide Followed by A Guanine Nucleotide |
| CIAP | Cellular Inhibitors of Apoptosis Protein |
| COX | Cyclooxygenase |
| CSCs | Cancer Stem-Like Cells |
| CTX | Cyclophosphamide |
| CXCR4 | CXC Motif Chemokine Receptor 4 |
| DMSO | Dimethyl Sulphoxide |
| DNA | Deoxyribonucleic Acid |
| DNMT-1 | DNA Methyltransferase-1 |
| EGFR | Epidermal Growth Factor Receptor |
| eNOS | Endothelial Nitric Oxide Synthase |
| ERK | Extracellular Signal–Regulated Kinase |
| FAK | Focal Adhesion Kinase |
| FGFR | Fibroblast Growth Factor Receptor |
| G1 phase | Gap 1 Phase |
| GTP | Guanosine-5′-triphosphate |
| HaCaT | Aneuploid Immortal Keratinocyte Cell Line |
| HIF-1α | Hypoxia-Inducible Factor 1-Alpha |
| IC50 | The Half Maximal Inhibitory Concentration |
| K562 | Human Chronic Myelogenous Leucemia Cell Line |
| LOVO | Human Colon (Supraclavicular Lymph Node Metastasis) |
| M | Mitosis |
| MAPK | Mitogen-Activated Protein Kinase |
| MDA-MB-231 | Human Adenocarcinoma Breast Cell Line |
| MMP | Matrix Metalloproteinase |
| mRNA | Messenger Ribonucleic Acid |
| MYCN | Myelocytomatosis |
| NDEA | N-nitrosodiethylamine |
| NF-ĸB | Nuclear Factor-kappa B |
| NOD | Nonobese Diabetic |
| Notch-2 | Neurogenic Locus Notch Homolog Protein-2 |
| NSG | NOD Scid Gamma |
| Oct-4 | Octamer-Binding Transcription Factor 4 |
| ODC | Ornithine Decarboxylase |
| OXPHOS | Oxidative Phosphorylation |
| p15INK4B | Cyclin-Dependent Kinase Inhibitor 4B |
| p21WAF1/CIP1 | Cyclin-Dependent Kinase Inhibitor 1 |
| p27KIP1 | Cyclin-Dependent Kinase Inhibitor 1B |
| p75NTR | Neurotrophin Receptor p75 |
| PARP | Poly (ADP-Ribose) Polymerase |
| PCNA | Proliferating Cell Nuclear Antigen |
| PDGFRα | Platelet-Derived Growth Factor Receptor α |
| p-mTOR | Phosphorylated Mammalian Target of Rapamycin |
| PI3K | Phosphoinositide 3-Kinase |
| PTK | Protein Tyrosine Kinase |
| RAF-1 | Murine Leukemia Viral Oncogene Homolog 1 |
| RAS | Rat Sarcoma |
| Rb | Retinoblastoma |
| ROS | Reactive Oxygen Species |
| S phase | Synthesis Phase |
| SCID | Severe Combined Immunodeficiency |
| Ser | Serine |
| SIV | Sub-Intestinal Vessel |
| SLUG | Human Embryonic Protein Snail Family Transcriptional Repressor 2 |
| Sox2 | (Sex Determining Region Y)-Box 2 |
| STAT3 | Signal Transducer and Activator of Transcription 3 |
| SW480 | Human Colorectal Adenocarcinoma Cell Line |
| TIMP | Tissue Inhibitor of Metalloproteinase |
| TRADD | Tumour Necrosis Factor Receptor Superfamily Member 1A Associated via Death Domain |
| TY-1 | Primary Effusion Lymphoma Cell Line |
| U251 | Human Brain Glioblastoma Cell Line |
| UVB | Ultraviolet Radiation B |
| VEGFR2 | Vascular Endothelial Growth Factor Receptor 2 |
| VIM | Vimentin |
| XIAP | X-Linked Inhibitor of Apoptosis Protein |
References
- IARC, International Agency for Research on Cancer, World Health Organization. Press Release nº 263, 12 September 2018. Available online: http://gco.iarc.fr/today/home (accessed on 14 September 2018).
- Bray, F. Transitions in human development and the global cancer burden. In World Cancer Report 2014; Stewart, B.W., Wild, C.P., Eds.; International Agency for Research on Cancer: Lyon, France, 2014; pp. 54–68. ISBN 978-92-832-0443-5. [Google Scholar]
- WCRFI, World Cancer Research Fund International. Available online: http://www.wcrf.org/int/cancer-facts-figures/worldwide-data (accessed on 30 June 2018).
- Mullard, A. FDA approves first immunotherapy combo. Nat. Rev. Drug Discov. 2015, 14, 739. [Google Scholar] [CrossRef]
- Nicolini, A.; Carpi, A.; Ferrari, P.; Biava, P.M.; Rossi, G. Immunotherapy and hormone-therapy in metastatic breast cancer: A review and an update. Curr. Drug Targets 2016, 17, 1127–1139. [Google Scholar] [CrossRef] [PubMed]
- ASCO, American Society of Clinical Oncology. Available online: https://www.asco.org/research-progress/reports-studies/clinical-cancer-advances-2018/advances-cancer-treatment (accessed on 14 September 2018).
- Lartigue, J. Tumor heterogeneity: A central foe in the war on cancer. J. Community Support. Oncol. 2018, 16, 167–174. [Google Scholar] [CrossRef]
- Nikolaou, N.; Pavlopoulou, A.; Georgakilas, A.G.; Kyrodimos, E. The challenge of drug resistance in cancer treatment: A current overview. Clin. Exp. Metastasis 2018, 35, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Feinberg, A.P.; Ohlsson, R.; Henikoff, S. The epigenetic progenitor origin of human cancer. Nat. Rev. Genet. 2006, 7, 21–33. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Cancer Facts and Figures 2017. Atlanta: American Cancer Society. 2017. Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf (accessed on 30 June 2018).
- Sun, W. Recent advances in cancer immunotherapy. J. Hemat. Oncol. 2017, 10, 96. [Google Scholar] [CrossRef] [PubMed]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct. Target Ther. 2018, 3, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Torres, V.; Encinar, J.A.; Herranz-López, M.; Pérez-Sánchez, A.; Galiano, V.; Barrajón-Catalán, E.; Micol, V. An updated review on marine anticancer compounds: The use of virtual screening for the discovery of small-molecules cancer drugs. Molecules 2017, 22, 1037. [Google Scholar] [CrossRef] [PubMed]
- Seca, A.M.L.; Pinto, D.C.G.A. Plant secondary metabolites as anticancer agents: Successes in clinical trials and therapeutic application. Int. J. Mol. Sci. 2018, 19, 263. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981–2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Gamal, A.A.E. Biological importance of marine algae. Saudi Pharm. J. 2010, 18, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Boopathy, N.S.; Kathiresan, K. Anticancer drugs from marine flora: An overview. J. Oncol. 2010, 2010, 18. [Google Scholar] [CrossRef]
- Pérez, M.J.; Falqué, E.; Domínguez, H. Antimicrobial action of compounds from marine seaweed. Mar. Drugs 2016, 14, 52. [Google Scholar] [CrossRef]
- Wan-Loy, C.; Siew-Moi, P. Marine algae as a potential source for anti-obesity agents. Mar. Drugs 2016, 14, 222. [Google Scholar] [CrossRef] [PubMed]
- Seca, A.M.L.; Pinto, D.C.G.A. Overview on the antihypertensive and anti-obesity effects of secondary metabolites from seaweeds. Mar. Drugs 2018, 16, 237. [Google Scholar] [CrossRef] [PubMed]
- Rebours, C.; Marinho-Soriano, E.; Zertuche-González, J.A.; Hayashi, L.; Vásquez, J.A.; Kradolfer, P.; Soriano, G.; Ugarte, R.; Abreu, M.U.; Bay-Larsen, I.; et al. Seaweeds: An opportunity for wealth and sustainable livelihood for coastal communities. J. Appl. Phycol. 2014, 26, 1939–1951. [Google Scholar] [CrossRef] [PubMed]
- Anis, M.; Ahmed, S.; Hasan, M.M. Algae as nutrition, medicine and cosmetic: The forgotten history, present status and future trends. World J. Pharm. Pharm. Sci. 2017, 6, 1934–1959. [Google Scholar] [CrossRef]
- Gouveia, V.; Seca, A.M.L.; Barreto, M.C.; Pinto, D.C.G.A. Di-and sesquiterpenoids from Cystoseira genus: Structure, intra-molecular transformations and biological activity. Mini-Rev. Med. Chem. 2013, 13, 1150–1159. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Hiu, J.; Yang, B.; Lin, X.-P.; Zhou, X.-F.; Yang, X.-W.; Liu, Y. Chemical composition of seaweeds. Seaweed Sustainability-Food and-Non-Food Applications, 1st ed.; Tiwari, B.K., Troy, D., Eds.; Elsevier: London, UK, 2015; pp. 79–124. [Google Scholar]
- Cardoso, S.M.; Pereira, O.R.; Seca, A.M.L.; Pinto, D.C.G.A.; Silva, A.M.S. Seaweeds as preventive agents for cardiovascular diseases: From nutrients to functional foods. Mar. Drugs 2015, 13, 6838–6865. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Hou, S.; Song, S.; Zhang, B.; Ai, C.; Chen, X.; Liu, N. Impact of acidic, water and alkaline extraction on structural features, antioxidant activities of Laminaria japonica polysaccharides. Int. J. Biol. Macromol. 2018, 112, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Folmer, F.; Jaspars, M.; Dicato, M.; Diederich, M. Photosynthetic marine organisms as a source of anticancer compounds. Phytochem. Rev. 2010, 9, 557–579. [Google Scholar] [CrossRef]
- Murphy, C.; Hotchkiss, S.; Worthington, J.; McKeown, S.R. The potential of seaweed as a source of drugs for use in cancer chemotherapy. J. Appl. Phycol. 2014, 26, 2211–2264. [Google Scholar] [CrossRef]
- Alves, C.; Silva, J.; Pinteus, S.; Gaspar, H.; Alpoim, M.C.; Botana, L.M.; Pedrosa, R. From marine origin to therapeutics: The antitumor potential of marine algae-derived compounds. Front. Pharmacol. 2018, 9, 777. [Google Scholar] [CrossRef] [PubMed]
- Liby, K.T.; Yore, M.M.; Sporn, M.B. Triterpenoids and rexinoids as multifunctional agents for the prevention and treatment of cancer. Nat. Rev. Cancer 2007, 7, 357–369. [Google Scholar] [CrossRef] [PubMed]
- Rabi, T.; Bishayee, A. Terpenoids and breast cancer chemoprevention. Breast Cancer Res. Treat. 2009, 115, 223–239. [Google Scholar] [CrossRef] [PubMed]
- Ayyad, S.-E.N.; Abdel-Halim, O.B.; Shier, W.T.; Hoye, T.R. Cytotoxic hydroazulene diterpenes from the brown alga Cystoseira myrica. Z. Natuforsch 2003, 38, 33–38. [Google Scholar] [CrossRef]
- Gedara, S.R.; Abdel-Halim, O.B.; El-Sharkawy, S.H.; Salama, O.M.; Shier, W.T.; Halim, A.F. Cytotoxic hydroazulene diterpenes from the brown alga Dictyota dichotoma. Z. Naturforsch 2003, 58, 17–22. [Google Scholar] [CrossRef]
- Gouveia, V.L.M.; Seca, A.M.L.; Barreto, M.C.; Neto, A.I.; Kijjoa, A.; Silva, A.M.S. Cytotoxic meroterpenoids from the macroalga Cystoseira abies-marina. Phytochem. Lett. 2013, 6, 593–597. [Google Scholar] [CrossRef]
- Bouaïcha, N.; Tringali, C.; Pesando, D.; Malléa, M.; Roussakis, C.; Verbist, J.F. Bioactive diterpenoids isolated from Dipophus ligulatus. Planta Med. 1993, 59, 256–258. [Google Scholar] [CrossRef] [PubMed]
- Dorta, E.; Cueto, M.; Brito, I.; Darias, J. New terpenoids from the brown alga Stypopodium zonale. J. Nat. Prod. 2002, 65, 1727–1730. [Google Scholar] [CrossRef] [PubMed]
- Sabry, O.M.M.; Andrews, S.; McPhail, K.L.; Goeger, D.E.; Yokochi, A.; LePage, K.T.; Murray, T.F.; Gerwick, W.H. Neurotoxic meroditerpenoids from the tropical marine brown alga Stypopodium flabelliforme. J. Nat. Prod. 2005, 68, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- Abatis, D.; Vagias, C.; Galanakis, D.; Norris, J.N.; Moreau, D.; Roussakis, C.; Roussis, V. Atomarianones A and B: Two cytotoxic meroditerpenes from the brown alga Taonia atomaria. Tetrahedron. Lett. 2005, 46, 8525–8529. [Google Scholar] [CrossRef]
- Pereira, D.M.; Cheel, J.; Areche, C.; San-Martin, A.; Rovirosa, J.; Silva, L.R.; Valentao, P.; Andrade, P.B. Anti-proliferative activity of meroditerpenoids isolated from the brown alga Stypopodium flabelliforme against several cancer cell lines. Mar. Drugs 2011, 9, 852–862. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.; Urban, S. Meroditerpenoids from the southern Australian marine brown alga Sargassum fallax. Phytochemistry 2009, 70, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.C.; Kwon, H.C.; Kim, S.N.; Kim, H.S.; Um, B.H. Plastoquinones from Sargassum yezoense; Chemical structure and effects on the activation of peroxisome proliferator-activated receptor gamma. Chem. Pharm. Bull. 2011, 59, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Shi, D.; Ma, M.; Li, S.; Wang, S.; Han, L.; Yang, Y.; Fan, X.; Shi, J.; He, L. Sesquiterpenes from the red alga Laurencia tristicha. J. Nat. Prod. 2005, 68, 915–919. [Google Scholar] [CrossRef] [PubMed]
- Shizuri, Y.; Yamada, K. Laurebiphenyl, a dimeric sesquiterpene of the cyclolaurane-type from the red Laurencia nidifica. Phytochemistry 1985, 24, 1385–1386. [Google Scholar] [CrossRef]
- Sun, J.; Shi, D.-Y.; Wang, S.-J.; Han, L.-J.; Fan, X.; Yang, Y.-C.; Shi, J.-G. Chemical constituents of the red alga Laurencia tristicha. J. Asian Nat. Prod. Res. 2007, 9, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Zaleta-Pinet, D.A.; Holland, I.P.; Muñoz-Ochoa, M.; Murillo-Alvarez, J.I.; Sakoff, J.A.; van Altena, I.A.; McCluskey, A. Cytotoxic compounds from Laurencia pacifica. Org. Med. Chem. Lett. 2014, 4, 8. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Kurosawa, E. New aromatic sesquiterpenoids from the red alga Laurencia okamurai Yamada. Tetrahedron. Lett. 1978, 19, 2503–2506. [Google Scholar] [CrossRef]
- Antunes, E.M.; Afolayan, A.F.; Chiwakata, M.T.; Fakee, J.; Knott, M.G.; Whibley, C.E.; Hendricks, D.T.; Bolton, J.J.; Beukes, D.R. Identification and in vitro anti-esophageal cancer activity of a series of halogenated monoterpenes isolated from the South African seaweeds Plocamium suhrii and Plocamium cornutum. Phytochemistry 2011, 72, 769–772. [Google Scholar] [CrossRef] [PubMed]
- Sabry, O.M.M.; Goeger, D.E.; Valeriote, F.A.; Gerwick, W.H. Cytotoxic halogenated monoterpenes from Plocamium cartilagineum. Nat. Prod. Res. 2017, 31, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Mynderse, J.S.; Faulkner, D.J. Polyhalogenated monoterpenes from the red alga Plocamium cartilagineum. Tetrahedron. 1975, 31, 1963–1967. [Google Scholar] [CrossRef]
- Crews, P. Monoterpene halogenation by the red alga Plocamium oregonum. J. Org. Chem. 1977, 42, 2634–2636. [Google Scholar] [CrossRef] [PubMed]
- Knott, M.G. The Natural Product Chemistry of South African Plocamium Species. Master’s Thesis, Rhodes University, Grahamstown, South Africa, 2013. [Google Scholar]
- Vogel, C.V.; Pietraszkiewicz, H.; Sabry, O.M.; Gerwick, W.H.; Valeriote, F.A.; Vanderwal, C.D. Enantioselective divergent syntheses of several polyhalogenated Plocamium monoterpenes and evaluation of their selectivity for solid tumors. Angew. Chem. 2014, 126, 12401–12405. [Google Scholar] [CrossRef]
- Fuller, R.W.; Cardellina, J.H., II; Kato, Y.; Brinen, L.S.; Clardy, J.; Snader, K.M.; Boyd, M.R. A pentahalogenated monoterpene from the red alga Portieria hornemannii produces a novel cytotoxicity profile against a diverse panel of human tumor cell line. J. Med. Chem. 1992, 35, 3007–3011. [Google Scholar] [CrossRef] [PubMed]
- Kladi, M.; Xenaki, H.; Vagias, C.; Papazafiri, P.; Roussis, V. New cytotoxic sesquiterpenes from the red algae Laurencia obtuse and Laurencia microcladia. Tetrahedron. 2006, 62, 182–189. [Google Scholar] [CrossRef]
- Kim, M.-M.; Mendis, E.; Kim, S.-K. Laurencia okamurai extract containing laurinterol induces apoptosis in melanoma cells. J. Med. Chem. 2008, 11, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, L.S.; Tschoeke, D.A.; Oliveira, A.S.; Hill, L.J.; Paradas, W.C.; Salgado, L.T.; Thompson, C.C.; Pereira, R.C.; Thompson, F.L. New insights on the terpenone of the red seaweed Laurencia dendroidea (Florideophyceae, Rhodophyra). Mar. Drugs 2015, 13, 879–902. [Google Scholar] [CrossRef] [PubMed]
- Fuller, R.W.; Cardellina, J.H., II; Jurek, J.; Scheuer, P.; Alvarado-Lindner, B.; McGuire, M.; Gray, G.N.; Steiner, J.R.; Clardy, J.; Menez, E.; et al. Isolation and structure/activity features of halomon-related antitumor monoterpenes from the red alga Portieria hornemannii. J. Med. Chem. 1994, 37, 4407–4411. [Google Scholar] [CrossRef] [PubMed]
- Andrianasolo, E.H.; France, D.; Cornell-Kennon, S.; Gerwick, W.H. DNA methyl transferase inhibiting halogenated monoterpenes from the Madagascar red marine alga Portieria hornemannii. J. Nat. Prod. 2006, 69, 576–579. [Google Scholar] [CrossRef] [PubMed]
- Schlama, T.; Baati, R.; Gouverneur, V.; Valleix, A.; Falck, J.R.; Mioskowski, C. Total synthesis of (±)-halomon by a Johnson-Claisen rearrangement. Angew. Chem. Int. Ed. 1998, 37, 2085–2087. [Google Scholar] [CrossRef]
- Sotokawa, T.; Noda, T.; Pi, S.; Hirama, M. A three-step synthesis of halomon. Angew. Chem. Int. Ed. 2000, 39, 3430–3432. [Google Scholar] [CrossRef]
- Bucher, C.; Deans, R.M.; Burns, N.Z. Highly selective synthesis of halomon, plocamenone and isoplocamenone. J. Am. Chem. Soc. 2015, 137, 12784–12787. [Google Scholar] [CrossRef] [PubMed]
- Landry, M.L.; Burns, N.Z. Catalytic enantioselective dehalogenation in total synthesis. Acc. Chem. Res. 2018, 51, 1260–1271. [Google Scholar] [CrossRef] [PubMed]
- Depix, M.S.; Martínez, J.; Santibañez, F.; Rovirosa, J.; Martín, A.S.; Maccioni, R.B. The compound 14-keto-stypodiol diacetate from the algae Stypopodium flabelliforme inhibits microtubules and cell proliferation in DU-145 human prostatic cells. Mol. Cell. Biochem. 1998, 187, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Forth, S.; Kapoor, T.M. The mechanics of microtubule networks in cell division. J. Cell Biol. 2017, 216, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Santibañez, J.F.; Maccioni, R.B.; Martinez, J. The secretion of urokinase-like plasminogen activator is inhibited by microtubule-interacting drugs. Cell Biochem. Funct. 1995, 13, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.L.; Kavallaris, M.; McCarroll, J.A. Microtubules and their role in cellular stress in cancer. Front. Oncol. 2014, 4, 153. [Google Scholar] [CrossRef] [PubMed]
- Vairappan, C.S.; Suzuki, M.; Abe, T.; Masuda, M. Halogenated metabolites with antibacterial activity from the Okinawan Laurencia species. Phytochemistry 2001, 58, 517–523. [Google Scholar] [CrossRef]
- Suzuki, M.; Kurosawa, E.; Irie, T. Three new sesquiterpenoids containing bromine, minor constituents of Laurencia glandulifera Kützing. Tetrahedron. Lett. 1974, 15, 821–824. [Google Scholar] [CrossRef]
- Fedorov, S.N.; Shubina, L.K.; Bode, A.M.; Stonik, V.A.; Dong, Z. Dactylone inhibits epidermal growth factor-induced transformation and phenotype expression of human cancer cells and induces G1-S arrest and apoptosis. Cancer Res. 2007, 67, 5914–5921. [Google Scholar] [CrossRef] [PubMed]
- Tarhouni-Jabberi, S.; Zakraoui, O.; Ioannou, E.; Riach-Chebbi, I.; Haoues, M.; Roussis, V.; Kharrat, R.; Essafi-Benkhadir, K. Mertensene, a halogenated monoterpene, induces G2/M cell cycle arrest and caspase dependent apoptosis of human colon adenocarcinoma HT29 cell line through the modulation of ERK-1/-2, AKT and NF-KB signalling. Mar. Drugs. 2017, 15, 221. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Hashimoto, T.; Shimizu, K.; Yoshida, T.; Sakai, T.; Sowa, Y.; Komoto, A.; Kanazawa, K. Fucoxanthin induces cell cycle arrest at G0/G1 phase in human colon carcinoma cells through up-regulation of p21WAF1/Cip1. Biochim. Biophys. Acta 2005, 1726, 328–335. [Google Scholar] [CrossRef] [PubMed]
- D’Orazio, N.; Gemello, E.; Gammone, M.A.; Girolamo, M.; Ficoneri, C.; Riccioni, G. Fucoxanthin: A treasure from the sea. Mar. Drugs 2012, 10, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Mikami, K.; Hosokawa, M. Biosynthetic pathway and health benefits of fucoxanthin, an algae-specific xanthophyll in brown seaweeds. Int. J. Mol. Sci. 2013, 14, 13763–13781. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Ooi, T.; Hiraoka, M.; Oka, N.; Hamada, H.; Tamura, M.; Kusumi, T. Fucoxanthin and its metabolites in edible brown algae cultivated in deep seawater. Mar. Drugs 2004, 2, 63–72. [Google Scholar] [CrossRef]
- Wang, S.; Li, Y.; White, W.; Lu, J. Extracts from New Zealand Undaria pinnatifida containing fucoxanthin as potential functional biomaterials against cancer in vitro. J. Funct. Biomater. 2014, 5, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Ren, R.; Hashimoto, T.; Kanazawa, K. Fucoxanthin induces apoptosis in osteoclast-like cells differentiated from RAW264.7 cells. J. Agric. Food Chem. 2010, 58, 6090–6095. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Yuan, J.-P.; Wu, C.-F.; Wang, J.-H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; D′Orazio, N. Anti-obesity activity of the marine carotenoid fucoxanthin. Mar. Drugs 2015, 13, 2196–214. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tang, Y.; Zhang, Y.; Zhang, S.; Qu, J.; Wang, X.; Kong, R.; Han, C.; Liu, Z. Fucoxanthin: A promising medicinal and nutritional ingredient. Evid. Based Complement. Alternat. Med. 2015, 2015, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.R.; Hosokawa, M.; Miyashita, K. Fucoxanthin: A marine carotenoid exerting anti-cancer effects by affecting multiple mechanisms. Mar. Drugs 2013, 11, 5130–5147. [Google Scholar] [CrossRef] [PubMed]
- Zorofchian Moghadamtousi, S.; Karimian, H.; Khanabdali, R.; Razavi, M.; Firoozinia, M.; Zandi, K.; Abdul Kadir, H. Anticancer and antitumor potential of fucoidan and fucoxanthin, two main metabolites isolated from brown algae. Sci. World J. 2014, 2014, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.J. Fucoxanthin and its metabolite fucoxanthinol in cancer prevention and treatment. Mar. Drugs 2015, 13, 4784–4798. [Google Scholar] [CrossRef] [PubMed]
- Satomi, Y. Antitumor and cancer-preventative function of fucoxanthin: A marine carotenoid. Anticancer Res. 2017, 37, 1557–1562. [Google Scholar] [CrossRef] [PubMed]
- Okuzumi, J.; Nishino, H.; Murakoshi, M.; Iwashima, A.; Tanaka, Y.; Yamane, T.; Fujita, Y.; Takahashi, T. Inhibitory effects of fucoxanthin, a natural carotenoid, on N-myc expression and cell cycle progression in human malignant tumor cells. Cancer Lett. 1990, 55, 75–81. [Google Scholar] [CrossRef]
- Hosokawa, M.; Wanezaki, S.; Miyauchi, K.; Kurihara, H.; Kohno, H.; Kawabata, J.; Odashima, S.; Takahashi, K. Apostosis-inducing effect of fucoxanthin on human leukemia cell line HL-60. Food Sci. Technol. Res. 1999, 5, 243–246. [Google Scholar] [CrossRef]
- Kotake-Nara, E.; Kushiro, M.; Zhang, H.; Sugawara, T.; Miyashita, K.; Nagao, A. Carotenoids affect proliferation of human prostate cancer cells. J. Nutr. 2001, 131, 3303–3306. [Google Scholar] [CrossRef] [PubMed]
- Kotake-Nara, E.; Asai, A.; Nagao, A. Neoxanthin and fucoxanthin induce apoptosis in PC3 human prostate cancer cells. Cancer Lett. 2005, 220, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Hosokawa, M.; Kudo, M.; Maeda, H.; Kohno, H.; Tanaka, T.; Miyashita, K. Fucoxanthin induces apoptosis and enhances the antiproliferative effect of the PPARγ ligand, troglitazone, on colon cancer cells. Biochim. Biophys. Acta 2004, 1675, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Satomi, Y.; Nishino, H. Implication of mitogen-activated protein kinase in the induction of G1 cell cycle arrest and gadd45 expression by the carotenoid fucoxanthin in human cancer cells. Biochim. Biophys. Acta 2009, 1790, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Hashimoto, T.; Kanazawa, K. Growth inhibition of human hepatic carcinoma HepG2 cells by fucoxanthin is associated with down-regulation of cyclin D. Biochim. Biophys. Acta 2008, 1780, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Zhang, P.J.; Hamada, M.; Takahashi, S.; Xing, G.Q.; Liu, J.Q.; Sugiura, N. Potential chemoprevention effect of dietary fucoxanthin on urinary bladder cancer EJ-1 cell line. Oncol. Rep. 2008, 20, 1099–1110. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.L.; Huang, Y.S.; Hosokawa, M.; Miyashita, K.; Hu, M.L. Inhibition of proliferation of a hepatoma cell line by fucoxanthin in relation to cell cycle arrest and enhanced gap junctional intercellular communication. Chem. Biol. Interact. 2009, 182, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-N.; Heo, S.-J.; Kang, S.-M.; Ahn, G.; Jeon, Y.-J. Fucoxanthin induces apoptosis in human leukemia HL-60 cells through a ROS-mediated Bcl-xL pathway. Toxicol. In Vitro 2010, 24, 1648–1654. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.X.; Hu, X.M.; Xu, S.Q.; Jiang, Z.J.; Yang, W. Effects of fucoxanthin on proliferation and apoptosis in human gastric adenocarcinoma MGC-803 cells via JAK/STAT signal pathway. Eur. J. Pharmacol. 2011, 657, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Satomi, Y. Fucoxanthin induces GADD45A expression and G1 arrest with SAPK/JNK activation in LNCap Human prostate cancer cells. Anticancer Res. 2012, 32, 807–814. [Google Scholar] [PubMed]
- Ganesan, P.; Matsubara, K.; Sugawara, T.; Hirata, T. Marine algal carotenoids inhibit angiogenesis by down-regulating FGF-2-mediated intracellular signals in vascular endothelial cells. Mol. Cell. Biochem. 2013, 380, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zeng, Y.; Liu, Y.; Hu, X.; Li, S.; Wang, Y.; Li, L.; Lei, Z.; Zhang, Z. Fucoxanthin induces growth arrest and apoptosis in human bladder cancer T24 cells by up-regulation of p21 and down-regulation of mortalin. Acta Biochim. Biophys. Sin. 2014, 46, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zheng, J.; Zhang, Y.; Wang, Z.; Yang, Y.; Bai, M.; Dai, Y. Fucoxanthin activates apoptosis via inhibition of PI3K/Akt/mTOR pathway and supresses invasion and migration by restriction of p38-MMP-2/9 in human glioblastoma cells. Neurochem. Res. 2016, 41, 2728–2751. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Qiu, S.; Shao, N.; Zheng, J. Fucoxanthin and tumour necrosis factor-related apoptosis-inducing ligand (TRAIL) synergistically promotes apoptosis of human cervical cancer cells by targeting PI3K/Akt/NF-B signalling pathway. Med. Sci. Monit. 2018, 24, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Cheng, J.; Min, Z.; Yin, T.; Zhang, R.; Zhang, W.; Hu, L.; Cui, Z.; Gao, C.; Xu, S.; et al. Effect of fucoxanthin on autophagy and apoptosis in SGC-7901 cells and the mechanism. J. Cell Biochem. 2018, 119, 7274–7284. [Google Scholar] [CrossRef] [PubMed]
- Beppu, F.; Niwano, Y.; Tsukui, T.; Hosokawa, M.; Miyashita, K. Single and repeated oral dose toxicity study of fucoxanthin (FX), a marine carotenoid, in mice. J. Toxicol. Sci. 2009, 34, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Almeida, T.P.; Ferreira, J.; Vettorazzi, A.; Azqueta, A.; Rocha, E.; Ramos, A.A. Cytotoxic activity of fucoxanthin, alone and in combination with the cancer drugs imatinib and doxorubicin, in CML cell lines. Environ. Toxicol. Pharmacol. 2018, 59, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Xu, L.; Zhu, X.; Hu, J.; Peng, H.; Zeng, Z.; Xiong, H. Stability and bioaccessibility of fucoxanthin in nanoemulsions prepared from pinolenic acid-contained structured lipid. Int. J. Food Eng. 2017, 13, 1–14. [Google Scholar] [CrossRef]
- Vo, D.T.; Saravana, P.S.; Woo, H.-C.; Chun, B.-S. Fucoxanthin-rich oil encapsulation using biodegradable polyethylene glycol and particles from gas-saturated solutions technique. J. CO2 Util. 2018, 26, 359–369. [Google Scholar] [CrossRef]
- Li, H.; Xu, Y.; Sun, X.; Wang, S.; Wang, J.; Zhu, J.; Wang, D.; Zhao, L. Stability, bioactivity, and bioaccessibility of fucoxanthin in zein-caseinate composite nanoparticles fabricated at neutral pH by antisolvent precipitation. Food Hydrocol. 2018, 84, 379–388. [Google Scholar] [CrossRef]
- Ravi, H.; Kurrey, N.; Manabe, Y.; Sugawara, T.; Baskaran, V. Polymeric chitosan-glycolipid nanocarriers for an effective delivery of marine carotenoid fucoxanthin for induction of apoptosis in human colon Check for cancer cells (Caco-2 cells). Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 91, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, T.; Baskaran, V.; Tsuzuki, W.; Nagao, A. Brown algae fucoxanthin is hydrolysed to fucoxanthinol during absorption by CaCo-2 human intestinal cells and mice. J. Nutr. 2002, 132, 946–951. [Google Scholar] [CrossRef] [PubMed]
- Konishi, I.; Hosokawa, M.; Sashima, T.; Kobayashi, H.; Miyashita, K. Halocynthiaxanthin and fucoxanthinol isolated from Halocynthia roretzi induce apoptosis in human leukemia, breast and colon cancer cells. Comp. Biochem. Physiol. C 2006, 142, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Rwigemera, A.; Mamelona, J.; Martin, L.J. Inhibitory effects of fucoxanthinol on the viability of human breast cancer cell lines MCF-7 and MDA-MB-231 are correlated with modulation of the NF-κB pathway. Cell Biol. Toxicol. 2014, 30, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Asai, A.; Sugawara, T.; Ono, H.; Nagao, A. Biotransformation of fucoxanthinol into amarouciaxanthin A in mice and HepG2 cells: Formation and cytotoxicity of fucoxanthin metabolites. Drug Metab. Dispos. 2004, 32, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, C.; Tafuku, S.; Kadekaru, T.; Sawada, S.; Tomita, M.; Okudaira, T.; Nakazato, T.; Toda, T.; Uchihara, J.-N.; Taira, N.; et al. Antiadult T-cell leukemia effects of brown algae fucoxanthin and its deacetylated product, fucoxanthinol. Int. J. Cancer 2008, 123, 2702–2712. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Ishikawa, C.; Katano, H.; Yasumoto, T.; Mori, N. Fucoxanthin and its deacetylated product, fucoxanthinol, induce apoptosis of primary effusion lymphomas. Cancer Lett. 2011, 300, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-X.; Wijesekara, I.; Li, Y.; Kim, S.-K. Phlorotannins as bioactive agents from brown algae. Process Biochem. 2011, 46, 2219–2224. [Google Scholar] [CrossRef]
- Gómez-Guzmán, M.; Rodríguez-Nogales, A.; Algieri, F.; Gálvez, J. Potential role of seaweed polyphenols in cardiovascular-associated disorders. Mar. Drugs 2018, 16, 250. [Google Scholar] [CrossRef]
- Gutiérrez-Rodríguez, A.G.; Juárez-Portilla, C.; Olivares-Bañuelos, T.; Zepeda, R.C. Anticancer activity of seaweeds. Drug Discov. Today 2018, 23, 434–447. [Google Scholar] [CrossRef] [PubMed]
- Cabrita, M.T.; Vale, C.; Rauter, A.P. Halogenated compounds from marine algae. Mar. Drugs 2010, 8, 2301–2317. [Google Scholar] [CrossRef] [PubMed]
- Catarino, M.D.; Silva, A.M.S.; Cardoso, S.M. Fucaceae: A source of bioactive phlorotannins. Int. J. Mol. Sci. 2017, 18, 1327. [Google Scholar] [CrossRef]
- Tanniou, A.; Vandanjon, L.; Incera, M.; Serrano, L.E.; Husa, V.; Grand, J.; Nicolas, J.L.; Poupart, N.; Kervarec, N.; Engelen, A.; et al. Assessment of the spatial variability of phenolic contents and associated bioactivities in the invasive alga Sargassum muticum sampled along its European range from Norway to Portugal. J. Appl. Phycol. 2013, 26, 1215–1230. [Google Scholar] [CrossRef]
- Abdala-Díaz, R.; Cabello-Pasini, A.; Pérez-Rodríguez, E.; Álvarez, R.; Figueroa, F. Daily and seasonal variations of optimum quantum yield and phenolic compounds in Cystoseira tamariscifolia (Phaeophyta). Mar. Biol. 2006, 148, 459–465. [Google Scholar] [CrossRef]
- Yoshie, Y.; Wang, W.; Petillo, D.; Suzuki, T. Distribution of catechins in Japanese seaweeds. Fish Sci. 2000, 66, 998–1000. [Google Scholar] [CrossRef]
- Rodríguez-Bernaldo de Quirós, A.; Lage-Yusty, M.A.; López-Hernández, J. Determination of phenolic compounds in macroalgae for human consumption. Food Chem. 2010, 121, 634–638. [Google Scholar] [CrossRef]
- Lopes, G.; Andrade, P.B.; Valentão, P. Phlorotannins: Towards new pharmacological interventions for diabetes mellitus type 2. Molecules 2017, 22, 56. [Google Scholar] [CrossRef] [PubMed]
- Glombitza, K.-W.; Pauli, K. Fucols and phlorethols from the brown alga Scytothamnus australis hook. et harv. (Chnoosporaceae). Bot. Mar. 2003, 46, 315–320. [Google Scholar] [CrossRef]
- Singh, I.P.; Bharate, S.B. Phloroglucinol compounds of natural origin. Nat. Prod. Rep. 2006, 23, 558–591. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.P.; Hofseth, L.J.; Harris, C.C. Radical causes of cancer. Nat. Rev. Cancer 2003, 3, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Wijesekara, I.; Yoon, N.Y.; Li, Y.; Kim, S.-K. Phlorotannins from Ecklonia cava (Phaeophyceae): Biological activities and potential health benefits. BioFactors 2010, 36, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Kong, C.-S.; Kim, J.-A.; Yoon, N.-Y.; Kim, S.-K. Induction of apoptosis by phloroglucinol derivative from Ecklonia Cava in MCF-7 human breast cancer cells. Food Chem. Toxicol. 2009, 47, 1653–1658. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.-R.; Shin, T.-S.; Lee, M.S.; Park, J.-Y.; Park, K.-E.; Yoon, N.-Y.; Kim, J.-S.; Choi, J.-S.; Jang, B.-C.; Byun, D.-S.; et al. Isolation and identification of phlorotannins from Ecklonia stolonifera with antioxidant and anti-inflammatory properties. J. Agric. Food Chem. 2009, 57, 3483–3489. [Google Scholar] [CrossRef] [PubMed]
- Ryu, B.; Ahn, B.N.; Kang, K.H.; Kim, Y.S.; Li, Y.X.; Kong, C.S.; Kim, S.K.; Kim, D.G. Dioxinodehydroeckol protects human keratinocyte cells from UVB-induced apoptosis modulated by related genes Bax/Bcl-2 and caspase pathway. J. Photochem. Photobiol. B 2015, 153, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.-K.; Tang, Y.; Kim, Y.-S.; Hwang, J.-W.; Choi, E.-J.; Lee, J.-H.; Lee, S.-H.; Jeon, Y.-J.; Park, P.-J. First evidence that Ecklonia cava-derived dieckol attenuated MCF-7 human breast carcinoma cell migration. Mar. Drugs 2015, 13, 1785–1797. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.-H.; Yang, Y.-I.; Lee, K.-T.; Choi, J.-H. Dieckol, isolated from the edible brown algae Ecklonia cava induces apoptosis of ovarian cancer cells and inhibits tumour xenograft growth. J. Cancer Res. Clin. Oncol. 2015, 141, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.-K.; Uddin, N.; Hyun, J.-W.; Kim, C.; Suh, Y.; Lee, S.-J. Novel anticancer activity of phloroglucinol against breast cancer stem-like cells. Toxicol. Appl. Pharm. 2015, 286, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.-K.; Suh, Y.; Yoo, K.C.; Cui, Y.-H.; Hwang, E.; Kim, H.-J.; Kang, J.-S.; Kim, M.-J.; Lee, Y.Y.; Lee, S.-J. Phloroglucinol suppresses metastatic ability of breast cancer cells by inhibition of epithelial-mesenchymal cell transition. Cancer Sci. 2015, 106, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Katsui, N.; Suzuki, Y.; Kitamura, S.; Irie, T. 5,6-dibromoprotocatechualdehyde and 2,3-dibromo-4,5-dihydroxybenzyl methyl ether: New dibromophenols from Rhodomela larix. Tetrahedron 1967, 23, 1185–1188. [Google Scholar] [CrossRef]
- Kurata, K.; Taniguchii, K.; Takashima, K.; Hayashi, I.; Suzuki, M. Feeding-deterrent bromophenols from Odonthalia corymbifera. Phytochemistry 1997, 45, 485–487. [Google Scholar] [CrossRef]
- Kurihara, H.; Mitani, T.; Kawabata, J.; Takahashi, K. Two new bromophenols from the red alga Odonthalia corymbifera. J. Nat. Prod. 1999, 62, 882–884. [Google Scholar] [CrossRef] [PubMed]
- Shoeib, N.A.; Bibby, M.C.; Blunden, G.; Linley, P.A.; Swaine, D.J.; Wheelhouse, R.T.; Wright, C.W. In-vitro cytotoxic activities of the major bromophenols of the red alga Polysiphonia lanosa and some novel synthetic isomers. J. Nat. Prod. 2004, 67, 1445–1449. [Google Scholar] [CrossRef] [PubMed]
- Barreto, M.; Meyer, J.J.M. Isolation and antimicrobial activity of a lanosol derivative from Osmundaria serrata (Rhodophyta) and a visual exploration of its biofilm covering. S. Afr. J. Bot. 2006, 72, 521–528. [Google Scholar] [CrossRef]
- Liu, M.; Hansen, P.E.; Lin, X. Bromophenols in marine algae and their bioactivities. Mar. Drugs 2011, 9, 1273–1292. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, B.; Rold, T.L.; Harrell, C.E.; Burns, M.W., III; Waaland, J.R. Re-examination of the bromophenols in the red alga Rhodomela Larix. Phytochemistry 1975, 14, 2667–2670. [Google Scholar] [CrossRef]
- Han, L.; Xu, N.; Shi, J.; Yan, X.; Zheng, C. Isolation and pharmacological activities of bromophenols from Rhodomela confervoides. Chin. J. Oceanol. Limn. 2005, 23, 226–229. [Google Scholar] [CrossRef]
- Xu, X.-L.; Fan, X.; Sing, F.-H.; Zhao, J.-L.; Han, L.-J.; Yang, Y.-C.; Shi, J.-G. Bromophenols from the brown alga Leathesia nana. J. Asian Nat. Prod. Res. 2004, 6, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Dayong, S.; Jing, L.; Shuju, G.; Hua, S.; Xiao, F. The antitumor effect of bromophenol derivatives in vitro and Leathesia nana extract in vivo. Chin. J. Oceanol. Limn. 2009, 27, 277–282. [Google Scholar] [CrossRef]
- Fan, X.; Xu, N.-J.; Shi, J.-G. Bromophenols from the red alga Rhodomela confervoides. J. Nat. Prod. 2003, 66, 455–458. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Luo, J.; Jiang, B.; Wang, L.; Wang, S.; Wang, C.; Fu, C.; Li, J.; Shi, D. Marine bromophenol bis (2,3-dibromo-4,5-dihydroxy-phenyl)-methane inhibits the proliferation, migration, and invasion of hepatocellular carcinoma cells via modulating β1-Integrin/FAK signaling. Mar. Drugs 2015, 13, 1010–1025. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, L.-J.; Jiang, B.; Wu, N.; Li, X.; Liu, S.; Luo, J.; Shi, D. Anti-angiogenic properties of BDDPM, a bromophenol from marine red alga Rhodomela confervoides, with multi receptor tyrosine kinase inhibition effects. Int. J. Mol. Sci. 2015, 16, 13548–13560. [Google Scholar] [CrossRef] [PubMed]
- Yared, M.A.; Middleton, L.P.; Meric, F.; Cristofanilli, M.; Sahin, A.A. Expression of c-kit proto-oncogene product in breast tissue. Breast J. 2004, 10, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhang, W.; Wei, J.; Qiu, L.; Lin, X. Marine bromophenol bis(2,3-dibromo-4,5-dihydroxybenzyl) ether, induces mitochondrial apoptosis in K562 cells and inhibits topoisomerase I in vitro. Toxicol. Lett. 2012, 211, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Seiter, K. Toxicity of the topoisomerase I inhibitors. Expert Opin. Drug Saf. 2005, 4, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Liu, G.; Qiu, L.; Lin, X.; Liu, M. Marine bromophenol bis(2,3-dibromo-4,5-dihydroxybenzyl) ether, represses angiogenesis in HUVEC cells and in zebrafish embryos via inhibiting the VEGF signal systems. Biomed. Pharmacother. 2015, 75, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Güven, K.C.; Percot, A.; Sezik, E. Alkaloids in marine algae. Mar. Drugs 2010, 8, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.; Valentão, P.; Andrade, P.B. Bioactive compounds from macroalgae in the new Millennium: Implications for neurodegenerative diseases. Mar. Drugs 2014, 12, 4934–4972. [Google Scholar] [CrossRef] [PubMed]
- Gross, H.; Goeger, D.E.; Hills, P.; Mooberry, S.L.; Ballantine, D.L.; Murray, T.F.; Valeriote, F.A.; Gerwick, W.H. Lophocladines, bioactive alkaloids from the red alga Lophocladia sp. J. Nat. Prod. 2006, 69, 640–644. [Google Scholar] [CrossRef] [PubMed]
- Prasad, V.V.T.S.; Gopalan, R.O.G. Continued use of MDA-MB-435, a melanoma cell line, as a model for human breast cancer, even in year, 2014. NPJ Breast Cancer 2015, 1. [Google Scholar] [CrossRef] [PubMed]
- Lotter, M.; Schilling, J.; Reimann, E.; Bracher, F. First total synthesis of the 2,7-naphthyridine alkaloids lophocladine A and B. Arch. Pharm. Chem. Life Sci. 2006, 339, 677–679. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Ding, C.; Cheng, C.; Yao, Q. Convenient synthesis of 2,7-naphthyridine lophocladines A and B and their analogues. J. Comb. Chem. 2007, 9, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Disadee, W.; Ploypradith, P.; Aree, T.; Chaichit, N.; Ruchirawat, S. A novel approach for the synthesis of lophocladines A, B and C1 analogues. Tetrahedron. Lett. 2011, 52, 6142–6144. [Google Scholar] [CrossRef]
- Theeramunkong, S.; Vajragupta, O.; Mudjupa, C. Synthesis and biological evaluation of simplified analogs of lophocladine B as potential antitumor agents. Med. Chem. Res. 2016, 25, 2959–2964. [Google Scholar] [CrossRef]
- Kochanowska-Karamyan, A.J.; Hamann, M.T. Marine indole alkaloids: Potential new drug leads for the control of depression and anxiety. Chem. Rev. 2010, 110, 4489–4497. [Google Scholar] [CrossRef] [PubMed]
- Netz, N.; Till, O. Marine indole alkaloids. Mar. Drugs 2015, 13, 4814–4914. [Google Scholar] [CrossRef] [PubMed]
- Lunagariya, J.; Bhadja, P.; Zhong, S.; Vekariya, R.; Xu, S. Marine natural product bis-indole alkaloid caulerpin: Chemistry and biology. MiniRev. Med. Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Vest, S.E.; Dawes, C.J.; Romeo, J.T. Distribution of caulerpin and caulerpicin in eight species of the green alga Caulerpa (Caulerpales). Bot. Mar. 1983, 26, 313–316. [Google Scholar] [CrossRef]
- Anjaneyulu, A.S.R.; Prakash, C.V.S.; Mallavadhani, U.V. Sterols and terpenes of the marine green algal species Caulerpa racemosa and Codium decorticatum. J. Indian Chem. Soc. 1991, 68, 480. [Google Scholar]
- Yan, S.; Su, J.; Wang, Y.; Zeng, L. Studies on chemical constituents of Halimeda incrassata. Trop. Ocean 1999, 18, 91–94. [Google Scholar]
- Govenkar, M.B.; Wahidulla, S. Constituents of Chondria armata. Phytochemistry 2000, 54, 979–981. [Google Scholar] [CrossRef]
- Xu, X.; Su, J. The separation, identification and bioassay of caulerpin. Ziran Kexueban 1996, 35, 64–66. [Google Scholar]
- Liu, Y.; Morgan, J.B.; Coothankandaswamy, V.; Liu, R.; Jekabsons, M.B.; Mahdi, F.; Nagle, D.G.; Zhou, Y.-D. The Caulerpa pigment caulerpin inhibits HIF-1 activation and mitochondrial respiration. J. Nat. Prod. 2009, 72, 2104–2109. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Zhang, H.; Dong, M.; Wu, Z.; Shen, Z.; Xie, Y.; Kong, Z.; Dai, X.; Xu, B. Metabolic reprogramming and AMPKα1 pathway activation by caulerpin in colorectal cancer cells. Int. J. Oncol. 2017, 50, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Ferramosca, A.; Conte, A.; Guerra, F.; Felline, S.; Rimoli, M.G.; Mollo, E.; Zara, V.; Terlizzi, A. Metabolites from invasive pests inhibit mitochondrial complex II: A potential strategy for the treatment of human ovarian carcinoma. Biochem. Biophys. Res. Commun. 2016, 473, 1133–1138. [Google Scholar] [CrossRef] [PubMed]
- Montopoli, M.; Bellanda, M.; Lonardoni, F.; Ragazzi, E.; Dorigo, P.; Froldi, G.; Mammi, S.; Caparrotta, L. Metabolic reprogramming in ovarian cancer cells resistant to cisplatin. Curr. Cancer Drug Tragets 2011, 11, 226–235. [Google Scholar] [CrossRef]
- Okuzumi, J.; Takahashi, T.; Yamane, T.; Kitao, Y.; lnagake, M.; Ohya, K.; Nishino, H.; Tanaka, Y. Inhibitory effects of fucoxanthin, a natural crotenoid, on N-ethyl-N’-nitro-N-nitrosoguanidine-induced mouse duodenal carcinogenesis. Cancer Lett. 1993, 68, 159–168. [Google Scholar] [CrossRef]
- Auvinen, M.; Laine, A.; Paasinen-Sohns, A.; Kangas, A.; Kangas, L.; Saksela, O.; Andersson, L.C.; Hölttä, E. Human ornithine decarboxylase-overproducing NIH3T3 cells induce rapidly growing, highly vascularized enphatiss in nude mice. Cancer Res. 1997, 57, 3016–3025. [Google Scholar] [PubMed]
- Shimoda, H.; Tanaka, J.; Shan, S.-J.; Maoka, T. Anti-pigmentary activity of fucoxanthin and its influence on skin mRNA expression of melanogenic molecules. J. Pharm. Pharmacol. 2010, 62, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, S.; Xu, S.; Yu, X.; Ma, D.; Hu, X.; Cao, X. In vivo induction of apoptosis by fucoxanthin, a marine carotenoid, associated with down-regulating STAT3/EGFR signalling in Sarcoma 180 (S180) xenografts-bearing mice. Mar. Drugs 2012, 10, 2055–2068. [Google Scholar] [CrossRef] [PubMed]
- Sadeeshkumar, V.; Duraikannu, A.; Ravichandran, S.; Kodisundaram, P.; Fredrick, W.S.; Gobalakrishnan, R. Modulatory efficacy of dieckol on xenobiotic-metabolizing enzymes, cell proliferation, apoptosis, invasion, and angiogenesis during NDEA-induced rat hepatocarcinogenesis. Mol. Cell Biochem. 2017, 433, 195–204. [Google Scholar] [CrossRef] [PubMed]
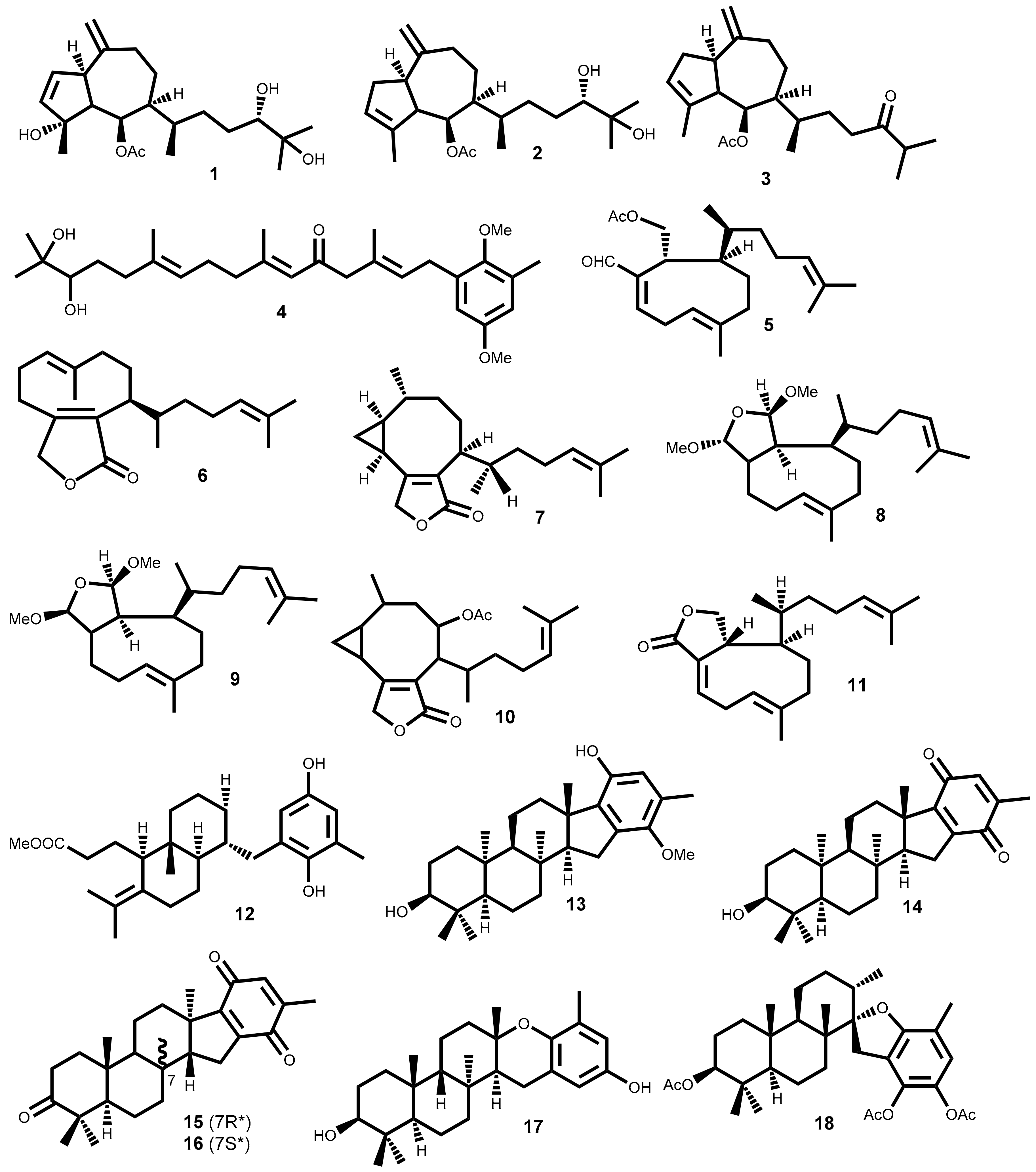
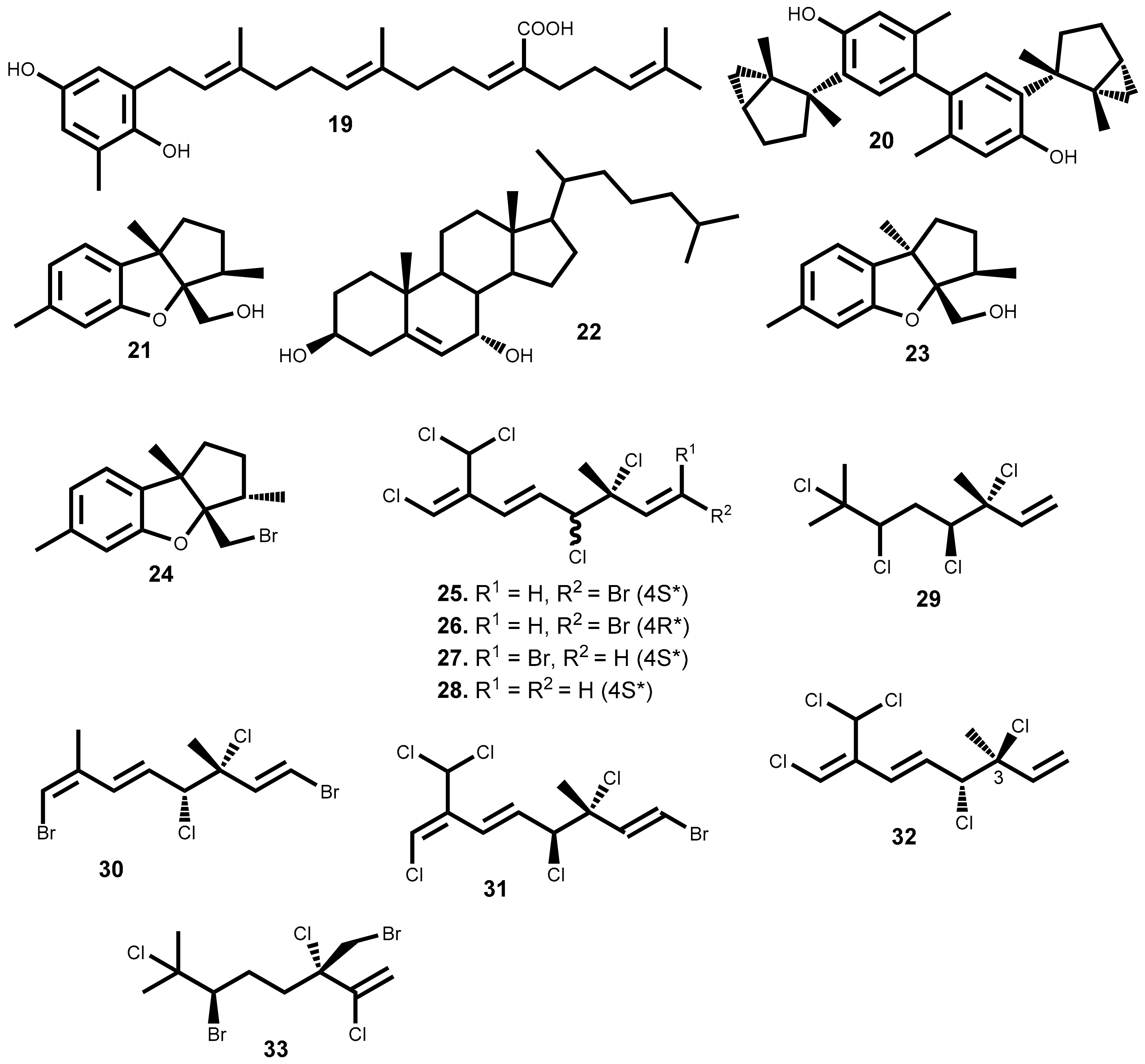
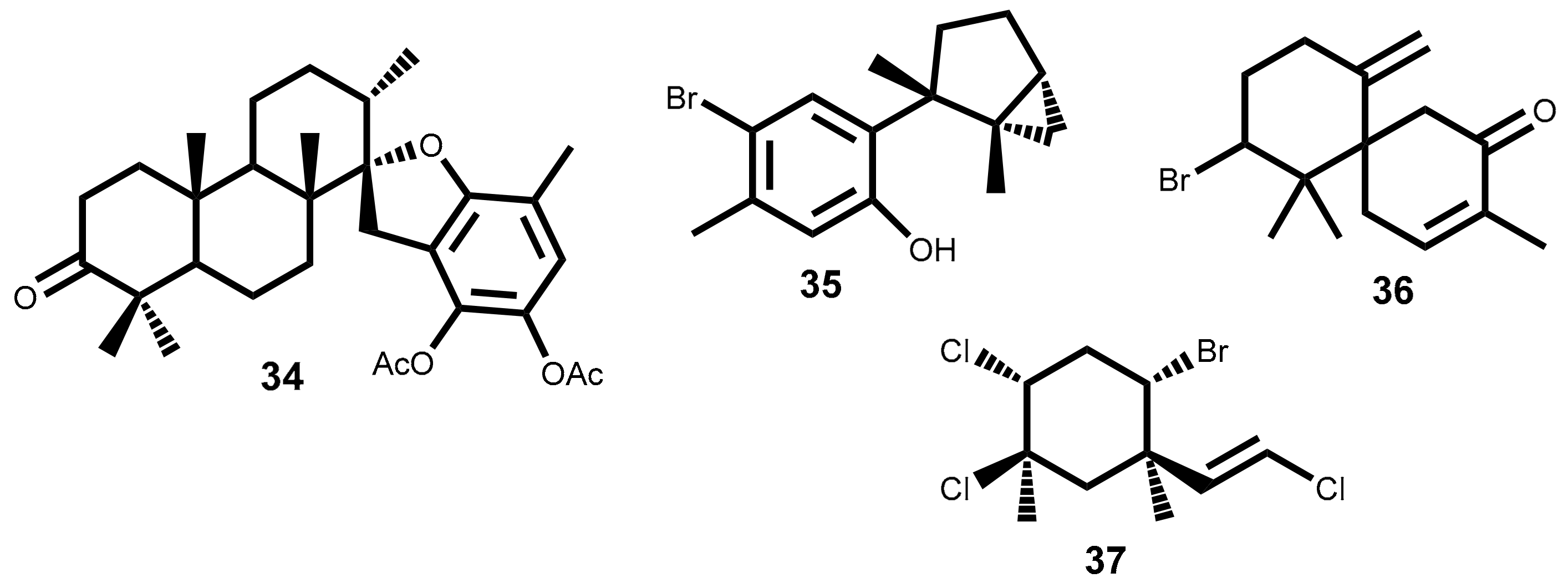
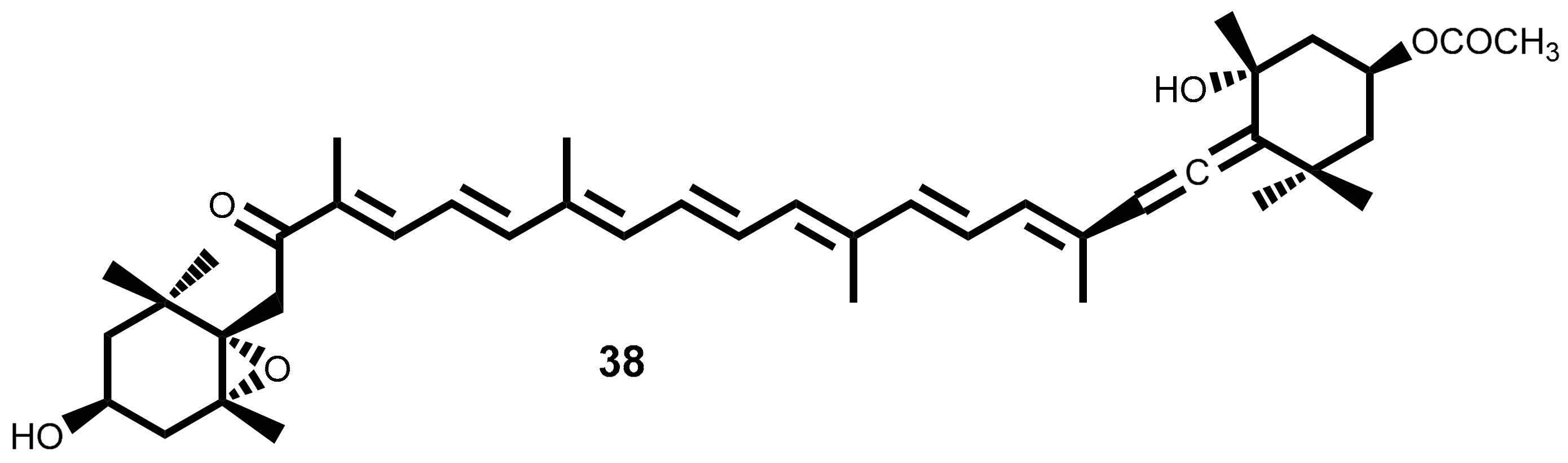
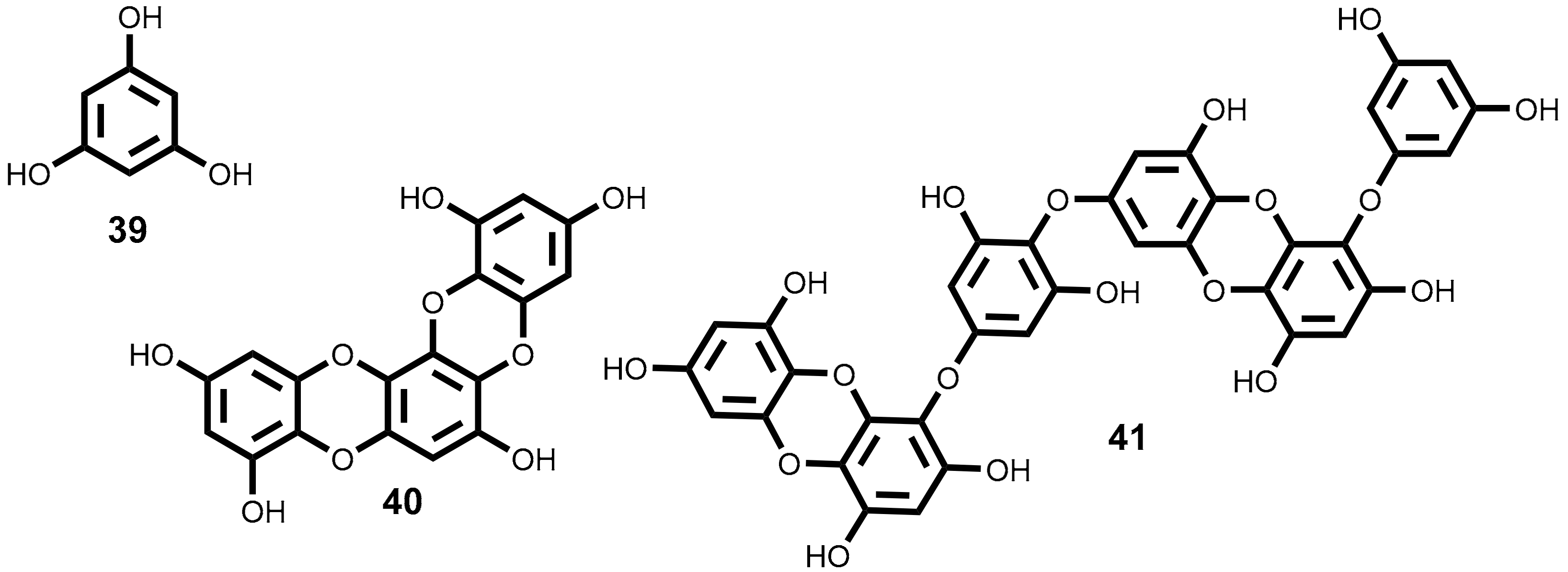
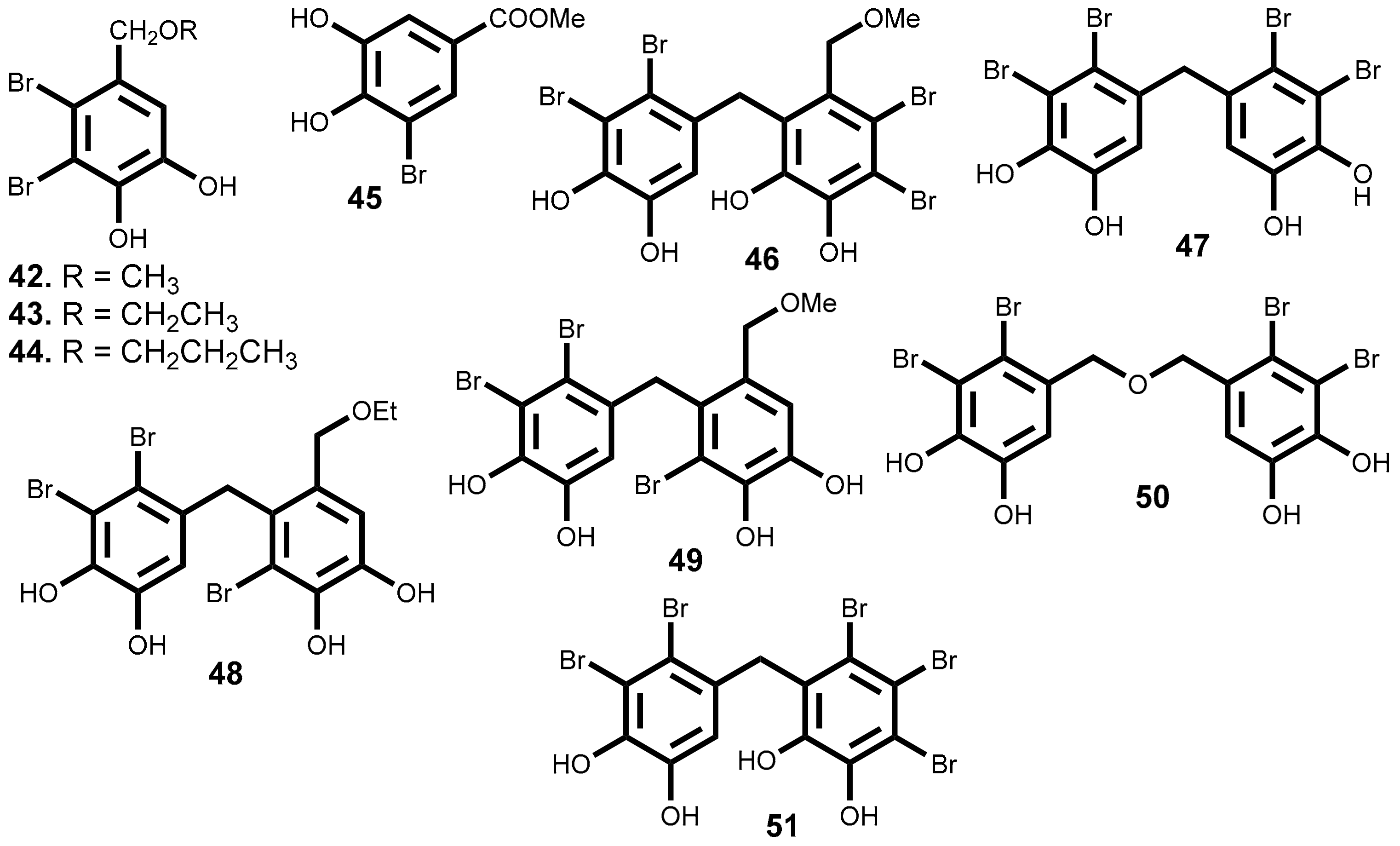
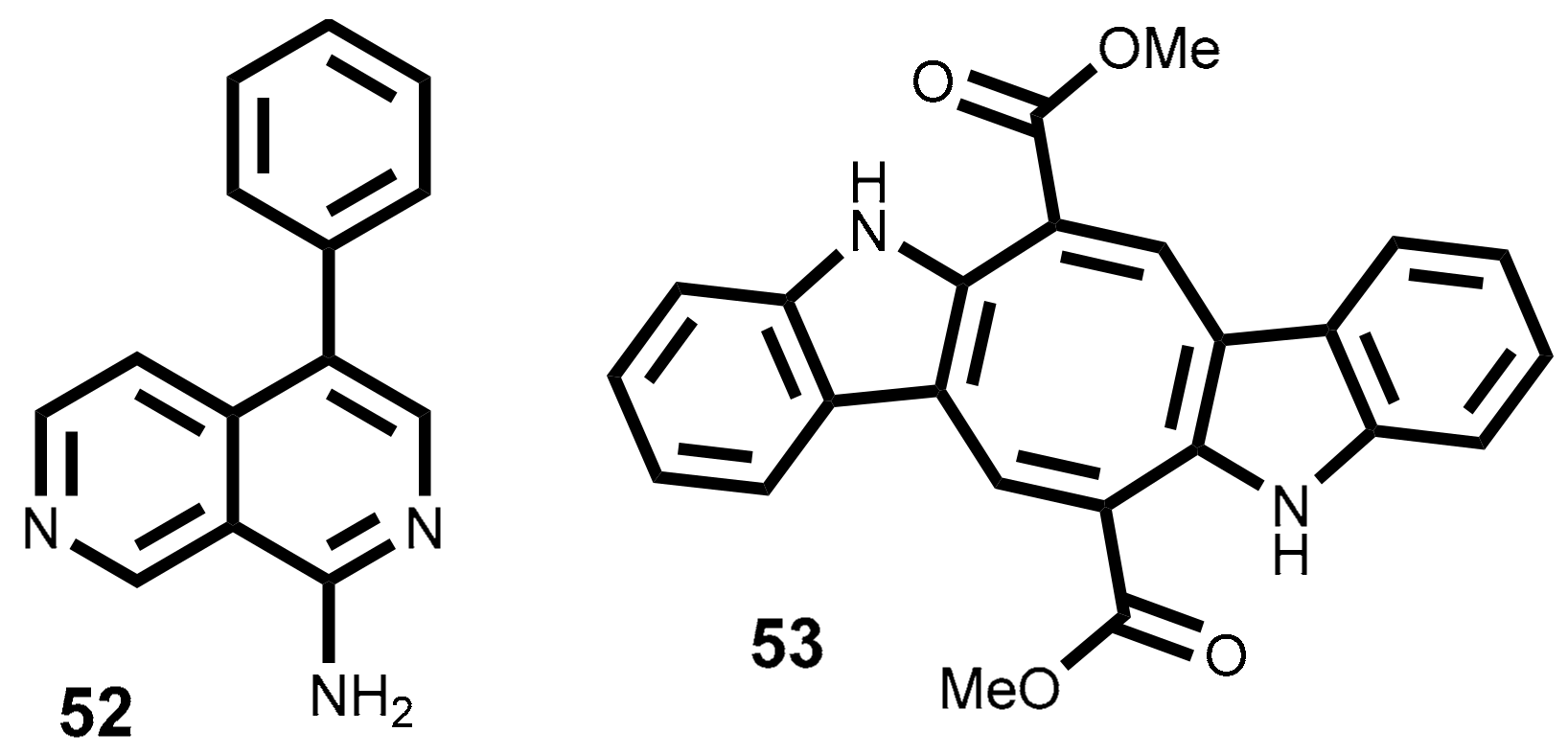
| Metabolites | Sources a [ref.] | Cell Lines Tested (IC50 Value µM) [ref.] |
|---|---|---|
| 1 | Cystoseira myrica (S.G.Gmelin) C.Agardh) [32] | KA3IT (13.1); NIH3T3 b (19.7) [32] |
| 2 | Cystoseira myrica (S.G.Gmelin) C.Agardh [32] | KA3IT (13.7); NIH3T3 b (20.6) [32] |
| 3 | Cystoseira myrica (S.G.Gmelin) C.Agardh [32]Dictyota dichotoma (Hudson) J.V.Lamouroux [33] | KA3IT (14.4); NIH3T3 b (43.3) [32] |
| 4 | Cystoseira abies-marina (S.G. Gmelin) C. Agardh [34] | HeLa–Log phase (5.6); Vero b–Log phase (14.6) [34] |
| 5 | Dilophus ligulatus (Kützing) Feldmann [35] | P388 (4.33–4.79) [35] |
| 6 | Dilophus ligulatus (Kützing) Feldmann [35] | P388 (11.2); P388/DOX (12.9); NSCLCN6-L16 (6.61) [35] |
| 7 | Dilophus ligulatus (Kützing) Feldmann [35] | P388 (3.64); P388/DOX (5.95); KB (12.2); NSCLCN6-L16 (3.31) [35] |
| 8 | Dilophus ligulatus (Kützing) Feldmann [35] | P388 (7.42); P388/DOX (9.41); KB (14.3), NSCLCN6-L16 (6.85) [35] |
| 9 | Dilophus ligulatus (Kützing) Feldmann [35] | P388 (10.3); P388/DOX (10.8) KB (6.85); NSCLCN6-L16 (1.71) [35] |
| 10 | Dilophus ligulatus (Kützing) Feldmann [35] | P388 (13.3); KB (14.4); NSCLCN6-L16 (4.44) [35] |
| 11 | Dilophus ligulatus (Kützing) Feldmann [35] | P388 (9.26); P388/DOX (7.93) NSCLCN6-L16 (0.99) [35] |
| 12 | Stypopodium zonale (J.V.Lamouroux) Papenfuss [36] | HT-29 (5.83); H-116 (5.83); A549 (5.83) [36] |
| 13 | Stypopodium flabelliforme Weber-van Bosse [37] | NCI-H460 (9) [37] |
| 14 | Stypopodium flabelliforme Weber-van Bosse [37] | NCI-H460 (14) [37] |
| 15 and 16 | Taonia atomaria (Woodward) J. Agardh [38] | NSCLC-N6 (7.35); A549 (7.35) [38] |
| 17 | Stypopodium flabelliforme Weber-van Bosse [39] | SH-SY5Y (12.2) [39] |
| 18 | Stypopodium flabelliforme Weber-van Bosse [39] | SH-SY5Y (14) [39] |
| 19 | Sargassum fallax Sonder [40]Sargassum yezoense (Yamada) Yoshida & T. Konno [41] | P388 (14) [40] |
| 20 | Laurencia tristicha Tseng, Chang, E.Z. et B.M. Xia [42]Laurencia nidifica J. Agardh [43] | A549 (3.94); HeLa (3.77); BGC-823 (2.86); Bel7402 (4.48); HCT-8 (4.15) [42] |
| 21 | Laurencia tristicha Tseng, Chang, E.Z. et B.M. Xia [44] | HeLa (15.5) [44] |
| 22 | Laurencia tristicha Tseng, Chang, E.Z. et B.M. Xia [44] | HeLa (0.3); BGC-823 (5.1); Bel7402 (0.5); HCT-8 (0.5); HT29 (9.1) [44] |
| 23 | Laurencia pacifica Kylin [45]Laurencia okamurae Yamada [46] | DU145 (6.8); HT-29 (9.1); A431 (9.6); A2780 (10); BE2-C (13); MCF-7 (14); SMA (14); SJ-G2 (15); MCF10A b (28) [45] |
| 24 | Laurencia pacifica Kylin [45]Laurencia okamurae Yamada [46] | HT-29 (15); DU145 (12); MCF10A b (46) [45] |
| 25 | Plocamium suhrii Kützing [47]Plocamium cartilagineum (Linnaeus) P.S.Dixon [48,49] | WHCO1 (6.6) [47]; CFU (3.36) [48] |
| 26 | Plocamium suhrii Kützing [47]Plocamium cartilagineum (Linnaeus) P.S.Dixon [48,49] | WHCO1 (9.9) [47] |
| 27 | Plocamium suhrii Kützing [47] | WHCO1 (9.3) [47] |
| 28 | Plocamium suhrii Kützing [47]Plocamium cartilagineum (Linnaeus) P.S.Dixon [49] Plocamium oregonum Doty [50] | WHCO1 (8.5) [47] |
| 29 | Plocamium suhrii Kützing [47]Plocamium corallorhiza (Turner) Hooker & Harvey [51] | WHCO1 (7.9) [47] |
| 30 | Plocamium suhrii Kützing [47] | WHCO1 (8.4) [47] |
| 31 | Plocamium cartilagineum (Linnaeus) P.S.Dixon [48,49] | CFU (3.36) [48] HCT-116 (3.36) [52] |
| 32 | Plocamium cartilagineum (Linnaeus) P.S.Dixon [48] | NCI-H460 (13) [48] |
| 33 | Portieria hornemannii (Lyngbye) P.C.Silva [53] | HCT-116 (0.92) [52,53] |
| Fucoxanthin Source | Cell Line Tested | Level of Activity | Activity/Mechanism | Target Molecules | Ref. |
|---|---|---|---|---|---|
| Hizikia fusiformis (Harvey) Okamura | GOTO | 38% growth inhibition at 15.2 µM | G1 cell cycle arrest | N-myc | [84] |
| Undaria pinnatifida (Harvey) Suringar | HL-60 | 46% cellular viability at 11.3 µM | Apoptosis induction | Caspase-3; caspase-7; caspase-9 | [85] |
| Unspecified source | PC3, DU145, LNCap | At 20 µM cellular viability was 14.9% of PC3, 5% of DU145 and 9.8% of LNCap cell lines | Apoptosis induction | Bcl-2; Bax; caspase-3 | [86,87] |
| Undaria pinnatifida (Harvey) Suringar | CaCo-2, DLD-1, HT29 | 36.8% cellular viability of CaCo-2 at 7.6 µM | Apoptosis and antiproliferative effect | Bcl-2 | [88] |
| Laminaria japonica Areschoug | WiDr, HCT116 | At 25 µM cell cycle arrest and phosphorylation of pRb | Cell cycle arrest G0/G1 | P21WAF1/Cip1 | [71] |
| Unspecified source | HepG2, DU145 | IC50 = 3 µM | Cell cycle arrest G1 | GADD45A | [89] |
| Laminaria japonica Areschoug | HepG2 | At 25 µM, inhibition in 29.5% of cell proliferation | Cell cycle arrest G1 | Cyclin D | [90] |
| Laminaria japonica Areschoug | EJ-1 | At 6.25 µM the cellular viability decreased to 52.62% | Apoptosis induction | Caspase-3 | [91] |
| Undaria pinnatifida (Harvey) Suringar | SK-Hep-1 | IC50 9.4 µM | Cell cycle arrest G1, apoptosis induction | Connexin 43; connexin-32 | [92] |
| Ishige okamurae Yendo | HL-60 | Inhibition of proliferation in 65% at 15 µM | Apoptosis induction | Caspase-3; caspase-7; PARP; Bcl-xL | [93] |
| Undaria pinnatifida (Harvey) Suringar | MGC-803 | At 75 µM apoptosis inducing effects like Paclitaxel (1 µM) | Cell cycle arrest G2/M, apoptosis induction | CyclinB1; survivin | [94] |
| Unspecified source | LNCap | IC50 = 2.5 µM | Cell cycle arrest G1 | GADD45A; SAPK/JNK | [95] |
| Undaria pinnatifida (Harvey) Suringar | HUVEC | At 5 µM inhibition of FGF-2 expression in 22% | Inhibition of angiogenesis | FGF-2; FGFR-1 | [96] |
| Unspecified source | T24 | Induced apoptosis at 40 µM and proliferation inhibition at 5–10 µM | Apoptosis induction; Cell cycle arrest G1 | Caspase-3; cyclin D1; cyclin E | [97] |
| Unspecified source | U87, U251 | At 25 µM the cellular viability decreased 52% and 43% in U87 and U251, respectively | Apoptosis induction. Inhibition of invasion and migration | Caspase-3; caspase-9; cleaved-PARP; P38-MMP-2/9 | [98] |
| Unspecified source | SiHa | Strong synergistic effect from combination with TRAIL | Apoptosis induction | Caspase-3; Bcl-2; Bax | [99] |
| Undaria pinnatifida (Harvey) Suringar | SGC7901 | IC50 = 9.80 ± 0.94 (48 h) | Apoptosis and autophagy induction | Beclin-1; caspase-3; Bcl-2 | [100] |
| Metabolites | Sources | Cell Lines Tested (IC50 Value µM) |
|---|---|---|
| 42 | Vertebrata lanosa (Linnaeus) T.A. Christensen [137], Neorhodomela larix (Turner) Masuda [134,140], Odonthalia corymbifera (S.G. Gmelin) Greville [135] | DLD-1 (14.6); HCT-116 (14.1) [137] |
| 43 | Vertebrata lanosa (Linnaeus) T.A.Christensen [137], Osmundaria serrata (Suhr) R.E. Norris [138] | DLD-1 (13.5); HCT-116 (2.51) [137] |
| 44 | Vertebrata lanosa (Linnaeus) T.A. Christensen [137] | DLD-1 (12.4); HCT-116 (1.32) [137] |
| 45 | Rhodomela confervoides (Hudson) P.C. Silva [141] | KB (12.5); Bel7402 (12.9); A549 (14.4); HELF a (25.9) [141] |
| 46 | Leathesia marina (Lyngbye) Decaisne [142,143] | A549 (2.5); BGC823 (8.8); MCF-7 (2.7); Bel7402 (4.8); B16-BL6 (7.3); HT-1080 (6.6); A2780 (2.7) [142,143] |
| 47 | Leathesia marina (Lyngbye) Decaisne [142], Rhodomela confervoides (Hudson) P.C. Silva [144] | A549 (1.8); BGC823 (3.8); MCF-7 (2.7); HCT-8 (2.2) [142] |
| 48 | Leathesia marina (Lyngbye) Decaisne [142,143] | BGC823 (4.6); MCF-7(3.4); Bel7402 (5.5); HCT-8 (2.8); B16-BL6 (3.3), HT-1080 (7.2); A2780 (7.1) [142,143] |
| 49 | Leathesia marina (Lyngbye) Decaisne [142,143] | BGC823 (8.6); B16-BL6 (15.4); HT-1080 (10.3) [142,143] |
| 50 | Leathesia marina (Lyngbye) Decaisne [142,143] | A549 (5.4); MCF-7 (4.6); Bel7402 (7.4); HCT-8 (5.9); HT-1080 (8.2); A2780 (8.6) [142,143] |
| 51 | Leathesia marina (Lyngbye) Decaisne [143] | A549 (1.6); BGC823 (3.3); MCF-7 (2.5); HCT-8 (1.9); B16-BL6 (3.2); A2780 (3.8) [143] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rocha, D.H.A.; Seca, A.M.L.; Pinto, D.C.G.A. Seaweed Secondary Metabolites In Vitro and In Vivo Anticancer Activity. Mar. Drugs 2018, 16, 410. https://doi.org/10.3390/md16110410
Rocha DHA, Seca AML, Pinto DCGA. Seaweed Secondary Metabolites In Vitro and In Vivo Anticancer Activity. Marine Drugs. 2018; 16(11):410. https://doi.org/10.3390/md16110410
Chicago/Turabian StyleRocha, Djenisa H. A., Ana M. L. Seca, and Diana C. G. A. Pinto. 2018. "Seaweed Secondary Metabolites In Vitro and In Vivo Anticancer Activity" Marine Drugs 16, no. 11: 410. https://doi.org/10.3390/md16110410
APA StyleRocha, D. H. A., Seca, A. M. L., & Pinto, D. C. G. A. (2018). Seaweed Secondary Metabolites In Vitro and In Vivo Anticancer Activity. Marine Drugs, 16(11), 410. https://doi.org/10.3390/md16110410