Marine Fungi as Producers of Benzocoumarins, a New Class of Inhibitors of Glycogen-Synthase-Kinase 3β
Abstract
:1. Introduction
2. Results
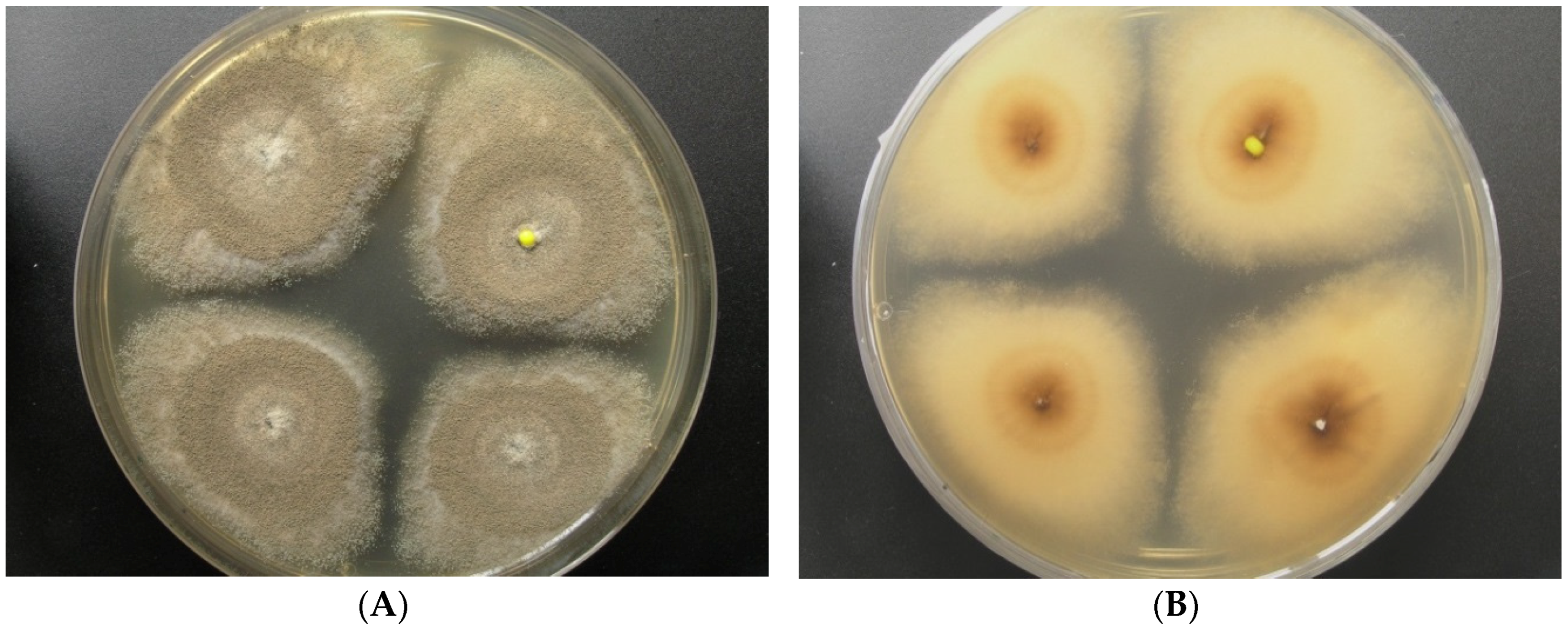
2.1. Origin and Classification of the Producer Strains
2.2. Production of Pannorin (1) by Aspergillus sp. LF660
2.3. Structural Elucidation
2.4. Biological Activities
3. Discussion
4. Materials and Methods
4.1. Isolation, Cultivation, and Storage of the Producer Strains LF660 and KF666
4.2. Identification of the Strains LF660 and KF666
4.3. Fermentation and Production of Extracts for the Purification of the Compounds 1, 2, and 3
4.4. Isolation of the Compounds 1, 2, and 3
4.5. Structure Elucidation of the Compounds 1, 2, and 3
4.6. Production of Pannorin (1) in a Stirred Tank Reactor
4.7. Biological Activities Assays
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Imhoff, J.F. Natural products from marine fungi—Still an underrepresented resource. Mar. Drugs 2016, 14, 19. [Google Scholar] [CrossRef] [PubMed]
- Imhoff, J.F.; Labes, A.; Wiese, J. Biomining the microbial treasures of the ocean: New natural products. Biotechnol. Adv. 2011, 29, 468–482. [Google Scholar] [CrossRef] [PubMed]
- Rateb, M.E.; Ebel, R. Secondary metabolites of fungi from marine habitats. Nat. Prod. Rep. 2011, 28, 290–344. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, O.; Nolan, Y. Glycogen synthase kinase-3 as a therapeutic target for cognitive dysfunction in neuropsychiatric disorders. CNS Drugs 2015, 29, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.; Reis, J.; Teixeira, J.; Borges, F. Alzheimer’s disease, enzyme targets and drug discovery struggles: From natural products to drug prototypes. Ageing Res. Rev. 2014, 15, 116–145. [Google Scholar] [CrossRef] [PubMed]
- Golpich, M.; Amini, E.; Hemmati, F.; Ibrahim, N.M.; Rahmani, B.; Mohamed, Z.; Raymond, A.A.; Dargahi, L.; Ghasemi, R.; Ahmadiani, A. Glycogen synthase kinase-3 beta (GSK-3β) signaling: Implications for Parkinson’s disease. Pharmacol. Res. 2015, 97, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Lee, K.S.; Lee, G.Y.; Jin, H.; Durrance, E.S.; Park, H.S.; Choi, S.H.; Park, K.S.; Kim, Y.-B.; Jang, H.C.; et al. Anti-diabetic efficacy of KICG1338, a novel glycogen synthase kinase-3β inhibitor, and its molecular characterization in animal models of type 2 diabetes and insulin resistance. Mol. Cell. Endocrinol. 2015, 409, 1–10. [Google Scholar] [CrossRef] [PubMed]
- McCubrey, J.A.; Steelman, L.S.; Bertrand, F.E.; Davis, N.M.; Sokolosky, M.; Abrams, S.L.; Montalto, G.; D’Assoro, A.B.; Libra, M.; Nicoletti, F.; et al. GSK-3 as potential target for therapeutic intervention in cancer. Oncotarget 2014, 5, 2881–2911. [Google Scholar] [CrossRef] [PubMed]
- Eldar-Finkelman, H.; Martinez, A. GSK-3 inhibitors: Preclinical and clinical focus on CNS. Front. Mol. Neurosci. 2011, 4. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Report on Diabetes; WHO Press: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization and Alzheimer’s Disease International. Dementia: A Public Health Priority; WHO Press: Geneva, Switzerland, 2012. [Google Scholar]
- Stewart, B.W.; Wild, C.P. World Cancer Report 2014; WHO Press: Geneva, Switzerland, 2014. [Google Scholar]
- Ogawa, H.; Hasumi, K.; Sakai, K.; Murakawa, S.; Endo, A. Pannorin, a new 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitor produced by Chrysosporium pannorum. J. Antibiot. 1991, 44, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Aly, A.H.; Edrada-Ebel, R.A.; Indriani, I.D.; Wray, V.; Müller, W.E.G.; Totzke, F.; Zirrgiebel, U.; Schächtele, C.; Kubbutat, M.H.G.; Lin, W.H. Cytotoxic metabolites from the fungal endophyte Alternaria sp. and their subsequent detection in its host plant Polygonum senegalense. J. Nat. Prod. 2008, 71, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Mikula, H.; Skrinjar, P.; Sohr, B.; Ellmer, D.; Hametner, C.; Fröhlich, J. Total synthesis of masked Alternaria mycotoxins-sulfates and glucosides of alternariol (AOH) and alternariol-9-methyl ether (AME). Tetrahedron 2013, 69, 10322–10330. [Google Scholar] [CrossRef]
- Kaur, A.; Raja, H.A.; Deep, G.; Agarwal, R.; Oberlies, N.H. Pannorin, B, a new naphthopyrone from an endophytic fungal isolate of Penicillium sp. Magn. Res. Chem. 2016, 54, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Vagstad, A.L.; Newman, A.G.; Storm, P.A.; Belecki, K.; Crawford, J.M.; Townsend, C.A. Combinatorial domain swaps provide insights into the rules of fungal polyketide synthase programming and the rational synthesis of non-native aromatic products. Angew. Chem. Int. Ed. Engl. 2013, 52, 1718–1721. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.G.; Vagstad, A.L.; Storm, P.A.; Townsend, C.A. Systematic domain swaps of iterative, nonreducing polyketide synthases provide mechanistic understanding and rationale for catalytic reprogramming. J. Am. Chem. Soc. 2014, 136, 7348–7362. [Google Scholar] [CrossRef] [PubMed]
- Solhaug, A.; Eriksen, G.S.; Holme, J.A. Mechanisms of action and toxicity of the mycotoxin alternariol: A review. Basic Clin. Pharmacol. Toxicol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.; Yu, R.; Wang, X.; Mao, Z.; Fu, L.; Liu, Y.; Zhou, L. Alternariol 9-methyl ether from the endophytic fungus Alternaria sp. Samif01 and its bioactivities. Braz. J. Microbiol. 2016, 47, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Baki, A.; Bielik, A.; Molnar, L.; Szendrei, G.; Keserü, G.M. A high through-put luminsescent assay for glycogen synthase kinase-3 beta inhibitors. Assay Drug Dev. Technol. 2007, 5, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Kramer, T.; Schmidt, B.; Lo Monte, F. Small-molecule inhibitors of GSK-3: Structural insights and their application to Alzheimer’s disease models. Int. J. Alzheimer’s Dis. 2012, 2012, 381029. [Google Scholar] [CrossRef] [PubMed]
- Wickerham, L.J. Taxonomy of yeasts. Tech. Bull. U. S. Dep. Agric. 1951, 29, 1–56. [Google Scholar]
- Wiese, J.; Ohlendorf, B.; Blümel, M.; Schmaljohann, R.; Imhoff, J.F. Phylogenetic identification of fungi isolated from the marine sponge Tethya aurantium and identification of their secondary metabolites. Mar. Drugs 2011, 9, 561–585. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Tatsuova, T.; Madden, T.L. Blast 2 sequences—A new tool for comparing protein and nucleotide sequences. FEMS Microbiol. Lett. 1999, 174, 247–250. [Google Scholar] [CrossRef]
- Ohlendorf, B.; Schulz, D.; Erhard, A.; Nagel, K.; Imhoff, J.F. Geranylphenazinediol, an acetylcholinesterase inhibitor produced by a Streptomyces species. J. Nat. Prod. 2012, 75, 1400–1404. [Google Scholar] [CrossRef] [PubMed]
- Schneemann, I.; Kajahn, I.; Ohlendorf, B.; Zinecker, H.; Erhard, A.; Nagel, K.; Wiese, J.; Imhoff, J.F. Mayamycin, a cytotoxic polyketide from a marine Streptomyces strain isolated from the marine sponge Halichondria panicea. J. Nat. Prod. 2010, 73, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
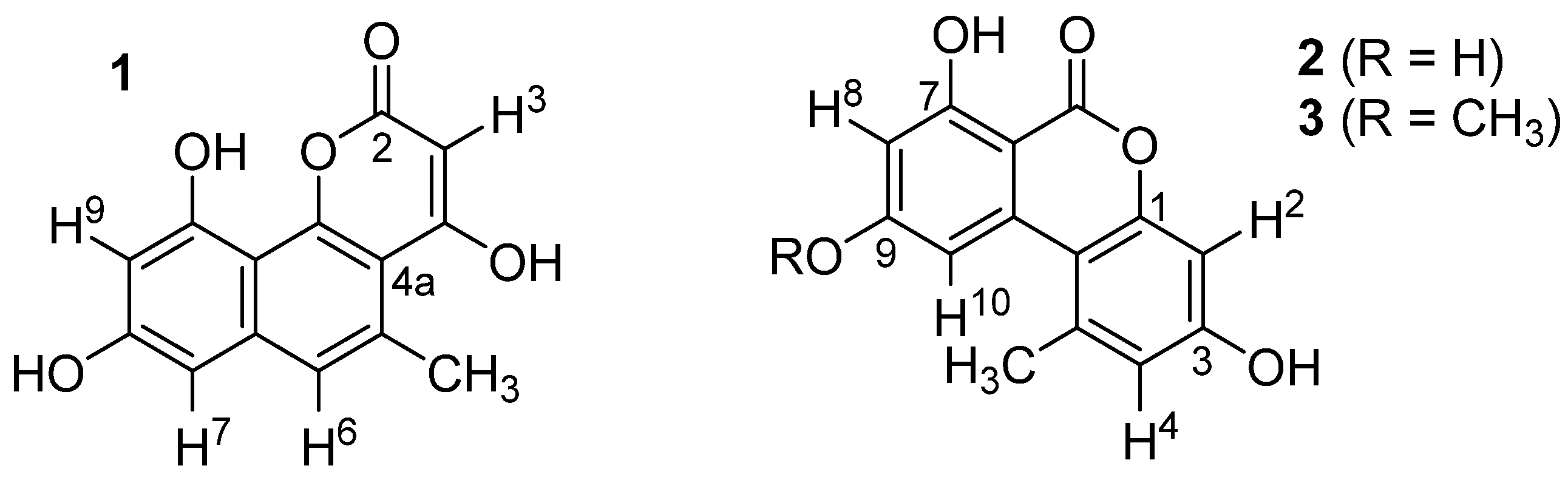
| Position | Alternariol (2) | Alternariol-9-methylether (3) | ||
|---|---|---|---|---|
| Isolated a | Literature a | Isolated a | Literature b | |
| H-2 | 6.55, d (2.6) | 6.55, d (2.5) | 6.72, d (2.6) | 6.72, d (2.3) |
| H-4 | 6.64, d (2.6) | 6.65, d (2.5) | 6.64, d (2.6) | 6.64, d (2.4) |
| H-8 | 6.31, d (2.1) | 6.32, d (2.0) | 6.61, d (2.2) | 6.61, d (2.3) |
| H-10 | 7.20, d (2.1) | 7.20, d (2.0) | 7.23, d (2.2) | 7.21, d (2.3) |
| 5-CH3 | 2.70, s | 2.71, s | 2.73, s | 2.73, s |
| O-CH3 | - | - | 3.90, s | 3.90, s |
| 3-OH | - | - | 10.38, brs | 10.36, brs |
| 7-OH | - | - | 11.82, brs | 11.81, brs |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiese, J.; Imhoff, J.F.; Gulder, T.A.M.; Labes, A.; Schmaljohann, R. Marine Fungi as Producers of Benzocoumarins, a New Class of Inhibitors of Glycogen-Synthase-Kinase 3β. Mar. Drugs 2016, 14, 200. https://doi.org/10.3390/md14110200
Wiese J, Imhoff JF, Gulder TAM, Labes A, Schmaljohann R. Marine Fungi as Producers of Benzocoumarins, a New Class of Inhibitors of Glycogen-Synthase-Kinase 3β. Marine Drugs. 2016; 14(11):200. https://doi.org/10.3390/md14110200
Chicago/Turabian StyleWiese, Jutta, Johannes F. Imhoff, Tobias A. M. Gulder, Antje Labes, and Rolf Schmaljohann. 2016. "Marine Fungi as Producers of Benzocoumarins, a New Class of Inhibitors of Glycogen-Synthase-Kinase 3β" Marine Drugs 14, no. 11: 200. https://doi.org/10.3390/md14110200
APA StyleWiese, J., Imhoff, J. F., Gulder, T. A. M., Labes, A., & Schmaljohann, R. (2016). Marine Fungi as Producers of Benzocoumarins, a New Class of Inhibitors of Glycogen-Synthase-Kinase 3β. Marine Drugs, 14(11), 200. https://doi.org/10.3390/md14110200







