The Anti-Inflammatory Effect of Algae-Derived Lipid Extracts on Lipopolysaccharide (LPS)-Stimulated Human THP-1 Macrophages
Abstract
:1. Introduction
2. Results
2.1. Fatty Acid Composition of Algal Lipid Extracts
| Extracts (% Total Fatty Acids) | Pavlova lutheri | Palmaria palmata | Porphyra dioica | Chondrus crispus | ||||
|---|---|---|---|---|---|---|---|---|
| Average | SD | Average | SD | Average | SD | Average | SD | |
| Saturated fatty acids (SFA) | ||||||||
| 12:0 | 0.19 | 0.14 | 0.16 | 0.05 | 0.32 | 0.13 | 0.42 | 0.15 |
| 14:0 | 14.26 | 0.47 | 4.51 | 0.35 | 0.53 | 0.10 | 1.98 | 0.39 |
| 16:0 | 12.84 | 0.19 | 22.27 | 0.40 | 28.12 | 1.19 | 27.33 | 0.50 |
| 18:0 | 0.68 | 0.02 | 1.69 | 0.07 | 2.16 | 0.11 | 2.71 | 0.05 |
| Sum of SFAs | 27.97 | 0.61 | 28.63 | 0.63 | 31.14 | 1.51 | 32.44 | 0.90 |
| Monounsaturated fatty acids (MUFA) | ||||||||
| 14:1 | 0.55 | 0.01 | 0.51 | 0.07 | 0.54 | 0.12 | 0.48 | 0.11 |
| 15:1 | 1.66 | 0.09 | 1.53 | 0.14 | 1.51 | 0.02 | 1.69 | 0.27 |
| 16:1 n-7 | 14.36 | 0.45 | 1.17 | 0.20 | 2.37 | 0.09 | 0.58 | 0.08 |
| 18:1 n-9 | 2.60 | 0.07 | 2.18 | 0.02 | 1.72 | 0.02 | 5.47 | 0.02 |
| 18:1 n-7 | 0.31 | 0.05 | 0.73 | 0.01 | 0.68 | 0.00 | 0.59 | 0.01 |
| 20:1 n-9 | n.d. | n.d. | 1.62 | 0.04 | n.d. | |||
| Sum of MUFAs | 19.49 | 0.54 | 6.12 | 0.11 | 8.44 | 0.06 | 8.82 | 0.24 |
| Polyunsaturated fatty acids (PUFA) | ||||||||
| 16:2 n-6 | 0.85 | 0.22 | n.d. | n.d. | n.d. | |||
| 16:2 n-4 | 1.68 | 0.11 | n.d. | n.d. | n.d. | |||
| 16:4 n-3 | 0.62 | 0.13 | n.d. | n.d. | n.d. | |||
| 18:2 n-6 | 0.59 | 0.02 | 0.65 | 0.01 | 1.64 | 0.01 | 1.58 | 0.05 |
| 18:3 n-6 | 2.09 | 0.04 | 0.26 | 0.01 | 0.56 | 0.01 | 0.49 | 0.01 |
| 18:3 n-3 | 1.13 | 0.01 | 0.65 | 0.01 | 1.34 | 0.02 | 0.23 | 0.00 |
| 18:4 n-3 | 4.98 | 0.08 | 2.16 | 0.04 | 1.58 | 0.01 | 0.25 | 0.01 |
| 20:2 n-6 | n.d. | 1.06 | 0.15 | 0.87 | 0.02 | 0.37 | 0.05 | |
| 20:3 n-6 | n.d. | n.d. | 2.21 | 0.35 | 0.19 | 0.16 | ||
| 20:4 n-6 | 0.60 | 0.02 | 0.67 | 0.01 | 3.03 | 0.07 | 19.85 | 0.82 |
| 20:5 n-3 | 27.67 | 0.37 | 57.94 | 0.89 | 46.35 | 0.91 | 33.47 | 0.21 |
| 22:5 n-3 | 1.17 | 0.20 | n.d. | n.d. | n.d. | |||
| 22:6 n-3 | 10.47 | 0.38 | 1.15 | 0.24 | n.d. | n.d. | ||
| Sum of PUFAs | 51.85 | 0.84 | 64.55 | 0.87 | 57.57 | 1.35 | 56.42 | 1.29 |
| n-3 | 46.04 | 1.05 | 61.91 | 0.71 | 49.26 | 0.93 | 33.94 | 0.20 |
| n-6 | 4.13 | 0.18 | 2.64 | 0.17 | 8.31 | 0.44 | 22.47 | 1.10 |
| Ratio n-6/ n-3 | 0.09 | 0.01 | 0.04 | 0.00 | 0.17 | 0.01 | 0.66 | 0.03 |
| Others | 0.69 | 0.30 | 0.70 | 0.12 | 2.85 | 0.14 | 2.32 | 0.61 |
2.2. Pigments
| Extracts | Pavlova lutheri | Palmaria palmata | Porphyra dioica | Chondrus crispus | ||||
|---|---|---|---|---|---|---|---|---|
| Pigments (% Molar) | Average | SD | Average | SD | Average | SD | Average | SD |
| chlorophyll a + deg. | 41.10 | 0.05 | 52.03 | 0.05 | 53.02 | 0.37 | 43.95 | 0.30 |
| β-carotene | 7.05 | 0.09 | 25.17 | 0.34 | 19.15 | 0.27 | 30.22 | 0.09 |
| zeaxanthin | n.d. | 22.80 | 0.38 | 27.53 | 0.59 | 25.84 | 0.40 | |
| diadinoxanthin + diatoxanthin + deg. | 16.11 | 0.03 | n.d. | n.d. | n.d. | |||
| cys-fucoxanthin | 5.51 | 0.01 | n.d. | n.d. | n.d. | |||
| chlorophyll c + deg. | 3.36 | 0.08 | n.d. | n.d. | n.d. | |||
| fucoxanthin + deg. | 26.87 | 0.07 | n.d. | n.d. | n.d. | |||
| Pigments (% crude extract) | 6.80 | 0.05 | 6.03 | 0.41 | 5.13 | 0.06 | 4.87 | 0.04 |
2.3. Lipid Class Composition and LC-PUFA Partitioning
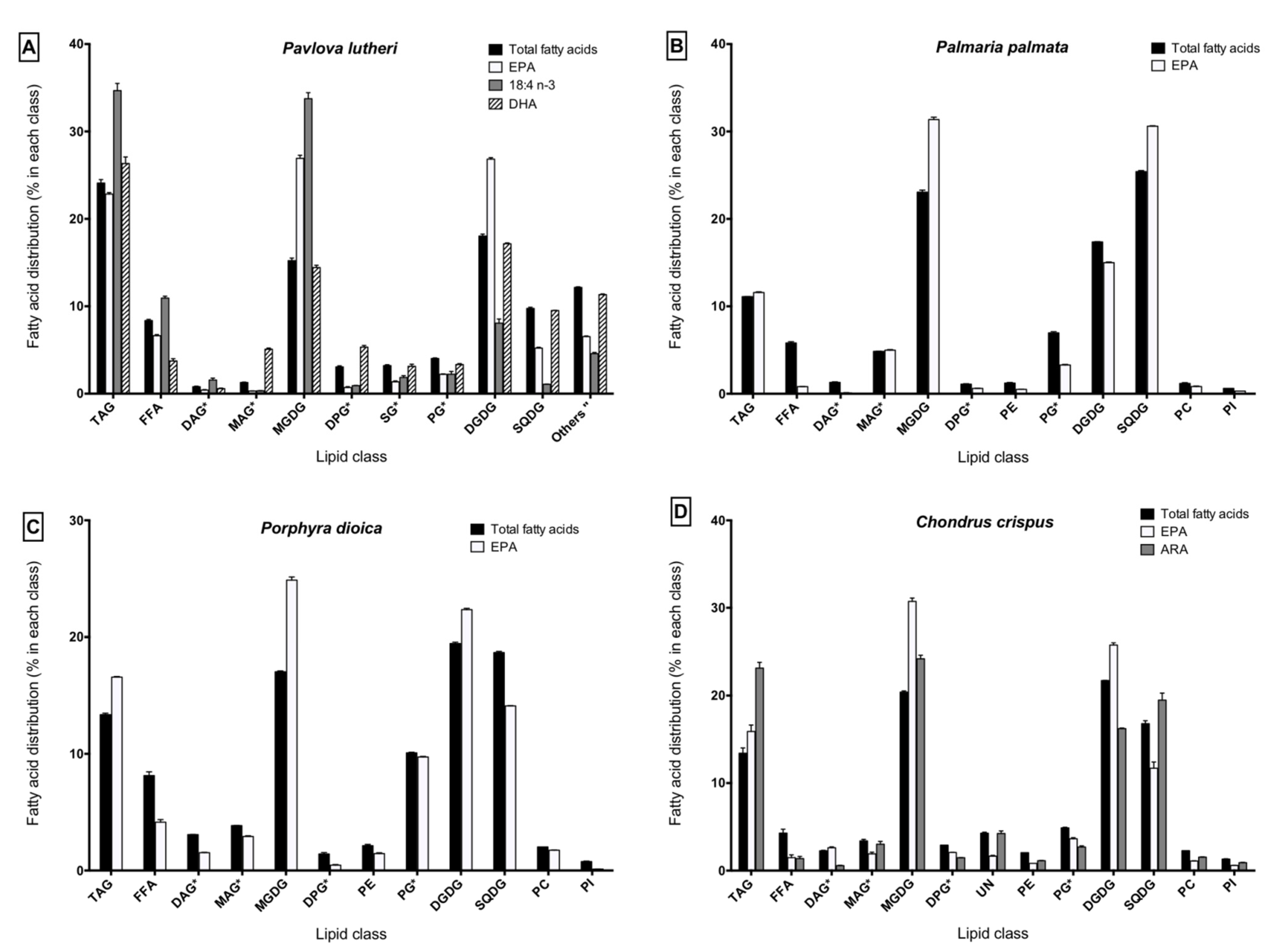
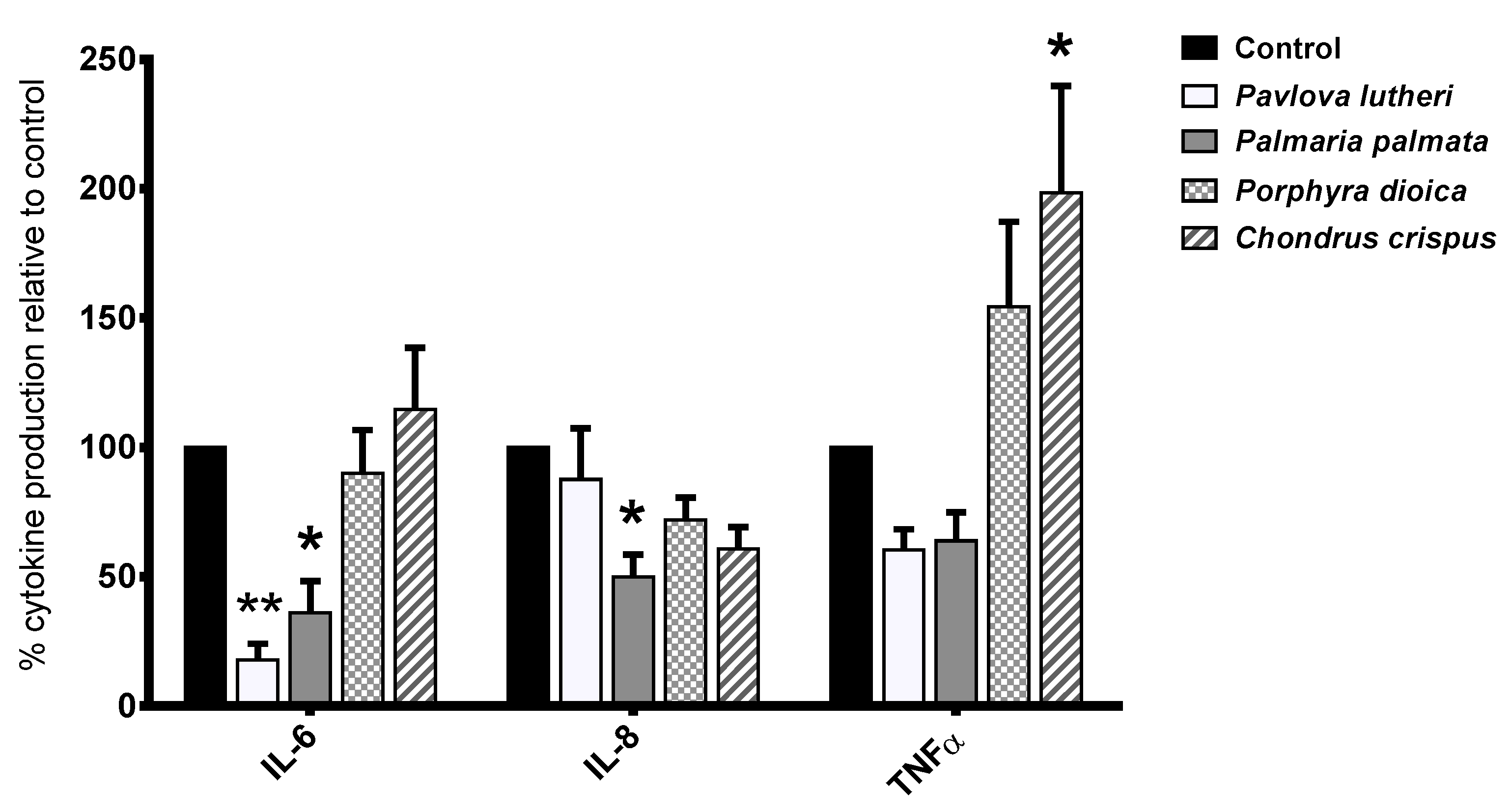
2.4. Inhibition of Inflammatory Cytokine Production in Lipopolysaccharide (LPS)-Stimulated THP-1 Macrophages
| Extract | Gene Symbol | Name | Fold Change | Gene Description and Function |
|---|---|---|---|---|
| P. lutheri | TLR8 | Toll-like receptor 8 | −3.33 | PAMP recognition and activation of innate immunity. Mediates cytokine production through activation of NF-κB |
| TLR1 | Toll-like receptor 1 | −4.16 | Interacts with TLR2 for immune activation through PAMP recognition. | |
| TRAF5 | TNF receptor-associated factor 5 | −2.69 | Mediates signal transduction of the TNF receptor family. Mediates NF-κB and JNK activation | |
| MAP3K1 | Mitogen-activated protein kinase 1 | −2.63 | Activates protein kinase signal transduction cascade such as the ERK and JNK kinase pathways and NF-κB pathway. | |
| PTGER1 | Prostaglandin E receptor 1 | +2.58 | Encodes a receptor for PGE2. Down-regulates COX2 and hence resolves inflammation. | |
| P. palmata | TLR8 | Toll-like receptor 8 | −2.91 | PAMP recognition and activation of innate immunity. Mediates cytokine production through activation of NF-κB |
| TLR1 | Toll-like receptor 1 | −3.99 | Interacts with TLR2 for immune activation through PAMP recognition. | |
| TRAF5 | TNF receptor-associated factor 5 | −2.73 | Mediates signal transduction of the TNF receptor family. Mediates NF-κB and JNK activation | |
| NOS2 | Nitric oxide synthase 2 | +2.83 | Encodes nitric oxide synthase which mediates tumoricidal and bactericidal actions in macrophages | |
| P. dioica | CCR1 | Chemokine (C-C motif) receptor 1 | −3.46 | Acts as a receptor for chemokines such as MIP-1α and MCP-3 which assist in immune cell recruitment |
| TLR8 | Toll-like receptor 8 | −3.29 | PAMP recognition and activation of innate immunity. Mediates cytokine production through activation of NF-κB | |
| TLR2 | Toll-like receptor 2 | −4.25 | Interacts with TLR1 for PAMP recognition leading to NF-κB activation and cytokine production | |
| TLR1 | Toll-like receptor 1 | −2.5 | Interacts with TLR2 for immune activation through PAMP recognition. | |
| TRAF5 | TNF receptor-associated factor 5 | −3.37 | Mediates signal transduction of the TNF receptor family. Mediates NF-κB and JNK activation | |
| TNFSF18 | TNF (ligand) superfamily, member 18 | −2.93 | Regulates T-cell activities by lowering the threshold for T-cell activation. | |
| TRAF6 | TNF receptor-associated factor 6 | −2.99 | Mediates signal transduction from TNF receptors and Toll/IL-1 receptors. | |
| MAP3K1 | Mitogen-activated protein 3 kinase 1 | −3.63 | Activates protein kinase signal transduction cascade such as the ERK and JNK kinase pathways and NF-κB pathway. | |
| STAT3 | Signal transducer and activator of transcription 3 | −3.56 | Activated by cytokines to create transcription factors that form part of JAK-STAT signaling cascade. | |
| CCR5 | Chemokine (C-C motif) receptor 5 | −2.72 | Chemokine receptor, expressed in macrophages involved in immune cell recruitment. | |
| TLR4 | Toll-like receptor 4 | −4.44 | PAMP recognition and activation of inflammatory cascade. Specifically recognises LPS | |
| IL6R | Interleukin 6 receptor | −2.54 | Binds with low affinity to the inflammatory cytokine IL-6 regulating immune response and acute phase reactions. | |
| PTGER1 | Prostaglandin E receptor 1 | +2.7 | Encodes a receptor for PGE2. Down-regulates COX2 and hence resolves inflammation. | |
| C. crispus | IL23 | Interleukin 23 | −4.19 | Activates STAT4 and stimulates production of IFNγ Associated with autoimmune inflammation and tumorigenesis. |
| TLR8 | Toll-like receptor 8 | −2.59 | PAMP recognition and activation of innate immunity. Mediates cytokine production through activation of NF-κB | |
| CCL17 | Chemokine (C-C motif) ligand 17 | −3.33 | Encodes a cytokine that is a chemotactic factor for T-lymphocytes. Recruitment and activation of mature T-cells | |
| TLR1 | Toll-like receptor 1 | −3.25 | Interacts with TLR2 for immune activation through PAMP recognition. | |
| TRAF5 | TNF receptor-associated factor 5 | −2.76 | Mediates signal transduction of the TNF receptor family. Mediates NF-κB and JNK activation | |
| TRAF6 | TNF receptor-associated factor 6 | −3.04 | Mediates signal transduction from TNF receptors and Toll/IL-1 receptors. |
2.5. Inhibition of Inflammatory Gene Expression by Algal Lipid Extracts
2.6. Validation of PCR Array Results through Quantitative PCR (qPCR)
| Gene Name | Primers | Nucleotide Sequence (5′ to 3′) | Tm (°C) |
|---|---|---|---|
| GAPDH | Forward | ACAGTTGCCATGTAGACC | 55.7 |
| Reverse | TTTTTGGTTGAGCACAGG | 59.9 | |
| TLR1 | Forward | CCCTACAAAAGGAATCTGTATC | 58.2 |
| Reverse | TGCTAGTCATTTTGGAACAC | 57.8 | |
| TLR8 | Forward | TGGAAAACATGTTCCTTCAG | 60.1 |
| Reverse | TGCTTTTTCTCATCACAAGG | 60.4 | |
| TRAF5 | Forward | GGAATGGCTTATTCAGAAGAG | 59.4 |
| Reverse | CCACAAACTGGTACTCTATAC | 52.8 |

3. Discussion
4. Experimental Section
4.1. Materials
4.2. Collection of Macroalgae and Biomass Preparation
4.3. Microalgal Strain, Culture Conditions and Biomass Preparation
4.4. Lipid Extraction
4.5. Lipid Class Separation
4.5.1. Lipid Separation by Silica Cartridge Chromatography
4.5.2. Neutral and Polar Lipid Fractionation by Thin-Layer Chromatography (TLC)
4.6. Fatty Acid Analysis
4.7. Pigment Analysis
4.8. Cell Culture
4.9. Cytotoxicity Assay
4.10. Cytokine Analysis
4.11. Preparation of RNA and cDNA Synthesis
4.12. Quantitative Real-Time PCR Analysis
4.13. Statistical Analysis
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Calder, P.C. n-3 Polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, 1505S–1519S. [Google Scholar] [PubMed]
- Hansson, G.K. Inflammation, Atherosclerosis, and Coronary Artery Disease. N. Engl. J. Med. 2005, 352, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Fiocchi, C. Inflammatory bowel disease: Etiology and pathogenesis. Gastroenterology 1998, 115, 182–205. [Google Scholar] [CrossRef]
- Choy, E.H.S.; Panayi, G.S. Cytokine Pathways and Joint Inflammation in Rheumatoid Arthritis. N. Engl. J. Med. 2001, 344, 907–916. [Google Scholar] [PubMed]
- Gregor, M.F.; Hotamisligil, G.S. Inflammatory mechanisms in obesity. Annu. Rev. Immunol. 2011, 29, 415–445. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Barger, S.; Barnum, S.; Bradt, B.; Bauer, J.; Cole, G.M.; Cooper, N.R.; Eikelenboom, P.; Emmerling, M.; Fiebich, B.L.; et al. Inflammation and Alzheimer’s disease. Neurobiol. Aging 2000, 21, 383–421. [Google Scholar] [CrossRef]
- Rallidis, L.S.; Paschos, G.; Liakos, G.K.; Velissaridou, A.H.; Anastasiadis, G.; Zampelas, A. Dietary alpha-linolenic acid decreases C-reactive protein, serum amyloid A and interleukin-6 in dyslipidaemic patients. Atherosclerosis 2003, 167, 237–242. [Google Scholar] [CrossRef]
- Calder, P.C.; Albers, R.; Antoine, J.-M.; Blum, S.; Bourdet-Sicard, R.; Ferns, G.A.; Folkets, G.; Friedmann, P.S.; Frost, G.S.; Guarner, F.; et al. Inflammatory Disease Processes and Interactions with Nutrition. Br. J. Nutr. 2009, 101 (Suppl. S1), S1–S45. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Marfella, R.; Ciotola, M.; Di Palo, C.; Giugliano, F.; Giugliano, G.; D’Armiento, M.; D’Andrea, F.; Giugliano, D. Effect of a mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome: A randomized trial. JAMA 2004, 292, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Núñez, B.; Pruimboom, L.; Dijck-Brouwer, D.A.; Muskiet, F.A. Lifestyle and nutritional imbalances associated with Western diseases: Causes and consequences of chronic systemic low-grade inflammation in an evolutionary context. J. Nutr. Biochem. 2013, 24, 1183–1201. [Google Scholar] [CrossRef] [PubMed]
- Esser, N.; Paquot, N.; Scheen, A.J. Anti-inflammatory agents to treat or prevent type 2 diabetes, metabolic syndrome and cardiovascular disease. Expert Opin. Investig. Drugs 2015, 24, 238–307. [Google Scholar] [CrossRef] [PubMed]
- Sostres, C.; Gargallo, C.J.; Arroyo, M.T.; Lanas, A. Adverse effects of non-steroidal anti-inflammatory drugs (NSAIDs, aspirin and coxibs) on upper gastrointestinal tract. Best Pract. Res. Clin. Gastroenterol. 2010, 24, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The role of marine omega-3 (n-3) fatty acids in inflammatory processes, atherosclerosis and plaque stability. Mol. Nutr. Food Res. 2012, 56, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. n-3 Fatty acids, inflammation and immunity: New mechanisms to explain old actions. Proc. Nutr. Soc. 2013, 72, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Weldon, S.M.; Mullen, A.C.; Loscher, C.E.; Hurley, L.A.; Roche, H.M. Docosahexaenoic acid induces an anti-inflammatory profile in lipopolysaccharide-stimulated human THP-1 macrophages more effectively than eicosapentaenoic acid. J. Nutr. Biochem. 2007, 18, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Mullen, A.; Loscher, C.E.; Roche, H.M. Anti-inflammatory effects of EPA and DHA are dependent upon time and dose-response elements associated with LPS stimulation in THP-1-derived macrophages. J. Nutr. Biochem. 2010, 21, 444–540. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef] [PubMed]
- González, R.; Ballester, I.; López-Posadas, R.; Suárez, M.D.; Zarzuelo, A.; Martínez-Augustin, O.; Sánchez de Medina, F. Effects of flavonoids and other polyphenols on inflammation. Crit. Rev. Food Sci. Nutr. 2011, 51, 331–362. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Yuan, J.-P.; Wu, C.-F.; Wang, J.-H. Fucoxanthin, a Marine Carotenoid Present in Brown Seaweeds and Diatoms: Metabolism and Bioactivities Relevant to Human Health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.-K.; Lee, S.-J.; Na, H.-J.; Ha, K.-S.; Han, J.-A.; Lee, H.; Kwon, Y.G.; Chung, C.K.; Kim, Y.M. Beta-carotene inhibits inflammatory gene expression in lipopolysaccharide-stimulated macrophages by suppressing redox-based NF-kappaB activation. Exp. Mol. Med. 2005, 37, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Lordan, S.; Ross, R.P.; Stanton, C. Marine bioactives as functional food ingredients: Potential to reduce the incidence of chronic diseases. Mar. Drugs 2011, 9, 1056–1100. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.; Guihéneuf, F.; Schmid, M.; Stengel, D.B.; Fitzgerald, G.; Ross, R.; Stanton, C. Algal-derived polyunsaturated fatty acids: Implications for human health. In Polyunsaturated Fatty Acids: Sources, Antioxidant Properties and Health Benefits; Catalá, A., Ed.; Nova Sciences Publishers, Inc.: Hauppauge, NY, USA, 2013; Chapter 3; pp. 45–99. [Google Scholar]
- Dawczynski, C. Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Schmid, M.; Guihéneuf, F.; Stengel, D. Fatty acid contents and profiles of 16 macroalgae collected from the Irish Coast at two seasons. J. Appl. Phycol. 2014, 26, 451–463. [Google Scholar] [CrossRef]
- Ratledge, C. Fatty acid biosynthesis in microorganisms being used for Single Cell Oil production. Biochimie 2004, 86, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Ward, O.P.; Singh, A. Omega-3/6 fatty acids: Alternative sources of production. Process Biochem. 2005, 40, 3627–3652. [Google Scholar] [CrossRef]
- Fisheries and Aquaculture Department; Food and Agriculture Organisation of the United Nations (FAO). The State of World Fisheries and Aquaculture 2010; FAO: Rome, Italy, 2010. [Google Scholar]
- Branch, T.A.; Jensen, O.P.; Ricard, D.; Ye, Y.; Hilborn, R. Contrasting global trends in marine fishery status obtained from catches and from stock assessments. Conserv. Biol. 2011, 25, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J. American Heart Association; Nutrition Committee. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef] [PubMed]
- Adarme-Vega, T.C.; Lim, D.K.; Timmins, M.; Vernen, F.; Li, Y.; Schenk, P.M. Microalgal biofactories: A promising approach towards sustainable omega-3 fatty acid production. Microb. Cell Fact. 2012, 11. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.A.; Chien, S.Y.; Chan, Y.L.; Lu, M.K.; Wu, C.H.; Kong, Z.L.; Wu, C.J. Inhibition of lipopolysaccharide (LPS)-induced inflammatory responses by Sargassum hemiphyllum sulfated polysaccharide extract in RAW 264.7 macrophage cells. J. Agric. Food Chem. 2011, 59, 2062–2068. [Google Scholar] [CrossRef] [PubMed]
- Jo, W.; Choi, Y.; Kim, H.; Nam, B.; Hong, S.; Lee, G.; Lee, S.; Seo, S.; Jeong, M. Anti-inflammatory effect of microalgal extracts from Tetraselmis suecica. Food Sci. Biotechnol. 2010, 19, 1519–1528. [Google Scholar] [CrossRef]
- Khan, M.N.; Cho, J.Y.; Lee, M.C.; Kang, J.Y.; Park, N.G.; Fujii, H.; Honh, Y.K. Isolation of two anti-inflammatory and one pro-inflammatory polyunsaturated fatty acids from the brown seaweed Undaria pinnatifida. J. Agric. Food Chem. 2007, 55, 6984–6988. [Google Scholar] [CrossRef] [PubMed]
- Vo, T.-S.; Kim, J.-A.; Wijesekara, I.; Kong, C.-S.; Kim, S.-K. Potent effect of brown algae (Ishige okamurae) on suppression of allergic inflammation in human basophilic KU812F cells. Food Sci. Biotechnol. 2011, 20, 1227–1234. [Google Scholar] [CrossRef]
- Park, Y.K.; Rasmussen, H.E.; Ehlers, S.J.; Blobaum, K.R.; Lu, F.; Schlegal, V.L.; Carr, T.P.; Lee, J.Y. Repression of proinflammatory gene expression by lipid extract of Nostoc commune var sphaeroides Kützing, a blue-green alga, via inhibition of nuclear factor-kappaB in RAW 264.7 macrophages. Nutr. Res. 2008, 28, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Hou, M.F.; Huang, H.W.; Chang, F.R.; Yeh, C.C.; Tang, J.Y.; Chang, H.W. Marine algal natural products with anti-oxidative, anti-inflammatory, and anti-cancer properties. Cancer Cell Int. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.J.; Moon, J.Y.; Kim, M.J.; Kim, D.S.; Kim, C.S.; Lee, W.J.; Lee, N.H.; Hyun, C.G. Inhibitory effect of Jeju endemic seaweeds on the production of pro-inflammatory mediators in mouse macrophage cell line RAW 264.7. J. Zhejiang Univ. Sci. B 2010, 11, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Nauroth, J.M.; Liu, Y.C.; Van Elswyk, M.; Bell, R.; Hall, E.B.; Chung, G.; Arterburn, L.M. Docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA n-6) algal oils reduce inflammatory mediators in human peripheral mononuclear cells in vitro and paw edema in vivo. Lipids 2010, 45, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Banskota, A.H.; Gallant, P.; Stefanova, R.; Melanson, R.; O’Leary, S.J. Monogalactosyldiacylglycerols, potent nitric oxide inhibitors from the marine microalga Tetraselmis chui. Nat. Prod. Res. 2013, 27, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Banskota, A.H.; Stefanova, R.; Gallant, P.; Osborne, J.A.; Melanson, R.; O’Leary, S.J. Nitric oxide inhibitory activity of monogalactosylmonoacylglycerols from a freshwater microalgae Chlorella sorokiniana. Nat. Prod. Res. 2013, 27, 1028–1031. [Google Scholar] [CrossRef] [PubMed]
- Banskota, A.; Stefanova, R.; Sperker, S.; Lall, S.; Craigie, J.; Hafting, J. Lipids isolated from the cultivated red alga Chondrus crispus inhibit nitric oxide production. J. Appl. Phycol. 2014, 26, 1565–1571. [Google Scholar] [CrossRef]
- Guedes, A.C.; Meireles, L.; Amaro, H.; Malcata, F.X. Changes in Lipid Class and Fatty Acid Composition of Cultures of Pavlova lutheri, in Response to Light Intensity. J. Am. Oil Chem. Soc. 2010, 87, 791–801. [Google Scholar] [CrossRef]
- Tatsuzawa, H.; Takizawa, E. Changes in lipid and fatty acid composition of Pavlova lutheri. Phytochemistry 1995, 40, 397–400. [Google Scholar] [CrossRef]
- Meireles, L.A.; Guedes, A.C.; Malcata, F.X. Lipid Class Composition of the Microalga Pavlova lutheri: Eicosapentaenoic and Docosahexaenoic Acids. J. Agric. Food Chem. 2003, 51, 2237–2241. [Google Scholar] [CrossRef] [PubMed]
- Volkman, J.K.; Jeffrey, S.W.; Nichols, P.D.; Rogers, G.I.; Garland, C.D. Fatty acid and lipid composition of 10 species of microalgae used in mariculture. J. Exp. Mar. Biol. Ecol. 1989, 128, 219–240. [Google Scholar] [CrossRef]
- Guihéneuf, F.; Fouqueray, M.; Mimouni, V.; Ulmann, L.; Jacquette, B.; Tremblin, G. Effect of UV stress on the fatty acid and lipid class composition in two marine microalgae Pavlova lutheri (Pavlovophyceae) and Odontella aurita (Bacillariophyceae). J. Appl. Phycol. 2010, 22, 629–638. [Google Scholar] [CrossRef]
- Sanina, N.M.; Goncharova, S.N.; Kostetsky, E.Y. Fatty acid composition of individual polar lipid classes from marine macrophytes. Phytochemistry 2004, 65, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Kuehl, F.A.; Egan, R.W. Prostaglandins, arachidonic acid, and inflammation. Science 1980, 210, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Choi, J.J.; Park, B.-K.; Yoon, S.J.; Choi, J.E.; Jin, M. Pheophytin a and chlorophyll a suppress neuroinflammatory responses in lipopolysaccharide and interferon-γ-stimulated BV2 microglia. Life Sci. 2014, 103, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.J.; Yoon, W.J.; Kim, K.N.; Ahn, G.N.; Kang, S.M.; Kang, D.H.; Affan, A.; Oh, C.; Jung, W.K.; Jeong, Y.J. Evaluation of anti-inflammatory effect of fucoxanthin isolated from brown algae in lipopolysaccharide-stimulated RAW 264.7 macrophages. Food Chem. Toxicol. 2010, 48, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Nidhi, B.; Sharavana, G.; Ramaprasad, T.R.; Vallikannan, B. Lutein derived fragments exhibit higher antioxidant and anti-inflammatory properties than lutein in lipopolysaccharide induced inflammation in rats. Food Funct. 2015, 6, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, K.; Murata, M.; Kaneniwa, M.; Saito, H.; Shinohara, K.; Maeda-Yamamoto, M. Inhibition of icosanoid production in MC/9 mouse mast cells by n-3 polyunsaturated fatty acids isolated from edible marine algae. Biosci. Biotechnol. Biochem. 1998, 62, 1412–1415. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.C.; Young, D.W.; Golenbock, D.T.; Christ, W.J.; Gusovsky, F. Toll-like receptor-4 mediates lipopolysaccharide-induced signal transduction. J. Biol. Chem. 1999, 274, 10689–10692. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Zhao, L.; Youn, H.S.; Weatherill, A.R.; Tapping, R.; Feng, L.; Lee, W.H.; Fitzgerald, K.A.; Hwang, D.H. Saturated Fatty Acid Activates but Polyunsaturated Fatty Acid Inhibits Toll-like Receptor 2 Dimerized with Toll-like Receptor 6 or 1. J. Biol. Chem. 2004, 279, 16971–16979. [Google Scholar] [CrossRef] [PubMed]
- Zeyda, M.; Szekeres, A.B.; Säemann, M.D.; Geyeregger, R.; Stockinger, H.; Zlabinger, G.J.; Waldhäusl, W.; Stulnig, T.M. Suppression of T Cell Signaling by Polyunsaturated Fatty Acids: Selectivity in Inhibition of Mitogen-Activated Protein Kinase and Nuclear Factor Activation. J. Immunol. 2003, 170, 6033–6039. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, M.; Tan, L.; Wang, C.; Ma, J.; Li, N.; Li, Y.; Xu, G.; Li, J. Docosahexaenoic acid changes lipid composition and interleukin-2 receptor signaling in membrane rafts. J. Lipid. Res. 2005, 46, 1904–1913. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.-J.; Lin, J.-T.; Chen, Y.-C.; Liu, S.-C.; Lu, F.-J.; Chang, T.-J.; Wang, M.; Lin, H.-W.; Chang, Y.-Y. Suppressive effect of carotenoid extract of Dunaliella salina alga on production of LPS-stimulated pro-inflammatory mediators in RAW264.7 cells via NF-κB and JNK inactivation. J. Funct. Foods 2013, 5, 607–615. [Google Scholar] [CrossRef]
- Wang, H.W.; Wu, T.; Qi, J.Y.; Wang, Y.Q.; Luo, X.P.; Ning, Q. Salidroside attenuates LPS-stimulated activation of THP-1 cell-derived macrophages through down-regulation of MAPK/NF-κB signaling pathways. J. Huazhong Univ. Sci. Technolog. Med. Sci. 2013, 33, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Chew, B.P.; Park, J.S. Carotenoid action on the immune response. J. Nutr. 2004, 134, 257S–261S. [Google Scholar] [PubMed]
- Cranmer-Byng, M.M.; Liddle, D.M.; de Boer, A.A.; Monk, J.M.; Robinson, L.E. Proinflammatory effects of arachidonic acid in a lipopolysaccharide-induced inflammatory microenvironment in 3T3-L1 adipocytes in vitro. Appl. Physiol. Nutr. Metab. 2015, 40, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Leiro, J.M.; Castro, R.; Arranz, J.A.; Lamas, J. Immunomodulating activities of acidic sulphated polysaccharides obtained from the seaweed Ulva rigida C. Agardh. Int. Immunopharmacol. 2007, 7, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Bendif, E.M.; Probert, I.; Hervé, A.; Billard, C.; Goux, D.; Lelong, C.; Cadoret, J.P.; Véron, B. Integrative Taxonomy of the Pavlovophyceae (Haptophyta): A Reassessment. Protist 2011, 162, 738–761. [Google Scholar] [CrossRef] [PubMed]
- Guillard, R.R.L. Culture of phytoplankton for feeding marine invertebrates. In Culture of Marine Invertebrate Animals; Smith, W.L., Chanley, M.H., Eds.; Plenum Press: New York, NY, USA, 1975; pp. 29–60. [Google Scholar]
- Guihéneuf, F.; Stengel, D. LC-PUFA-Enriched Oil Production by Microalgae: Accumulation of Lipid and Triacylglycerols Containing n-3 LC-PUFA Is Triggered by Nitrogen Limitation and Inorganic Carbon Availability in the Marine Haptophyte Pavlova lutheri. Mar. Drugs 2013, 11, 4246–4266. [Google Scholar] [CrossRef] [PubMed]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, A.; Carmeli, Y.; Berner, T. Regulation of fatty acid composition by irradiance level in the Eustigmatophyte Nannochloropsis sp.1. J. Phycol. 1989, 25, 686–692. [Google Scholar] [CrossRef]
- Williams, J.P. Glycerolipids and fatty acids on algae. In Handbook of Phycological Methods; Hellebust, J.A., Craigie, J.S., Eds.; Cambridge University Press: Cambridge, UK, 1978; pp. 99–107. [Google Scholar]
- Christie, W.W. Lipid Analysis, 2nd ed.; Pergamon Books: Oxford, UK, 1982. [Google Scholar]
- Henderson, R.J.; Tocher, D.R. Thin-layer chromatography. In Lipid Analysis: A Practical Approach; Hamilton, R.J., Hamilton, S., Eds.; IRL Press: Oxford, UK, 1992; pp. 65–111. [Google Scholar]
- Wright, S.W.; Jeffrey, S.W.; Mantoura, R.F.C.; Llewellyn, C.A.; Bjornland, T.; Repeta, D.; Welschmeyer, N. Improved HPLC method for the analysis of chlorophylls and carotenoids from marine phytoplankton. Mar. Ecol. Prog. Ser. 1991, 77, 183–196. [Google Scholar] [CrossRef]
- Bidigare, R.R.; Van Heukelem, L.; Trees, C.C. Analysis of algal pigments by high-performance liquid chromatography. In Algal Culturing Techniques; Andersen, R.A., Ed.; Academic Press: London, UK, 2005; pp. 327–345. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robertson, R.C.; Guihéneuf, F.; Bahar, B.; Schmid, M.; Stengel, D.B.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. The Anti-Inflammatory Effect of Algae-Derived Lipid Extracts on Lipopolysaccharide (LPS)-Stimulated Human THP-1 Macrophages. Mar. Drugs 2015, 13, 5402-5424. https://doi.org/10.3390/md13085402
Robertson RC, Guihéneuf F, Bahar B, Schmid M, Stengel DB, Fitzgerald GF, Ross RP, Stanton C. The Anti-Inflammatory Effect of Algae-Derived Lipid Extracts on Lipopolysaccharide (LPS)-Stimulated Human THP-1 Macrophages. Marine Drugs. 2015; 13(8):5402-5424. https://doi.org/10.3390/md13085402
Chicago/Turabian StyleRobertson, Ruairi C., Freddy Guihéneuf, Bojlul Bahar, Matthias Schmid, Dagmar B. Stengel, Gerald F. Fitzgerald, R. Paul Ross, and Catherine Stanton. 2015. "The Anti-Inflammatory Effect of Algae-Derived Lipid Extracts on Lipopolysaccharide (LPS)-Stimulated Human THP-1 Macrophages" Marine Drugs 13, no. 8: 5402-5424. https://doi.org/10.3390/md13085402
APA StyleRobertson, R. C., Guihéneuf, F., Bahar, B., Schmid, M., Stengel, D. B., Fitzgerald, G. F., Ross, R. P., & Stanton, C. (2015). The Anti-Inflammatory Effect of Algae-Derived Lipid Extracts on Lipopolysaccharide (LPS)-Stimulated Human THP-1 Macrophages. Marine Drugs, 13(8), 5402-5424. https://doi.org/10.3390/md13085402






