Antioxidant, Anti-Nephrolithe Activities and in Vitro Digestibility Studies of Three Different Cyanobacterial Pigment Extracts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Extracts
| Compounds (mg·g−1 dry mass) | Pseudanabaena sp. | Spirulina sp. | Lyngbya sp. | |
|---|---|---|---|---|
| Phycobiliproteins (Water extract) | C-PC a | 6.96 ± 0.34 | 11.62 ± 0.3 | 107.92 ± 1.79 |
| A-PC a | 2.94 ± 0.2 | 3.87 ± 0.11 | 21.64 ± 0.75 | |
| C-PE a | 9.45 ± 0.45 | 0.93 ± 0.02 | 108.76 ± 2.18 | |
| Carotenoids (Methanol extract) | Myxoxanthophyll | 0.05 ± 0.01 | 0.02 ± 0.00 | 0.05 ± 0.02 |
| Zeaxanthin | 0.29 ± 0.02 | 0.12 ± 0.01 | 0.15 ± 0.13 | |
| Canthaxanthin | 0.11 ± 0.01 | 0.04 ± 0.00 | 0.04 ± 0.01 | |
| Lycopene | 0.12 ± 0.01 | 0.17 ± 0.02 | ND a | |
| α-carotene | 0.08 ± 0.00 | 0.09 ± 0.01 | 0.10 ± 0.02 | |
| β-carotene | 0.27 ± 0.02 | 0.23 ± 0.02 | 0.17 ± 0.03 | |
| Echinenone | ND a | 0.38 ± 0.04 | ND a | |
| Chlorophyll (Methanol extract) | Chlorophyll a | 0.70 ± 0.03 | 4.80 ± 0.51 | 1.5 ± 0.03 |
2.2. Antioxidant Activities
2.2.1.Total Phenolic Content (TPC)

2.2.2. Total Antioxidant Capacity (TAC)
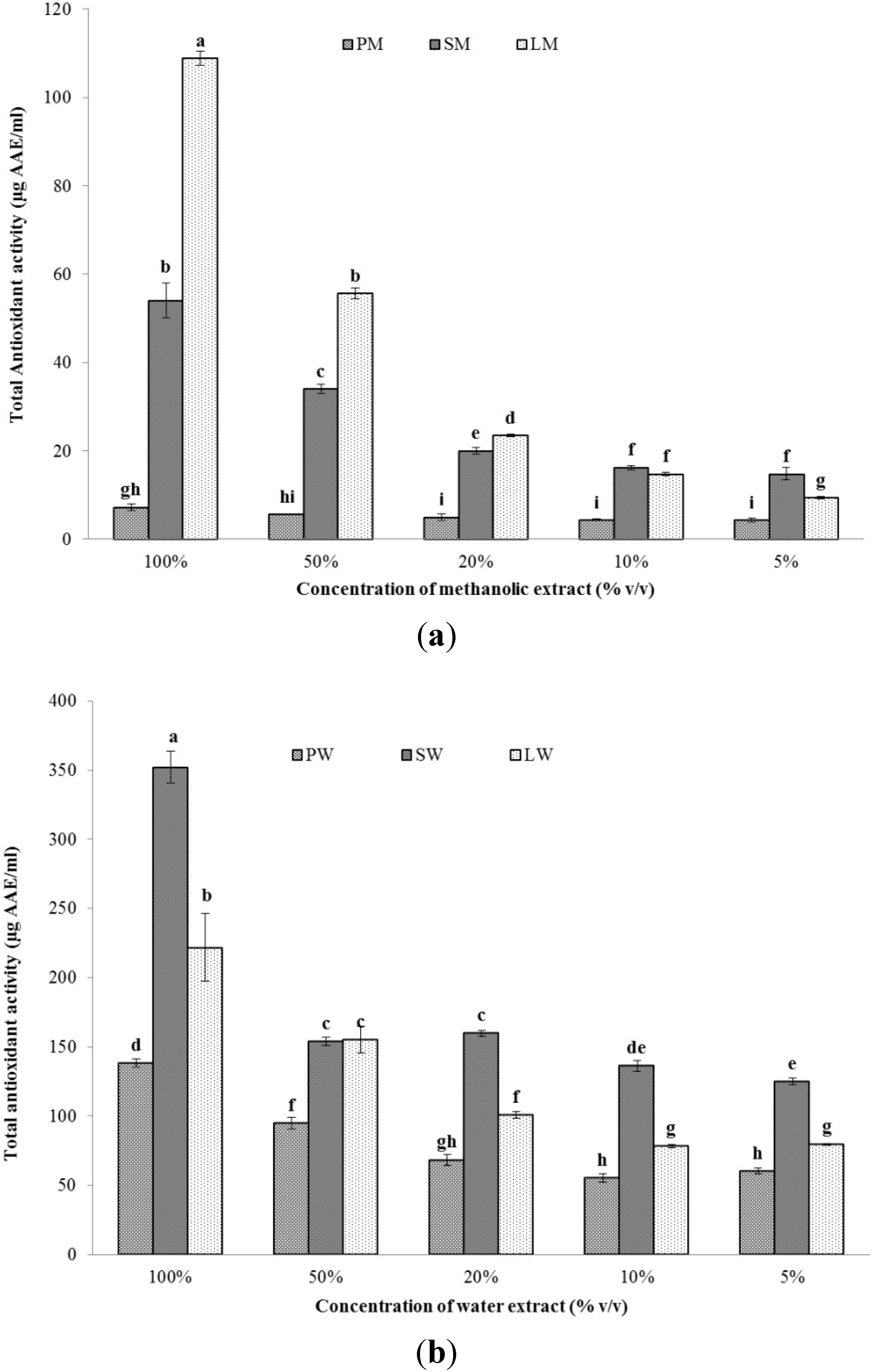
2.2.3. DPPH Scavenging Activity and EC50 Value
| Sample | TPC (mg GAE g−1 dry mass) | TAC (mg AAE g−1 dry mass) | EC50 DPPH (mg dry mass mg−1 DPPH) |
|---|---|---|---|
| PW | 58.94 ± 0.34 | 49.92 ± 1.11 | 42.73 ± 4.29 |
| SW | 119.19 ± 2.09 | 22.59 ± 0.74 | 148.46 ± 21.17 |
| LW | 278.73 ± 1.24 | 169.42 ± 18.54 | 18.78 ± 1.57 |
| PM | 0.65 ± 0.24 | 2.61 ± 0.27 | 1544.43 ± 207.76 |
| SM | 0.66 ± 0.3 | 3.46 ± 0.26 | 728.55 ± 667.73 |
| LM | 12.77 ± 2.23 | 83.11 ± 1.19 | 59.56 ± 37.38 |
2.3. Iso-Bolographic Studies
| Combination of Extracts | Fraction | Theoretical EC50 (mg mg−1 DPPH) | Experimental EC50 (mg mg−1 DPPH) | Synergistic Rate (%) |
|---|---|---|---|---|
| LW-PW | F1/9 | 40.33 | 185.21 ± 9.23 a | −359.20 |
| F5/5 | 30.75 | 55.88 ± 22.17 a | −81.71 | |
| F9/1 | 21.17 | 26.96 ± 11.99 a | −27.36 | |
| PW-SW | F1/9 | 137.89 | 261.88 ± 111.67 a | −89.92 |
| F5/5 | 95.59 | 130.93 ± 55.45 a,b | −36.97 | |
| F9/1 | 53.30 | 88.15 ± 63.01 b | −65.38 | |
| LW-SW | F1/9 | 135.49 | 242.33 ± 120.34 a | −78.85 |
| F5/5 | 83.62 | 29.60 ± 5.18 b | 64.60 | |
| F9/1 | 31.75 | 32.34 ± 17.66 b | −1.88 | |
| LM-SM | F1/9 | 661.65 | 164.23 ± 84.79 a | 75.18 |
| F5/5 | 394.06 | 52.60 ± 17.10 a | 86.65 | |
| F9/1 | 126.46 | 53.63 ± 30.31 a | 57.60 | |
| LM-PM | F1/9 | 1395.94 | 5371.82 ± 628.95 a | −284.82 |
| F5/5 | 802.00 | 883.11 ± 89.96 a | −10.11 | |
| F9/1 | 208.05 | 1388.47 ± 320.20 a | −567.37 | |
| SM-PM | F1/9 | 1462.84 | 1141.63 ± 427.18 a | 21.96 |
| F5/5 | 1136.49 | 726.50 ± 58.38 a | 36.07 | |
| F9/1 | 810.14 | 463.86 ± 82.31 a | 42.74 | |
| LW-SM | F1/9 | 657.57 | 587.30 ± 93.65 a | 10.69 |
| F5/5 | 373.66 | 595.09 ± 112.33 a | −59.26 | |
| F9/1 | 89.75 | 71.10 ± 52.77 a | 20.79 | |
| SW-PM | F1/9 | 1404.83 | 718.10 ± 82.43 a | 48.88 |
| F5/5 | 846.45 | 1242.09 ± 204.02 a | −46.74 | |
| F9/1 | 288.06 | 450.80 ± 198.58 a | −56.50 | |
| PW-LM | F1/9 | 57.88 | 50.28 ± 23.22 a | 13.13 |
| F5/5 | 51.15 | 176.53 ± 26.72 a | −245.15 | |
| F9/1 | 44.41 | 53.35 ± 18.67 a | −20.13 | |
| LM-SW | F1/9 | 139.57 | 171.74 ± 74.87 a | −23.05 |
| F5/5 | 104.01 | 690.94 ± 57.20 a,b | −564.29 | |
| F9/1 | 68.45 | 70.23 ± 13.17 b | −2.60 | |
| PM-LW | F1/9 | 171.34 | 220.12 ± 45.49 a | −28.47 |
| F5/5 | 781.60 | 858.59 ± 166.33 a | −9.85 | |
| F9/1 | 1391.87 | 1908.85 ± 388.08 a | −37.14 | |
| SM-PW | F1/9 | 111.31 | 94.51 ± 51.36 a | 15.09 |
| F5/5 | 385.64 | 68.65 ± 25.04 a | 82.20 | |
| F9/1 | 659.97 | 111.03 ± 78.97 a | 83.18 |
2.4. In Vitro Digestibility Studies
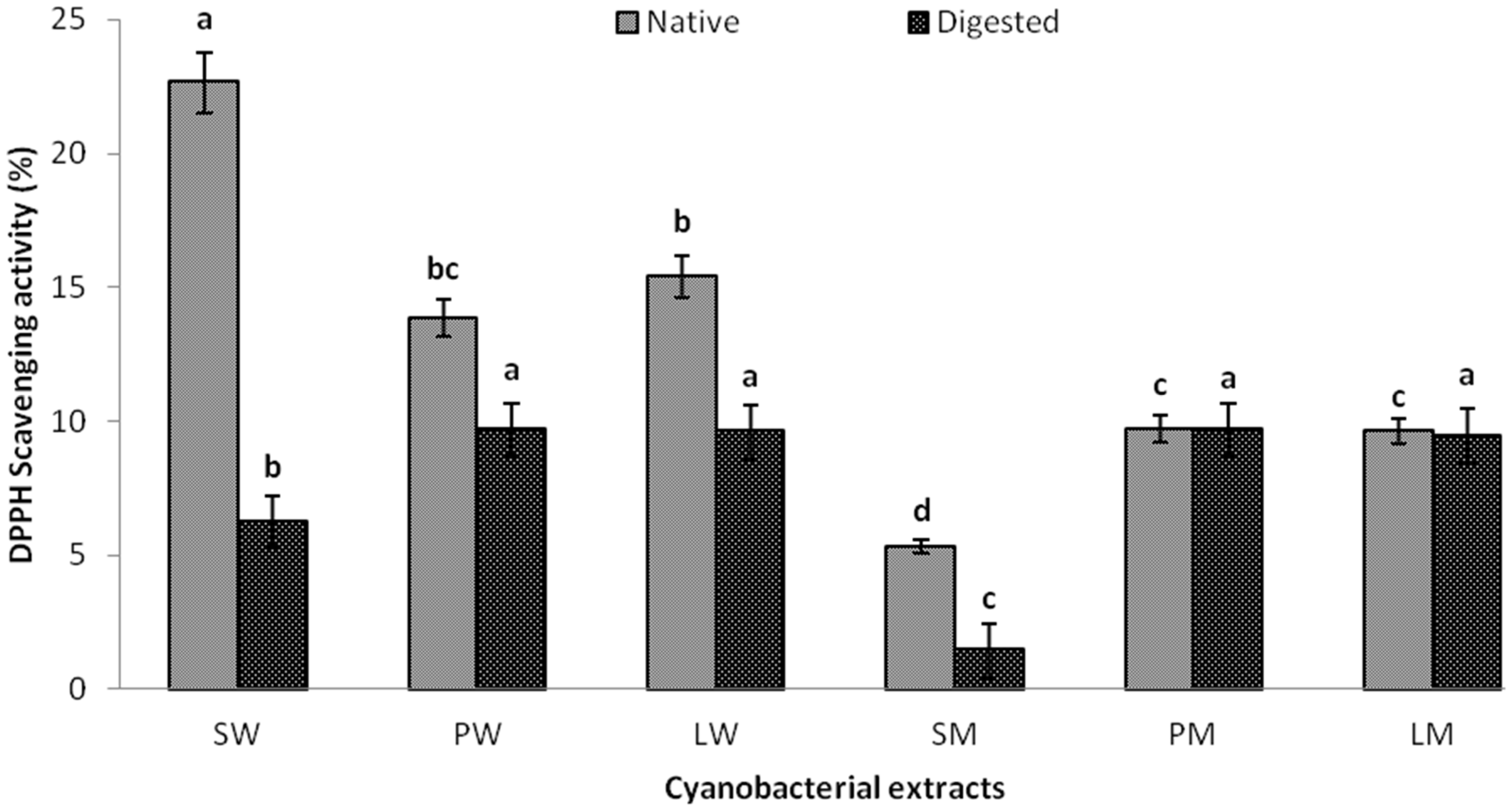
2.5. Effect of Water and Methanolic Extracts on Calcium Oxalate Crystallization
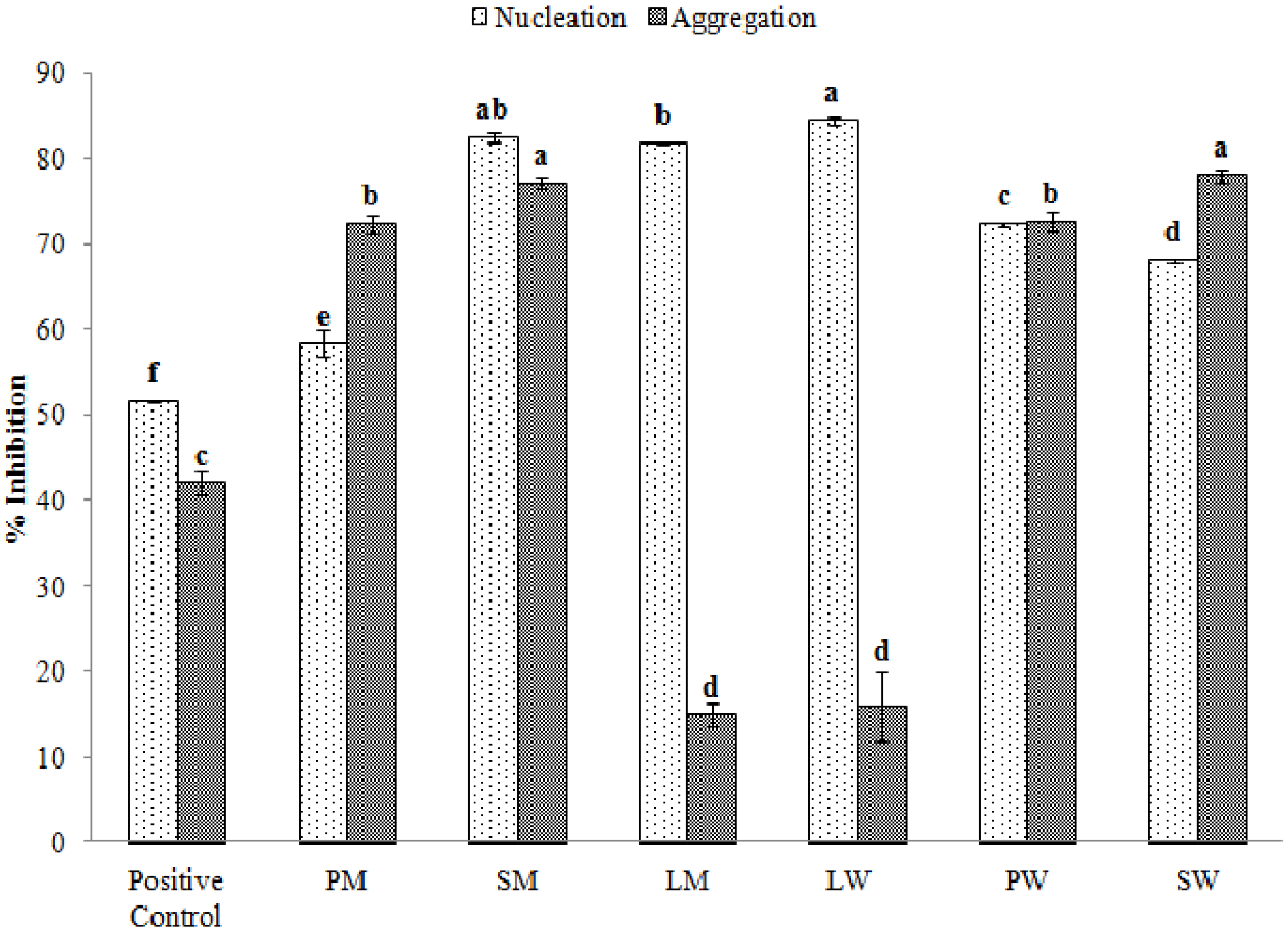
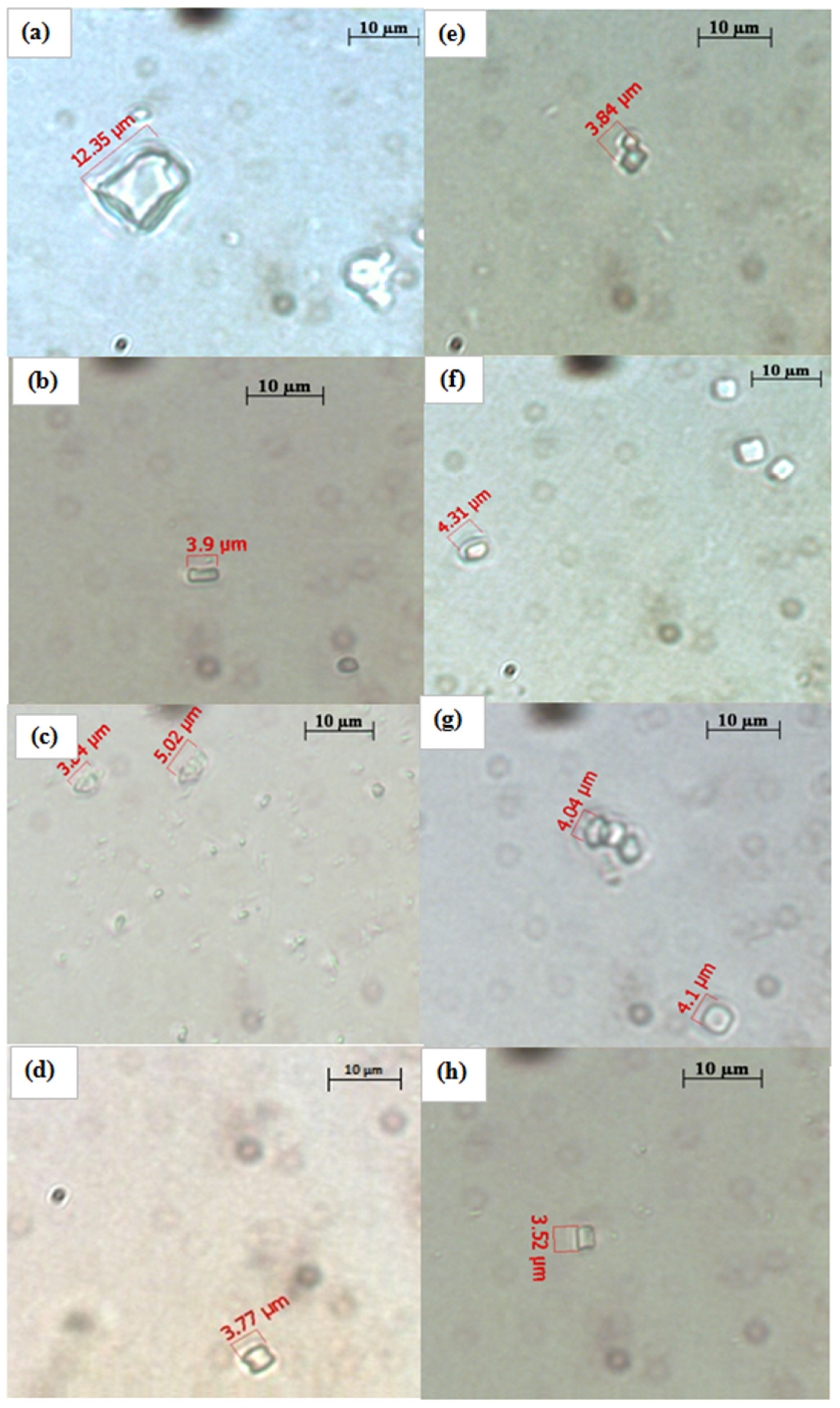
3. Experimental Section
3.1. Preparation of Extracts
3.2. Characterization of Water and Methanol Extracts
3.3. Antioxidant Activities
3.3.1. Total Phenolic Content
3.3.2. Total Antioxidant Activity
3.3.3. DPPH Assay
3.4. Iso-Bolographic Studies
3.5. In Vitro Digestibility of Different Extracts
3.6. Calcium Oxalate Crystallization Assay
3.7. Statistical Analysis
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflict of Interest
References
- Shishido, T.; Humisto, A.; Jokela, J.; Liu, L.; Wahlsten, M.; Tamrakar, A.; Fewer, D.; Permi, P.; Andreote, A.; Fiore, M.; et al. Antifungal compounds from cyanobacteria. Mar. Drugs 2015, 13, 2124–2140. [Google Scholar] [CrossRef] [PubMed]
- Guschina, I.A.; Harwood, J.L. Lipids and lipid metabolism in eukaryotic algae. Prog. Lipid Res. 2006, 45, 160–186. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, K. Renewable Biological Systems for Alternative Sustainable Energy Production; FAO: Osaka, Japan, 1997. [Google Scholar]
- Sekar, S.; Chandramohan, M. Phycobiliproteins as a commodity: Trends in applied research, patents and commercialization. J. Appl. Phycol. 2008, 20, 113–136. [Google Scholar] [CrossRef]
- Fabregas, J.; Herrero, C. Vitamin content of four marine microalgae. Potential use as source of vitamins in nutrition. J. Ind. Microbiol. 1990, 5, 259–263. [Google Scholar] [CrossRef]
- Mishra, A.; Kavita, K.; Jha, B. Characterization of extracellular polymeric substances produced by micro-algae Dunaliella salina. Carbohydr. Polym. 2011, 83, 852–857. [Google Scholar] [CrossRef]
- Hirschberg, J.; Chamovitz, D. Carotenoids in cyanobacteria. In The Molecular Biology of Cyanobacteria; Bryant, D.A., Ed.; Advances in Photosynthesis and Respiration; Springer: Dordrecht, The Netherlands, 2004; Volume 1, pp. 559–579. [Google Scholar]
- Koller, M.; Muhr, A.; Braunegg, G. Microalgae as versatile cellular factories for valued products. Algal Res. 2014, 6, 52–63. [Google Scholar] [CrossRef]
- Blanco, A.; Moreno, J.; Del Campo, J.; Rivas, J.; Guerrero, M. Outdoor cultivation of lutein-rich cells of Muriellopsis sp. in open ponds. Appl. Microbiol. Biotechnol. 2007, 73, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Sonani, R.; Singh, N.; Awasthi, A.; Prasad, B.; Kumar, J.; Madamwar, D. Phycoerythrin extends life span and health span of Caenorhabditis elegans. AGE 2014, 36, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sonani, R.R.; Singh, N.K.; Kumar, J.; Thakar, D.; Madamwar, D. Concurrent purification and antioxidant activity of phycobiliproteins from Lyngbya sp. A09DM: An antioxidant and anti-aging potential of phycoerythrin in Caenorhabditis elegans. Process Biochem. 2014, 49, 1757–1766. [Google Scholar] [CrossRef]
- Wang, Y.; Cai, C.; Li, B.; Liu, C.; He, P. Photodynamic effect of two kinds of phycobiliproteins on human liver cancer cell line SMMC-7721 in vitro. Sheng Wu Gong Cheng Xue Bao 2009, 25, 1417–1423. [Google Scholar] [PubMed]
- Zhang, C.-Y.; Wu, W.-H.; Wang, J.; Lan, M.-B. Antioxidant properties of polysaccharide from the brown seaweed Sargassum graminifolium (Turn.), and its effects on calcium oxalate crystallization. Mar. Drugs 2012, 10, 119–130. [Google Scholar] [CrossRef] [PubMed]
- McClements, D.J.; Decker, E.A.; Park, Y.; Weiss, J. Structural design principles for delivery of bioactive components in nutraceuticals and functional foods. Crit. Rev. Food Sci. Nutr. 2009, 49, 577–606. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Liu, H.-J.; Mei, X.-Y.; Nakajima, M.; Yin, L.-J. Preliminary study into the factors modulating β-carotene micelle formation in dispersions using an in vitro digestion model. Food Hydrocoll. 2012, 26, 427–433. [Google Scholar] [CrossRef]
- Golding, M.; Wooster, T.J. The influence of emulsion structure and stability on lipid digestion. Curr. Opin. Coll. Interface Sci. 2010, 15, 90–101. [Google Scholar] [CrossRef]
- Morist, A.; Montesinos, J.; Cusidó, J.; Gòdia, F. Recovery and treatment of Spirulina platensis cells cultured in a continuous photobioreactor to be used as food. Process Biochem. 2001, 37, 535–547. [Google Scholar] [CrossRef]
- Mishra, S.K.; Shrivastav, A.; Mishra, S. Preparation of highly purified C-phycoerythrin from marine cyanobacterium Pseudanabaena sp. Protein Expr. Purif. 2011, 80, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, H.; Rivas, J.; Guerrero, M.G.; Losada, M. Nitrogen-fixing cyanobacterium with a high phycoerythrin content. Appl. Environ. Microbiol. 1989, 55, 758–760. [Google Scholar] [PubMed]
- Aakermann, T.; Skulberg, O.M.; Liaaen-Jensen, S. A comparison of the carotenoids of strains of Oscillatoria and Spirulina (Cyanobacteria). Biochem. Syst. Ecol. 1992, 20, 761–769. [Google Scholar] [CrossRef]
- Singh, O.A. Diversity Analysis and PCR Based Molecular Characterization of Carotenoid Rich Oscillatorian Cyanobacteria of North Eastern (NE) Region of India. Ph.D. Thesis, Assam University, Assam, India, 27 February 2015. [Google Scholar]
- Jiang, H.; Li, H.; Yu, C.; Yang, T.; Hu, J.; Liu, R.; Deng, Z.-Y. The evaluation of antioxidant interactions among 4 common vegetables using isobolographic analysis. J. Food Sci. 2015, 80, C1162–C1169. [Google Scholar] [CrossRef] [PubMed]
- Machu, L.; Misurcova, L.; Vavra Ambrozova, J.; Orsavova, J.; Mlcek, J.; Sochor, J.; Jurikova, T. Phenolic content and antioxidant capacity in algal food products. Molecules 2015, 20, 1118–1133. [Google Scholar] [CrossRef] [PubMed]
- Ismaiel, M.M.S.; El-Ayouty, Y.M.; Piercey-Normore, M.D. Antioxidants characterization in selected cyanobacteria. Ann. Microbiol. 2014, 64, 1223–1230. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, P.; Kumar, C.S.; Bhaskar, N. Antioxidant properties of methanol extract and its solvent fractions obtained from selected Indian red seaweeds. Bioresour. Technol. 2008, 99, 2717–2723. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Kumari, P.; Trivedi, N.; Shukla, M.; Gupta, V.; Reddy, C.R.K.; Jha, B. Minerals, PUFAs and antioxidant properties of some tropical seaweeds from Saurashtra coast of India. J. Appl. Phycol. 2011, 23, 797–810. [Google Scholar] [CrossRef]
- Cervantes-Paz, B.; Yahia, E.M.; de Jesús Ornelas-Paz, J.; Victoria-Campos, C.I.; Ibarra-Junquera, V.; Pérez-Martínez, J.D.; Escalante-Minakata, P. Antioxidant activity and content of chlorophylls and carotenoids in raw and heat-processed Jalapeño peppers at intermediate stages of ripening. Food Chem. 2014, 146, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, W.; Rui, X.; Chen, X.; Jiang, M.; Dong, M. Structural characterization and bioactivity of released exopolysaccharides from Lactobacillus plantarum 70810. Int. J. Biol. Macromol. 2014, 67, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Bláha, L.; Babica, P.; Maršálek, B. Toxins produced in cyanobacterial water blooms—Toxicity and risks. Interdiscip. Technol. 2009, 2, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, W.W.; Mahmood, N.A.; Hyde, E.G. Natural Toxins from Cyanobacteria (Blue-Green Algae). In Marine Toxins; Hall, S., Strichartz, G., Eds.; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 1990; Volume 418, pp. 87–106. [Google Scholar]
- Wu, Q.; Fu, X.-P.; Sun, L.-C.; Zhang, Q.; Liu, G.-M.; Cao, M.-J.; Cai, Q.-F. Effects of physicochemical factors and in vitro gastrointestinal digestion on antioxidant activity of R-phycoerythrin from red algae Bangia fusco-purpurea. Int. J. Food Sci. Technol. 2015, 50, 1445–1451. [Google Scholar] [CrossRef]
- Teodosio Melo, K.R.; Gomes Camara, R.B.; Queiroz, M.F.; Jacome Vidal, A.A.; Machado Lima, C.R.; Melo-Silveira, R.F.; Almeida-Lima, J.; Oliveira Rocha, H.A. Evaluation of sulfated polysaccharides from the brown seaweed Dictyopteris justii as antioxidant agents and as inhibitors of the formation of calcium oxalate crystals. Molecules 2013, 18, 14543–14563. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Mishra, S.; Pawar, R.; Ghosh, P.K. Purification and characterization of C-Phycocyanin from cyanobacterial species of marine and freshwater habitat. Protein Expr. Purif. 2005, 40, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Pancha, I.; Chokshi, K.; George, B.; Ghosh, T.; Paliwal, C.; Maurya, R.; Mishra, S. Nitrogen stress triggered biochemical and morphological changes in the microalgae Scenedesmus sp. CCNM 1077. Bioresour. Technol. 2014, 156, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.; Bogorad, L. Complementary chromatic adaptation in a filamentous blue-green alga. J. Cell Biol. 1973, 58, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Andersson, S.C.; Rumpunen, K.; Johansson, E.; Olsson, M.E. Carotenoid content and composition in rose hips (Rosa spp.) during ripening, determination of suitable maturity marker and implications for health promoting food products. Food Chem. 2011, 128, 689–696. [Google Scholar] [CrossRef]
- Dudonné, S.; Vitrac, X.; Coutière, P.; Woillez, M.; Mérillon, J.-M. Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem. 2009, 57, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Šiler, B.; Živković, S.; Banjanac, T.; Cvetković, J.; Nestorović Živković, J.; Ćirić, A.; Soković, M.; Mišić, D. Centauries as underestimated food additives: Antioxidant and antimicrobial potential. Food Chem. 2014, 147, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Hur, S.J.; Decker, E.A.; McClements, D.J. Influence of initial emulsifier type on microstructural changes occurring in emulsified lipids during in vitro digestion. Food Chem. 2009, 114, 253–262. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paliwal, C.; Ghosh, T.; Bhayani, K.; Maurya, R.; Mishra, S. Antioxidant, Anti-Nephrolithe Activities and in Vitro Digestibility Studies of Three Different Cyanobacterial Pigment Extracts. Mar. Drugs 2015, 13, 5384-5401. https://doi.org/10.3390/md13085384
Paliwal C, Ghosh T, Bhayani K, Maurya R, Mishra S. Antioxidant, Anti-Nephrolithe Activities and in Vitro Digestibility Studies of Three Different Cyanobacterial Pigment Extracts. Marine Drugs. 2015; 13(8):5384-5401. https://doi.org/10.3390/md13085384
Chicago/Turabian StylePaliwal, Chetan, Tonmoy Ghosh, Khushbu Bhayani, Rahulkumar Maurya, and Sandhya Mishra. 2015. "Antioxidant, Anti-Nephrolithe Activities and in Vitro Digestibility Studies of Three Different Cyanobacterial Pigment Extracts" Marine Drugs 13, no. 8: 5384-5401. https://doi.org/10.3390/md13085384
APA StylePaliwal, C., Ghosh, T., Bhayani, K., Maurya, R., & Mishra, S. (2015). Antioxidant, Anti-Nephrolithe Activities and in Vitro Digestibility Studies of Three Different Cyanobacterial Pigment Extracts. Marine Drugs, 13(8), 5384-5401. https://doi.org/10.3390/md13085384







