Biological Properties of Fucoxanthin in Oil Recovered from Two Brown Seaweeds Using Supercritical CO2 Extraction
Abstract
:1. Introduction
2. Results and Discussion
2.1. Conventional Solvent Extraction
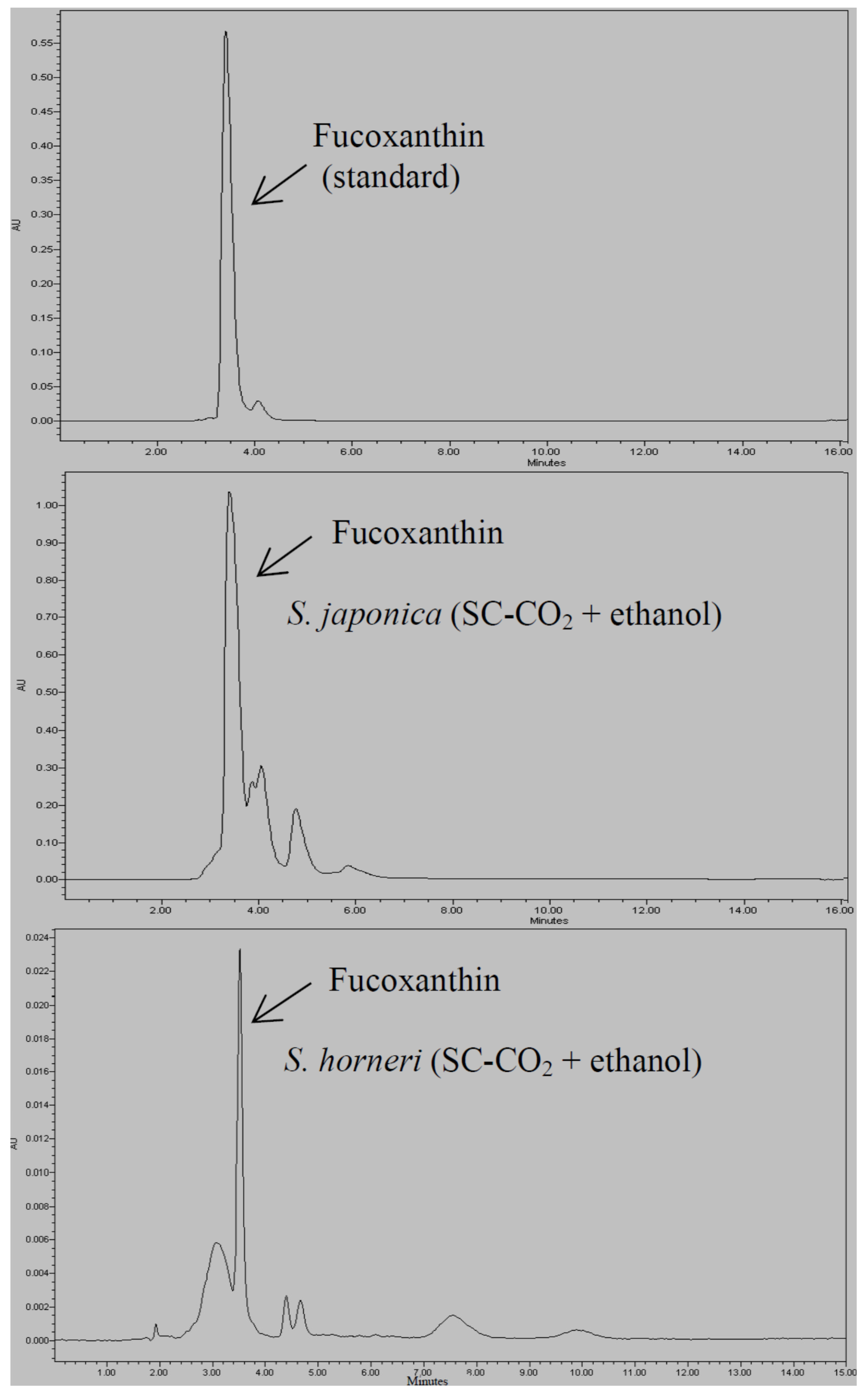
2.2. Extraction of Oil Using SC-CO2 with Ethanol as Co-Solvent
| Solvent | Extraction Yield (g/100 g DW) | Fucoxanthin Content (mg/g) | ||
|---|---|---|---|---|
| S. japonica | S. horneri | S. japonica | S. horneri | |
| SC-CO2 + Ethanol | 1.09 ± 0.56 a | 1.41 ± 0.15 b | 0.41 ± 0.05 a | 0.77 ± 0.07 a |
| Acetone mix methanol | 1.19 ± 0.21 a | 1.29 ± 0.05 a | 0.48 ± 0.10 b | 0.71 ± 0.05 b |
| Hexane | 1.24 ± 0.06 a | 1.42 ± 0.08 c | 0.16 ± 0.01 a | 0.05 ± 0.03 c |
| Ethanol | 1.22 ± 0.12 a | 1.36 ± 0.14 c | 0.12 ± 0.02 a,b | 0.08 ± 0.05 c |
2.3. Measurement of FA Composition and Fucoxanthin Content
| Fatty acid compositions | S. japonica (mg/g in Extracted Oil) | S. horneri (mg/g in Extracted Oil) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| SC-CO2 + Ethanol | Acetone mix methanol | Hexane | Ethanol | SC-CO2 + Ethanol | Acetone mix methanol | Hexane | Ethanol | ||
| Saturated fatty acid (SFA) | C10:0 | N.D | N.D | N.D | N.D | 49.1 ± 0.70 | 51.4 ± 0.50 | 211.7 ± 3.1 | N.D |
| C11:0 | N.D | N.D | N.D | N.D | N.D | 8.5 ± 0.30 | N.D | 250.10 ± 3.50 | |
| C12:0 | N.D | N.D | N.D | N.D | 17.4 ± 0.10 | 20.1 ± 0.20 | 27.9 ± 0.30 | N.D | |
| C13:0 | 14.10 ± 1.0 | N.D | N.D | N.D | 34.3 ± 0.50 | 29.90 ± 0.20 | 23.00 ± 0.50 | N.D | |
| C14:0 | 141.40 ± 8.00 | 123.8 ± 1.00 | 130.50 ± 1.40 | 114.80 ± 0.20 | 75.50 ± 0.80 | 65.80 ± 0.40 | 13.00 ± 0.30 | 38.60 ± 0.40 | |
| C15:0 | 4.60 ± 0.10 | 10.20 ± 0.10 | 14.50 ± 0.20 | 4.30 ± 0.10 | 41.60 ± 0.50 | 52.40 ± 0.40 | 21.20 ± 0.40 | 16.30 ± 0.30 | |
| C16:0 | N.D | N.D | N.D | N.D | 214.80 ± 3.40 | 185.40 ± 2.40 | 180.70 ± 1.80 | 233.2 ± 2.90 | |
| C17:0 | N.D | N.D | N.D | 18.40 ± 0.10 | N.D | 20.10 ± 0.10 | 22.50 ± 0.60 | N.D | |
| C18:0 | N.D | N.D | N.D | N.D | 17.10 ± 0.20 | 19.50 ± 0.20 | 26.00 ± 0.40 | N.D | |
| C20:0 | N.D | N.D | 48.10 ± 0.10 | 51.70 ± 0.20 | 8.50 ± 0.10 | 12.10 ± 0.20 | 8.60 ± 0.10 | 29.80 ± 0.40 | |
| C21:0 | 29.80 ± 0.10 | 107.10 ± 0.10 | 91.90 ± 1.00 | 102.70 ± 0.50 | 62.30 ± 0.80 | 61.10 ± 0.60 | 44.30 ± 0.40 | 39.30 ± 0.50 | |
| C23:0 | N.D | N.D | 7.20 ± 0.10 | 5.80 ± 0.10 | N.D | 7.60 ± 0.20 | N.D | 11.30 ± 0.40 | |
| Total SFA | 189.90 ± 9.20 | 241.10 ± 1.20 | 292.20 ± 2.80 | 297.70 ± 1.20 | 520.60 ± 7.10 | 533.90 ± 5.70 | 578.90 ± 7.90 | 618.60 ± 8.40 | |
| Monounsaturated fatty acid (MUFA) | C15:1 | 252.70 ± 0.10 | 179.80 ± 0.10 | 178.00 ± 1.00 | 200.90 ± 0.10 | 11.30 ± 0.20 | 15.90 ± 0.30 | 28.40 ± 0.50 | N.D |
| C14:1 | 5.00 ± 0.10 | 29.90 ± 0.00 | 8.60 ± 0.50 | 21.30 ± 0.40 | 16.60 ± 0.20 | 21.40 ± 0.30 | 21.10 ± 0.20 | 56.40 ± 0.50 | |
| C16:1 | 18.70 ± 0.10 | 45.30 ± 0.10 | 45.80 ± 0.10 | 40.10 ± 0.70 | 26.90 ± 0.40 | 23.10 ± 0.20 | 13.10 ± 0.30 | 56.00 ± 0.80 | |
| C17:1 | 26.90 ± 0.10 | 29.60 ± 0.40 | 10.20 ± 0.10 | 19.60 ± 0.10 | 6.20 ± 0.10 | 29.60 ± 0.40 | 37.90 ± 0.70 | N.D | |
| C18:1n9c | N.D | N.D | N.D | N.D | 122.90 ± 0.50 | 112.40 ± 0.70 | 89.80 ± 0.70 | 83.70 ± 1.10 | |
| C18:1n9t | 340.10 ± 0.10 | 182.90 ± 0.50 | 177.50 ± 1.00 | 168.90 ± 0.20 | 16.90 ± 0.20 | 18.50 ± 0.50 | 23.40 ± 0.30 | N.D | |
| C22:1n9 | N.D | N.D | N.D | N.D | N.D | 8.30 ± 0.20 | N.D | N.D | |
| Total MUFA | 644.30 ± 0.50 | 467.50 ± 1.10 | 420.10 ± 2.70 | 450.80 ± 1.50 | 200.80 ± 1.60 | 229.20 ± 2.60 | 213.70 ± 2.70 | 196.10 ± 2.40 | |
| Polyunsaturated fatty acid (PUFA) | C18:2n6c | 10.40 ± 0.10 | 53.80 ± 0.20 | 78.70 ± 0.10 | 83.50 ± 0.20 | 7.30 ± 0.10 | 12.50 ± 0.10 | 11.70 ± 0.20 | 38.60 ± 0.40 |
| C18:2n6t | 4.10 ± 0.10 | N.D | N.D | N.D | 64.30 ± 0.50 | 70.10 ± 0.40 | 20.60 ± 0.40 | N.D | |
| C18:3n6 | 23.20 ± 0.10 | 47.80 ± 0.10 | 45.00 ± 0.10 | 45.90 ± 0.50 | 24.60 ± 0.30 | 16.90 ± 0.50 | 9.50 ± 0.10 | N.D | |
| C20:2 | 23.50 ± 0.10 | 56.40 ± 1.00 | N.D | N.D | 10.50 ± 0.10 | 18.70 ± 0.30 | 10.60 ± 0.20 | 22.0 ± 0.40 | |
| C18:3n3 | N.D | N.D | N.D | N.D | N.D | 4.50 ± 0.10 | 5.10 ± 0.20 | 30.20 ± 0.60 | |
| C20:3n6 | 27.80 ± 0.20 | 62.80 ± 0.50 | 107.00 ± 1.40 | 104.80 ± 0.80 | 65.80 ± 0.90 | 64.50 ± 0.50 | 66.70 ± 0.70 | 60.10 ± 0.80 | |
| C22:2 | N.D | N.D | N.D | N.D | 13.90 ± 0.30 | 9.50 ± 0.40 | 8.00 ± 0.10 | N.D | |
| C20:5n3 | 57.70 ± 0.40 | 70.60 ± 0.10 | 57.00 ± 0.20 | 17.30 ± 0.20 | 80.80 ± 0.90 | 54.70 ± 1.20 | 75.20 ± 0.80 | 34.40 ± 0.50 | |
| C22:6n3 | N.D | N.D | N.D | N.D | N.D | N.D | N.D | N.D | |
| Total PUFA | 146.70 ± 1.00 | 291.40 ± 1.90 | 287.70 ± 1.80 | 251.50 ± 1.70 | 267.20 ± 3.10 | 251.40 ± 3.50 | 202.30 ± 2.70 | 185.30 ± 2.70 | |
2.4. Antioxidant Properties and Total Phenolic Content
2.4.1. Hydrogen Peroxide Scavenging (H2O2) Assay
| H2O2 IC50 (μg/mL) | β-carotene Bleaching Assay (%) | Total Antioxidant Capacity (mg/g α-tocopherol DW) | Total Phenolic Content (mg/g GAE DW) | |||||
|---|---|---|---|---|---|---|---|---|
| Solvent | S. japonica | S. horneri | S. japonica | S. horneri | S. japonica | S. horneri | S. japonica | S. horneri |
| SC-CO2 + ethanol | 600.73 ± 0.15 a | 686.31 ± 0.20 a | 72.00 ± 0.74 a | 75.03 ± 1.56 a | 30.74 ± 0.13 a | 38.91 ± 3.79 a | 0.60 ± 0.05 a | 0.64 ± 0.01 a |
| Acetone mix methanol | 450.49 ± 1.42 b | 546.10 ± 1.05 b | 65.10 ± 3.03 b | 69.51 ± 0.35 b | 28.74 ± 1.38 b | 28.05 ± 0.72 b | 0.56 ± 0.04 b | 0.60 ± 0.01 b |
| Hexane | 100.15 ± 2.17 c | 125.18 ± 0.35 c | 31.81 ± 0.76 c | 36.58 ± 0.65 c | 16.67 ± 2.41 c | 19.77 ± 0.86 c | 0.28 ± 0.01 c | 0.42 ± 0.02 c |
| Ethanol | 200.02 ± 4.21 d | 220.62 ± 1.57 d | 42.66 ± 0.85 d | 46.71 ± 2.62 d | 22.5 ± 0.10 d | 25.63 ± 0.93 d | 0.34 ± 0.05 d | 0.43 ± 0.01 c |
| Ascorbic acid | 448.19 ± 0.78 | |||||||
| Trolox | 84.56 ± 0.34 | |||||||
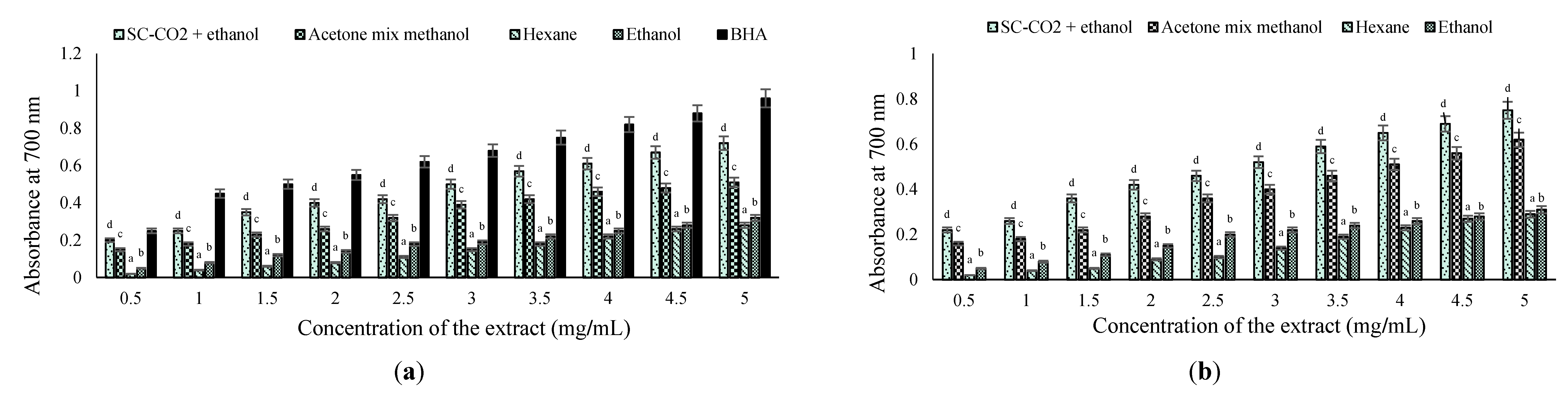
2.4.2. Reducing Power (RP) Method
2.4.3. β-Carotene Linoleic Acid Method
2.4.4. Total Antioxidant Activity
2.4.5. Total Phenolic Content
2.5. Antimicrobial Studies
| Inhibition Zone (mm) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Microorganisms | S. japonica extracts | S. horneri extracts | ||||||
| SC-CO2 + Ethanol | Acetone mix Methanol | Hexane | Ethanol | SC-CO2 + Ethanol | Acetone Mix Methanol | Hexane | Ethanol | |
| E. coli | 18 ± 0.10 d | 21 ± 0.50 e | 15 ± 0.10 b | 16 ± 0.70 c | 21 ± 1.10 f | 28 ± 1.50 c | 18 ± 0.90 c | 19 ± 0.90 c |
| P. aeruginosa | N.D | N.D | N.D | N.D | N.D | N.D | N.D | N.D |
| S. typhimurium | N.D | N.D | N.D | N.D | N.D | N.D | N.D | N.D |
| L. monocytogenes | 12 ± 0.05 b | 15 ± 0.20 a | 8 ± 0.25 a | 9 ± 0.08 b | 14 ± 0.20 b | 24 ± 0.10 b | 8 ± 0.02 a | 10 ± 0.02 b |
| B. cereus | 10 ± 0.01 a | 16 ± 0.60 b | N.D | N.D | 12 ± 0.60 a | 17 ± 0.30 a | N.D | N.D |
| S. aureus | 18 ± 0.50 d | 20 ± 0.50 d | 8 ± 0.40 a | 6 ± 0.05 a | 20 ± 0.45 e | 24 ± 0.10 b | 10 ± 0.15 b | 7 ± 0.04 a |
| C. albicans | 12 ± 0.20 b | 18 ± 0.10 c | N.D | N.D | 16 ± 0.80 c | 24 ± 0.55 b | N.D | N.D |
| A. brasiliensis | 16 ± 0.00 c | 20 ± 0.10 d | N.D | N.D | 18 ± 0.50 d | 28 ± 0.85 c | N.D | N.D |
2.6. Antihypertensive Activity
| IC50 (μg/mL) | ||
|---|---|---|
| Solvent | S. japonica Extracts | S. horneri Extracts |
| SC-CO2 + ethanol | 0.89 ± 0.07 b | 0.97 ± 0.11 b |
| Acetone mix methanol | 1.05 ± 0.14 a | 1.28 ± 0.50 a |
| Hexane | 0.03 ± 0.01 c | 0.01 ± 0.00 c |
| Ethanol | 0.05 ± 0.01 c | 0.07 ± 0.01 c |
| Captopril | 0.08 ± 0.02 μg/mL | |
3. Experimental Section
3.1. Materials
3.2. Sample Preparation
3.3. Conventional Solvent Extraction
3.4. Supercritical Fluid Extraction with Carbon Dioxide
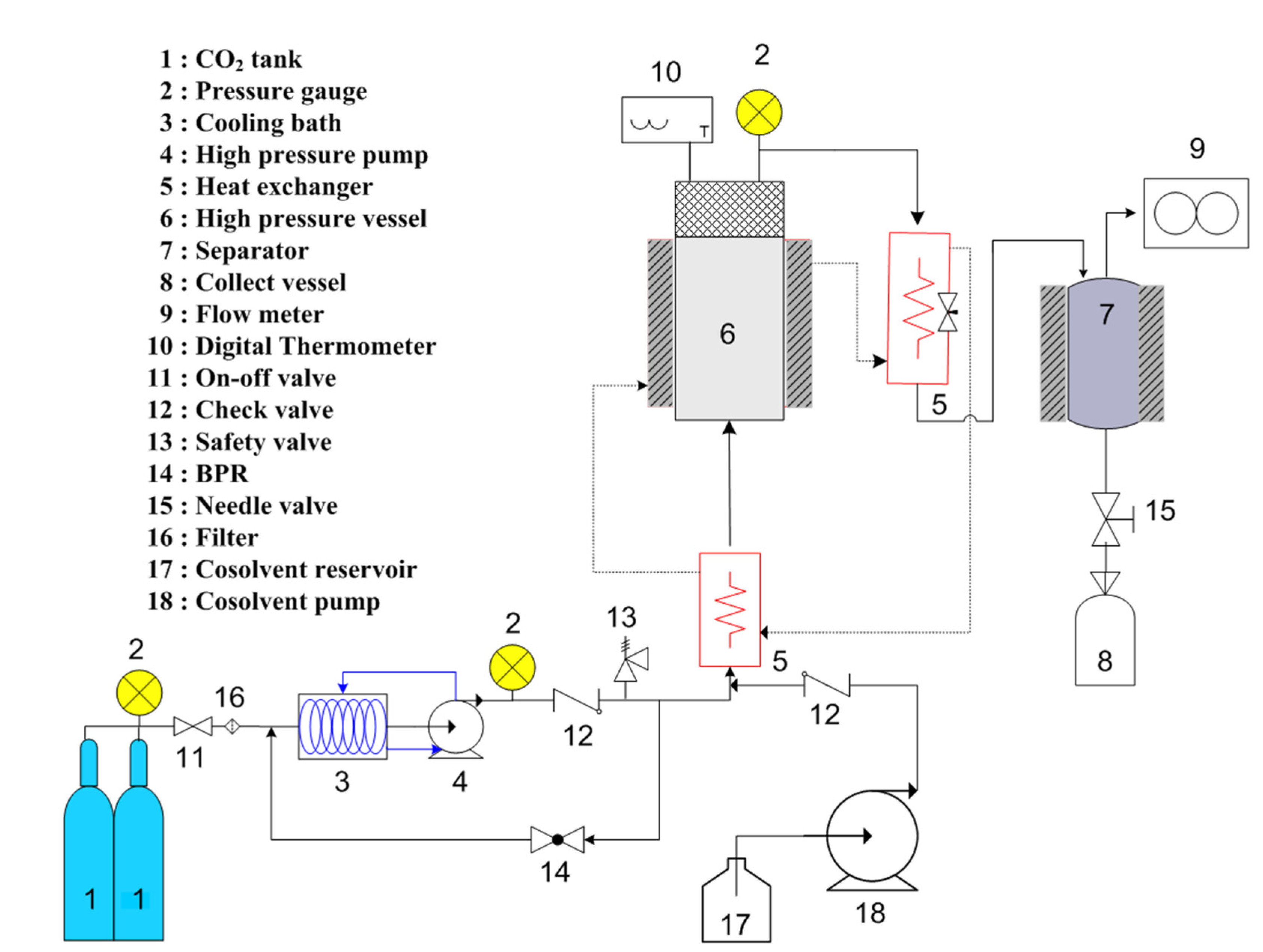
3.5. Fucoxanthin Analysis by HPLC
3.6. FA Composition Analysis
3.7. Antioxidant Activity Assessment
3.7.1. H2O2 Scavenging Assay
3.7.2. RP Method
3.7.3. β-Carotene Linoleic Acid Method/Conjugated Diene Assay
3.7.4. Total Antioxidant Activity
3.8. TPC Assay
3.9. Antimicrobial Activity
3.10. Determination of Antihypertensive Activity
3.11. Statistical Analysis
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miyashita, K.; Mikami, N.; Hosokawa, M. Chemical and nutritional characteristics of brown seaweed lipids: A review. J. Funct. Foods. 2013, 5, 1507–1517. [Google Scholar] [CrossRef]
- Rodriguez-Jasso, R.M.; Mussatto, S.I.; Pastrana, L.; Aguilar, C.N.; Teixeira, J.A. Microwave-assisted extraction of sulfated polysaccharides (fucoidan) from brown seaweed. Carbohydr. Polym. 2011, 86, 1137–1144. [Google Scholar] [CrossRef]
- Hong, D.; Hien, H.; Son, P. Seaweeds from Vietnam used for functional food, medicine and biofertilizer. J. Appl. Phycol. 2007, 19, 817–826. [Google Scholar] [CrossRef]
- Synytsya, A.; Kim, W.-J.; Kim, S.-M.; Pohl, R.; Synytsya, A.; Kvasnička, F.; Čopíková, J.; Park, Y.I. Structure and antitumour activity of fucoidan isolated from sporophyll of Korean brown seaweed Undaria pinnatifida. Carbohydr. Polym. 2010, 81, 41–48. [Google Scholar] [CrossRef]
- Wang, J.; Guo, H.; Zhang, J.; Wang, X.; Zhao, B.; Yao, J.; Wang, Y. Sulfated modification, characterization and structure antioxidant relationships of Artemisia sphaerocephala polysaccharides. Carbohydr. Polym. 2010, 81, 897–905. [Google Scholar] [CrossRef]
- Jang, S.-S.; Shirai, Y.; Uchida, M.; Wakisaka, M. Production of mono sugar from acid hydrolysis of seaweed. Afr. J. Biotechnol. 2014, 11, 1953–1963. [Google Scholar]
- Eom, S.-H.; Kim, Y.-M.; Kim, S.-K. Antimicrobial effect of phlorotannins from marine brown algae. Food Chem. Toxicol. 2012, 50, 3251–3255. [Google Scholar] [PubMed]
- Miyashita, K.; Nishikawa, S.; Beppu, F.; Tsukui, T.; Abe, M.; Hosokawa, M. The allenic carotenoid fucoxanthin, a novel marine nutraceutical from brown seaweeds. J. Sci. Food Agric. 2011, 91, 1166–1174. [Google Scholar] [CrossRef] [PubMed]
- Hosokawa, M.; Okada, T.; Mikami, N.; Konishi, I.; Miyashita, K. Bio-functions of marine carotenoids. Food Sci. Biotechnol. 2009, 18, 1–11. [Google Scholar]
- Quitain, A.T.; Kai, T.; Sasaki, M.; Goto, M. Supercritical carbon dioxide extraction of fucoxanthin from Undaria pinnatifida. J. Agric. Food Chem. 2013, 61, 5792–5797. [Google Scholar] [CrossRef] [PubMed]
- Athukorala, Y.; Jeon, Y.-J. Screening for angiotensin 1-converting enzyme inhibitory activity of Ecklonia cava. J. Food Sci. Nutr. 2005, 10, 134–139. [Google Scholar]
- Yoshiki, M.; Tsuge, K.; Tsuruta, Y.; Yoshimura, T.; Koganemaru, K.; Sumi, T.; Matsui, T.; Matsumoto, K. Production of new antioxidant compound from mycosporine-like amino acid, porphyra-334 by heat treatment. Food Chem. 2009, 113, 1127–1132. [Google Scholar] [CrossRef]
- Tierney, M.S.; Croft, A.K.; Hayes, M. A review of antihypertensive and antioxidant activities in macroalgae. Bot. Mar. 2010, 53, 387–408. [Google Scholar] [CrossRef]
- Lee, J.-H.; Chun, B.-S. Effect of antioxidant activity of mixture obtained from brown seaweed and wheat germ oils using different extraction methods. Food. Sci. Biotechnol. 2013, 22, 9–17. [Google Scholar] [CrossRef]
- Conde, E.; Moure, A.; Domínguez, H. Supercritical CO2 extraction of fatty acids, phenolics and fucoxanthin from freeze-dried Sargassum muticum. J. Appl. Phycol. 2014, 27, 957–964. [Google Scholar] [CrossRef]
- Terasaki, M.; Hirose, A.; Narayan, B.; Baba, Y.; Kawagoe, C.; Yasui, H.; Saga, N.; Hosokawa, M.; Miyashita, K. Evaluation of recoverable functional lipid components of several brown seaweeds (Phaeophyta) from Japan with special reference to fucoxanthin and fucosterol contents. J. Phycol. 2009, 45, 974–980. [Google Scholar] [CrossRef]
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Calder, P.C. Polyunsaturated fatty acids, inflammation, and immunity. Lipids 2001, 36, 1007–1024. [Google Scholar] [CrossRef] [PubMed]
- Alles, M.S.; Eussen, S.R.B.M.; van der Beek, E.M. Nutritional challenges and opportunities during the weaning period and in young childhood. Ann. Nutr. Metab. 2014, 64, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Erkkilä, A.T.; Lehto, S.; Pyörälä, K.; Uusitupa, M.I. n-3 Fatty acids and 5-y risks of death and cardiovascular disease events in patients with coronary artery disease. Am. J. Clin. Nutr. 2003, 78, 65–71. [Google Scholar] [PubMed]
- Airanthi, M.; Hosokawa, M.; Miyashita, K. Comparative antioxidant activity of edible Japanese brown seaweeds. J. Food Sci. 2011, 76, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, K.; Ozaki, Y.; Hashimoto, T.; Das, S.K.; Matsushita, S.; Hirano, M.; Okada, T.; Komoto, A.; Mori, N.; Nakatsuka, M. Commercial-scale preparation of biofunctional fucoxanthin from waste parts of brown sea algae Laminalia japonica. Food. Sci. Technol. Res. 2008, 14, 573–582. [Google Scholar] [CrossRef]
- Gupta, S.; Abu-Ghannam, N. Bioactive potential and possible health effects of edible brown seaweeds. Trends Food. Sci. Technol. 2011, 22, 315–326. [Google Scholar] [CrossRef]
- Athiperumalsami, T.; Rajeswari, V.D.; Poorna, S.H.; Kumar, V.; Jesudass, L.L. Antioxidant activity of seagrasses and seaweeds. Bot. Mar. 2010, 53, 251–257. [Google Scholar] [CrossRef]
- Kumaran, A.; Karunakaran, R.J. In vitro antioxidant activities of methanol extracts of five Phyllanthus species from India. LWT-Food. Sci. Technol. 2007, 40, 344–352. [Google Scholar] [CrossRef]
- Chandini, S.K.; Ganesan, P.; Bhaskar, N. In vitro antioxidant activities of three selected brown seaweeds of India. Food Chem. 2008, 107, 707–713. [Google Scholar] [CrossRef]
- Martins, C.D.L.; Ramlov, F.; Carneiro, N.P.N.; Gestinari, L.M.; dos Santos, B.F.; Bento, L.M.; Lhullier, C.; Gouvea, L.; Bastos, E.; Horta, P.A. Antioxidant properties and total phenolic contents of some tropical seaweeds of the Brazilian coast. J. Appl. Phycol. 2013, 25, 1179–1187. [Google Scholar] [CrossRef]
- Kelman, D.; Posner, E.K.; McDermid, K.J.; Tabandera, N.K.; Wright, P.R.; Wright, A.D. Antioxidant activity of Hawaiian marine algae. Mar. Drugs 2012, 10, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Chew, Y.L.; Lim, Y.Y.; Omar, M.; Khoo, K.S. Antioxidant activity of three edible seaweeds from two areas in South East Asia. LWT Food. Sci. Technol. 2008, 41, 1067–1072. [Google Scholar] [CrossRef]
- Yuan, Y.V.; Bone, D.E.; Carrington, M.F. Antioxidant activity of dulse (Palmaria palmata) extract evaluated in vitro. Food. Chem. 2005, 91, 485–494. [Google Scholar] [CrossRef]
- Luo, H.-Y.; Wang, B.; Yu, C.-G.; Qu, Y.; Su, C. Evaluation of antioxidant activities of five selected brown seaweeds from China. Med. Plants. Res. 2010, 4, 2557–2565. [Google Scholar]
- Zhang, W.-W.; Duan, X.-J.; Huang, H.-L.; Zhang, Y.; Wang, B.-G. Evaluation of 28 marine algae from the Qingdao coast for antioxidative capacity and determination of antioxidant efficiency and total phenolic content of fractions and subfractions derived from Symphyocladia latiuscula (Rhodomelaceae). J. Appl. Phycol. 2007, 19, 97–108. [Google Scholar] [CrossRef]
- De Quirós, A.R.-B.; Lage-Yusty, M.; López-Hernández, J. Determination of phenolic compounds in macroalgae for human consumption. Food. Chem. 2010, 121, 634–638. [Google Scholar]
- Zubia, M.; Fabre, M.S.; Kerjean, V.; Le Lann, K.; Stiger-Pouvreau, V.; Fauchon, M.; Deslandes, E. Antioxidant and antitumoural activities of some Phaeophyta from Brittany coasts. Food. Chem. 2009, 116, 693–701. [Google Scholar] [CrossRef]
- Jiménez-Escrig, A.; Gómez-Ordóñez, E.; Rupérez, P. Brown and red seaweeds as potential sources of antioxidant nutraceuticals. J. Appl. Phycol. 2012, 24, 1123–1132. [Google Scholar] [CrossRef]
- Wang, T.; Jonsdottir, R.; Ólafsdóttir, G. Total phenolic compounds, radical scavenging and metal chelation of extracts from Icelandic seaweeds. Food Chem. 2009, 116, 240–248. [Google Scholar] [CrossRef]
- Jo, J.-H.; Kim, D.; Lee, S.; Lee, T.K. Total phenolic contents and biological activities of Korean seaweed extracts. Food Sci. Biotechnol. 2005, 14, 798–802. [Google Scholar]
- Val, A.; Platas, G.; Basilio, A.; Cabello, A.; Gorrochategui, J.; Suay, I.; Vicente, F.; Portillo, E.; Río, M.; Reina, G. Screening of antimicrobial activities in red, green and brown macroalgae from Gran Canaria (Canary Islands, Spain). Int. Microbiol. 2001, 4, 35–40. [Google Scholar]
- Rajauria, G.; Jaiswal, A.K.; Abu-Gannam, N.; Gupta, S. Antimicrobial, antioxidant and free radical-scavenging capacity of brown seaweed Himanthalia elongata from western coast of Ireland. J. Food. Biochem. 2013, 37, 322–335. [Google Scholar] [CrossRef]
- Katsumi, I.; Akiko, K.; Hiroko, M.; Miyuki, W.; Hiroko, N.; Junko, H.; Takahisa, N. Effect of Undaria pinnatifida (Wakame) on the development of cardiovascular disease in strokeprone spontaneously hypertensive rats. Clin. Exp. Pharmacol. Physiol. 2003, 30, 44–48. [Google Scholar]
- Jung, H.A.; Hyun, S.K.; Kim, H.R.; Choi, J.S. Angiotensin-converting enzyme I inhibitory activity of phlorotannins from Ecklonia stolonifera. Fish. Sci. 2006, 72, 1292–1299. [Google Scholar] [CrossRef]
- Cha, S.; Lee, K.; Jeon, Y. Screening of extracts from red algae in Jeju for potentials marine angiotensin-I converting enzyme (ACE) inhibitory activity. Algae 2006, 21, 343. [Google Scholar] [CrossRef]
- Roh, M.-K.; Uddin, M.S.; Chun, B.-S. Extraction of fucoxanthin and polyphenol from Undaria pinnatifida using supercritical carbon dioxide with co-solvent. Biotechnol. Bioprocess Eng. 2008, 13, 724–729. [Google Scholar] [CrossRef]
- Lee, S.M.; Asaduzzaman, A.; Chun, B.S. Characterization of lecithin isolated from anchovy (Engraulis japonica) residues deoiled by supercritical carbon dioxide and organic solvent extraction. J. Food Sci. 2012, 77, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Firestone, D. Official Methods and Recommended Practices of the AOCS; AOCS: Urbana, IL, USA, 2009. [Google Scholar]
- Xia, S.; Gao, B.; Li, A.; Xiong, J.; Ao, Z.; Zhang, C. Preliminary characterization, antioxidant properties and production of chrysolaminarin from marine diatom Odontella aurita. Mar. Drugs 2014, 12, 4883–4897. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Hu, Z.; Fu, H.; Hu, M.; Xu, X.; Chen, J. Chemical analysis and antioxidant activity in vitro of a β-d-glucan isolated from Dictyophora indusiata. Int. J. Biol. Macromol. 2012, 51, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.-G.; Zhang, W.-W.; Duan, X.-J.; Li, X.-M. In vitro antioxidative activities of extract and semi-purified fractions of the marine red alga, Rhodomela confervoides (Rhodomelaceae). Food. Chem. 2009, 113, 1101–1105. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, K.; Huang, S.; Wang, H.; Mu, X.; He, C.; Ji, X.; Zhang, J.; Huang, F. Antioxidant activity of microwave-assisted extract of longan (Dimocarpus longan Lour.) peel. Food. Chem. 2008, 106, 1264–1270. [Google Scholar] [CrossRef]
- Li, H.-B.; Wong, C.-C.; Cheng, K.-W.; Chen, F. Antioxidant properties in vitro and total phenolic contents in methanol extracts from medicinal plants. LWT Food. Sci. Technol. 2008, 41, 385–390. [Google Scholar] [CrossRef]
- Meillisa, A.; Siahaan, E.A.; Park, J.-N.; Woo, H.-C.; Chun, B.-S. Effect of subcritical water hydrolysate in the brown seaweed Saccharina japonica as a potential antibacterial agent on food-borne pathogens. J. Appl. Phycol. 2013, 25, 763–769. [Google Scholar] [CrossRef]
- Ko, S.-C.; Kang, N.; Kim, E.-A.; Kang, M.C.; Lee, S.-H.; Kang, S.-M.; Lee, J.-B.; Jeon, B.-T.; Kim, S.-K.; Park, S.-J. A novel angiotensin I-converting enzyme (ACE) inhibitory peptide from a marine Chlorella ellipsoidea and its antihypertensive effect in spontaneously hypertensive rats. Process. Biochem. 2012, 47, 2005–2011. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sivagnanam, S.P.; Yin, S.; Choi, J.H.; Park, Y.B.; Woo, H.C.; Chun, B.S. Biological Properties of Fucoxanthin in Oil Recovered from Two Brown Seaweeds Using Supercritical CO2 Extraction. Mar. Drugs 2015, 13, 3422-3442. https://doi.org/10.3390/md13063422
Sivagnanam SP, Yin S, Choi JH, Park YB, Woo HC, Chun BS. Biological Properties of Fucoxanthin in Oil Recovered from Two Brown Seaweeds Using Supercritical CO2 Extraction. Marine Drugs. 2015; 13(6):3422-3442. https://doi.org/10.3390/md13063422
Chicago/Turabian StyleSivagnanam, Saravana Periaswamy, Shipeng Yin, Jae Hyung Choi, Yong Beom Park, Hee Chul Woo, and Byung Soo Chun. 2015. "Biological Properties of Fucoxanthin in Oil Recovered from Two Brown Seaweeds Using Supercritical CO2 Extraction" Marine Drugs 13, no. 6: 3422-3442. https://doi.org/10.3390/md13063422
APA StyleSivagnanam, S. P., Yin, S., Choi, J. H., Park, Y. B., Woo, H. C., & Chun, B. S. (2015). Biological Properties of Fucoxanthin in Oil Recovered from Two Brown Seaweeds Using Supercritical CO2 Extraction. Marine Drugs, 13(6), 3422-3442. https://doi.org/10.3390/md13063422







