Potential Threats Posed by Tetrodotoxins in UK Waters: Examination of Detection Methodology Used in Their Control
Abstract
:1. Introduction
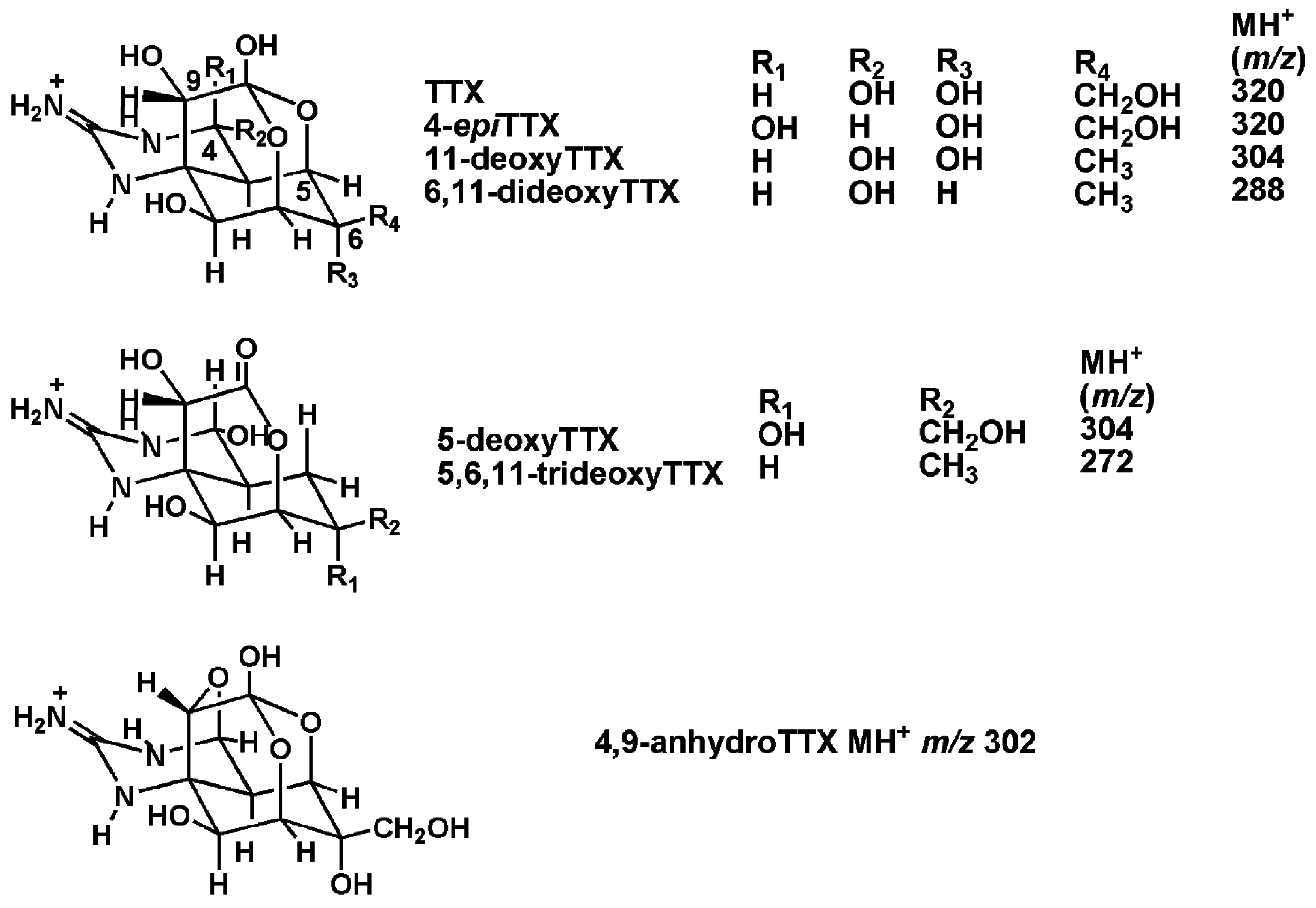
2. Prevalence of Tetrodotoxins
3. Tetrodotoxin Threats for UK Waters
4. Toxin Testing Methods
4.1. Animal Bioassays
4.2. Chemical Methods
4.2.1. Conventional Chromatography Methods
4.2.2. Mass Spectrometric Methods
4.3. Biomolecular Methods
4.3.1. Cytotoxicity Assay
4.3.2. Receptor Binding Assays
4.3.3. Immunoassays
4.3.4. Biosensor Methods
| Method | Advantages | Disadvantages |
|---|---|---|
| Mouse bioassay (MBA) |
|
|
| Cytotoxicity assay |
|
|
| Receptor binding assay (RBA) |
|
|
| ELISA |
|
|
| Conventional chromatography |
|
|
| LC-MS(MS) |
|
|
| Biosensor methods |
|
|
5. Suitability of Toxin Testing Methods
5.1. Suitability of Existing and Potential Methods for TTX Testing
- Given the detection of TTXs in two marine sites within Southern England, to conduct retrospective and ongoing testing of bivalves throughout the UK to assess the occurrence and prevalence of TTXs.
- To assess the performance of commercial TTX ELISA and/or any other suitable immunoassay available in kit form for applicability to samples as a screening test for end-product testing.
- To continue the validation of the SPR method (developed at Queens University Belfast and validated at the Agri-Foods and Biosciences Institute Northern Ireland (AFBINI) as part of the Interreg project ATLANTOX) for TTX in all species of relevance.
- To extend the validation of the PSP and TTX LC-MS/MS method to external laboratories, generating performance data through collaborative study.
- To conduct a UK-wide assessment of the above methods on a range of suitable samples and to make subsequent recommendations on performance and applicability.
5.2. Identification of Knowledge Gaps Which Might be Addressed Through Further Research or Method Development
- Identification of sources of TTXs in UK waters, both those present now and potentially in the future
- Ongoing analysis of bacterial cultures by suitable methods for assessment of presence of TTXs in water samples
- The determination of marine species of relevance that may accumulate TTXs and associated depuration rates
- The determination of specific TTX profile studies in relevant species
- Relationship of toxicity to specific fish species and fish size
- Evaluation of MBA-replacement screening methods, in particular the commercial ELISA and SPR biosensor
- Develop understanding of TTX and TTX metabolites toxicity in relation to human exposure, including long-term assessment of intoxicated people to determine potential long-term affects
- Interlaboratory validation of LC-MS/MS TTX method for applicability to samples of relevance to the UK
6. Conclusions: Proposed Options for Routine Monitoring to Meet Legal Requirements
- Analysis of bacterial cultures and contaminated marine organisms for the continued identification of sources of TTXs in UK waters
- Evaluation of MBA-replacement screening methods, in particular the commercial ELISA and SPR biosensor
- Interlaboratory validation of the quantitative confirmatory LC-MS/MS method for applicability to samples of relevance for the UK
Acknowledgments
Conflicts of Interest
References
- Isbister, G.K.; Kiernan, M.C. Neurotoxic marine poisoning. Lancet Neurol. 2005, 4, 219–228. [Google Scholar] [CrossRef]
- Arakawa, O.; Hwang, D.-F.; Taniyama, S.; Takatani, T. Toxins of pufferfish that cause human intoxications. Coast. Environ. Ecosyst. Issues East China Sea 2010, 1, 227–244. [Google Scholar]
- Itoi, S.; Yoshimkawa, S.; Asahina, K.; Suzuki, M.; Ishizuka, K.; Takimoto, N.; Mitsuoka, R.; Naoto, Y.; Detake, A.; Takayanagi, C.; et al. Larval pufferfish protected by maternal tetrodotoxin. Toxicon 78, 35–40. [CrossRef] [PubMed]
- Yasumoto, T.; Endo, A.; Yasumura, D.; Nagai, H.; Murata, M.; Yotsu, M. Bacterial production of Tetrodotoxin and its derivatives. Mar. Toxin Prog. Venom Toxin Res. 1987, 26, 310–313. [Google Scholar]
- Wu, Z.; Xie, L.; Xia, G.; Zhang, J.; Nie, Y.; Hu, J.; Wang, S.; Zhang, A. A new tetrodotoxin-producing actinomycete, Norcardiopsis dassonvillei, isolated from the ovaries of puffer fish Fugu rubripes. Toxicon 2005, 45, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-J.; Yu, R.-C.; Luo, X.; Zhou, M.-J.; Lin, X.-T. Toxin-screening and identification of bacteria from highly toxic marine gastropod Nassarius semiplicatus. Toxicon 2008, 52, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Arakawa, O. Tetrodotoxin—Distribution and accumulation in aquatic organisms, and cases of human intoxication. Mar. Drugs 2008, 6, 220–242. [Google Scholar] [CrossRef] [PubMed]
- Vlamis, A.; Katikou, P.; Rodriguez, I.; Rey, V.; Alfonso, A.; Papazachariou, A.; Zacharaki, T.; Botana, M.A.; Botana, M.L. First detection of Tetrodotoxin in Greek shellfish by UPLC-MS/MS potentially linked to the presence of the dinoflagellate Prorocentrum minimum. Toxins 2015, 7, 1779–1807. [Google Scholar] [CrossRef] [PubMed]
- Chau, R.; Kalaitzis, A.J.; Neilan, A.B. On the origin and biosynthesis of tetrodotoxin. Aqutic Toxicol. 2011, 104, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Khor, S.; Wood, S.A.; Salvitti, L.; Taylor, D.I.; Adamson, J.; McNabb, P.; Cary, S.C. Investigating diet as the source of tetrodotoxin in Pleurobranchaea maculate. Mar. Drugs 2014, 12, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Alcaraz, A.; Whiplle, R.E.; Gregg, H.R.; Anderson, B.D.; Grant, P.M. Analysis of Tetrodotoxin. Forensic Sci. Int. 1999, 99, 35–45. [Google Scholar] [CrossRef]
- Noguchi, T.; Miyazawa, K.; Daigo, K.; Arakawa, O. Paralytic shellfish poisoning (PSP) toxin–And/or Tetrodotoxin-contaminated crabs and food poisoning by them. Toxin Rev. 2011, 30, 91–102. [Google Scholar] [CrossRef]
- Yasumoto, T. Historic Considerations Regarding Seafood Safety. In Seafood and Freshwater Toxins: Pharmacology, Physiology and Detection; Botana, L.M., Ed.; Dekker: New York, NY, USA, 2000; pp. 1–17. [Google Scholar]
- Yu, C.F.; Yu, P.H.F.; Chan, P.L.; Yan, Q.; Wong, P.K. Two novel species of tetrodotoxin-producing bacteria isolated from toxic marine puffer fishes. Toxicon 2004, 44, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Teramoto, N.; Yotsu-Yamashita, M. Selective blocking effects of 4,9-anhydrotetrodotoxin, purified from a crude mixture of tetrodotoxin analogues, on NaV1.6 channels and its chemical aspects. Mar. Drugs 2015, 13, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Jang, J-H.; Lee, J-S.; Totsu-Yamashita, M. LC/MS analysis of Tetrodotoxin and its deoxy analogs in the marine puffer fish Fugu niphobles from the Southern coast of Korea, and in the brackish water puffer fishes Tetraodon nigroviridis and Tetraodon biocellatus from Southeast Asia. Mar. Drugs 2010, 8, 1049–1058. [Google Scholar]
- Chulanetra, M.; Sookrung, N.; Srimanote, P.; Indrawattana, N.; Thanongsaksrikul, J.; Sakolvaree, Y.; Congsa-Nguan, M.; Kurazono, H.; Chaicumpa, W. Toxic marine puffer fish in Thailand Seas and Tetrodotoxin they contained. Toxins 2011, 3, 1249–1262. [Google Scholar] [CrossRef] [PubMed]
- Leung, K.S-Y.; Fong, B.M-W.; Tsoi, Y-K. Analytical challenges: Determination of tetrodotoxin in human urine and plasma by LC-MS/MS. Mar. Drugs 2011, 9, 2291–2303. [Google Scholar] [CrossRef] [PubMed]
- Jen, H.-C.; Lin, S.-J.; Lin, S.-Y.; Huang, I.Y.W.; Liao, I.C.; Arakawa, O.; Hwang, D.F. Occurrence of tetrodotoxin and paralytic shellfish poisons in a gastropod implicated in food poisoning in southern Taiwan. Food Addit. Contam. 2007, 24, 902–909. [Google Scholar] [CrossRef] [PubMed]
- Jen, H.-C.; Lin, S.-J.; Tsai, Y.-H.; Chen., C.-H.; Lin, Z.-C.; Hwang, D.F. Tetrodotoxin poisoning evidenced by solid-phase extraction combining with liquid chromatography-tandem-mass spectrometry. J. Chrom. A 2008, 871, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Jen, H.-C.; Nguyen, T.A.-T.; Wu, Y.-J.; Hoang, T.; Arakawa, O.; Lin, W.-F.; Hwang, D.-F. Tetrodotoxin and paralytic shellfish poisons in gastropod species from Vietnam analysed by high performance liquid chromatography-tandem mass spectrometry. J. Food Drug Anal. 2014, 22, 178–188. [Google Scholar] [CrossRef]
- Sato, S.; Ogata, T.; Kodama, M. Trace amounts of saxitoxins in the viscera of chum salmon Oncorhynchus keta. Mar. Ecol. Prog. Ser. 1998, 175, 295–298. [Google Scholar] [CrossRef]
- You, J.; Yue, Y.; Xing, F.; Xia, W.; Lai, S.; Zhang, F. Tetrodotoxin poisoning caused by Goby fish consumption in southeast China: A retrospective case series analysis. Clinics 2015, 70, 24–29. [Google Scholar] [CrossRef]
- Hwang, D.F.; Noguchi, T. Tetrodotoxin poisoning. Adv. Food Nutr. Res. 2007, 52, 141–236. [Google Scholar] [PubMed]
- Hwang, P.-A.; Tsai, Y.-H.; Lin, H.-P.; Hwang, D.-F. Tetrodotoxin-binding proteins isolated from five species of toxic gastropods. Food Chem. 2007, 103, 1153–1158. [Google Scholar] [CrossRef]
- Hwang, P.-A.; Tsai, Y.-H.; Lin, H.-P.; Hwang, D.-F. The gastropod possessing TTX and/or PSP. Food Rev. Int. 2007, 23, 321–340. [Google Scholar] [CrossRef]
- McNabb, P.; Selwood, A.I.; Munday, R.; Wood, S.A.; Taylor, D.I.; MacKenzie, L.A.; van Ginkel, R.; Rhodes, L.L.; Cornelisen, C.; Heasman, K.; et al. Detection of Tetrodotoxin from the grey side-gilled sea slug—Pleurobranchaea maculate, and associated dog neurotoxicosis on beaches adjacent to the Hauraki Gulf, Auckland, New Zealand. Toxicon 2010, 56, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Oglivie, S.; Taylor, D.; McNabb, P.; Hamon, D.; Nathan, P.; Anderson, A. Tetrodotoxin in Kaimoana: Science and Matauranga Mitigating Health Risks from a Lethal Neurotoxin; Report No. 2219; Cawthron Institute: Auckland, New Zealand, August 2012. [Google Scholar]
- Wood, S.A.; Taylor, D.I.; McNabb, P.; Walker, J.; Adamson, J.; Cary, S.C. Tetrodotoxin concentrations in Pleurobranchaea manculata: Temporal, Spatial and Individual variability from New Zealand populations. Mar. Drugs 2012, 10, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Salvitti, L.; Wood, S.A.; Taylor, D.I.; McNabb, P.; Cary, S.C. First identification of tetrodotoxin in the flatworm Stylochoplana sp.; a source of TTX for the sea slug Pleurobranchaea maculate. Toxicon 2015, 95, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Farias, N.E.; Obernat, S.; Goya, A.B. Outbreak of a nuerotoxic side-gilled sea slug (Pleurobranchaea sp.) in Argentinean coasts. N. Z. J. Zool. 2015, 2015, 1–6. [Google Scholar]
- Kodama, M.; Sato, S.; Ogata, T. Alexandrium tamarense as a source of Tetrodotoxin in the scallop Patinopecten yessoensis. Toxic Phytoplankton Blooms in the Sea. In Proceeding of the 5th International Conference on Toxic Marine Phytoplankton, Newport, RI, USA, 28 October–1 November 1991; Smayda, T.J., Shimizu, Y., Eds.; Elsevier: Amsterdam, The Netherlands; pp. 401–406.
- McNabb, P.S.; Taylor, D.I.; Ogilvie, S.C.; Wilkinson, L.; Anderson, A.; Hamon, D.; Wood, S.A.; Peake, B.M. First detection of Tetrodotoxin in the bivalve Paphies autralis by liquid chromatography coupled to triple quadrupole mass spectrometry with and without pre-column reaction. J. AOAC Int. 2014, 97, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Bentur, Y.; Ashkar, J.; Lurie, Y.; Levy, Y.; Azzam, Z.S.; Litmanovich, M. Lessepsian migration and tetrodotoxin poisoning due to Lagocephalus sceleratus in the Eastern Mediterranean. Toxicon 2008, 52, 964–968. [Google Scholar] [CrossRef] [PubMed]
- Katikou, P.; Georgantelis, D.; Sinouris, N.; Petsi, A.; Fotaras, T. First report on toxicity assessment of the Lessepesian migrant pufferfish Lagocephalus scleratus (Gmelin, 1789) from European waters (Aegean Sea, Greece). Toxicon 2009, 54, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Nader, M.; Indary, S.; Boustany, L. The Puffer Fish Lagocephalus Sceleratus (Gmelin, 1789) in the Eastern Mediterranean; GCP/INT/041/EC-GRE-ITA/TD-10; FAO EastMed: Athens, Greece, 2012; p. 39. [Google Scholar]
- El Masry, S.K.; Fawzi, M.M. Tetrodotoxin versus ciguatera fish poisoning in the Mediterranean. Ind. J. Forensic Med. Toxicol. 2011, 5, 50–53. [Google Scholar]
- Jribi, I.; Bradai, N. First record of the Lessepsian migrant species Lagocephanus sceleratus (Gmelin, 1789) (Actinopterygii: Tetraodontidae) in the Central Mediterranean. BioInvasions Rec. 2012, 1, 49–52. [Google Scholar] [CrossRef]
- Silva, M.; Azavedo, J.; Rodriguez, P.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. New gastropod vectors and tetrodotoxin potential expansion in temperate waters of the Atlantic Ocean. Mar. Drugs 2012, 10, 712–726. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.; Alfonso, A.; Vale, C.; Alfonso, C.; Vale, P.; Tellez, A.; Botana, L.M. First toxicity report of tetrodotoxin and 5,6,11-TrideoxyTTX in the Trumpet Shell Charonia lampas lampas in Europe. Anal. Chem. 2008, 80, 5622–5629. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Ortega, J.F.; Morales-de los Santos, J.M.; Herrera-Gutierrez, M.E.; Fernandez-Sanchez, V.; Rodriguez Loureo, P.; Rancano, A.A.; Tellez-Andrade, A. Seafood intoxication by tetrodotoxin: First case in Europe. J. Emerg. Med. 2010, 39, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Baptista, M.; Silva, M.; Sabour, B.; Hassouani, M.; Barreiro, A.; Otero, P.; Azevedo, J.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. Emergent Marine Toxins in the Temperate North Atlantic Coast. In Proceeding of the ICES Annual Science Conference, A Coruna, Spain, 15–19 September 2014; Available online: https://www.researchgate.net/publication/272499301_Emergent_marine_toxins_in_the_temperate_north_Atlantic_coast (accessed on 4 September 2015).
- Kodama, M.; Sato, S.; Sakamoto, S.; Ogata, T. Occurrence of Tetrodotoxin in Alexandirum tamarense, a causative dinoflagellate of paralytic shellfish poisoning. Toxicon 1996, 34, 1101–1105. [Google Scholar] [CrossRef]
- Hess, P. Phytoplankton and Biotoxin Monitoring Programs for the Safe Exploitation of Shellfish in Europe. In New Trends in Marine and Freshwater Toxins; Cabado, A.G., Vietes, A.G., Eds.; Nova Science: New York, NY, USA, 2012; pp. 347–377. [Google Scholar]
- Bane, V.; Lehane, M.; Dikshit, M.; O’Riordan, A.; Furey, A. Tetrodotoxin: Chemistry, toxicity, source, distribution and detection. Toxins 2014, 6, 693–755. [Google Scholar] [CrossRef] [PubMed]
- Peck, I. Want to Eat the World’s Most Deadly Dish? Join the Club. Available online: http://www.independent.co.uk/life-style/food-and-drink/news/want-to-eat-the-worlds-most-deadly-dish-join-the-club-2292393.html (accessed on 10 December 2015).
- Cohen, N.J.; Deeds, J.R.; Wong, E.S.; Hanner, R.H.; Yancy, H.F.; White, K.D.; Thompson, T.M.; Wahl, M.; Pham, T.D.; Guichard, F.M.; et al. Public health response to puffer fish (Tetrodotoxin) poisoning from mislabelled product. J. Food Prot. 2009, 72, 810–817. [Google Scholar] [PubMed]
- Salvitti, L.; Wood, S.A.; McNabb, P.; Cary, S.C. No evidence for a culturable bacterial Tetrodotoxin producer in Pleurobranchaea maculata (Gastropoda: Pleurobranchidae) and Stylochoplana sp. (Platyhelminthes: Polycladida). Toxins 2015, 7, 255–273. [Google Scholar] [CrossRef] [PubMed]
- Lowe, J.A.; Howard, T.; Pardaens, A.; Tinker, J.; Jenkins, G.; Ridley, J.; Leake, J.; Holt, J.; Wakelin, S.; Wolf, J.; et al. UK Climate Projections Science Report: Marine and Coastal Projections; DEFRA: London, UK, 2009. Available online: http://ukclimateprojections.metoffice.gov.uk/22530 (accessed on 25 September 2015).
- Turner, A.D.; Powell, A.; Schofield, A.; Lees, D.N.; Baker-Austin, C. Detection of the pufferfish toxin Tetrodotoxin in European bivalves, England, 2013 to 2014. Eurosurveillance 2015, 20. [Google Scholar] [CrossRef]
- Powell, A.; Baker-Austin, C.; Wagley, S.; Bayley, A.; Hartnell, R. Isolation of Pandemic Vibrio parahaemolyticus from UK Water and Shellfish Produce. Microb. Ecol. 2013, 65, 924–927. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, K.; Ikuma, S.; Kawamura, R.; Tachikawa, K.; Sakai, C.; Tamura, C.; Amakasu, D. Tetrodotoxin. VII. On the structures of Tetrodotoxin and its derivatives. Chem. Pharm. Bull. 1964, 12, 1357–1374. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, T. Assay Method for Tetrodotoxin. In Food Hygiene Examination Manual, Vol. II; Japan Food Hygiene Association: Tokyo, Japan, 1978; pp. 232–240. [Google Scholar]
- Yasumoto, T. Manual for Methods for Food Sanitation Tests, Vol for Chemistry, Bureau for Environmental Health, Ministry for Health and Welfare; Japan Food Hygenic Association: Tokyo, Japan, 1991; p. 232. [Google Scholar]
- Hungerford, J.M. Committee on Natural Toxins and Food Allergens: Marine and Freshwater Toxins. General Referee Reports. J. AOAC Int. 2006, 89, 248–269. [Google Scholar] [PubMed]
- Sato, S.; Ogata, T.; Borja, V.; Gonzales, C.; Fukuyo, Y.; Kodama, M. Frequent occurrence of paralytic shellfish poisoning toxins as dominant toxins in marine puffer from tropical water. Toxicon 2000, 38, 1101–1109. [Google Scholar] [CrossRef]
- Hwang, D.-F.; Cheng, C.-A.; Chen, H.-C.; Jeng, S.-S.; Noguchi, T.; Ohwada, K.; Hashimoto, K. Microflora and Tetrodotoxin-producing bacteria in the lined moon shell Natica lineata. Fish. Sci. 1994, 60, 567–571. [Google Scholar]
- Wang, J.W.; Wang, D.B.; Luo, X.Y.; Ji, R.; Zhang, J.; Cao, M.H. Preparation and characterisation of monoclonal antibodies to Tetrodotoxin. J. Hyg. Res. 1996, 25, 308–311. [Google Scholar]
- Asakawa, M.; Shida, Y.; Miyazawa, K.; Noguchi, T. Instrumental Analysis of Tetrodotoxin. In Chromatography—The Most Versatile Method of Chemical Analysis; InTech Open Science: Rijeka, Croatia, 2012; Available online: http://cdn.intechopen.com/pdfs/40373/InTech-Instrumental_analysis_of_tetrodotoxin.pdf (accessed on 10 December 2015).
- Yasumoto, T.; Nakamura, M.; Oshima, Y.; Takahata, J. Construction of a continuous tetrodotoxins analyser. Bull. Jpn. Soc. Sci. Fish. 1982, 48, 1481–1483. [Google Scholar] [CrossRef]
- Yasumoto, T.; Michishita, T. Fluorometric determination of tetrodotoxin by high performance liquid chromatography. Agric. Biol. Chem. 1985, 49, 3077–3080. [Google Scholar] [CrossRef]
- Yotsu, M.; Endo, A.; Yasumoto, T. An improved Tetrodotoxin analyser. Agric. Biol. Chem. 1989, 53, 893. [Google Scholar] [CrossRef]
- Mebs, D.; Yotsu-Yamashita, M.; Yasumoto, T.; Lotters, S.; Schluter, A. Further report of the occurrence of tetrodotoxin in Atelopus species (Family: Bufonidae). Toxicon 1995, 33, 246–249. [Google Scholar] [CrossRef]
- Chen, X.-W.; Liu, H.-X.; Jin, Y.-B.; Li, S.-F.; Bi, X.; Chung, S.; Zhang, S.-S.; Jang, Y.-Y. Separation, identification and quantification of Tetrodotoxin and its analogs by LC-MS without calibration of individual analogs. Toxicon 2011, 57, 938–943. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, C.T.; Yotsu-Yamashita, M.; Yasumoto, T.; Brodie, E.D.; Brodie, E.D. Toxicity of dangerous prey: Variation of tetrodotoxin levels within and among populations of the newt Taricha granulosa. J. Chem. Ecol. 1999, 25, 2161–2173. [Google Scholar] [CrossRef]
- Yotsu-Yamashita, M.; Mebs, D. Occurrence of 11-oxotetrodotoxin in the red-spotted newt, Notophthalmus viridescens, and further studies on the levels of tetrodotoxin and its analogues in the newt’s efts. Toxicon 2003, 41, 893–897. [Google Scholar] [CrossRef]
- Yotsu-Yamashita, M.; Gilhen, J.; Russell, R.W.; Krysko, K.L.; Melaun, C.; Kurz, A.; Kauferstein, S.; Kordis, D.; Mebs, D. Variability of tetrodotoxin and of its analogues in the red-spotted newt, Notophthalmus viridescens (Amphibia: Urodela: Salamandridae). Toxicon 2012, 59, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Kawatsu, K.; Shibata, T.; Hamano, Y. Application of immunoaffinity chromatography for detection of tetrodotoxin from urine samples of poisoned patients. Toxicon 1999, 37, 325–333. [Google Scholar] [CrossRef]
- O’Leary, M.A.; Schneider, J.J.; Isbister, G.K. Use of high performance liquid chromatography to measure tetrodotoxin in serum and urine of poisoned patients. Toxicon 2004, 44, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.H.; Yu, C.F.; Tam, S.; Yu, P.H.F. Rapid screening of tetrodotoxin in urine and plasma of patients with pufferfish poisoning by HPLC with creatinine correction. Food Addit. Contam. 2010, 27, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Suenaga, K.; Kotoku, S. Detection of Tetrodotoxin in autopsy material by gas chromatography. Arch. Toxicol. 1980, 44, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.; Ma, Y.J.; Yan, J.D.; Tao, W.U. Determination of Tetrodotoxin in swell by capillary zone electrophoresis. Chin. J. Biochem. Pharm. 2003, 24, 176–178. [Google Scholar]
- Shoji, Y.; Yotsu-Yamashita, M.; Miyazawa, T.; Yasumoto, T. Electrospray ionisation mass spectrometry of Tetrodotoxin and its analogs: Liquid chromatography/mass soectrometry, tandem mass spectrometry, and liquid chromatography/tandem mass spectrometry. Anal. Biochem. 2001, 290, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, Y.; Nishio, S.; Noguchi, T.; Arakawa, O.; Kanoh, S.; Hashimoto, K. Detection of Tetrodotoxin by thin-layer chromatography/fast atom bombardment mass spectrometry. Anal. Biochem. 1988, 175, 258–262. [Google Scholar] [CrossRef]
- Pleasance, S.; Ayer, S.W.; Laycoc, M.V.; Thibault, P. Ionspray mass spectrometry of marine toxins. III. Analysis of paralytic shellfish poisoning toxins by flow-injection analysis, liquid chromatography/mass spectrometry and capillary electrophoresis/mass spectrometry. Rapid Commun. Mass Spectrom. 1992, 6, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Nishibori, N.; Nishio, S. Purification of Tetrodotoxin-like substances in the xanthid crab Atergatis floridus collected from Asakawa Bay, Tokushima. Bull. Shikoku Univ. 1994, 2, 93–100. [Google Scholar]
- Diener, M.; Christian, B.; Ahmed, M.S.; Luckas, B. Determination of Tetrodotoxin and its analogs in the puffer fish Takifugu oblongus from Bangladesh by hydrophilic interaction chromatography and mass-spectrometric detection. Anal. Bioanal. Chem. 2007, 389, 1997–2002. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, T.; Jang, J.; Yotsu-Yamashita, M. Hydrophilic interaction liquid chromatography electrospray ionisation mass spectrometry of Tetrodotoxin and its analogs. Anal. Biochem. 2006, 352, 142–144. [Google Scholar] [CrossRef] [PubMed]
- Horie, M.; Kabayashi, S.; Shimizu, S.; Nakazawa, H. Determination of tetrodotoxin in Pufferfish by liquid chromatography electrospray ionization mass spectrophotometry. Analyst 2002, 127, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Thattiyaphong, A.; Unahalekhaka, J.; Mekha, N.; Nispa, W.; Kluengklangdon, P.; Rojanapantip, L. Efficiency of a rapid test for detection of Tetrodotoxin in Puffer fish. J. Immunoassay. Immunochem. 2014, 35, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Yotsu-Yamashita, M.; Jang, J.-H.; Cho, Y.; Konoki, K. Optimisation of simultaneous analysis of Tetrodotoxin, 4-epitetrodotoxin, 4,9-anhdrotetrodotoxin, and 5, 6, 11-trideoxytetrodotoxin by hydrophilic interaction liquid chromatography-tandem mass spectrometry. Forensic Toxicol. 2011, 29, 61–64. [Google Scholar] [CrossRef]
- Jang, J.; Yotsu-Yamashita, M. Distribution of Tetrodotoxin, saxitoxin and their analogs among the tissues of the puffer fish Fugu pardalis. Toxicon 2006, 48, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Salvitti, L.; Wood, S.A.; Winsor, L.; Cary, S.C. Intracellular immunohistochemical detection of Tetrodotoxin in Pleurobranchaea maculate (Gastropoda) and Stylochoplana sp. (Turbellaria). Mar. Drugs 2015, 13, 756–769. [Google Scholar] [CrossRef] [PubMed]
- Boundy, M.J.; Selwood, A.I.; Harwood, D.T.; McNabb, P.S.; Turner, A.D. Development of a sensitive and selective liquid chromatography-mass spectrometry method for high throughput analysis of paralytic shellfish toxins using graphitized carbon solid phase extraction. J. Chromatogr. A 2015, 1387. [Google Scholar] [CrossRef] [PubMed]
- Deeds, J.R.; White, K.D.; Etheridge, S.M.; Landsberg, J.H. Concentrations of saxitoxin and Tetrodotoxin in three species of puffers from the Indian River Lagoon, Florida, the location for multiple cases of saxitoxin puffer poisoning from 2002 to 2004. Trans. Am. Fish. Soc. 2008, 137, 1317–1326. [Google Scholar] [CrossRef]
- Yotsu-Yamashita, M.; Abe, Y.; Kudo, Y.; Ritson-Williams, R.; Paul, V.J.; Konoki, K.; Cho, Y.; Adachi, M.; Imazu, T.; Nishikawa, T.; Isobe, M. First identification of 5,11-dideoxytetrodotoxin in marine animals and characterization of major fragment ions of tetrodotoxin and its analogs by high resolution ESI-MS/MS. Mar. Drugs 2013, 11, 2799–2813. [Google Scholar] [CrossRef] [PubMed]
- Akaki, K. Determination of Tetrodotoxin in puffer-fish tissues, and in serum and urine of intoxicated humans by liquid chromatography with tandem mass spectrometry. J. Food Hyg. Soc. Jpn. 2006, 47, 46–50. [Google Scholar] [CrossRef]
- Tsai, Y.H.; Hwang, D.F.; Cheng, C.A.; Hwang, C.C.; Deng, J.F. Determination of Tetrodotoxin in human urine and blood using C18 cartridge column, ultrailtration and LC-MS. J. Chromatogr. A 2006, 832, 75–80. [Google Scholar]
- Hayashida, M.; Hayakawa, H.; Wada, K.; Yamada, T.; Nihira, M.; Ohno, Y. A column-switching LC/MS/ESI method for detecting Tetrodotoxin and Aconitum alkaloids in serum. Legal Med. 2003, 5, S101–S104. [Google Scholar] [CrossRef]
- Saito, T.; Miura, N.; Namera, A.; Ota, S.; Miyazaki, S.; Inokuchi, S. A rapid sample preparation procedure using monospin CBA and amide columns for tetrodotoxin detection in serum and urine using LC-MS/MS analysis. Chromatographia 2014, 77, 687–693. [Google Scholar] [CrossRef]
- Kogure, K.; Tamplin, M.L.; Simidu, Y.; Colwell, R. A tissue culture assay for tetrodotoxin, saxi-toxin and related toxins. Toxicon 1988, 26, 191–197. [Google Scholar] [CrossRef]
- Hamasaki, K.; Kogure, K.; Ohwada, K. A biological method for the quantitative measurement of Tetrodotoxin (TTX): Tissue culture bioassay in combination with a water-soluble tetrazolium salt. Toxicon 1996, 4, 490–495. [Google Scholar] [CrossRef]
- Hamasaki, K.; Kogure, K.; Ohwada, K. An improved method of tissue culture bioassay for Tetrodotoxin. Fish. Sci. 1996, 62, 825–829. [Google Scholar]
- Davio, S.R.; Fontelo, P.A. A competitive displacement assay to detect saxitoxin and Tetrodotoxin. Anal. Biochem. 1984, 141, 199–204. [Google Scholar] [CrossRef]
- Doucette, G.J.; Powell, C.L.; Do, E.U.; Byon, C.Y.; Cleves, F.; McClain, S.G. Evaluation of 11-[3H]-tetrodotoxin use in a heterologous receptor binding assay for PSP toxins. Toxicon 2000, 38, 1465–1474. [Google Scholar] [CrossRef]
- Doucette, G.J.; Logan, M.M.; Ramsdell, J.S.; van Dolah, F.M. Development and preliminary validation of a microtiter plate-based receptor binding assay for paralytic shellfish poison toxins. Toxicon 1997, 35, 625–636. [Google Scholar] [CrossRef]
- Yasumoto, T.; (Japan Food Research Laboratories, Tokyo, Japan). Personal communication, 2013.
- Raybould, T.J.G.; Bignami, G.S.; Inouye, L.K.; Simpson, S.B.; Byrnes, J.B.; Grothaus, P.G.; Vann, D.C. A monoclonal antibody-based immunoassay for detecting Tetrodotoxin in biological samples. J. Clin. Lab. Anal. 1992, 6, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Neagu, D.; MIcheli, L.; Palleschi, G. Study of a toxin-alkaline phosphatase conjugate for the development of an immunosensor for Tetrodotoxin determination. Anal. Bioanal. Chem. 2006, 385, 1068–1074. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, Y.-S.; Pan, F.-G.; Liu, Z.-S.; Wang, Z. The development and optimisation of ELISA for the determination of Tetrodotoxin. J. Med. Coll. PLA 2007, 22, 347–351. [Google Scholar] [CrossRef]
- Kawatsu, K.; Hamano, Y.; Yoda, T.; Terano, Y.; Shibata, T. Rapid and highly sensitive enzyme immunoassay for quantitative determination of Tetrodotoxin. Jpn. J. Med. Sci. Biol. 1997, 50, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Wei, W.J.; Nan, L.; Lei, L.H.; Hui, H.C.; Fen, G.X.; Jun, L.Y.; Jing, Z.; Rong, J. Development of competitive indirect ELISA for the detection of Tetrodotoxin and a survey of the distribution of Tetrodotoxin in the tissues of wild puffer fish in the waters of southeast China. Food Addit. Contam 2010, 27, 1589–1597. [Google Scholar] [CrossRef] [PubMed]
- Stokes, A.N.; Williams, B.L.; French, S.S. An improved competitive inhibition enzymatic immunoassay method for Tetrodotoxin quantification. Biol. Proced. Online 2012, 14. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.Z.; Ji, R.; Jiang, T.; Yang, J. Development of ELISA-kit of quantitative analysis for Tetrodotoxin. China Public Health 2005, 21, 1423–1424. [Google Scholar]
- Zhong, Q.; Huang, A.; Wang, B.; Dong, X. Development of direct competitive ELISA kit for the detection of Tetrodotoxin using HRP Labeled Antigen. Adv. Mater. Res. 2011, 236–238, 2820–2824. [Google Scholar] [CrossRef]
- Reagen. Tetrodotoxin ELISA Test Kit. Available online: http://www.reagen.us/products.asp?id=769 (accessed on 10 December 20115).
- Reverté, L.; de la Iglesia, P.; del Río, V.; Campbell, K.; Elliott, C.T.; Kawatsu, K.; Katikou, P.; Diogène, J.; Campàs, M. Detection of tetrodotoxins in puffer fish by a self-assembled monolayer-based immunoassay and comparison with surface plasmon resonance, LC-MS/MS and mouse bioassay. Anal. Chem. 2015, 87, 10839–10847. [Google Scholar] [CrossRef] [PubMed]
- Chuen, B.S.; Loughran, M.; Hayashi, T.; Nagashima, Y.; Watanabe, E. Use of a channel biosensor for the assay of paralytic shellfish toxins. Toxicon 1998, 36, 1371–1381. [Google Scholar] [CrossRef]
- Barnes, P.; Campbell, K.; Haughey, S.; Elliott, C.; Higgins, C.H.; Botana, L.M.; Vasconcelos, V. Single laboratory validation of a screening method for detection of Tetrodotoxin in Charonica lampas lampas using a Surface Plasmon Resonance Biosensor. In Proceeding of the AOAC Marine and Freshwater Taskforce Meeting, Vigo, Spain, 1–5 May 2011.
- Taylor, A.D.; Vaisocherova, H.; Deeds, J.; deGrasse, S.; Jiang, S. Tetrodotoxin detection by surface Plasmon resonance sensor in Pufferfish matrices and urine. J. Sens. 2011, 2011. [Google Scholar] [CrossRef]
- Yakes, B.J.; Kanyuck, K.M.; deGrasse, S.L. First report of a Direct Surface Plasmon Resonance Immunosensor for a small molecule seafood toxin. Anal. Chem. 2014, 86, 9251–9255. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.C.; Jen, H.C.; Chen, C.L.; Hwang, D.F.; Chang, R.; Hwang, J.S.; Chiang, H.P. SERS study of Tetrodotoxin (TTX) by using silver nanoparticles arrays. Plasmonics 2009, 4, 187–192. [Google Scholar] [CrossRef]
- Yakes, B.J.; Etheridge, S.M.; Mulvaney, S.P.; Tamanaha, C.R. Fluidic force discrimination assays: A new technology for Tetrodotoxin detection. Mar. Drugs 2010, 8, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Gessner, B.D. Neurotoxic Toxins. In Seafood and Freshwater Toxins: Pharmacology, Physiology and Detection; Botana, L.M., Ed.; Dekker: New York, NY, USA, 2000; pp. 65–90. [Google Scholar]
- Paredes, I.; Rietjens, I.M.C.M.; Vietes, J.M.; Cabado, A.G. Update of risk assessments of main marine biotoxins in the European Union. Toxicon 2011, 58, 336–354. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turner, A.D.; Higgins, C.; Higman, W.; Hungerford, J. Potential Threats Posed by Tetrodotoxins in UK Waters: Examination of Detection Methodology Used in Their Control. Mar. Drugs 2015, 13, 7357-7376. https://doi.org/10.3390/md13127070
Turner AD, Higgins C, Higman W, Hungerford J. Potential Threats Posed by Tetrodotoxins in UK Waters: Examination of Detection Methodology Used in Their Control. Marine Drugs. 2015; 13(12):7357-7376. https://doi.org/10.3390/md13127070
Chicago/Turabian StyleTurner, Andrew D., Cowan Higgins, Wendy Higman, and James Hungerford. 2015. "Potential Threats Posed by Tetrodotoxins in UK Waters: Examination of Detection Methodology Used in Their Control" Marine Drugs 13, no. 12: 7357-7376. https://doi.org/10.3390/md13127070
APA StyleTurner, A. D., Higgins, C., Higman, W., & Hungerford, J. (2015). Potential Threats Posed by Tetrodotoxins in UK Waters: Examination of Detection Methodology Used in Their Control. Marine Drugs, 13(12), 7357-7376. https://doi.org/10.3390/md13127070







