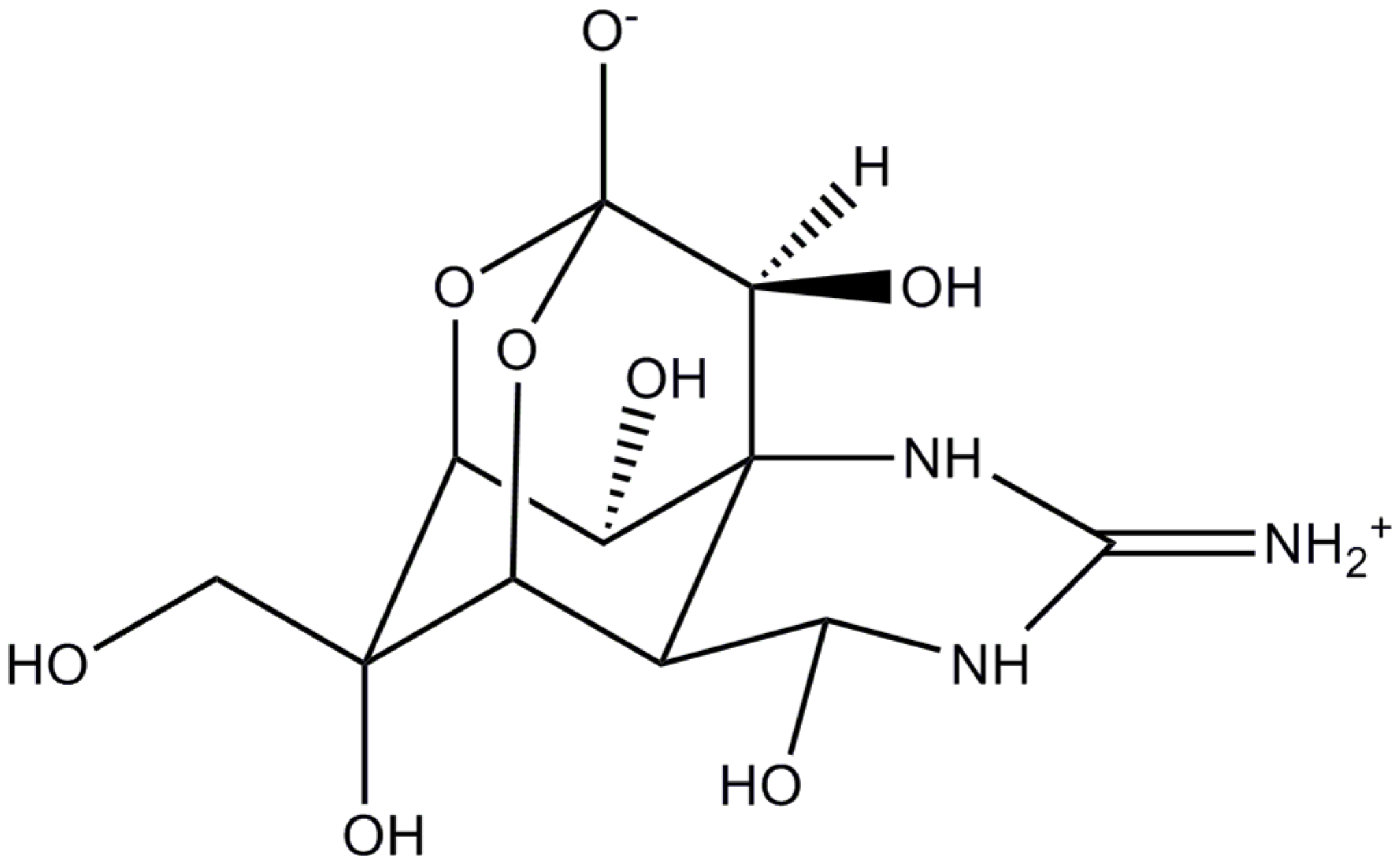
Tetrodotoxin, an Extremely Potent Marine Neurotoxin: Distribution, Toxicity, Origin and Therapeutical Uses
Abstract
:1. Introduction
2. Distribution in Nature
2.1. Aquatic Animals
Pufferfish
| Organisms | Species | Bacteria | Reference |
|---|---|---|---|
| Red algae | Jania sp. | Vibrio, Alteromonas and Shewanella | [47] |
| Crustacean: Copepods (Parasites of Pufferfish) | Pseudocaligus fugu and Taeniacanthus sp. | Roseobacter | [48] |
| Crustacean: xanthid crab | Atergatis floridus | Vibrio strain | [14] |
| Mollusc gastropod | Niotha Clathrata | Vibrio and Pseudomonas | [49] |
| Mollusc cephalopod | Octopus maculosus | Bacillus, Pseudomonas alteromonas and Vibrio spp. | [43] |
| Echinoderms: starfish | Astropecten polyacanthus | Vibrio alginolyticus | [50] |
| Vertebrates: pufferfish | Takifugu snyderi | Vibrio strain | [51] |
| Pufferfish | Takifugu obscurus | Aeromonas | [52] |
| Pufferfish | Fugu poecilonotus | Vibrio, Alteromonas and Shewanella | [47] |
| Pufferfish | Takifugu niphobles | Raoultella terrigena | [41] |
| Pufferfish | Fugu obscurus | Bacillus spp. | [53] |
| Pufferfish | Fugu rubripes | Bacillus and Actinomycetes | [54] |
| Pufferfish | Chelonodon patoca | Microbacterium arabinogalactanolyticum, Serratia marcescens, Vibrio alginolyticus | [55] |
| Pufferfish | Fugu vermicularis radiatus | Vibrio strain LM-1 | [56] |
2.2. Terrestrial Animals
2.3. Origin of STX vs. TTX
3. Toxicity and Mechanism of Action
| Grade | Symptoms |
|---|---|
| 1 | Neuromuscular symptoms (paresthesia around the mouth, headache, diaphoresis, pupillary constriction) and mild gastrointestinal symptoms (nausea, vomiting, hypersalivation, hyperemesis, hematemesis, hypermotility, diarrhea, abdominal pain). |
| 2 | Paresthesia spreading to the trunk and extremities, early motor paralysis and lack of coordination. |
| 3 | Increased neuromuscular symptoms (dysarthria, dysphagia, aphagia, lethargy, incoordination, ataxia, floating sensation, cranial nerve palsies, muscular fasciculations) cardiovascular/pulmonary symptoms (hypotension or hypertension, vasomotor blockade, cardiac arrhythmias including sinus bradycardia, asystole, tachycardia, and atrioventricular node conduction abnormalities; cianosis, pallor, dyspnea); dermatologic symptoms (exfoliative dermatitis, petechiae, blistering) hypotension, and aphonia. |
| 4 | Impaired conscious state, respiratory paralysis, severe hypotension, and cardiac arrhythmia. |
4. Human Cases of Intoxication
4.1. Asian Countries
4.2. Other Continents
4.2.1. America
4.2.2. Oceania
4.2.3. Europe
| Cases | Species Implicated | Country/Region Contaminated Shellfish | Date | Amount of Toxin or Fish Ingested | Symptoms Started/Deaths | Reference |
|---|---|---|---|---|---|---|
| 1 | Charonia sauliae | Shimizu, Shizuoka, Japan | December 1979 | 17,000 MU | 30 min after ingestion. Fully recovered in 5 days | [77] |
| 1 | Diodon hystrix | Hawaii, USA | 1986 | NR | He recovered within 1 week | [79] |
| 3 | fugu (pufferfish) brought from Japan | California, USA | April 1996 | a middle-quarter of fugu | 2–20 min after ingestion | [87] |
| 1 | Takifugu poecilonotus | Nagasaki, Japan | October 1996 | 10,000 MU | 30 to 60 min after ingestion. He died during the following hour. | [78] |
| 4 | NR | Nosy Be, Madagascar | July 1998 | 16 MU/g | NR | [88] |
| 5 | Takifugu niphoble | Chungua, Taiwan | January 2000 | 11 g of the cooked fish liver (280 MU/g) | NR | [67] |
| 6 | NR | Taiwan Strait | April 2001 | NR | 2 to 3 h after ingestion | [89] |
| 6 | Nassarius glans | Tungsa Island, Taiwan | April 2004 | digestive gland (2048 MU/g) and muscle (2992 MU/g) | NR | [90] |
| 202 | eight strains of Shewanella spp. | China | September 2007 | NR | 1 to 4 h after ingestion | [91] |
| 156 | 13 species of pufferfish | Bangladesh | 1998–2008 | NR | 10 min–15 h after ingestion. 40 deaths | [76] |
| NR | Lagocephalus inermis | Nagasaki, Japan | October 2008 | residual liver sample showed toxicity as high as 1230 MU/g | NR | [92] |
| 2 | Octopus Hapalochlaena fasciata | Taipei, Taiwan | December 2010 | The concentration of TTX was 31.8–94.3 μg/g (39.1–83.4 ng/mL in the urine and <0.1 ng/mL in plasma). | The symptoms subsided within five days and the patient fully recovered. | [93] |
| 26 | Amoyacaninus and Yongeichthys nebulosus | Guangdong, China | March 2012 | 100–300 g of fish consumed. The amount of TTX found in muscle and viscera was 9.69 MU/g and 10.42 MU/g in the case of A. canicus, and 14.51 MU/g and 15.47 MU/g in the case of Y. nebulosus | NR | [94] |
| 12 | Lagocephalus sceleratus | coast of Reunion Island (Southwest Indian Ocean) | September 2013 | NR | fully recovered within a few days | [95] |
| 2 | Lagocephalus lunaris | Minneapolis, Minnesota | June 2014 | 5.7–72.3 ppm | Thirty minutes after consumption. After 6 h his symptoms improved | [81] |
| 71 | Carcinoscorpius rotundicauda | Chon Buri, Thailand | NR | NR | Nineteen patients required artificial ventilation and there were two deaths. | [96] |
5. Therapies for TTX Intoxication
6. Medical Applications of Tetrodotoxin
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Suehiro, M. Historical review on chemical and medical studies of globefish toxin before World War II. Yakushigaku Zasshi 1994, 29, 428–434. [Google Scholar] [PubMed]
- Mosher, H.S.; Fuhrman, F.A.; Buchwald, H.D.; Fischer, H.G. Tarichatoxin-tetrodotoxin, a potent neurotoxin. Science 1964, 144, 1100–1110. [Google Scholar] [CrossRef] [PubMed]
- Asakawa, M.; Toyoshima, T.; Shida, Y.; Noguchi, T.; Miyazawa, K. Paralytic toxins in a ribbon worm Cephalothrix species (Nemertean) adherent to cultured oysters in Hiroshima Bay, Hiroshima Prefecture, Japan. Toxicon 2000, 38, 763–773. [Google Scholar] [CrossRef]
- Chulanetra, M.; Sookrung, N.; Srimanote, P.; Indrawattana, N.; Thanongsaksrikul, J.; Sakolvaree, Y.; Chongsa-Nguan, M.; Kurazono, H.; Chaicumpa, W. Toxic marine puffer fish in Thailand seas and tetrodotoxin they contained. Toxins (Basel) 2011, 3, 1249–1262. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Yotsu-Yamashita, M. Distribution of tetrodotoxin, saxitoxin, and their analogs among tissues of the puffer fish Fugu pardalis. Toxicon 2006, 48, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, Y.; Tanu, M.B.; Noguchi, T. First occurrence of a food poisoning incident due to ingestion of Takifugu oblongus, along with a toxicological report on three marine puffer species in Bangladesh. Shokuhin Eiseigaku Zasshi 1999, 40, 473–480. [Google Scholar] [CrossRef]
- Noguchi, T.; Narita, H.; Maruyama, J.; Hashimoto, K. Tetrodotoxin in the starfish Astropecten polyacanthus, in association with toxification of a trumpet shell “Boshubora” Charonia sauliae. Nippon Suisan Gakkaishi 1982, 48, 1173–1177. [Google Scholar] [CrossRef]
- Sheumack, D.D.; Howden, M.E.H. Maculotoxin: A neurotoxin from the venom glands of the octopus Hapalochlaena maculosa identified as tetrodotoxin. Science 1978, 199, 188–189. [Google Scholar] [CrossRef] [PubMed]
- Sui, L.M.; Chen, K.; Hwang, P.A.; Hwang, D.F. Identification of tetrodotoxin in marine gastropods implicated in food poisoning. J. Nat. Toxins 2002, 11, 213–220. [Google Scholar] [PubMed]
- Kim, Y.H.; Brown, G.B.; Mosher, F.A. Tetrodotoxin: Occurrence in atelopid frogs of Costa Rica. Science 1975, 189, 151–152. [Google Scholar] [CrossRef] [PubMed]
- Yasumoto, T.; Yotsu, M.; Murate, M.; Naoki, H. New tetrodotoxin analogues from the newt Cynops ensicauda. J. Am. Chem. Soc. 1988, 110, 2344–2345. [Google Scholar] [CrossRef]
- Yotsu-Yamashita, M.; Gilhen, J.; Russell, R.W.; Krysko, K.L.; Melaun, C.; Kurz, A.; Kauferstein, S.; Kordis, D.; Mebs, D. Variability of tetrodotoxin and of its analogues in the red-spotted newt, Notophthalmus viridescens (Amphibia: Urodela: Salamandridae). Toxicon 2012, 59, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Yotsu-Yamashita, M.; Mebs, D.; Kwet, A.; Schneider, M. Tetrodotoxin and its analogue 6-epitetrodotoxin in newts (Triturus spp.; Urodela, Salamandridae) from southern Germany. Toxicon 2007, 50, 306–309. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Jeon, J.K.; Arakawa, O.; Sugita, H.; Deguchi, Y.; Shida, Y.; Hashimoto, K. Occurrence of tetrodotoxin and anhydrotetrodotoxin in Vibrio sp. isolated from the intestines of a xanthid crab, Atergatis floridus. J. Biochem. 1986, 99, 311–314. [Google Scholar] [PubMed]
- Matsui, T.; Sato, H.; Hamada, S.; Shimizu, C. Comparison of toxicity of the cultured and wild puffer fish Fugu niphobles. Bull. Jpn. Soc. Sci. Fish. 1982, 48, 253. [Google Scholar]
- Noguchi, T.; Arakawa, O.; Takatani, T. Toxicity of pufferfish Takifugu rubripes cultured in netcages at sea or aquaria on land. Comp. Biochem. Physiol. Part D 2006, 1, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Maruyama, J.; Kanoh, S.; Jeon, J.K.; Noguchi, T.; Harada, T.; Murata, O.; Hashimoto, K. Toxicity of the cultured pufferfish Fugu rubripes rubripes along with their resistibility against tetrodotoxin. Bull. Jpn. Soc. Sci. Fish. 1984, 50, 1573–1575. [Google Scholar] [CrossRef]
- Honda, S.; Arakawa, O.; Takatani, T.; Tachibana, K.; Yagi, M.; Tanigawa, A.; Noguchi, T. Toxification of cultured puffer fish Takifugu rubripes by feeding on tetrodotoxin-containing diet. Nippon Suisan Gakkaishi 2005, 71, 815–820. [Google Scholar] [CrossRef]
- Matsui, T.; Hamada, S.; Konosu, S. Difference in accumulation of puffer fish toxin and crystalline tetrodotoxin in the puffer fish, Fugu rubripes rubripes. Bull. Jpn. Soc. Sci. Fish. 1981, 47, 535–537. [Google Scholar] [CrossRef]
- Yamamori, K.; Kono, M.; Furukawa, K.; Matsui, T. The toxification of juvenile cultured kusafugu Takifugu niphobles by oral administration of crystalline tetrodotoxin. Shokuhin Eiseigaku Zasshi 2004, 45, 73–75. [Google Scholar] [CrossRef] [PubMed]
- Bentur, Y.; Ashkar, J.; Lurie, Y.; Levy, Y.; Azzam, Z.S.; Litmanovich, M.; Golik, M.; Gurevych, B.; Golani, D.; Eisenman, A. Lessepsian migration and tetrodotoxin poisoning due to Lagocephalus sceleratus in the eastern Mediterranean. Toxicon 2008, 52, 964–968. [Google Scholar] [CrossRef] [PubMed]
- Katikou, P.; Georgantelis, D.; Sinouris, N.; Petsi, A.; Fotaras, T. First report on toxicity assessment of the Lessepsian migrant pufferfish Lagocephalus sceleratus (Gmelin, 1789) from European waters (Aegean Sea, Greece). Toxicon 2009, 54, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.A.; Mossa, A.E.W. Red Sea puffer fish poisoning: Emergency diagnosis and management of human intoxication. Egypt. J. Aquat. Res. 2005, 31, 370–378. [Google Scholar]
- Denac, H.; Mevissen, M.; Scholtysik, G. Structure, function and pharmacology of voltage-gated sodium channels. Naunyn-Schmiedebergs Arch. Pharmacol. 2000, 362, 453–479. [Google Scholar] [CrossRef] [PubMed]
- Homaira, N.; Rahman, M.; Luby, S.P.; Rahman, M.; Haider, M.S.; Faruque, L.I.; Khan, D.; Parveen, S.; Gurley, E.S. Multiple outbreaks of puffer fish intoxication in Bangladesh, 2008. Am. J. Trop. Med. Hyg. 2010, 83, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Ebesu, J.S.M. Puffer poisoning: Epidemiology and treatment. J. Toxicol. Toxin Rev. 2001, 20, 1–10. [Google Scholar] [CrossRef]
- EC. Regulation (EC) No 854/2004 of the European Parliament and of the Council of 29 April 2004 Laying down Specific Rules for the Organisation of Official Controls on Products of Animal Origin Intended for Human Consumption. Available online: https://www.food.gov.uk/sites/default/files/multimedia/pdfs/h3ojregulation.pdf (accessed on 5 August 2015).
- EC. Regulation (EC) No 853/2004 of the European Parliament and of the Council of 29 April 2004 Laying down Specific Hygiene Rules for Food of Animal Origin. Available online: https://www.fsai.ie/uploadedFiles/Food_Business/Reg853_2004.pdf (accessed on 5 August 2015).
- Hagen, N.A.; du Souich, P.; Lapointe, B.; Ong-Lam, M.; Dubuc, B.; Walde, D.; Love, R.; Ngoc, A.H. Tetrodotoxin for moderate to severe cancer pain: A randomized, double blind, parallel design multicenter study. J. Pain Symptom Manag. 2008, 35, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.K.; Mikusa, J.P.; Hernandez, G.; Baker, S.; Shieh, C.C.; Neelands, T.; Zhang, X.F.; Niforatos, W.; Kage, K.; Han, P.; et al. Involvement of the TTX-resistant sodium channel Nav 1.8 in inflammatory and neuropathic, but not post-operative, pain states. Pain 2006, 123, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Marcil, J.; Walczak, J.S.; Guindon, J.; Ngoc, A.H.; Lu, S.; Beaulieu, P. Antinociceptive effects of tetrodotoxin (TTX) in rodents. Br. J. Anaesth. 2006, 96, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Liu, T.T.; Wang, X.; Epstein, D.H.; Zhao, L.Y.; Zhang, X.L.; Lu, L. Tetrodotoxin reduces cue-induced drug craving and anxiety in abstinent heroin addicts. Pharmacol. Biochem. Behav. 2009, 92, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Lehman, E.M.; Brodie, E.D., Jr.; Brodie, E.D., 3rd. No evidence for an endosymbiotic bacterial origin of tetrodotoxin in the newt Taricha granulosa. Toxicon 2004, 44, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Bane, V.; Lehane, M.; Dikshit, M.; O’Riordan, A.; Furey, A. Tetrodotoxin: Chemistry, toxicity, source, distribution and detection. Toxins (Basel) 2014, 6, 693–755. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Arakawa, O. Tetrodotoxin—Distribution and accumulation in aquatic organisms, and cases of human intoxication. Mar. Drugs 2008, 6, 220–242. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, C.T. The chemical and evolutionary ecology of tetrodotoxin (TTX) toxicity in terrestrial vertebrates. Mar. Drugs 2010, 8, 577–593. [Google Scholar] [CrossRef] [PubMed]
- Cardall, B.L.; Brodie, E.D., Jr.; Brodie, E.D., 3rd; Hanifin, C.T. Secretion and regeneration of tetrodotoxin in the rough-skin newt (Taricha granulosa). Toxicon 2004, 44, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Daly, J.W. Marine toxins and nonmarine toxins: Convergence or symbiotic organisms? J. Nat. Prod. 2004, 67, 1211–1215. [Google Scholar] [CrossRef] [PubMed]
- Daly, J.W.; Myers, C.W.; Whittaker, N. Further classification of skin alkaloids from neotropical poison frogs (Dendrobatidae), with a general survey of toxic/noxious substances in the amphibia. Toxicon 1987, 25, 1023–1095. [Google Scholar] [CrossRef]
- Wood, S.A.; Taylor, D.I.; McNabb, P.; Walker, J.; Adamson, J.; Craig, C.S. Tetrodotoxin concentrations in Pleurobranchaea maculata: Temporal, spatial and individual variability from New Zealand populations. Mar. Drugs 2012, 10, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Yu, V.C.; Yu, P.H.; Ho, K.C.; Lee, F.W. Isolation and identification of a new tetrodotoxin-producing bacterial species, Raoultella terrigena, from Hong Kong marine puffer fish Takifugu niphobles. Mar. Drugs 2011, 9, 2384–2396. [Google Scholar] [CrossRef] [PubMed]
- Chau, R.; Kalaitzis, J.A.; Neilan, B.A. On the origins and biosynthesis of tetrodotoxin. Aquat. Toxicol. 2011, 104, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.F.; Arakawa, O.; Saito, T.; Noguchi, T.; Simidu, U.; Tsukamoto, K.; Shida, Y.; Hashimoto, K. Tetrodotoxin-producing bacteria from the blue-ringed octopus Octopus maculosus. Mar. Biol. 1989, 100, 327–332. [Google Scholar] [CrossRef]
- Matsumoto, T.; Nagashima, Y.; Kusuhara, H.; Sugiyama, Y.; Ishizaki, S.; Shimakura, K.; Shiomi, K. Involvement of carrier-mediated transport system in uptake of tetrodotoxin into liver tissue slices of puffer fish Takifugu rubripes. Toxicon 2007, 50, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Yotsu-Yamashita, M.; Sugimoto, A.; Terakawa, T.; Shoji, Y.; Miyazawa, T.; Yasumoto, T. Purification, characterization, and cDNA cloning of a novel soluble saxitoxin and tetrodotoxin binding protein from plasma of the puffer fish, Fugu pardalis. Eur. J. Biochem. 2001, 268, 5937–5946. [Google Scholar] [CrossRef] [PubMed]
- Jal, S.; Khora, S.S. An overview on the origin and production of tetrodotoxin, a potent neurotoxin. J. Appl. Microbiol. 2015, 119, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Simidu, U.; Kita-Tsukamoto, K.; Yasumoto, T.; Yotsu, M. Taxonomy of four marine bacterial strains that produce tetrodotoxin. Int. J. Syst. Evol. Microbiol. 1990, 40, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Venmathi Maran, B.A.; Iwamoto, E.; Okuda, J.; Matsuda, S.; Taniyama, S.; Shida, Y.; Asakawa, M.; Ohtsuka, S.; Nakai, T.; Boxshall, G.A. Isolation and characterization of bacteria from the copepod Pseudocaligus fugu ectoparasitic on the panther puffer Takifugu pardalis with the emphasis on TTX. Toxicon 2007, 50, 779–790. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.A.; Hwang, D.F.; Tsai, Y.H.; Chen, H.C.; Jeng, S.S.; Noguchi, T.; Ohwada, K.; Hasimoto, K. Microflora and tetrodotoxin-producing bacteria in a gastropod, Niotha clathrata. Food Chem. Toxicol. 1995, 33, 929–934. [Google Scholar] [CrossRef]
- Narita, H.; Matsubara, S.; Miwa, N.; Akahane, S.; Murakami, M.; Goto, T.; Nara, M.; Noguchi, T.; Saito, T.; Shida, Y. Vibrio alginolyticus, a TTX-producing bacterium isolated from the starfish Astropecten polyacanthus. Nippon Suisan Gakkaishi 1987, 53, 617–621. [Google Scholar] [CrossRef]
- Hashimoto, K.; Noguchi, T.; Watabe, S. New aspects of tetrodotoxin. In Microbial Toxins in Foods and Feeds. Cellular and Molecular Modes of Action; Pohland, A.E., Dowell, V.R., Jr., Richard, J.L., Cole, R.J., Eklund, M.W., Green, S.S., Norred, W.P., III, Potter, M.E., Eds.; Plenum Press: New York, NY, USA, 1990; pp. 575–588. [Google Scholar]
- Yang, G.; Xu, J.; Liang, S.; Ren, D.; Yan, X.; Bao, B. A novel TTX-producing Aeromonas isolated from the ovary of Takifugu obscurus. Toxicon 2010, 56, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Fan, Y. Isolation and characterization of a Bacillus species capable of producing tetrodotoxin from the puffer fish Fugu obscurus. World J. Microbiol. Biotechnol. 2010, 26, 1755–1760. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, Y.; Xie, L.; Xia, G.; Hu, J.; Wang, S.; Zhang, R. Toxicity and distribution of tetrodotoxin-producing bacteria in puffer fish Fugu rubripes collected from the Bohai Sea of China. Toxicon 2005, 46, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.F.; Yu, P.H.F.; Chan, P.L.; Yan, Q.; Wong, P.K. Two novel species of tetrodotoxin-producing bacteria isolated from toxic marine puffer fishes. Toxicon 2004, 44, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-J.; Jeong, D.-Y.; Kim, W.-S.; Kim, H.-D.; Kim, C.-H.; Park, W.-W.; Park, Y.-H.; Kim, K.-S.; Kim, H.-M.; Kim, D.-S. A tetrodotoxin-producing Vibrio strain, LM-1, from the puffer fish Fugu vermicularis radiatus. Appl. Environ. Microbiol. 2000, 66, 1698–1701. [Google Scholar] [CrossRef] [PubMed]
- Moczydlowski, E.G. The molecular mystique of tetrodotoxin. Toxicon 2013, 63, 165–183. [Google Scholar] [CrossRef] [PubMed]
- Stüken, A.; Orr, R.J.S.; Kellmann, R.; Murray, S.A.; Neilan, B.A.; Jakobsen, K.S. Discovery of nuclear-encoded genes for the neurotoxin saxitoxin in dinoflagellates. PLoS ONE 2011, 6, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Vlamis, A.; Katikou, P.; Rodriguez, I.; Rey, V.; Alfonso, A.; Papazachariou, A.; Zacharaki, T.; Botana, A.M.; Botana, L.M. First detection of tetrodotoxin in Greek shellfish by UPLC-MS/MS potentially linked to the presence of the dinoflagellate Prorocentrum minimum. Toxins 2015, 7, 1779–1807. [Google Scholar] [CrossRef] [PubMed]
- Kodama, M.; Sato, S.; Sakamoto, S.; Ogata, T. Occurrence of tetrodotoxin in Alexandrium tamarense, a causative dinoflagellate of paralytic shellfish poisoning. Toxicon 1996, 34, 1101–1105. [Google Scholar] [CrossRef]
- Murray, S.A.; Mihali, T.K.; Neilan, B.A. Extraordinary conservation, gene loss, and positive selection in the evolution of an ancient neurotoxin. Mol. Biol. Evol. 2011, 28, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.; Nikkon, F.; Pervin, F.; Rahman, M.M.; Khatun, S.; Hossain, T.; Khan, A.; Sarker, S.K.; Mosaddik, A.; Absar, N. Biochemical and histopathological effects of tetrodotoxin isolated from puffer fish Tetraodon patoca available in Bangladesh. Res. J. Med. Med. Sci. 2008, 3, 177–181. [Google Scholar]
- Saoudi, M.; Rabeh, F.B.; Jammoussi, K.; Abdelmouleh, A.; Belbahri, L.; Feki, A.E. Biochemical and physiological responses in Wistar rat after administration of puffer fish (Lagocephalus lagocephalus) flesh. J. Food Agric. Environ. 2007, 5, 107–111. [Google Scholar]
- Zimmer, T. Effects of tetrodotoxin on the mammalian cardiovascular system. Mar. Drugs 2010, 8, 741–762. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.F.; Williams, S.R.; Nordt, S.P.; Manoguerra, A.S. A review of selected seafood poisonings. Undersea Hyperb. Med. 1999, 26, 175–184. [Google Scholar] [PubMed]
- Sorokin, M. Puffer fish poisoning. Med. J. Aust. 1973, 1, 957. [Google Scholar] [PubMed]
- Hwang, D.F.; Noguchi, T. Tetrodotoxin poisoning. Adv. Food Nutr. Res. 2007, 52, 141–236. [Google Scholar] [PubMed]
- Fukuda, A.; Tani, A. Records of puffer poisonings report 3. Nippon Igaku Oyobi Kenko Hoken 1941, 3528, 7–13. [Google Scholar]
- Xu, Q.; Huang, K.; Gao, L.; Zhang, H.; Rong, K. Toxicity of tetrodotoxin towards mice and rabbits. Wei Sheng Yan Jiu 2003, 32, 371–374. [Google Scholar] [PubMed]
- Rodrigue, D.C.; Etzel, R.A.; Hall, S.; de Porras, E.; Velasquez, O.H.; Tauxe, R.V.; Kilbourne, E.M.; Blake, P.A. Lethal paralytic shellfish poisoning in Guatemala. Am. J. Trop. Med. Hyg. 1990, 42, 267–271. [Google Scholar] [PubMed]
- Yang, C.C.; Liao, S.C.; Deng, J.F. Tetrodotoxin poisoning in Taiwan: An analysis of poison center data. Vet. Hum. Toxicol. 1996, 38, 282–286. [Google Scholar] [PubMed]
- William, S.H.; Shepherd, S. Scombroid, ciguatera, and other seafood intoxications. In Clinical Toxicology, 1st ed.; Ford, M.D., Delaney, K.A., Ling, L.J., Erickson, T., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2001; pp. 959–968. [Google Scholar]
- Lin, S.Z.; Hwang, D.F. Distribution and sources of tetrodotoxin. Sci. Dev. 2007, 419, 20–25. [Google Scholar]
- Chew, S.K.; Goh, C.H.; Wang, K.W.; Mah, P.K.; Tan, B.Y. Puffer fish (tetrodotoxin) poisoning: Clinical report and role of anti-cholinesterase drugs in therapy. Singap. Med. J. 1983, 24, 168–171. [Google Scholar]
- Ahasan, H.A.; Mamun, A.A.; Karim, S.R.; Bakar, M.A.; Gazi, E.A.; Bala, C.S. Paralytic complications of puffer fish (tetrodotoxin) poisoning. Singap. Med. J. 2004, 45, 73–74. [Google Scholar]
- Islam, Q.T.; Razzak, M.A.; Islam, M.A.; Bari, M.I.; Basher, A.; Chowdhury, F.R.; Sayeduzzaman, A.B.; Ahasan, H.A.; Faiz, M.A.; Arakawa, O.; et al. Puffer fish poisoning in Bangladesh: Clinical and toxicological results from large outbreaks in 2008. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Narita, H.; Noguchi, T.; Maruyama, J.; Ueda, Y.; Hashimoto, K.; Watanabe, Y.; Hida, K. Occurrence of tetrodotoxin in a trumpet shell, “boshubora” Charonia sauliae. Bull. Jpn. Soc. Sci. Fish. 1981, 47, 935–941. [Google Scholar] [CrossRef]
- Noguchi, T.; Akaeda, H. Pufferfish poisoning. Jpn. J. Toxicol. 1998, 11, 339–345. [Google Scholar]
- Sims, J.K.; Ostman, D.C. Emergency diagnosis and management of mild human tetrodotoxin. Ann. Emerg. Med. 1986, 15, 1094–1098. [Google Scholar] [CrossRef]
- Cohen, N.J.; Deeds, J.R.; Wong, E.S.; Hanner, R.H.; Yancy, H.F.; White, K.D.; Thompson, T.M.; Wahl, M.; Pham, T.D.; Guichard, F.M.; et al. Public health response to puffer fish (Tetrodotoxin) poisoning from mislabeled product. J. Food Prot. 2009, 72, 810–817. [Google Scholar] [PubMed]
- Cole, J.B.; Heegaard, W.G.; Deeds, J.R.; McGrath, S.C.; Handy, S.M. Tetrodotoxin poisoning outbreak from imported dried puffer fish—Minneapolis, Minnesota, 2014. Centers for Disease Control and Prevention (CDC). MMWR Morb. Mortal Wkly. Rep. 2015, 63, 1222–1225. [Google Scholar] [PubMed]
- De Souza Simões, E.M.; Mendes, T.M.; Adão, A.; Haddad Junior, V. Poisoning after ingestion of pufferfish in Brazil: Report of 11 cases. J. Venom. Anim. Toxins Incl. Trop. Dis. 2014, 20, 54. [Google Scholar] [CrossRef] [PubMed]
- Cavazzoni, E.; Lister, B.; Sargent, P.; Schibler, A. Blue-ringed octopus (Hapalochlaena sp.) envenomation of a 4-year-old boy: A case report. Clin. Toxicol. (Phila.) 2008, 46, 760–761. [Google Scholar] [CrossRef] [PubMed]
- McNabb, P.; Selwood, A.; Munday, R.; Wood, S.A.; Taylor, D.; Mackenzie, L.A.; van Ginkel, R.; Rhodes, L.L.; Cornelisen, C.; Heasman, K.; et al. Detection of tetrodotoxin from the grey side-gilled sea slug—Pleurobranchaea maculata, and associated dog neurotoxicosis on beaches adjacent to the Hauraki Gulf, Auckland, New Zealand. Toxicon 2010, 56, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ortega, J.F.; Morales-de los Santos, J.M.; Herrera-Gutiérrez, M.E.; Fernández-Sánchez, V.; Rodríguez Louro, P.; Rancaño, A.A.; Téllez-Andrade, A. Seafood intoxication by tetrodotoxin: First case in Europe. J. Emerg. Med. 2010, 39, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.; Alfonso, A.; Vale, C.; Alfonso, C.; Vale, P.; Tellez, A.; Botana, L.M. First toxicity report of tetrodotoxin and 5,6,11-trideoxyTTX in the trumpet shell Charonia lampas lampas in Europe. Anal. Chem. 2008, 80, 5622–5629. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Tetrodotoxin poisoning associated with eating puffer fish transported from Japan—California, 1996. MMWR Morb. Mortal Wkly. Rep. 1996, 45, 389–391. [Google Scholar]
- Ravaonindrina, N.; Andriamaso, T.H.; Rasolofonirina, N. Puffer fish poisoning in Madagascar: Four case reports. Arch. Inst. Pasteur Madag. 2001, 67, 61–64. [Google Scholar]
- How, C.K.; Chern, C.H.; Huang, Y.C.; Wang, L.M.; Lee, C.H. Tetrodotoxin poisoning. Am. J. Emerg. Med. 2003, 21, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.A.; Tsai, Y.H.; Deng, J.F.; Cheng, C.A.; Ho, P.H.; Hwang, D.F. Identification of tetrodotoxin in a marine gastropod (Nassarius glans) responsible for human morbidity and mortality in Taiwan. J. Food. Prot. 2005, 68, 1696–1701. [Google Scholar] [PubMed]
- Wang, D.; Wang, Y.; Huang, H.; Lin, J.; Xiao, D.; Kan, B. Identification of tetrodotoxin-producing Shewanella spp. from feces of food poisoning patients and food samples. Gut Pathog. 2013, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, Y.; Matsumoto, T.; Kadoyama, K.; Ishizaki, S.; Taniyama, S.; Takatani, T.; Arakawa, O.; Terayama, M. Tetrodotoxin poisoning due to smooth-backed blowfish, Lagocephalus inermis and the toxicity of L. inermis caught off the Kyushu coast, Japan. Shokuhin Eiseigaku Zasshi 2012, 53, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.J.; Lin, C.L.; Chen, C.H.; Hsieh, C.H.; Jen, H.C.; Jian, S.J.; Hwang, D.F. Toxin and species identification of toxic octopus implicated into food poisoning in Taiwan. Toxicon 2014, 91, 96–102. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Yue, Y.; Xing, F.; Xia, W.; Lai, S.; Zhang, F. Tetrodotoxin poisoning caused by Goby fish consumption in southeast China: A retrospective case series analysis. Clinics (Sao Paulo) 2015, 70, 24–29. [Google Scholar] [CrossRef]
- Puech, B.; Batsalle, B.; Roget, P.; Turquet, J.; Quod, J.P.; Allyn, J.; Idoumbin, J.P.; Chane-Ming, J.; Villefranque, J.; Mougin-Damour, K.; et al. Family tetrodotoxin poisoning in Reunion Island (Southwest Indian Ocean) following the consumption of Lagocephalus sceleratus (Pufferfish). Bull. Soc. Pathol. Exot. 2014, 107, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Kanchanapongkul, J.; Krittayapoositpot, P. An epidemic of tetrodotoxin poisoning following ingestion of the horseshoe crab Carcinoscorpius rotundicauda. Southeast Asian J. Trop. Med. Public Health 1995, 26, 364–367. [Google Scholar] [PubMed]
- Chew, S.K.; Chew, L.S.; Wang, K.W.; Mah, P.K.; Tan, B.Y. Anticholinesterase drugs in the treatment of tetrodotoxin poisoning. Lancet 1984, 2, 108. [Google Scholar] [CrossRef]
- Huot, R.I.; Armstrong, D.L.; Chanh, T.C. Protection against nerve toxicity by monoclonal antibodies to the sodium channel blocker tetrodotoxin. J. Clin. Investig. 1989, 83, 1821–1826. [Google Scholar] [CrossRef] [PubMed]
- Rivera, V.R.; Poli, M.A.; Bignami, G.S. Prophylaxis and treatment with a monoclonal antibody of tetrodotoxin poisoning in mice. Toxicon 1995, 33, 1231–1237. [Google Scholar] [CrossRef]
- Matsumura, K. In vivo neutralization of tetrodotoxin by a monoclonal antibody. Toxicon 1995, 33, 1239–1241. [Google Scholar] [CrossRef]
- Fukiya, S.; Matsumura, K. Active and passive immunization for tetrodotoxin in mice. Toxicon 1992, 30, 1631–1634. [Google Scholar] [CrossRef]
- Kaufman, B.; Wright, D.C.; Ballou, W.R.; Monheit, D. Protection against tetrodotoxin and saxitoxin intoxication by a cross-protective rabbit anti-tetrodotoxin antiserum. Toxicon 1991, 29, 581–587. [Google Scholar] [CrossRef]
- Matsumura, K. A monoclonal antibody against tetrodotoxin that reacts to the active group for the toxicity. Eur. J. Pharmacol. Environ. Toxicol. Pharmacol. 1995, 293, 41–45. [Google Scholar] [CrossRef]
- Xu, Q.H.; Wei, C.H.; Huang, K.; Rong, K.T. Toxin-neutralizing effect and activity-quality relationship for mice tetrodotoxin-specific polyclonal antibodies. Toxicology 2005, 206, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.H.; Rong, K.T.; Yun, L.H. Protection against tetrodotoxin poisoning by an experimental vaccine. In Proceedings of the 5th Congress of Toxicology in Developing Countries, Guilin, China, 10–13 November 2003.
- Xu, Q.H.; Rong, K.T.; Yun, L.H.; Wei, C.H.; Gao, L.S.; Huang, K. New artificial antigen for tetrodotoxin and its high efficiency on antitoxic effect. J. Immunol. 2003, 19, 424–428. [Google Scholar]
- Xu, Q.H.; Wei, C.H.; Huang, K.; Gao, L.S.; Rong, K.T.; Yun, L.H. An experimental vaccine against tetrodotoxin with longer term of validity. Chin. J. Immunol. 2003, 19, 339–342. [Google Scholar]
- Arakawa, O.; Hwang, D.F.; Tanyama, S.; Takatani, T. Toxins of pufferfish that cause human intoxications. In Coastal Environmental and Ecosystem Issues of the East China Sea; Ishimatsu, A., Lie, H.-J., Eds.; TERRAPUB and Nagasaki University: Tokyo, Japan, 2010; pp. 227–244. [Google Scholar]
- Chowdhury, F.R.; Ahasan, H.A.; Rashid, A.K.; Mamun, A.A.; Khaliduzzaman, S.M. Tetrodotoxin poisoning: A clinical analysis, role of neostigmine and short-term outcome of 53 cases. Singap. Med. J. 2007, 48, 830–833. [Google Scholar]
- Hegyi, B.; Komáromi, I.; Kistamás, K.; Ruzsnavszky, F.; Váczi, K.; Horváth, B.; Magyar, J.; Bányász, T.; Nánási, P.P.; Szentandrássy, N. Tetrodotoxin blockade on canine cardiac l-type Ca2+ channels depends on pH and redox potential. Mar. Drugs 2013, 11, 2140–2153. [Google Scholar] [CrossRef] [PubMed]
- Guzmán, A.; Fernández de Henestrosa, A.R.; Marín, A.-P.; Ho, A.; González Borroto, J.I.; Carasa, I.; Pritchard, L. Evaluation of the genotoxic potential of the natural neurotoxin Tetrodotoxin (TTX) in a battery of in vitro and in vivo genotoxicity assays. Mutat. Res./Genet. Toxicol. Environ. 2007, 634, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. The influence of natural products upon drug discovery. Nat. Prod. Rep. 2000, 17, 215–234. [Google Scholar] [CrossRef] [PubMed]
- Sakai, R.; Swanson, G.T. Recent progress in neuroactive marine natural products. Nat. Prod. Rep. 2014, 31, 273–309. [Google Scholar] [CrossRef] [PubMed]
- Münchau, A.; Bhatia, K.P. Uses of botulinum toxin injection in medicine today. Br. Med. J. 2000, 320, 161–165. [Google Scholar] [CrossRef]
- Burroughs, J.R.; Anderson, R.L. Cosmetic botulinum toxin applications: General considerations and dosing. In Pearls and Pitfalls in Cosmetic Oculoplastic Surgery; Hartstein, M.E., Massry, G.G., Holds, J.B., Eds.; Springer: New York, NY, USA, 2015; pp. 393–394. [Google Scholar]
- Anderson, P.D.; Bokor, G. Conotoxins: Potential weapons from the sea. J. Bioterror. Biodef. 2012, 3, 120. [Google Scholar] [CrossRef]
- Dias, E.; Paulino, S.; Pereira, P. Cyanotoxins: From poisoning to healing—A possible pathway? Limnetica 2015, 34, 159–172. [Google Scholar]
- Martínez, A.; Garrido-Maestú, A.; Ben-Gigirey, B.; Chapela, M.J.; González, V.; Vieites, J.M.; Cabado, A.G. Marine biotoxins. In Handbook of Marine Biotechnology; Kim, S.-K., Ed.; Springer: Berlin, Germany, 2015; pp. 869–904. [Google Scholar]
- Nieto, F.R.; Cobos, E.J.; Tejada, M.Á.; Sánchez-Fernández, C.; González-Cano, R.; Cendán, C.M. Tetrodotoxin (TTX) as a therapeutic agent for pain. Mar. Drugs 2012, 10, 281–305. [Google Scholar] [CrossRef] [PubMed]
- Sztriha, L.; Lestringant, G.G.; Hertecant, J.; Frossard, P.M.; Masouyé, I. Congenital insensitivity to pain with anhidrosis. Pediatr. Neurol. 2001, 25, 63–66. [Google Scholar] [CrossRef]
- Fozzard, H.A.; Lipkind, G.M. The tetrodotoxin binding site is within the outer vestibule of the sodium channel. Mar. Drugs 2010, 8, 219–234. [Google Scholar] [CrossRef] [PubMed]
- Cervenka, R.; Zarrabi, T.; Lukacs, P.; Todt, H. The outer vestibule of the Na+ channel-toxin receptor and modulator of permeation as well as gating. Mar. Drugs 2010, 8, 1373–1393. [Google Scholar] [CrossRef] [PubMed]
- Berde, C.B.; Athiraman, U.; Yahalom, B.; Zurakowski, D.; Corfas, G.; Bognet, C. Tetrodotoxin-bupivacaine-epinephrine combinations for prolonged local anesthesia. Mar. Drugs 2011, 9, 2717–2728. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.R.; Novick, P.A.; Parsons, W.H.; McGregor, M.; Zablocki, J.; Pande, V.S.; du Bois, J. Marked difference in saxitoxin and tetrodotoxin affinity for the human nociceptive voltage-gated sodium channel (Nav1.7). Proc. Natl. Acad. Sci. USA 2012, 109, 18102–18107. [Google Scholar] [CrossRef] [PubMed]
- Vornanen, M.; Hassinen, M.; Haverinen, J. Tetrodotoxin sensitivity of the vertebrate cardiac Na+ current. Mar. Drugs 2011, 9, 2409–2422. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wood, J.N. The roles of sodium channels in nociception: Implications for mechanisms of neuropathic pain. Pain Med. 2011, 12, S93–S99. [Google Scholar] [CrossRef] [PubMed]
- Burliński, P.J.; Burlińska, A.M.; Gonkowski, S.; Całka, J. Resiniferatoxin and tetrodotoxin induced NPY and TH immunoreactivity changes within the paracervical ganglion neurons supplying the urinary bladder. J. Mol. Neurosci. 2013, 49, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Grimm, J.W.; See, R.E. Dissociation of primary and secondary reward-relevant limbic nuclei in an animal model of relapse. Neuropsychopharmacology 2000, 22, 473–479. [Google Scholar] [CrossRef]
- Green, P.G.; Alvarez, P.; Levine, J.D. Topical tetrodotoxin attenuates photophobia induced by corneal injury in the rat. J. Pain 2015, 16, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Lodge, D.J.; Grace, A.A. Aberrant hippocampal activity underlies the dopamine dysregulation in an animal model of schizophrenia. J. Neurosci. 2007, 27, 11424–11430. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Marine-sourced anti-cancer and cancer pain control agents in clinical and late preclinical development. Mar. Drugs 2014, 12, 255–278. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Li, J.; Lu, C.-L.; Kang, L.; Xie, L.; Zhang, Y.-Y.; Zhou, X.-B.; Zhong, S. Tetrodotoxin alleviates acute heroin withdrawal syndrome: A multicentre, randomized, double-blind, placebo-controlled study. Clin. Exp. Pharmacol. Physiol. 2011, 38, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.S. Natural products to drugs: Natural product derived compounds in clinical trials. Nat. Prod. Rep. 2005, 22, 162–195. [Google Scholar] [CrossRef] [PubMed]
- Mantegazza, M.; Curia, G.; Biagini, G.; Ragsdale, D.S.; Avoli, M. Voltage-gated sodium channels as therapeutic targets in epilepsy and other neurological disorders. Lancet Neurol. 2010, 9, 413–424. [Google Scholar] [CrossRef]
- Belardinelli, L.; Dhalla, A. Sodium Channel Blockers Reduce Glucagon Secretion. U.S. Patent US 20140221286 A1, 20 September 2012. [Google Scholar]
- Kalaitzis, J.A.; Chau, R.; Kohli, G.S.; Murray, S.A.; Neilan, B.A. Biosynthesis of toxic naturally-occurring seafood contaminants. Toxicon 2010, 56, 244–258. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Akai, S.; Shoji, H.; Sugita, N.; Yoshida, S.; Nagai, Y.; Suzuki, K.; Nakamura, Y.; Kajihara, Y.; Funabashi, M.; et al. Stereoselective and efficient total synthesis of optically active tetrodotoxin from d-glucose. J. Org. Chem. 2008, 73, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Chau, J.; Ciufolin, M.A. The chemical synthesis of tetrodoxin: An ongoing quest. Mar. Drugs 2011, 9, 2046–2074. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lago, J.; Rodríguez, L.P.; Blanco, L.; Vieites, J.M.; Cabado, A.G. Tetrodotoxin, an Extremely Potent Marine Neurotoxin: Distribution, Toxicity, Origin and Therapeutical Uses. Mar. Drugs 2015, 13, 6384-6406. https://doi.org/10.3390/md13106384
Lago J, Rodríguez LP, Blanco L, Vieites JM, Cabado AG. Tetrodotoxin, an Extremely Potent Marine Neurotoxin: Distribution, Toxicity, Origin and Therapeutical Uses. Marine Drugs. 2015; 13(10):6384-6406. https://doi.org/10.3390/md13106384
Chicago/Turabian StyleLago, Jorge, Laura P. Rodríguez, Lucía Blanco, Juan Manuel Vieites, and Ana G. Cabado. 2015. "Tetrodotoxin, an Extremely Potent Marine Neurotoxin: Distribution, Toxicity, Origin and Therapeutical Uses" Marine Drugs 13, no. 10: 6384-6406. https://doi.org/10.3390/md13106384
APA StyleLago, J., Rodríguez, L. P., Blanco, L., Vieites, J. M., & Cabado, A. G. (2015). Tetrodotoxin, an Extremely Potent Marine Neurotoxin: Distribution, Toxicity, Origin and Therapeutical Uses. Marine Drugs, 13(10), 6384-6406. https://doi.org/10.3390/md13106384