New Antioxidative Secondary Metabolites from the Fruits of a Beibu Gulf Mangrove, Avicennia marina
Abstract
:1. Introduction

2. Results and Discussion
| Position | 1 | 2 | |||
|---|---|---|---|---|---|
| δC, Mult | δH (J in Hz) | δC, Mult | δH (J in Hz) | ||
| phenylethyl | 1 | 115.9, C | 121.4, C | ||
| 2 | 147.6, C | 148.6, C | |||
| 3 | 130.6, C | 103.9, CH | 6.16 (s) | ||
| 4 | 156.8, C | 143.7, C | |||
| 5 | 116.0, CH | 6.71 (d, 8.7) | 143.5, C | ||
| 6 | 131.0, CH | 7.07 (d, 8.7) | 110.1, CH | 6.41 (s) | |
| 7 | 36.3, CH2 | 2.81–2.84 (m) | 34.6, CH2 | 2.83–2.87 (m) | |
| 8 | 72.3, CH2 | 4.02–4.06 (m) | 72.3, CH2 | 4.00–4.04 (m) | |
| 3.70–3.74 (m) | 3.71–3.75 (m) | ||||
| glucosyl | 1′ | 104.2, CH | 4.38 (d, 7.9) | 103.5, CH | 4.78 (d, 7.3) |
| 2′ | 76.2, CH | 3.38 (dd, 9.1, 7.9) | 74.5, CH | 3.40 (dd, 9.1, 7.3) | |
| 3′ | 81.3, CH | 3.81 (t, 9.1) | 81.1, CH | 3.84 (t, 9.1) | |
| 4′ | 70.6, CH | 4.91 (t, 9.1) | 70.5, CH | 4.94 (t, 9.1) | |
| 5′ | 76.0, CH | 3.52–3.57 (m) | 76.1, CH | 3.50–3.55 (m) | |
| 6′ | 62.3, CH2 | 3.58–3.63 (m) | 60.3, CH2 | 3.61–3.65 (m) | |
| 3.49–3.54 (m) | 3.52–3.56 (m) | ||||
| rhamnosyl | 1″ | 103.0, CH | 5.19 (d, 1.6) | 102.0, CH | 4.58 (d, 1.6) |
| 2″ | 72.0, CH | 3.91 (dd, 3.5, 1.6) | 72.1, CH | 3.92 (dd, 3.1, 1.2) | |
| 3″ | 72.2, CH | 3.56 (dd; 9.5, 3.5) | 72.3, CH | 3.58 (dd; 9.7, 3.1) | |
| 4″ | 73.8, CH | 3.28 (t, 9.5) | 73.5, CH | 3.25 (t, 9.7) | |
| 5″ | 70.4, CH | 3.54–3.58 (m) | 70.6, CH | 3.55–3.59 (m) | |
| 6″ | 18.4, CH3 | 1.08 (d, 6.2) | 18.2, CH3 | 1.05 (d, 6.2) | |
| cinnamoyl in 1 | 1‴ | 127.8, C | 127.7, C | ||
| caffeoyl in 2 | 2‴ | 147.5, C | 115.3, CH | 7.00 (d, 1.5) | |
| 3‴ | 133.4, C | 143.6, C | |||
| 4‴ | 161.5, C | 145.6, C | |||
| 5‴ | 117.7, CH | 6.82 (d, 8.7) | 119.5, CH | 6.94 (d, 6.5) | |
| 6‴ | 130.9, CH | 7.48 (d, 8.7) | 123.2, CH | 6.60 (dd, 6.5, 1.5) | |
| 7‴ | 147.8, CH | 7.67 (d, 15.9) | 144.5, CH | 7.46 (d, 15.8) | |
| 8‴ | 115.6, CH | 6.35 (d, 15.9 ) | 115.3, CH | 6.39 (d, 15.8 ) | |
| 9‴ | 168.3, C | 169.2, C | |||
| Position | 3 | 4 | |||
|---|---|---|---|---|---|
| δC, Mult | δH (J in Hz) | δC, Mult | δH (J in Hz) | ||
| phenylethyl in 3 | 1 | 125.8, C | 134.3, C | ||
| cinnamoyl in 4 | 2 | 155.2, C | 127.9, CH | 7.60 (d, 7.6) | |
| 3 | 112.2, CH | 6.61 (s) | 128.6, CH | 7.40-7.45 (m, overlap) | |
| 4 | 144.4, C | 130.1, CH | 7.40-7.45 (m, overlap) | ||
| 5 | 144.2, C | 128.6, CH | 7.40-7.45 (m, overlap) | ||
| 6 | 115.1, CH | 6.64 (s) | 127.9, CH | 7.60 (d, 7.6) | |
| 7 | 35.1, CH2 | 2.78–2.82 (m) | 145.1, CH | 7.73 (d, 15.6) | |
| 8 | 60.8, CH2 | 3.92–3.96 (m) | 117.3, CH | 6.59 (d, 15.6) | |
| 3.68–3.72 (m) | |||||
| 9 | 166.7, C | ||||
| glucosyl | 1′ | 103.1, CH | 4.34 (d, 7.5) | 105.2, CH | 4.28 (d, 7.3) |
| 2′ | 74.0, CH | 3.37 (dd, 9.1, 7.5) | 74.6, CH | 3.33 (dd, 9.1, 7.3) | |
| 3′ | 76.5, CH | 3.81 (t, 9.1) | 82.4, CH | 4.09 (t, 9.1) | |
| 4′ | 70.3, CH | 4.91 (t, 9.1) | 70.6, CH | 4.51 (t, 9.1) | |
| 5′ | 73.6, CH | 3.54–3.57 (m) | 73.1, CH | 3.76–3.80 (m) | |
| 6′ | 63.2, CH2 | 3.58–3.63 (m) | 63.8, CH2 | 3.70–3.74 (m) | |
| 3.49–3.54 (m) | 3.54–3.59 (m) | ||||
| caffeoyl in 3 | 1″ | 129.1, C | 104.1, CH | 4.41 (d, 7.6) | |
| glucosyl in 4 | 2″ | 114.7, CH | 7.03 (d, 1.5) | 73.1, CH | 3.29 (dd, 9.1, 7.6) |
| 3″ | 145.1, C | 77.7, CH | 4.02 (t, 9.1) | ||
| 4″ | 147.8, C | 70.4, CH | 4.49 (t, 9.1) | ||
| 5″ | 115.1, CH | 7.01 (d, 7.5) | 71.7, CH | 3.72–3.76 (m) | |
| 6″ | 121.7, CH | 6.67 (dd, 7.5, 1.5) | 62.7, CH2 | 3.69–3.72 (m) | |
| 3.55–3.59 (m) | |||||
| 7″ | 145.6, CH | 7.56 (d, 17.2) | |||
| 8″ | 114.7, CH | 6.30 (d, 17.2) | |||
| 9″ | 167.7, C |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation
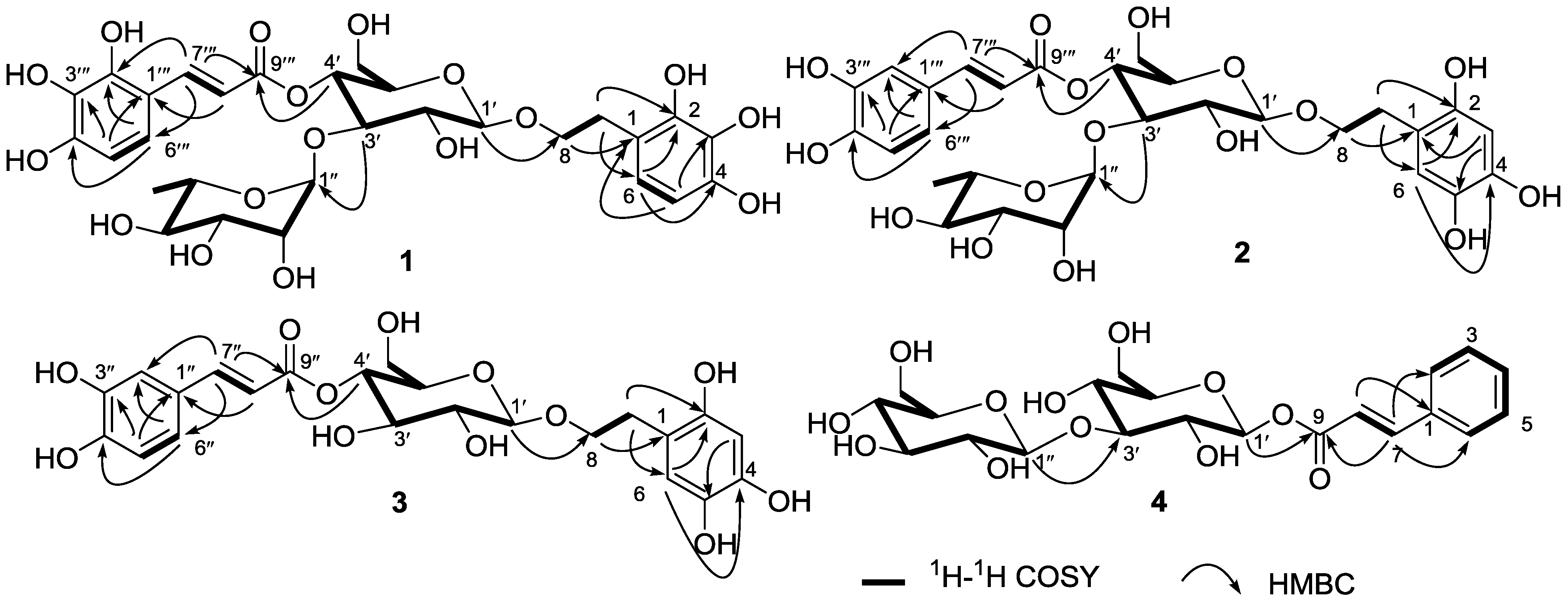
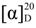 −24.7° (c 0.74, MeOH); UV (MeOH) λmax (log ε) nm: 214 (4.12), 246 (4.00), 291 (4.12), 331 (3.75); IR (KBr) νmax 3381, 1691, 1621 and 1602 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 1; HRESIMS m/z 657.2029 (calcd. for C29H36O17 + H, 657.2031).
−24.7° (c 0.74, MeOH); UV (MeOH) λmax (log ε) nm: 214 (4.12), 246 (4.00), 291 (4.12), 331 (3.75); IR (KBr) νmax 3381, 1691, 1621 and 1602 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 1; HRESIMS m/z 657.2029 (calcd. for C29H36O17 + H, 657.2031).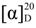 −35.5° (c 0.39, MeOH); UV (MeOH) λmax (log ε) nm: 215 (3.75), 241 (3.97), 289 (4.01), 332 (3.67); IR (KBr) νmax 3386, 1693, 1622 and 1600 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 1; HRESIMS: m/z 663.1898 (calcd. for C29H36O16 + Na, 663.1901).
−35.5° (c 0.39, MeOH); UV (MeOH) λmax (log ε) nm: 215 (3.75), 241 (3.97), 289 (4.01), 332 (3.67); IR (KBr) νmax 3386, 1693, 1622 and 1600 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 1; HRESIMS: m/z 663.1898 (calcd. for C29H36O16 + Na, 663.1901).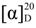 −32.2° (c 0.46, MeOH); UV (MeOH) λmax (log ε) nm: 214 (4.09), 245 (4.01), 290 (4.12), 332 (3.23); IR (KBr) νmax 3392, 1693, 1624 and 1600 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 2; HRESIMS: m/z 495.1501 (calcd. for C23H26O12 + H, 495.1503).
−32.2° (c 0.46, MeOH); UV (MeOH) λmax (log ε) nm: 214 (4.09), 245 (4.01), 290 (4.12), 332 (3.23); IR (KBr) νmax 3392, 1693, 1624 and 1600 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 2; HRESIMS: m/z 495.1501 (calcd. for C23H26O12 + H, 495.1503).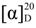 +13.2° (c 0.57, MeOH); UV (MeOH) λmax (log ε) nm: 211 (3.98), 245 (3.91), 289 (4.10); IR (KBr) νmax 3402, 1690, 1620 and 1602 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 2; HRESIMS: m/z 495.1475 (calcd. for C21H28O12 + Na, 495.1473).
+13.2° (c 0.57, MeOH); UV (MeOH) λmax (log ε) nm: 211 (3.98), 245 (3.91), 289 (4.10); IR (KBr) νmax 3402, 1690, 1620 and 1602 cm−1; 1H (CD3OD, 600 MHz) and 13C (CD3OD, 150 MHz) NMR; see Table 2; HRESIMS: m/z 495.1475 (calcd. for C21H28O12 + Na, 495.1473).3.4. Acid Hydrolysis of 1–4
3.5. Cellular Antioxidant Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yi, X.X.; Chen, Y.; Xie, W.P.; Xu, M.B.; Chen, Y.N.; Gao, C.H.; Huang, R.M. Four new jacaranone analogs from the fruits of a Beibu Gulf mangrove Avicennia marina. Mar. Drugs 2014, 12, 2515–2525. [Google Scholar] [CrossRef]
- Tanaka, T.; Ikeda, T.; Kaku, N.; Zhu, X.H.; Okawa, M.; Yokomizo, K.; Uyeda, M.; Nohara, T. A new lignan glycoside and phenylethanoid glycosides from Strobilanthes cusia BREMEK. Chem. Pharm. Bull. 2004, 52, 1242–1245. [Google Scholar] [CrossRef]
- Ono, M.; Yoshida, A.; Ito, Y.; Nohara, T. Phenethyl alcohol glycosides and isopentenol glycoside from fruit of Bupleurum falcatum. Phytochemistry 1999, 51, 819–823. [Google Scholar]
- Matsumoto, M.; Koga, S.; Shoyama, Y.; Nishioka, I. Phenolic glycoside composition of leaves and callus-cultures of Digitalis purpurea. Phytochemistry 1987, 26, 3225–3227. [Google Scholar]
- Faller, A.L.K.; Fialho, E.; Liu, R.H. Cellular antioxidant activity of Feijoada whole meal coupled with an in vitro digestion. J. Agric. Food Chem. 2012, 60, 4826–4832. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gao, C.-H.; Yi, X.-X.; Xie, W.-P.; Chen, Y.-N.; Xu, M.-B.; Su, Z.-W.; Yu, L.; Huang, R.-M. New Antioxidative Secondary Metabolites from the Fruits of a Beibu Gulf Mangrove, Avicennia marina. Mar. Drugs 2014, 12, 4353-4360. https://doi.org/10.3390/md12084353
Gao C-H, Yi X-X, Xie W-P, Chen Y-N, Xu M-B, Su Z-W, Yu L, Huang R-M. New Antioxidative Secondary Metabolites from the Fruits of a Beibu Gulf Mangrove, Avicennia marina. Marine Drugs. 2014; 12(8):4353-4360. https://doi.org/10.3390/md12084353
Chicago/Turabian StyleGao, Cheng-Hai, Xiang-Xi Yi, Wen-Pei Xie, Yin-Ning Chen, Ming-Ben Xu, Zhi-Wei Su, Lian Yu, and Ri-Ming Huang. 2014. "New Antioxidative Secondary Metabolites from the Fruits of a Beibu Gulf Mangrove, Avicennia marina" Marine Drugs 12, no. 8: 4353-4360. https://doi.org/10.3390/md12084353
APA StyleGao, C.-H., Yi, X.-X., Xie, W.-P., Chen, Y.-N., Xu, M.-B., Su, Z.-W., Yu, L., & Huang, R.-M. (2014). New Antioxidative Secondary Metabolites from the Fruits of a Beibu Gulf Mangrove, Avicennia marina. Marine Drugs, 12(8), 4353-4360. https://doi.org/10.3390/md12084353




