In Vitro Antitumor Activity of Stellettin B, a Triterpene from Marine Sponge Jaspis stellifera, on Human Glioblastoma Cancer SF295 Cells
Abstract
:1. Introduction
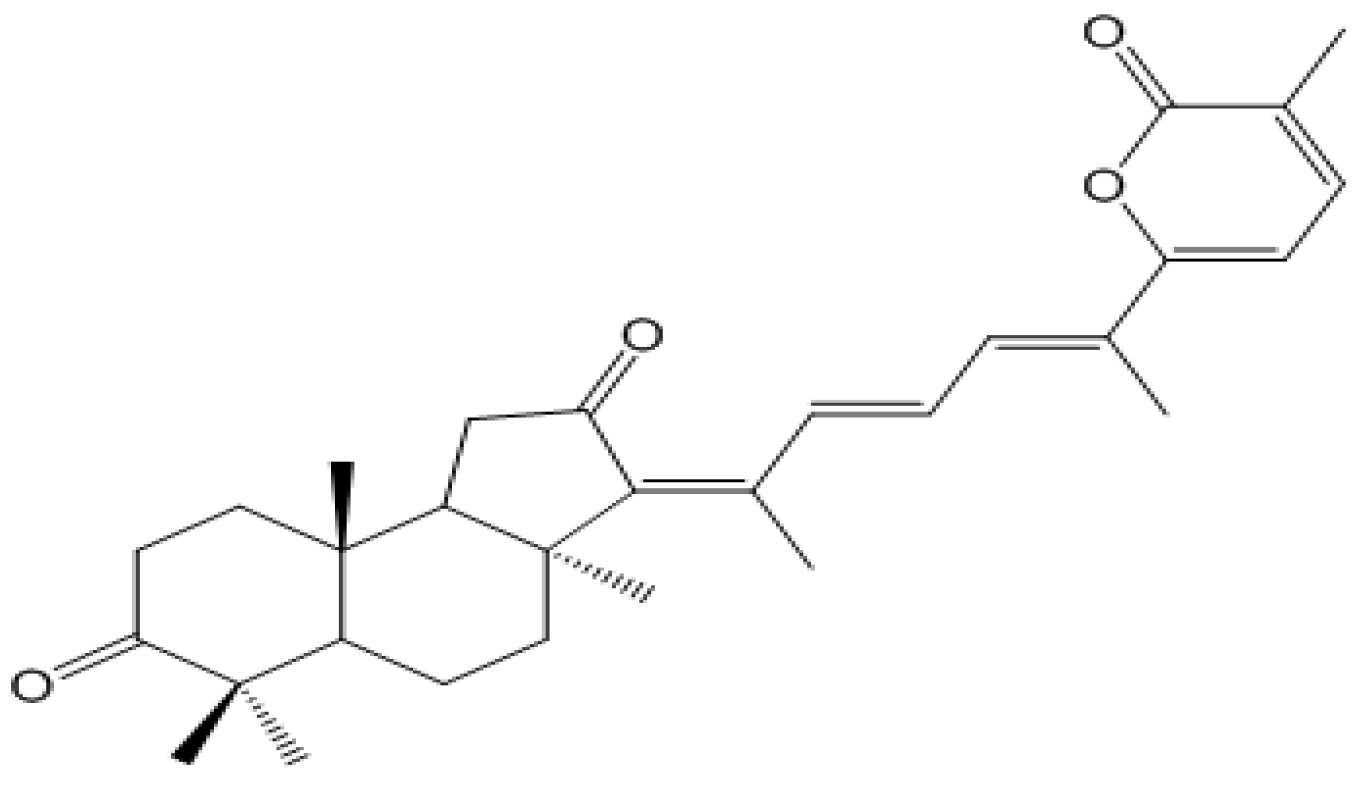
2. Results and Discussion
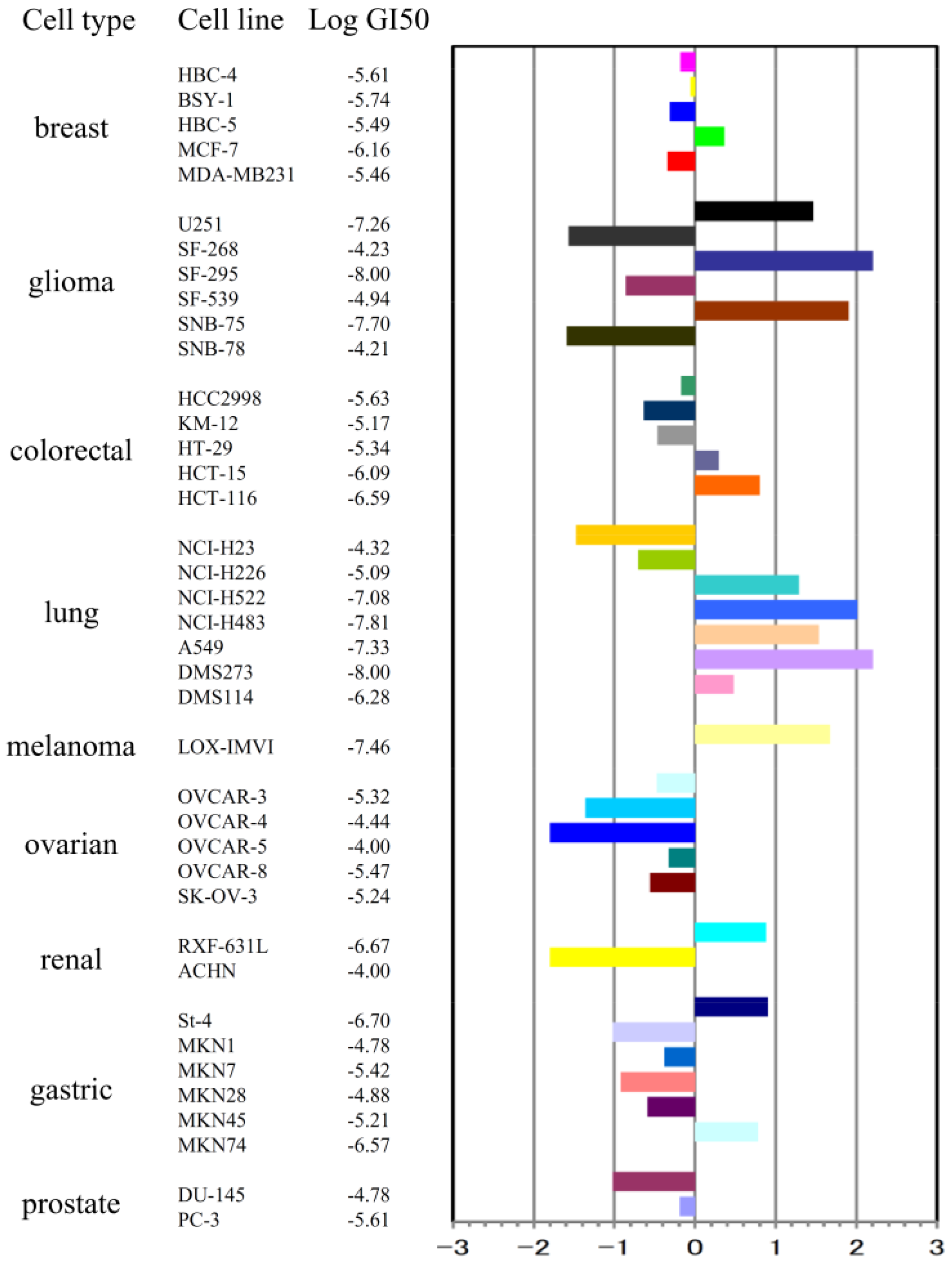
2.1. Stellettin B Inhibited Cell Growth of Various Tumor Cell Lines Including SF295
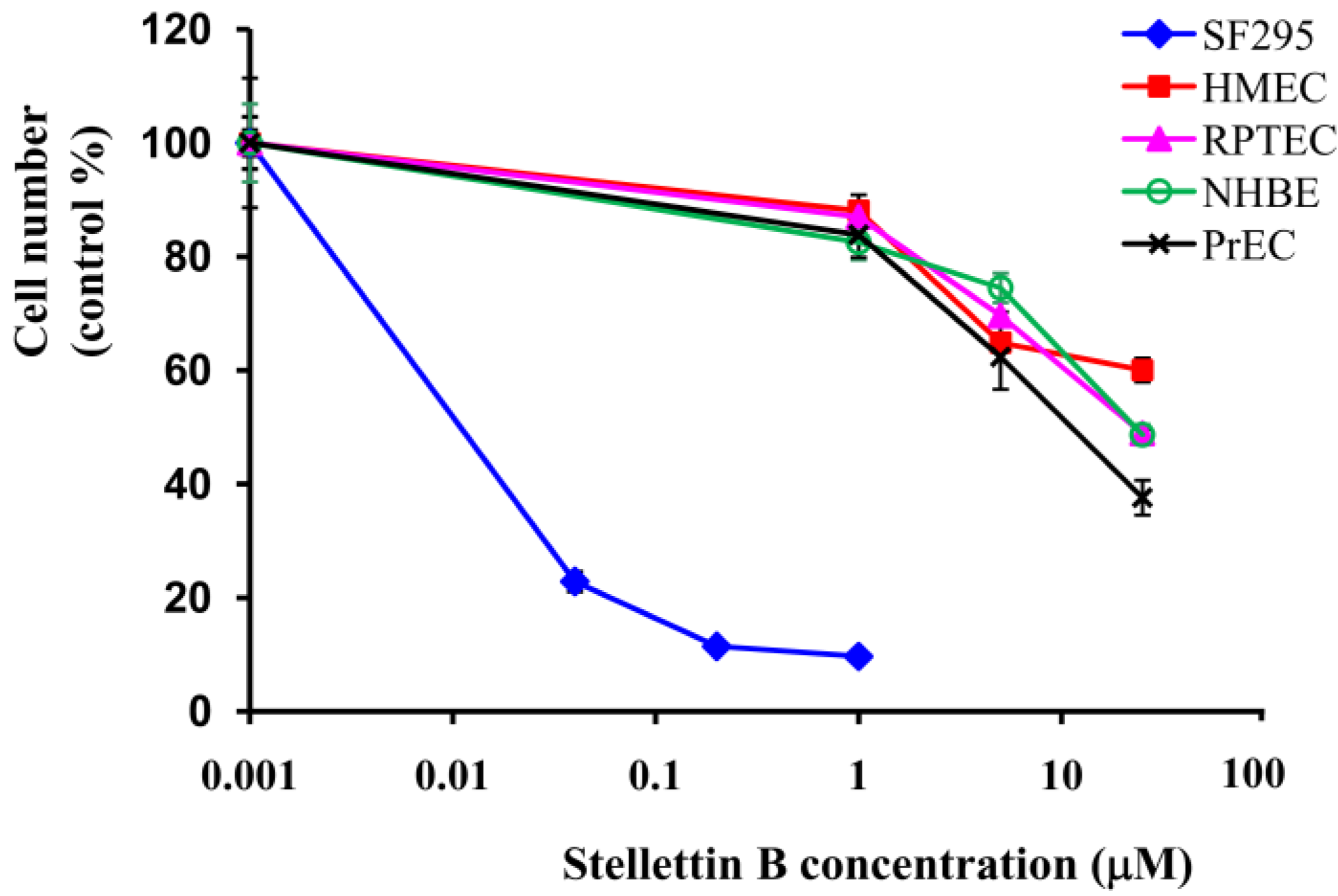
2.2. Stellettin B Showed High Selectivity in Growth Inhibition against SF295 Cells Compared with Normal Cells
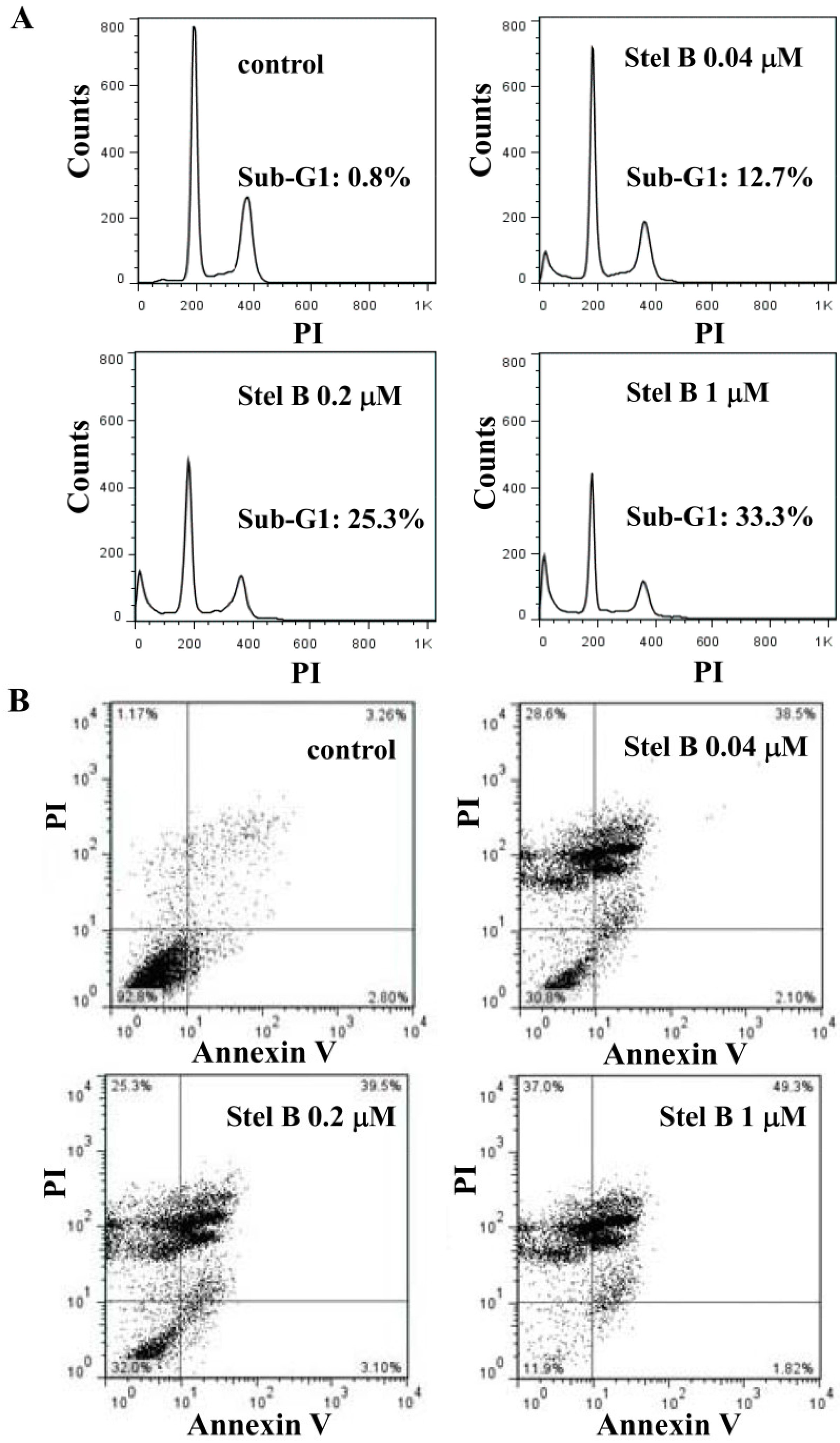
2.3. Stellettin B Induced Apoptosis in SF295 Cells
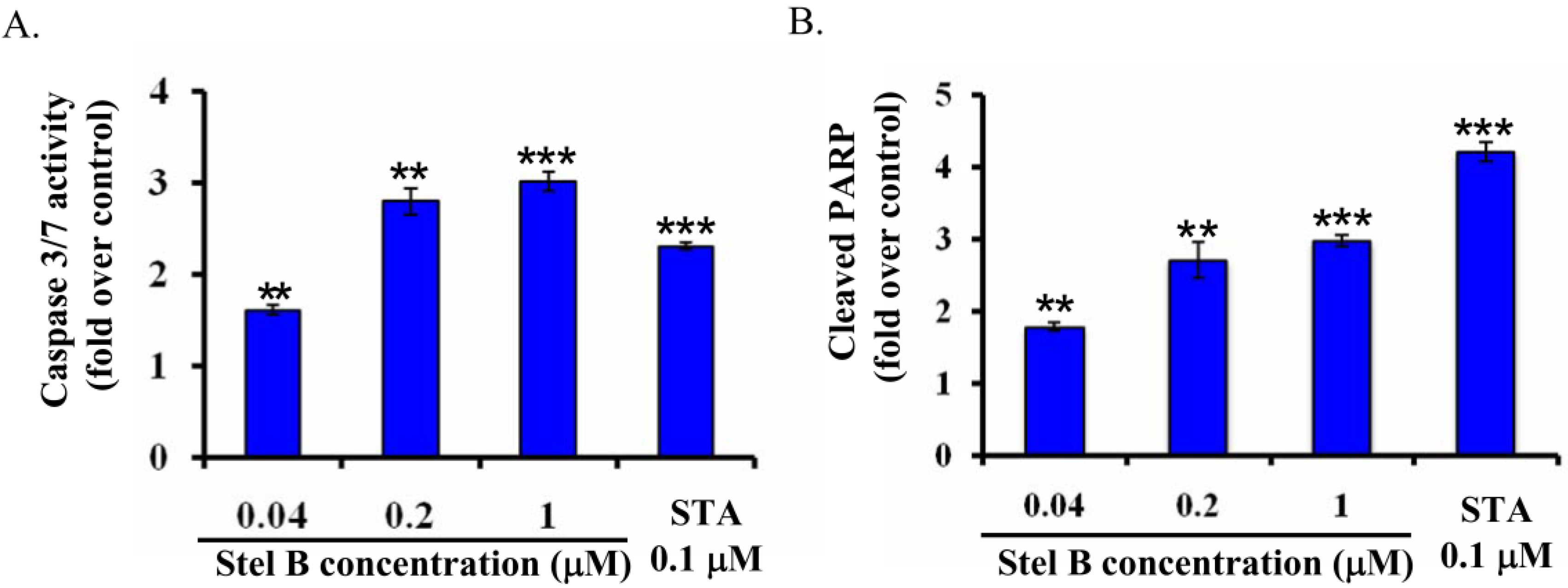
2.4. Stellettin B Increased Caspase 3/7 Activity and the Cleavage of Poly-(ADP-Ribose) Polymerase (PARP) in SF295 Cells
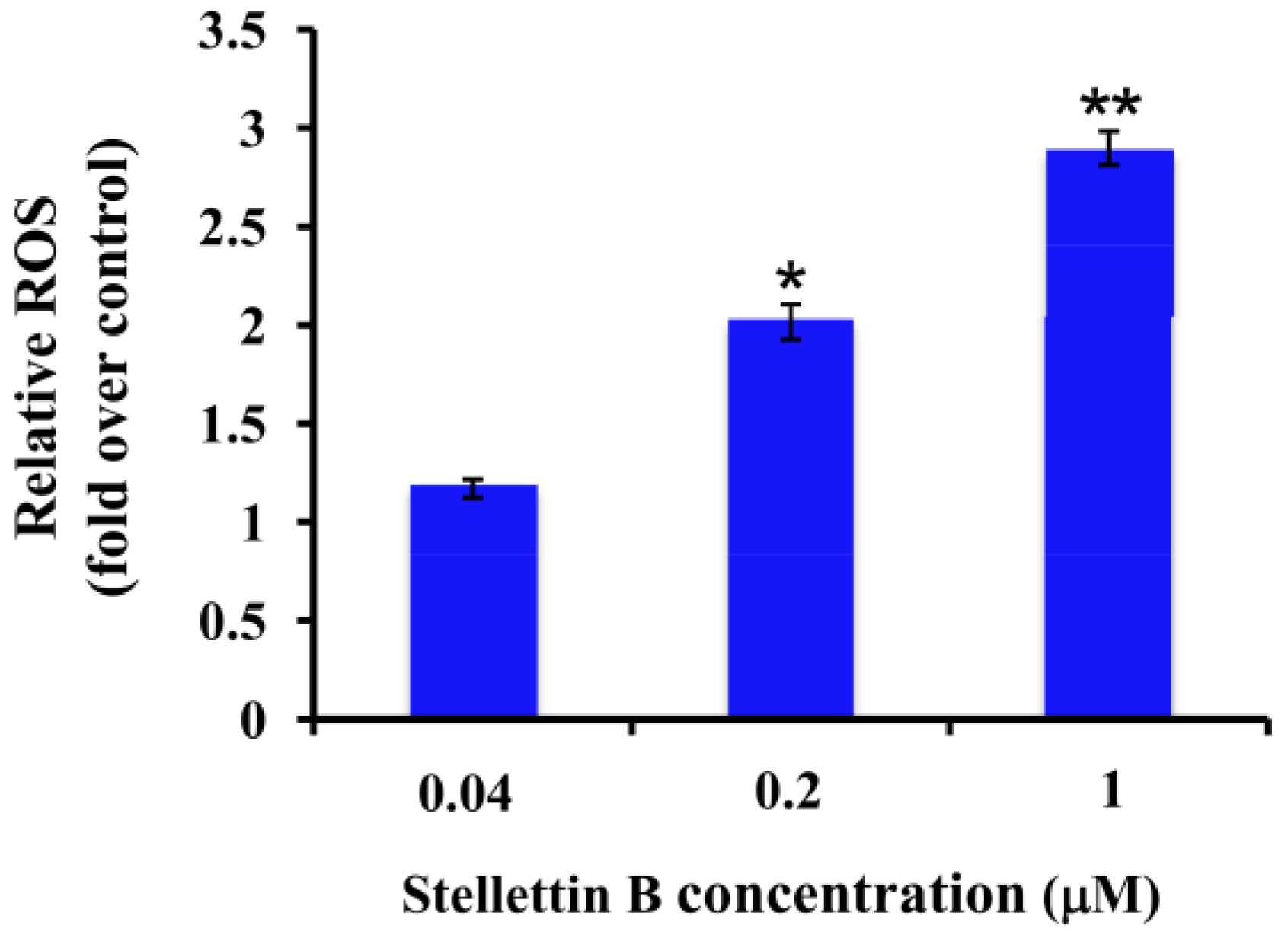
2.5. Stellettin B Increased the Reactive Oxygen Species (ROS) in SF295 Cells
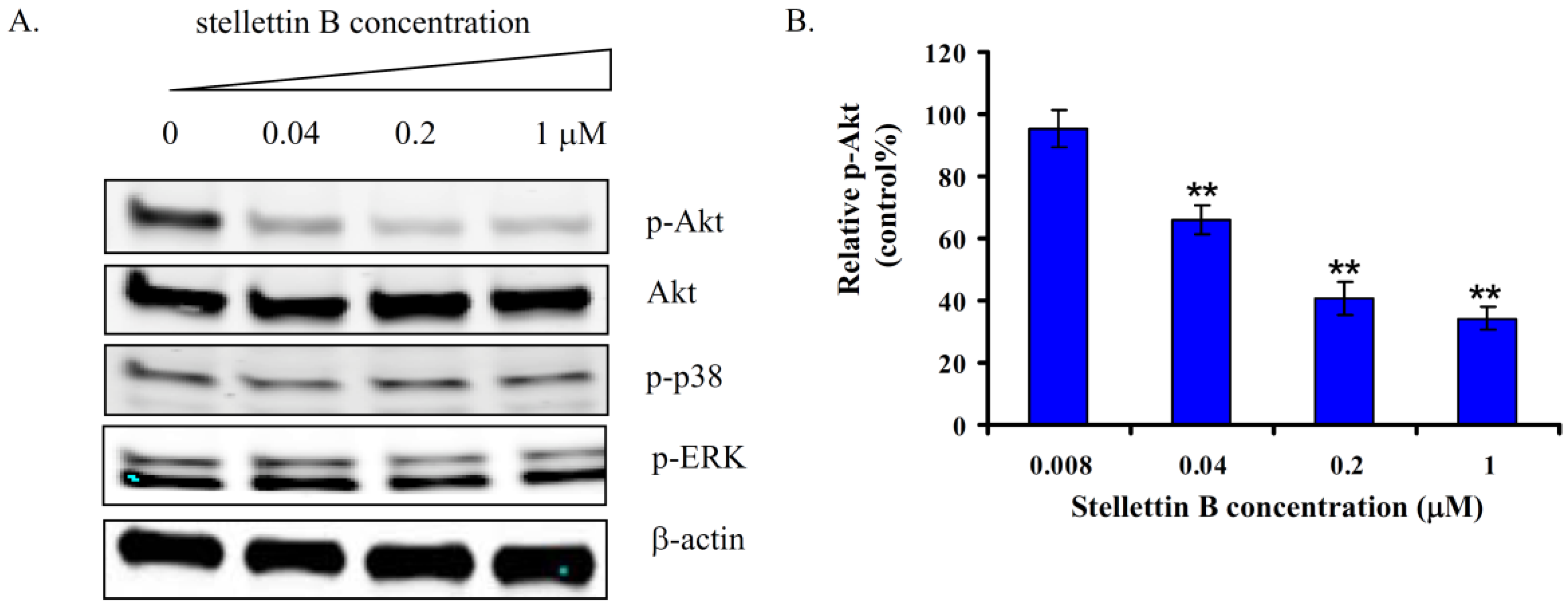
2.6. Stellettin B Inhibited Phosphorylation of Akt in SF295 Cells
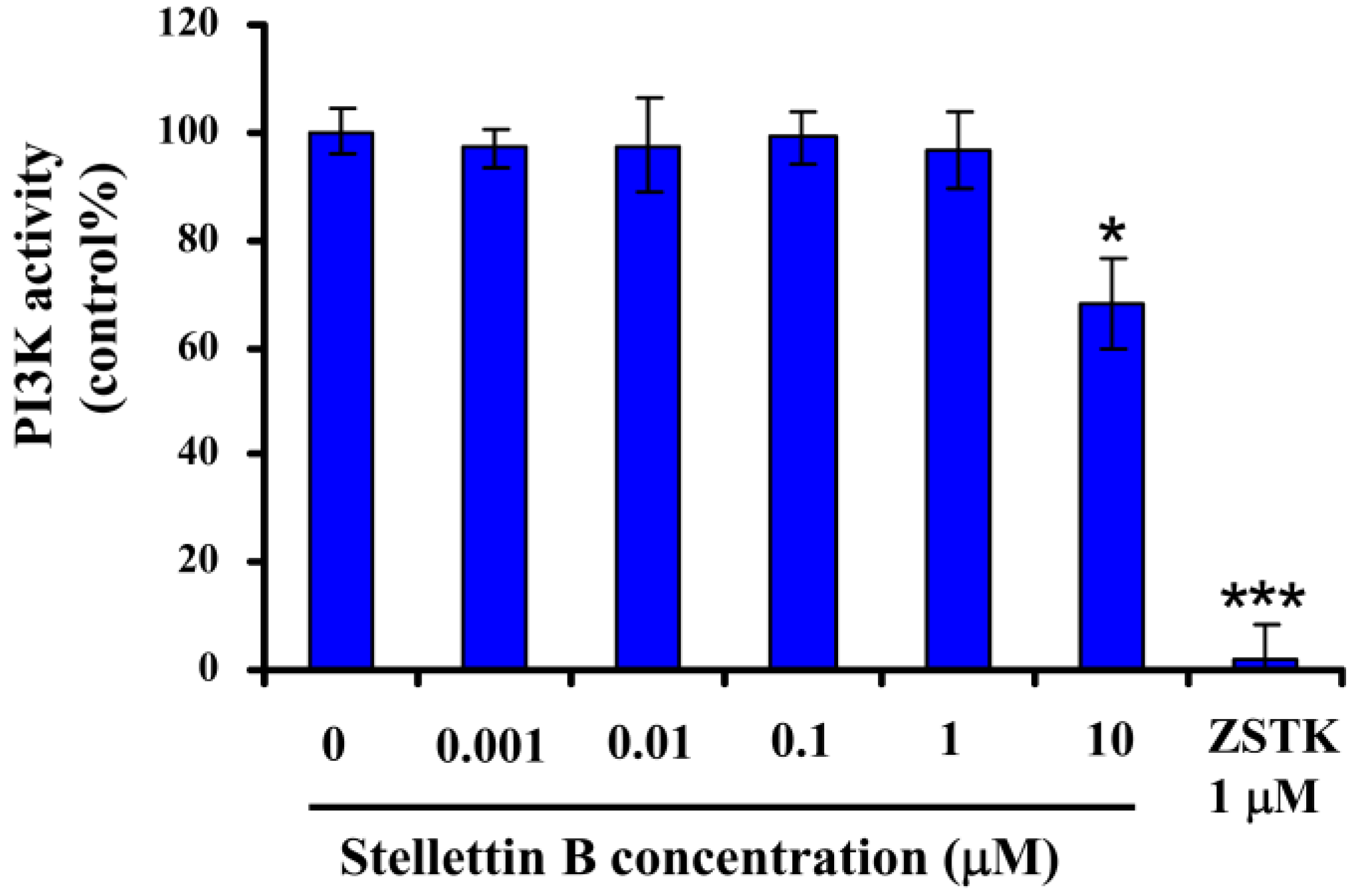
3. Experimental Section
3.1. Materials
3.2. Cell Lines and Cell Culture
3.3. Isolation and Identification of Stellettin B
3.4. Determination of Inhibitory Activity on Cell Growth of 39 Human Cancer Cell Lines and Plotting of JFCR39 Fingerprint
3.5. Determination of Inhibitory Activity on Growth of Normal Cells as well as SF295 Cancer Cells by WST Assay
3.6. Flow Cytometric Analysis of Cell Cycle Distribution and Apoptosis
3.7. Annexin V/PI Assay for Apoptosis
3.8. Caspase-Glo Assay
3.9. PathScan ELISA Assay
3.10. ROS Production Assay
3.11. Western Blot Analysis
3.12. Homogenous Time-Resolved Fluorescence (HTRF) PI3K Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kong, D.; Aoki, S.; Sowa, Y.; Sakai, T.; Kobayashi, M. Smenospongine, a sesquiterpene aminoquinone from a marine sponge, induces G1 arrest or apoptosis in different leukemia cells. Mar. Drugs 2008, 6, 480–488. [Google Scholar]
- Jin, M.; Zhao, W.; Zhang, Y.; Kobayashi, M.; Duan, H.; Kong, D. Antiproliferative effect of aaptamine on human chronic myeloid leukemia k562 cells. Int. J. Mol. Sci. 2011, 12, 7352–7359. [Google Scholar] [CrossRef]
- Kong, D.; Yamori, T.; Kobayashi, M.; Duan, H. Antiproliferative and antiangiogenic activities of smenospongine, a marine sponge sesquiterpene aminoquinone. Mar. Drugs 2011, 9, 154–161. [Google Scholar] [CrossRef]
- Preusser, M.; de Ribaupierre, S.; Wohrer, A.; Erridge, S.C.; Hegi, M.; Weller, M.; Stupp, R. Current concepts and management of glioblastoma. Ann. Neurol. 2011, 70, 9–21. [Google Scholar] [CrossRef]
- Kong, D.; Dan, S.; Yamazaki, K.; Yamori, T. Inhibition profiles of phosphatidylinositol 3-kinase inhibitors against PI3K superfamily and human cancer cell line panel JFCR39. Eur. J. Cancer 2010, 46, 1111–1121. [Google Scholar] [CrossRef]
- Kong, D.; Yamori, T. JFCR39, a panel of 39 human cancer cell lines, and its application in the discovery and development of anticancer drugs. Bioorg. Med. Chem. 2012, 20, 1947–1951. [Google Scholar] [CrossRef]
- Edwards, L.A.; Thiessen, B.; Dragowska, W.H.; Daynard, T.; Bally, M.B.; Dedhar, S. Inhibition of ILK in PTEN-mutant human glioblastomas inhibits PKB/Akt activation, induces apoptosis, and delays tumor growth. Oncogene 2005, 24, 3596–3605. [Google Scholar]
- Guo, W.Z.; Shiina, I.; Wang, Y.; Umeda, E.; Watanabe, C.; Uetake, S.; Ohashi, Y.; Yamori, T.; Dan, S. Ridaifen-SB8, a novel tamoxifen derivative, induces apoptosis via reactive oxygen species-dependent signaling pathway. Biochem. Pharmacol. 2013, 86, 1272–1284. [Google Scholar] [CrossRef]
- Zhang, X.D.; Gillespie, S.K.; Hersey, P. Staurosporine induces apoptosis of melanoma by both caspase-dependent and -independent apoptotic pathways. Mol. Cancer Ther. 2004, 3, 187–197. [Google Scholar] [CrossRef]
- Jagtap, P.; Szabo, C. Poly(ADP-ribose) polymerase and the therapeutic effects of its inhibitors. Nat. Rev. Drug Discov. 2005, 4, 421–440. [Google Scholar] [CrossRef]
- Circu, M.L.; Aw, T.Y. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic. Biol. Med. 2010, 48, 749–762. [Google Scholar] [CrossRef]
- Franke, T.F.; Kaplan, D.R.; Cantley, L.C. PI3K: Downstream AKTion blocks apoptosis. Cell 1997, 88, 435–437. [Google Scholar] [CrossRef]
- Franke, T.F. PI3K/Akt: Getting it right matters. Oncogene 2008, 27, 6473–6488. [Google Scholar] [CrossRef]
- Kong, D.; Yamori, T. Advances in development of phosphatidylinositol 3-kinase inhibitors. Curr. Med. Chem. 2009, 16, 2839–2854. [Google Scholar] [CrossRef]
- Kong, D.; Yamori, T. Phosphatidylinositol 3-kinase inhibitors: Promising drug candidates for cancer therapy. Cancer Sci. 2008, 99, 1734–1740. [Google Scholar]
- Kong, D.; Yamori, T. ZSTK474 is an ATP-competitive inhibitor of class I phosphatidylinositol 3 kinase isoforms. Cancer Sci. 2007, 98, 1638–1642. [Google Scholar]
- Liu, W.K.; Ho, J.C.; Che, C.T. Apoptotic activity of isomalabaricane triterpenes on human promyelocytic leukemia HL60 cells. Cancer Lett. 2005, 230, 102–110. [Google Scholar] [CrossRef]
- Tasdemir, D.; Mangalindan, G.C.; Concepcion, G.P.; Verbitski, S.M.; Rabindran, S.; Miranda, M.; Greenstein, M.; Hooper, J.N.; Harper, M.K.; Ireland, C.M. Bioactive isomalabaricane triterpenes from the marine sponge Rhabdastrella globostellata. J. Nat. Prod. 2002, 65, 210–214. [Google Scholar] [CrossRef]
- Lv, F.; Deng, Z.; Li, J.; Fu, H.; van Soest, R.W.; Proksch, P.; Lin, W. Isomalabaricane-type compounds from the marine sponge Rhabdastrella aff. distincta. J. Nat. Prod. 2004, 67, 2033–2036. [Google Scholar]
- Wei, S.Y.; Li, M.; Tang, S.A.; Sun, W.; Xu, B.; Cui, J.R.; Lin, W.H. Induction of apoptosis accompanying with G(1) phase arrest and microtubule disassembly in human hepatoma cells by jaspolide B, a new isomalabaricane-type triterpene. Cancer Lett. 2008, 262, 114–122. [Google Scholar] [CrossRef]
- Kong, D.X.; Yamori, T. ZSTK474, a novel phosphatidylinositol 3-kinase inhibitor identified using the JFCR39 drug discovery system. Acta Pharmacol. Sin. 2010, 31, 1189–1197. [Google Scholar] [CrossRef]
- Yamori, T.; Matsunaga, A.; Sato, S.; Yamazaki, K.; Komi, A.; Ishizu, K.; Mita, I.; Edatsugi, H.; Matsuba, Y.; Takezawa, K.; et al. Potent antitumor activity of MS-247, a novel DNA minor groove binder, evaluated by an in vitro and in vivo human cancer cell line panel. Cancer Res. 1999, 59, 4042–4049. [Google Scholar]
- McCabe Terrence, C.J. A triterpenoid pigment with the isomalabaricane skeleton from the marine sponge stelletta sp. Tetrahedron Lett. 1982, 23, 3307–3310. [Google Scholar] [CrossRef]
- Kong, D.; Okamura, M.; Yoshimi, H.; Yamori, T. Antiangiogenic effect of ZSTK474, a novel phosphatidylinositol 3-kinase inhibitor. Eur. J. Cancer 2009, 45, 857–865. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tang, S.-A.; Zhou, Q.; Guo, W.-Z.; Qiu, Y.; Wang, R.; Jin, M.; Zhang, W.; Li, K.; Yamori, T.; Dan, S.; et al. In Vitro Antitumor Activity of Stellettin B, a Triterpene from Marine Sponge Jaspis stellifera, on Human Glioblastoma Cancer SF295 Cells. Mar. Drugs 2014, 12, 4200-4213. https://doi.org/10.3390/md12074200
Tang S-A, Zhou Q, Guo W-Z, Qiu Y, Wang R, Jin M, Zhang W, Li K, Yamori T, Dan S, et al. In Vitro Antitumor Activity of Stellettin B, a Triterpene from Marine Sponge Jaspis stellifera, on Human Glioblastoma Cancer SF295 Cells. Marine Drugs. 2014; 12(7):4200-4213. https://doi.org/10.3390/md12074200
Chicago/Turabian StyleTang, Sheng-An, Qianxiang Zhou, Wen-Zhi Guo, Yuling Qiu, Ran Wang, Meihua Jin, Wenjing Zhang, Ke Li, Takao Yamori, Shingo Dan, and et al. 2014. "In Vitro Antitumor Activity of Stellettin B, a Triterpene from Marine Sponge Jaspis stellifera, on Human Glioblastoma Cancer SF295 Cells" Marine Drugs 12, no. 7: 4200-4213. https://doi.org/10.3390/md12074200
APA StyleTang, S.-A., Zhou, Q., Guo, W.-Z., Qiu, Y., Wang, R., Jin, M., Zhang, W., Li, K., Yamori, T., Dan, S., & Kong, D. (2014). In Vitro Antitumor Activity of Stellettin B, a Triterpene from Marine Sponge Jaspis stellifera, on Human Glioblastoma Cancer SF295 Cells. Marine Drugs, 12(7), 4200-4213. https://doi.org/10.3390/md12074200



