Abstract
The prevailing paradigm states that cancer cells acquire multiple genetic mutations in oncogenes or tumor suppressor genes whose respective activation/up-regulation or loss of function serve to impart aberrant properties, such as hyperproliferation or inhibition of cell death. However, a tumor is now considered as an organ-like structure, a complex system composed of multiple cell types (e.g., tumor cells, inflammatory cells, endothelial cells, fibroblasts, etc.) all embedded in an inflammatory stroma. All these components influence each other in a complex and dynamic cross-talk, leading to tumor cell survival and progression. As the microenvironment has such a crucial role in tumor pathophysiology, it represents an attractive target for cancer therapy. In this review, we describe the mechanism of action of trabectedin and plitidepsin as an example of how these specific drugs of marine origin elicit their antitumor activity not only by targeting tumor cells but also the tumor microenvironment.
1. Introduction
According to the prevailing paradigm of carcinogenesis, normal cells become cancer cells by acquiring multiple genetic mutations in oncogenes or tumor suppressor genes in which respective activation/up-regulation or loss of function served to impart aberrant properties, such as hyperproliferation, blockade of differentiation or inhibition of cell death [1,2,3]. However, a tumor is now considered as an organ-like structure, a complex system composed of multiple cell types (e.g., tumor cells, inflammatory cells, endothelial cells, fibroblasts, etc.) all embedded in an inflammatory stroma. All these components influence each other in a complex and dynamic cross-talk, leading to tumor survival and progression [4]. For example, growth factors and cytokines released by non-tumor cells support malignant cell progression and contribute to suppress the immune system inside tumors. Therefore, the microenvironment of a tumor is an integral part of its anatomy and physiology, and functionally, one cannot dissociate this microenvironment from what have traditionally been called “cancer cells” [5,6].
As the microenvironment has such a crucial role in tumor pathophysiology, it represents an attractive target for cancer therapy. There is already a wealth of information about specific cells and molecules in the tumor microenvironment that can be used as targets for cancer therapy at present [7,8]. For this reason, new molecules with antitumor features are looked at with new eyes. Anti-angiogenic therapies, for example, have been shown to improve clinical outcome in patients with different tumor types; similarly, signaling by the extracellular matrix (ECM) and its receptors are also becoming attractive targets of therapy, especially, as it is clear that ECM receptors and growth factor receptors cooperate to maintain tissue specificity. In this review, we describe the mechanism of action of trabectedin and plitidepsin as an example of how drugs of marine origin elicit their antitumor activity by targeting tumor cells but also the tumor microenvironment.
2. The Tumor Microenvironment
Organs are composed of parenchymal cells that perform the main organ function and the stroma that gives the supportive framework. In normal tissues, the parenchyma is formed by cells inter-connected by cell-cell junctions that define cell morphology, proliferation, migration, adhesion, differentiation, and cell death [9]. On the other hand, the stroma includes the ECM, fibroblasts, migratory immune cells, and neural elements supported by a vascular network, all within a milieu of cytokines and growth factors. The interstitial matrix is an intricate and highly dynamic network of fibers composed of glycosaminoglycan (GAG)-containing glycoproteins. Different types of fibrous collagen together with fibronectin, hyaluronan, and proteoglycans confer mechanical strength, elasticity and a precise spatial organization to tissues. Organ architecture and function are maintained by dynamic interactions between parenchymal cells and their microenvironment [10]. Both components, cells and stroma, communicate via complex autocrine, juxtacrine, and paracrine mechanisms consisting of soluble molecules, including growth factors, cytokines, hormones, and proteases, insoluble factors, such as ECM components, and direct cell-cell interactions. For example, the ECM contains a wide range of growth factors that are bound in an inactive form to matricellular proteins, but can be rapidly released and activated in case of need, for example during tissue repair.
As any organ, tumors are also composed of cells and stroma. However, both these components are altered. Tumor cells are characterized by the presence of mutations that alter their normal functioning but also their relationship with the surrounding microenvironment. For example, as cancer cells begin to expand, they produce factors that activate myofibroblasts and recruit tumor-associated fibroblasts. These mesenchymal cells, as well as adipocytes, are responsible for many of the tumor-associated changes in the extracellular matrix that promote and foster tumor progression [11]. Normal cells are also active participants that shape the features of tumors. Inflammatory cells, including neutrophils, and macrophages, are frequently the first immune cells recruited to the tumor, and may be either tumor-promoting or tumor-inhibiting depending on their degree of activation and polarization [5,12]. Another inflammatory cell type, the mast cell, is also recruited early and promotes tumor progression by releasing proteases that activate angiogenesis [13]. Myeloid-derived suppressor cells inhibit T-cell activation and promote angiogenesis, cancer cell invasion, and metastasis [14]. Natural killer cells and different types of T cells may have either pro- or antitumor functions, depending on their mode of activation. Immunoglobulins released by B cells promote tumor growth by initiating the inflammatory response. Finally, cytokines and chemokines produced by tumor cells, leukocytes and other cell types are key orchestrators of the cancer-related inflammation and have long been associated with the recruitment of leukocytes in tumors and therefore with the sustained presence of inflammatory cells [15,16,17]. These molecules are involved in important biological processes that govern proliferation and migration of different cells, regulation of innate and adaptive immune responses, and angiogenic and repair programs of the tissues.
On the other hand, the nature of the ECM in the tumor is different from that of the host tissue. The tumor stroma is characterized by a remarkable subversion of the tissue architecture and by a different composition of some ECM components. For instance, whereas the host organ may have been relatively soft and pliable, tumors are often tough and fibrotic. It has recently been shown that these alterations in the mechanical properties of the tissue contribute to the malignant phenotype [18,19,20]. Ultrastructural and immunohistochemical analyses also revealed the up-regulation of several proteins, such as tenascin, decorin, byglican, α-smooth muscle actin, osteopontin, fibulin-1, fibronectin, and the appearance of spliced protein isoforms that are not normally expressed [21,22]. This changed architecture may promote cell invasion by enabling cells to migrate along the collagen fibers or by enhancing integrin signaling [19,23]. Moreover, these matricellular proteins are degraded by specific proteases, such as matrix metalloproteases (MMPs), cathepsins, hyaluronidases, heparanase, elastase, urokinase-type plasminogen activator (uPA), plasmin and others in which activity is generally increased in tumors [24,25]. Finally, tumor blood vessels are also different from those of the normal vasculature as they are irregular and dilated. Furthermore, perivascular cells in tumors are loosely associated with endothelial cells [26,27]. These changes result in abnormal blood flow and leaky blood vessels with extravasation of excess fluid and proteins from the capillaries. The increase in the interstitial fluid pressure generates areas of hypoxia and acidosis inside tumors.
In summary, both normal (fibroblasts, endothelial cells, lymphocytes, macrophages, mast cells, and other cell types) and cancer cells inhabit a complex cellular and matrix ecosystem all of which may interact via juxtacrine and paracrine mechanisms. The constant disruption of homeostasis by proliferating tumor cells produces a chronic inflammatory reaction [28]. However, the classic players in acute inflammation (granulocytes, macrophages, endothelial cells, and fibroblasts) that ordinarily lead to the resolution of a wound through an orderly series of events, instead react paradoxically to the presence of dysfunctional epithelial cells by promoting their survival and replication [28]. This process also includes inflammatory angiogenesis and tissue remodeling. Thus, from this perspective, the microenvironment becomes an integral, essential part of a tumor [29,30].
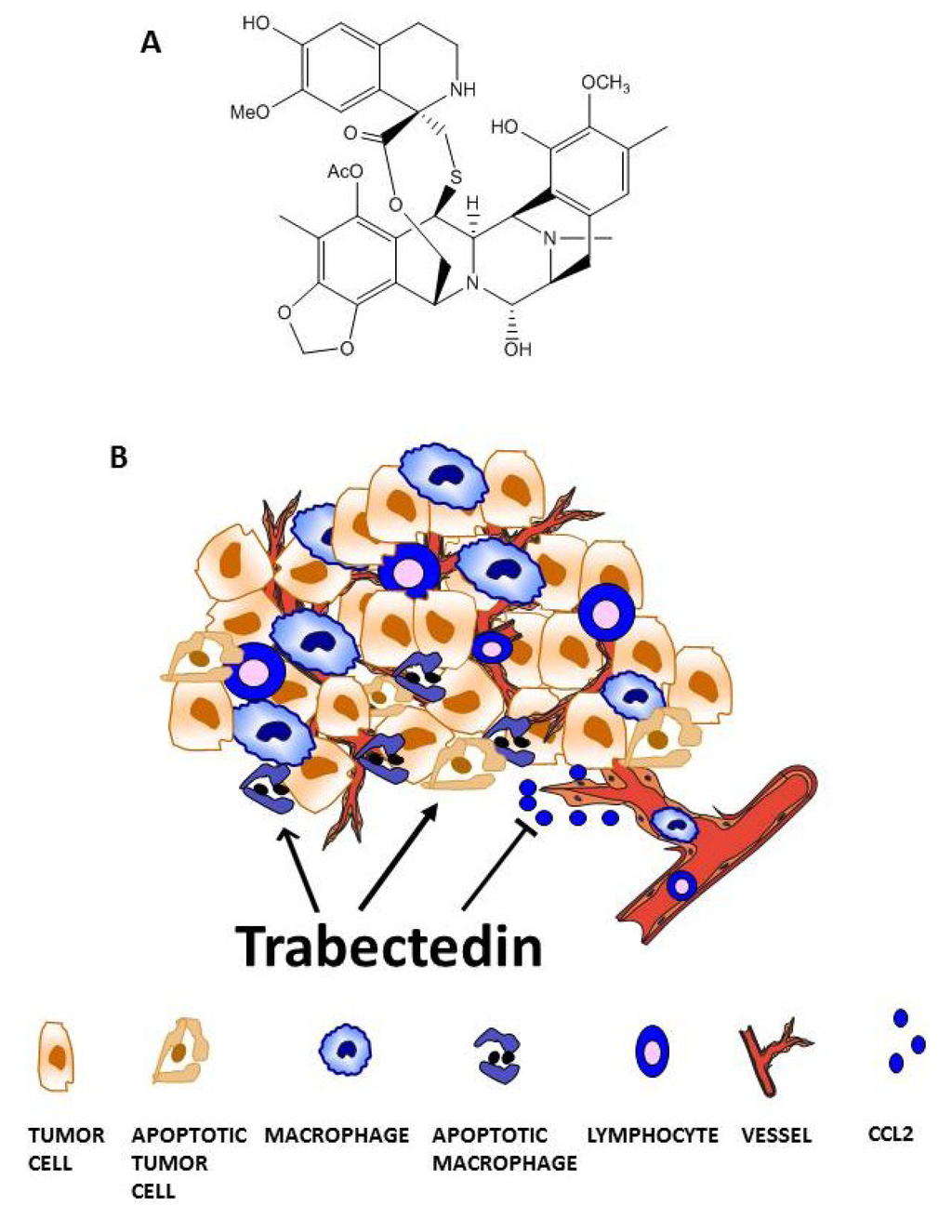
Figure 1.
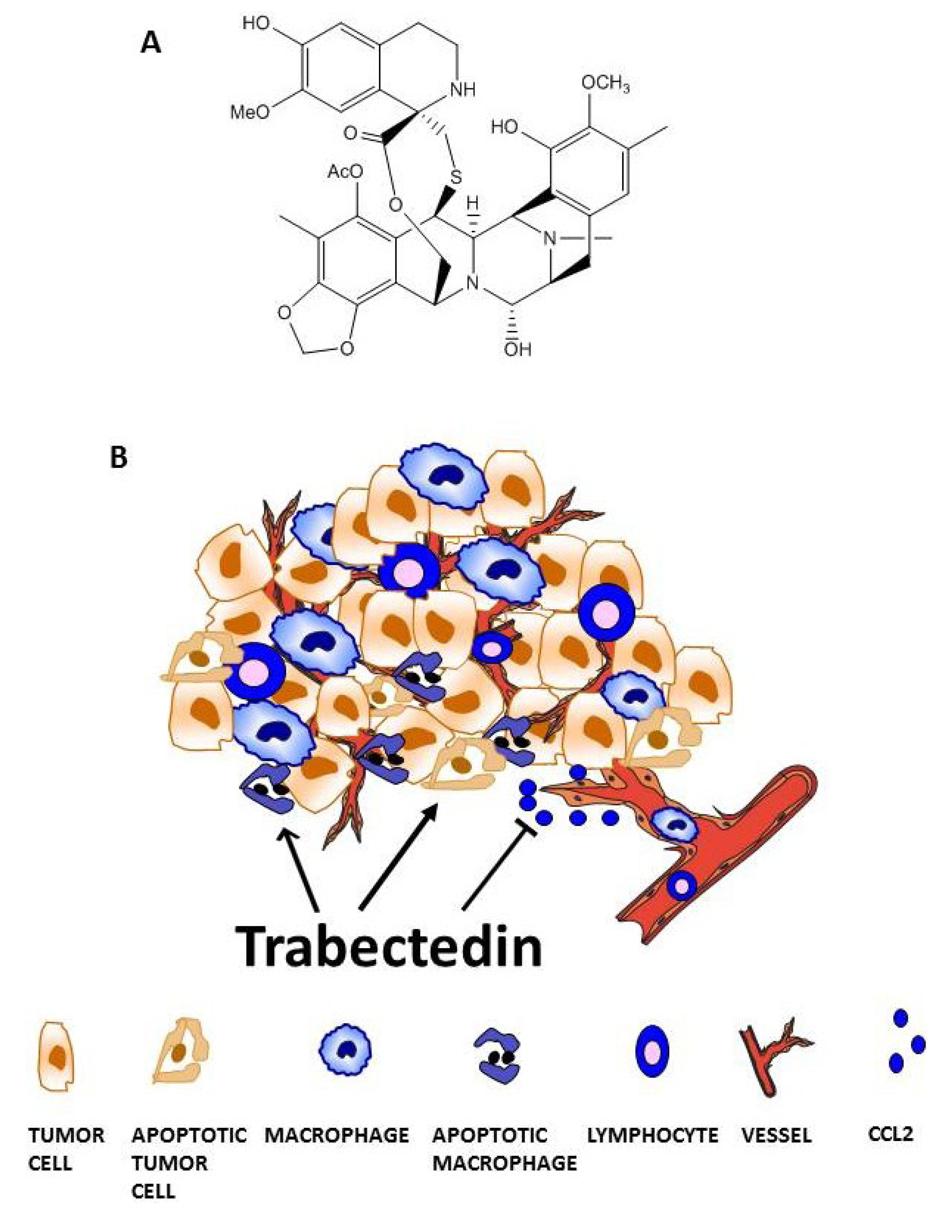
Trabectedin targets tumor-associated macrophages (TAMs). (A) Chemical structure. (B) Trabectedin acts on the tumor microenvironment by directly affecting monocytes and TAMs or indirectly by inhibiting the secretion of inflammatory mediators involved in different pathophysiological processes, such as inflammatory cell recruitment or tumor angiogenesis.
Figure 1.
Trabectedin targets tumor-associated macrophages (TAMs). (A) Chemical structure. (B) Trabectedin acts on the tumor microenvironment by directly affecting monocytes and TAMs or indirectly by inhibiting the secretion of inflammatory mediators involved in different pathophysiological processes, such as inflammatory cell recruitment or tumor angiogenesis.
3. Trabectedin Targets Tumor-Associated Macrophages (TAMs)
Trabectedin is an anticancer drug that was originally isolated from the marine Caribbean tunicate Ecteinascidia turbinata and now is produced synthetically (Figure 1) [31]. Trabectedin is used for the treatment of soft tissue sarcoma patients and, in combination with pegylated liposomal doxorubicin, for relapsed platinum-sensitive ovarian cancer patients [32,33]. It is a well-tolerated drug; the most common adverse effects observed after administration are reversible neutropenia and reversible AST/ALT elevation. The mechanism of action of trabectedin on tumor cells has been reviewed elsewhere [31]. Of note, the drug shows unique and high-specific inhibition of the transcription process. Transcription inhibition, that affects both the transcribing and the non-transcribing strands, occurs by different mechanisms such as the displacement of specific transcription factors from their promoters, the stabilization of the DNA duplex structure, a direct interaction with RNA Pol II or the induction of the massive degradation of transcribing Pol II [34,35,36,37]. For example, through transcription inhibition, trabectedin modulates the expression of downstream targets, alters tumor biology, and induces the resumption of natural cellular differentiation in sarcomas resulting from the dysregulation of transcription factors, such as EWS-Fli1 fusion protein and FUS-CHOP [31,38,39,40].
In addition to its activity on tumor cells, trabectedin also targets key processes in the biology of tumors, indicating that the drug is more versatile than currently available chemotherapeutic agents. First evidence came from the finding that, among all leukocytes, blood human monocytes and macrophages were hypersensitive to the drug [41]. This finding prompted a series of experiments to understand the exquisite selectivity of trabectedin for mononuclear phagocytes. It was demonstrated that the drug rapidly triggered the activation of caspase 8, downstream of membrane TRAIL receptors (TRAIL-R) [42]. Leukocyte subsets have different sets of TRAIL-R. Monocytes and macrophages mainly express the signaling TRAIL-Rs and are sensitive to trabectedin. In contrast, neutrophils and T lymphocytes preferentially express the non-signaling TRAIL-R (which prevents caspase 8 activation) and are therefore non-susceptible to trabectedin. On the other hand, low, non-cytotoxic concentrations of trabectedin not only inhibit monocyte differentiation into TAMs, but also the production of specific inflammatory mediators, such as CCL2, IL-6, VEGF, and CXC chemokine ligand-8 (CXCL8). This effect was observed particularly in monocytes, TAMs, myxoid liposarcoma cells and ovarian cancer cells [12,31,41]. Other chemokines involved in monocyte recruitment are also transcriptionally affected by trabectedin treatment (e.g., CCL7, CCL3, and CCL14). Importantly, all these effects are not reported for chemotherapeutic agents other than trabectedin (e.g., cisplatin, doxorubicin) [31,41]. Trabectedin also affected the expression of ECM-related genes produced by TAMs and fibroblast, such as fibronectin, osteopontin and matrix-metallo protease-2 (MMP2) or collagen type 1 [43,44]. These results indicate that trabectedin may reduce the high turnover of the tumor stroma.
As described previously, macrophages are a major cellular component of human tumors, where they are commonly termed tumor-associated macrophages (TAMs). These TAMs are derived from monocytes recruited into tumors by chemokines secreted by both malignant and stromal cells [45]. As macrophages, TAMs are versatile cells that are capable of displaying different functional activities. Based on their plasticity, macrophages can be classified in two extreme types: “classical” (or M1) and “alternative” (or M2) [46,47]. After stimulation with interferon gamma (IFN-γ), granulocyte-monocyte-colony stimulating factor (GM-CSF) and tumor necrotic factor-α (TNF-α), M1 macrophages secrete high levels of pro-inflammatory cytokines, such as interleukin-12 (IL-12), interleukin-1 (IL-1), and interleukin-6 (IL-6) and have potent antitumor efficacy [48]. Alternatively, monocytes exposed to interleukin-4 (IL-4) and interleukin-13 (IL-13) become polarized toward the M2-type. This is characterized by higher production of the anti-inflammatory cytokine interleukin-10 (IL-10) and low expression of pro-inflammatory cytokines and amplification of metabolic pathways that can suppress adaptive immune responses [47,49,50]. M2-related activities favor disease progression [51,52,53]. For instance, M2-type TAMs promote tumor angiogenesis by releasing several angiogenic factors, such as vascular endothelial growth factor (VEGF) or platelet-derived growth factor (PDGF) [54]. TAMs also release chemokines (e.g., CCL17, CCL18, CCL22), which increase intratumoral recruitment of lymphoid cells without cytotoxic activity (T-helper 2 lymphocytes; Th2) or with suppressive activity (regulatory T cells; Treg) [55]. The impressive array of tumor-promoting functions is consistent with clinical studies showing high macrophage density in many human cancer types to be associated with increased tumor angiogenesis and metastasis, and/or a poor prognosis [56,57,58,59,60,61,62,63,64]. Thus, TAMs are key players of the tumor microenvironment that promote disease progression [12,53,65,66].
The above-mentioned inhibitory activity of trabectedin on TAMs is not only observed in vitro but also in various animal tumor models [42,67]. In those models, trabectedin significantly decreased the number of blood monocytes and of tumor-associated macrophages but not of other leukocyte subsets (e.g., neutrophils or lymphocytes) [42]. To prove that the cytotoxic activity on mononuclear phagocytes is an important mechanism of its anti-tumor efficacy, tumor cells resistant to trabectedin in vitro were injected into mice [42]. Interestingly, in the in vivo setting, trabectedin still showed anti-tumor activity, in spite of the confirmed tumor cell resistance to the drug when exposed in vitro. Tumor growth was significantly restored after administration of myeloid cells from tumor-bearing untreated mice after each drug treatment cycle. Therefore, macrophage targeting in vivo appear to be a key component of the anti-tumor activity of trabectedin. Effects other than macrophage depletion also account for its antitumor efficacy. Using human liposarcoma xenografts grown in nude mice, Trabectedin induces a significant decrease in the expression of relevant tumor mediators such as the chemokines CCL2 and CXCL8, IL-6 or the angiogenic factor VEGF, and an overall effect on the angiogenic network [42,67]. Thus, in addition to direct activity on mononuclear phagocytes, trabectedin reduces the secretion of inflammatory mediators affecting main tumor pathophysiological processes such as the recruitment of circulating monocytes into tumors or angiogenesis [41,42].
The key evidence that trabectedin presents selective activity against monocytes and TAMs finally came from experiments conducted on human samples from patients receiving the drug as part of their therapy. Firstly, a decrease in monocytes was observed within few days after injection of trabectedin in most patients with soft tissue sarcoma [42]. Furthermore, by comparing tumor sections collected before and after neo-adjuvant therapy, a dramatic decrease of macrophage infiltration and vessel network was evidenced, reinforcing the concept that this compound strikes both the neoplastic compartment and the tumor micro-environment. Similarly, monocytes and TAMs from ovarian cancer biopsies were sensitive to trabectedin at low nM concentrations and were much more sensitive than lymphocytes. Moreover, at those concentrations, trabectedin significantly inhibited chemokine (CCL2), inflammatory cytokine (IL-6), and angiogenic factor (e.g., angiopoietin-2, VEGF) production by monocytes, macrophages, and TAMs [31,41].
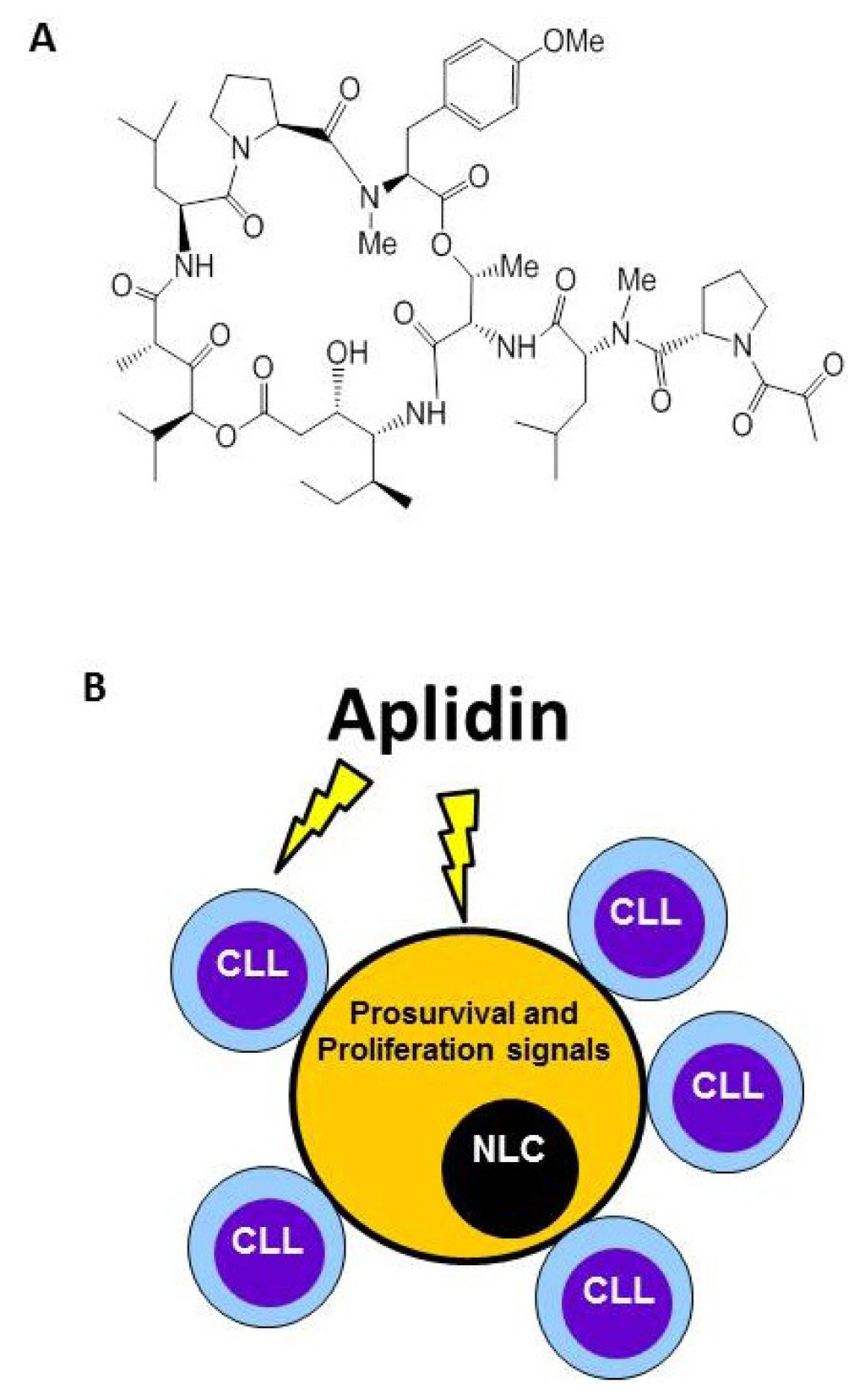
Figure 2.
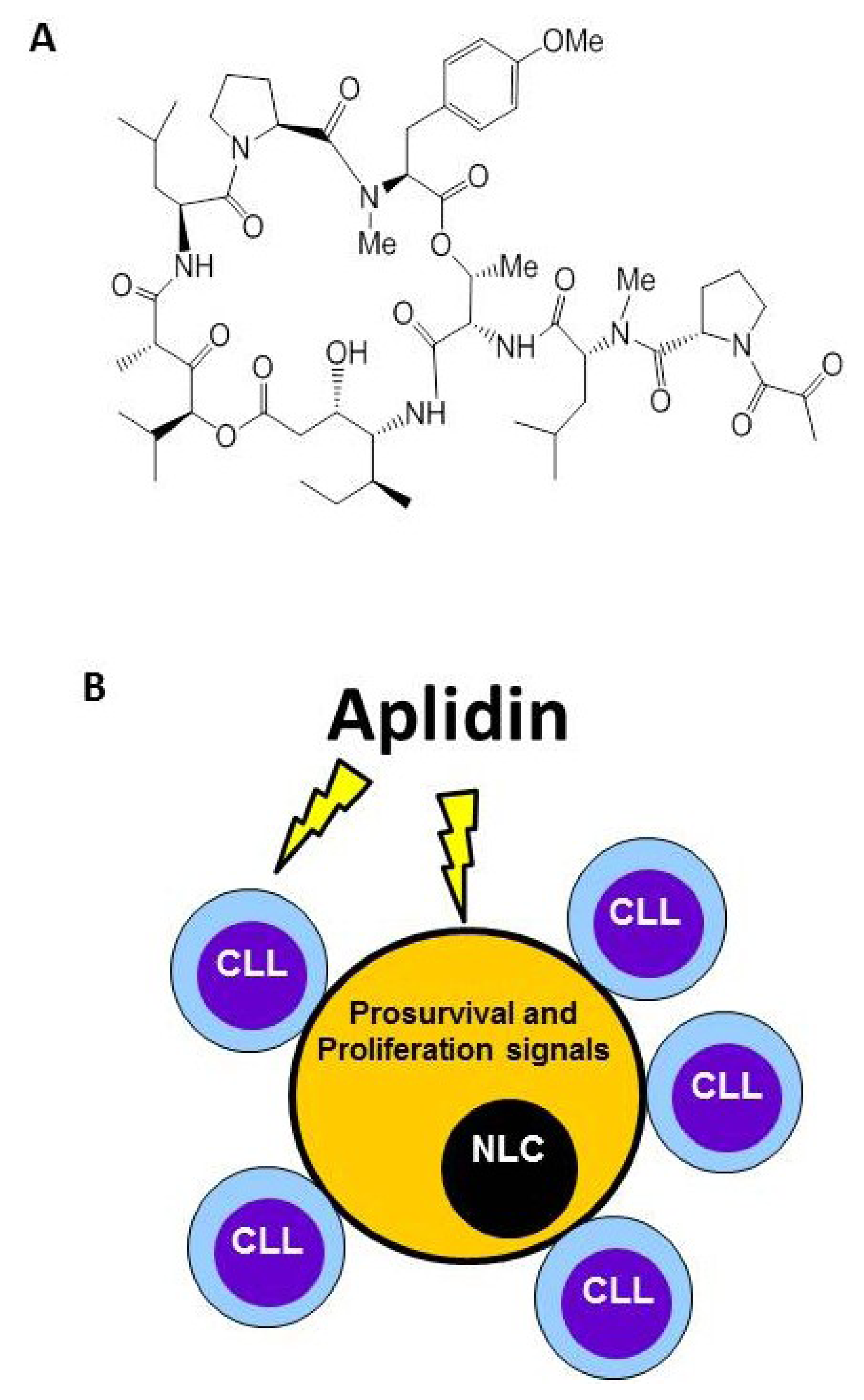
Plitidepsin (Aplidin) targets tumor-associated Nurse-Like cells (NLCs). (A) Chemical structure. (B) Besides inducing apoptosis of Chronic Lymphocytic Leukemia (CLL) cells, Plitidepsin also acts on monocytes and NLCs. NLCs promote survival of CLL cells by releasing pro-survival factors such as CXCL12.
Figure 2.
Plitidepsin (Aplidin) targets tumor-associated Nurse-Like cells (NLCs). (A) Chemical structure. (B) Besides inducing apoptosis of Chronic Lymphocytic Leukemia (CLL) cells, Plitidepsin also acts on monocytes and NLCs. NLCs promote survival of CLL cells by releasing pro-survival factors such as CXCL12.
4. Plitidepsin Targets Monocytes and Nurse-Like Cells (NLCs)
Plitidepsin (Aplidin) is a synthetic depsipeptide originally isolated from the tunicate Aplidium albicans and now obtained by total synthesis (Figure 2). The compound is currently being evaluated in a Phase III trial for the treatment of refractory/relapsed multiple myeloma patients (clinicaltrial.gov code: NCT01102426). Plitidepsin is a well-tolerated drug. The most common plitidepsin-related adverse events are nausea, fatigue, and myalgia. The mechanism of action of plitidepsin on tumor cells was reviewed elsewhere [68]. Briefly, after interacting with a moderately high-affinity binding site in the cell membrane, plitidepsin led to the rapid activation of Rac1 and the sustained activation of JNK and p38/MAPK that will finally provoke the appearance of a caspase-dependent apoptosis [68,69,70,71]. Results of several studies associated the activation of JNK by plitidepsin with an increased oxidative stress characterized by a rise in the levels of reactive oxygen species (ROS) and a fall in reduced glutathion GSH [71,72]. The oxidative stress induced by Plitidepsin also provokes an endoplasmic reticulum (ER) stress associated to cell death [73]. After cell treatment, plitidepsin triggers the activation of several key molecular components of the classical ER stress-induced unfolded protein response (UPR). These include the proteolytic processing of ATF6 and the alternative splicing of XBP1, as well as the phosphorylation of eIF2α and JNK. Plitidepsin also induces the decrease of CHOP protein levels, due to its rapid degradation by the ubiquitin/proteasome machinery.
In addition to the previously mentioned effects, plitidepsin is also acting on the tumor microenvironment. This is clearly observed on chronic lymphocytic leukemia models (CLL). It was recently published that besides its direct effects on CLL cells, plitidepsin exhibits a potent activity against monocytes and NLCs, a subset of cells derived from peripheral monocytes of CLL patients [74]. The high sensitivity of monocyte and monocytic-derived cells to plitidepsin indirectly affects leukemic cell survival by impairing the delivery of pro-survival signals from these cell populations. As with TAMs in solid tumors, NLCs promote survival of CLL cells and play a crucial role for CLL cell progression. Among other pro-survival factors, NLCs secrete CXCL12, a chemokine that not only induces the migration of CLL cells to lymphoid tissues but also protects them from spontaneous and drug-induced apoptosis in a contact-dependent fashion [75,76]. NLCs can be found in the spleen and secondary lymphoid tissue of patients with CLL [77]. In line with the protective function of NLCs on CLL cells, it was also observed that the previous treatment of isolated NLCs with Plitidepsin impairs the survival of CLL cells. Plitidepsin induced monocyte and NLCs death by triggering apoptosis, as evidenced by early exposure of phosphatidylserine in the outer leaflet of plasma membrane, the activation of caspase-3 and subsequent cleavage of PARP [74]. The production of ROS induced by plitidepsin plays a central role in monocyte death, as drug-induced apoptosis was blocked when preincubating monocytes with ebselen, a compound that increases intracellular GSH levels. It is currently known that ROS and reactive nitrogen species (RNS) regulate the molecular and biochemical pathways responsible for human monocyte survival and that any disbalance in this strict regulation (e.g., induction of oxidative stress), can be detrimental for these cells [78]. In this regard, it was reported that monocytes are more susceptible than lymphocytes to cell death triggered by oxidative stress [79]. These data indicate that, besides its direct antitumor activity on CLL cells, plitidepsin also affects other main drivers of CLL biology, such as monocytic-derived cells.
5. Conclusions
In the last decades the concept that the tumor microenvironment is simply a supporting structure for the preservation of tissue architecture has dramatically changed. Indeed, microenvironmental components provide signals affecting cell adhesion, migration, proliferation, differentiation, and death. Monocytic-derived cells are key players of the cancer-related inflammation present at tumor sites. Such a reactive milieu eventually supports tumor cell proliferation and the full-blown development of neo-angiogenesis. There is increasing evidence that successful anti-cancer therapies are not only dependent on the cancer phenotype but also on the normalization of the tumor stroma. In this view, the findings showing that trabectedin and plitidepsin have wider mechanisms of action acting not only on tumor cells but also modifying the whole microenvironment is of great interest and may contribute to the development of new drugs from marine origin as anticancer therapies.
The opportunity to combine direct antiproliferative activity on tumor cells with the capacity to counteract the pro-tumoral properties of the tumor microenvironment is emerging as an especially appealing therapeutic effect of some drugs of marine origin. As reviewed above, the antitumor activity of trabectedin and plitidepsin is not only related to their effects on tumor cells, but also on their ability to affect the tumor microenvironment, in particular monocytic-derived cell (TAMs and NLCs, respectively) and their pro-tumoral functions. Of note, the effects of trabectedin on tumor microenvironment are in line with response patterns evident in several patients, such as tumor shrinkage or delayed response and prolonged stabilization even in the absence of evident tumor shrinkage [80]. Trabectedin can therefore be considered a particularly important antitumor agent with mechanisms of action that make it especially appropriate for targeting key processes in the biology of cancer. Similar considerations can be taken into account for plitidepsin.
To our knowledge, there are no other reports showing this specific activity with other marine-derived drugs. Therefore, whether this is a common feature of marine natural products or a specific mechanism for trabectedin and plitidepsin remains to be elucidated.
6. Future Directions
Several questions need to be further addressed concerning the activity of drugs of marine origin on tumor microenvironment. We first need to understand if the activities observed for trabectedin or plitidepsin on the tumor microenvironment can be observed for other marine-derived drugs. A second question is whether these drugs are also affecting other stromal components besides the myeloid-derived compartment. We also need to understand whether the depletion of monocytic-derived cells is similar in all treated patients or if individual variability may result in different therapeutic efficacy. Another important question is whether, by affecting myeloid cells, marine-derived drugs independently influence main pathophysiological pathways of a given tumor. For example, it would be of interest to know if the anti-angiogenic activity of trabectedin is due to a direct effect on the vessel network or is mediated via the reduction of pro-angiogenic cytokines released by TAMs. While it is clear that marine-derived drugs have favorable mechanisms on tumor cells and the tumor microenvironment, additional research into the following actions would be beneficial. All these questions need to be addressed in order to improve the drug discovery and developmental process that would translate into more effective treatments with drugs of marine origin.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Farber, E. The multistep nature of cancer development. Cancer Res. 1984, 44, 4217–4223. [Google Scholar]
- Weinberg, R.A. Oncogenes, antioncogenes, and the molecular bases of multistep carcinogenesis. Cancer Res. 1989, 49, 3713–3721. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Albini, A.; Sporn, M.B. The tumour microenvironment as a target for chemoprevention. Nat. Rev. Cancer 2007, 7, 139–147. [Google Scholar]
- Egeblad, M.; Nakasone, E.S.; Werb, Z. Tumors as organs: Complex tissues that interface with the entire organism. Dev. Cell 2010, 18, 884–901. [Google Scholar] [CrossRef]
- Mueller, M.M.; Fusenig, N.E. Friends or foes—bipolar effects of the tumour stroma in cancer. Nat. Rev. Cancer 2004, 4, 839–849. [Google Scholar] [CrossRef]
- Joyce, J.A. Therapeutic targeting of the tumor microenvironment. Cancer Cell 2005, 7, 513–520. [Google Scholar] [CrossRef]
- O’Brien, L.E.; Zegers, M.M.; Mostov, K.E. Opinion: Building epithelial architecture: Insights from three-dimensional culture models. Nat. Rev. Mol. Cell Biol. 2002, 3, 531–537. [Google Scholar] [CrossRef]
- Bissell, M.J.; Weaver, V.M.; Lelievre, S.A.; Wang, F.; Petersen, O.W.; Schmeichel, K.L. Tissue structure, nuclear organization, and gene expression in normal and malignant breast. Cancer Res. 1999, 59, 1757s–1763s. [Google Scholar]
- Kalluri, R.; Zeisberg, M. Fibroblasts in cancer. Nat. Rev. Cancer 2006, 6, 392–401. [Google Scholar] [CrossRef]
- Allavena, P.; Mantovani, A. Immunology in the clinic review series; focus on cancer: Tumour-associated macrophages: Undisputed stars of the inflammatory tumour microenvironment. Clin. Exp. Immunol. 2012, 167, 195–205. [Google Scholar] [CrossRef]
- Coussens, L.M.; Raymond, W.W.; Bergers, G.; Laig-Webster, M.; Behrendtsen, O.; Werb, Z.; Caughey, G.H.; Hanahan, D. Inflammatory mast cells up-regulate angiogenesis during squamous epithelial carcinogenesis. Genes Dev. 1999, 13, 1382–1397. [Google Scholar] [CrossRef]
- Gabrilovich, D.I.; Nagaraj, S. Myeloid-derived suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 2009, 9, 162–174. [Google Scholar] [CrossRef]
- Allavena, P.; Germano, G.; Marchesi, F.; Mantovani, A. Chemokines in cancer related inflammation. Exp. Cell Res. 2011, 317, 664–673. [Google Scholar] [CrossRef]
- Mantovani, A. Molecular pathways linking inflammation and cancer. Curr. Mol. Med. 2010, 10, 369–373. [Google Scholar] [CrossRef]
- Germano, G.; Mantovani, A.; Allavena, P. Targeting of the innate immunity/inflammation as complementary anti-tumor therapies. Ann. Med. 2011, 43, 581–593. [Google Scholar] [CrossRef]
- Paszek, M.J.; Zahir, N.; Johnson, K.R.; Lakins, J.N.; Rozenberg, G.I.; Gefen, A.; Reinhart-King, C.A.; Margulies, S.S.; Dembo, M.; Boettiger, D.; et al. Tensional homeostasis and the malignant phenotype. Cancer Cell 2005, 8, 241–254. [Google Scholar]
- Levental, K.R.; Yu, H.; Kass, L.; Lakins, J.N.; Egeblad, M.; Erler, J.T.; Fong, S.F.; Csiszar, K.; Giaccia, A.; Weninger, W.; et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 2009, 139, 891–906. [Google Scholar] [CrossRef]
- Provenzano, P.P.; Eliceiri, K.W.; Campbell, J.M.; Inman, D.R.; White, J.G.; Keely, P.J. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 2006, 4, 38:1–38:16. [Google Scholar]
- Hynes, R.O. The extracellular matrix: Not just pretty fibrils. Science 2009, 326, 1216–1219. [Google Scholar] [CrossRef]
- Allen, M.; Louise Jones, J. Jekyll and Hyde: the role of the microenvironment on the progression of cancer. J. Pathol. 2011, 223, 162–176. [Google Scholar]
- Condeelis, J.; Pollard, J.W. Macrophages: Obligate partners for tumor cell migration, invasion, and metastasis. Cell 2006, 124, 263–266. [Google Scholar] [CrossRef]
- Roycik, M.D.; Fang, X.; Sang, Q.X. A fresh prospect of extracellular matrix hydrolytic enzymes and their substrates. Curr. Pharm. Des. 2009, 15, 1295–1308. [Google Scholar] [CrossRef]
- Giraudo, E.; Inoue, M.; Hanahan, D. An amino-bisphosphonate targets MMP-9-expressing macrophages and angiogenesis to impair cervical carcinogenesis. J. Clin. Invest. 2004, 114, 623–633. [Google Scholar]
- Bergers, G.; Benjamin, L.E. Tumorigenesis and the angiogenic switch. Nat. Rev. Cancer 2003, 3, 401–410. [Google Scholar] [CrossRef]
- Tredan, O.; Galmarini, C.M.; Patel, K.; Tannock, I.F. Drug resistance and the solid tumor microenvironment. J. Natl. Cancer Inst. 2007, 99, 1441–1454. [Google Scholar] [CrossRef]
- Albini, A.; Tosetti, F.; Benelli, R.; Noonan, D.M. Tumor inflammatory angiogenesis and its chemoprevention. Cancer Res. 2005, 65, 10637–10641. [Google Scholar] [CrossRef]
- Bissell, M.J.; Labarge, M.A. Context, tissue plasticity, and cancer: Are tumor stem cells also regulated by the microenvironment? Cancer Cell 2005, 7, 17–23. [Google Scholar]
- Bhowmick, N.A.; Neilson, E.G.; Moses, H.L. Stromal fibroblasts in cancer initiation and progression. Nature 2004, 432, 332–337. [Google Scholar] [CrossRef]
- D’Incalci, M.; Galmarini, C.M. A review of trabectedin (ET-743): a unique mechanism of action. Mol. Cancer Ther. 2010, 9, 2157–2163. [Google Scholar] [CrossRef]
- Demetri, G.D.; Chawla, S.P.; von Mehren, M.; Ritch, P.; Baker, L.H.; Blay, J.Y.; Hande, K.R.; Keohan, M.L.; Samuels, B.L.; Schuetze, S.; et al. Efficacy and safety of trabectedin in patients with advanced or metastatic liposarcoma or leiomyosarcoma after failure of prior anthracyclines and ifosfamide: results of a randomized phase II study of two different schedules. J. Clin. Oncol. 2009, 27, 4188–4196. [Google Scholar] [CrossRef]
- Monk, B.J.; Herzog, T.J.; Kaye, S.B.; Krasner, C.N.; Vermorken, J.B.; Muggia, F.M.; Pujade-Lauraine, E.; Lisyanskaya, A.S.; Makhson, A.N.; Rolski, J.; et al. Trabectedin plus pegylated liposomal Doxorubicin in recurrent ovarian cancer. J. Clin. Oncol. 2010, 28, 3107–3114. [Google Scholar] [CrossRef]
- Aune, G.J.; Takagi, K.; Sordet, O.; Guirouilh-Barbat, J.; Antony, S.; Bohr, V.A.; Pommier, Y. Von Hippel-Lindau-coupled and transcription-coupled nucleotide excision repair-dependent degradation of RNA polymerase II in response to trabectedin. Clin. Cancer Res. 2008, 14, 6449–6455. [Google Scholar] [CrossRef]
- Bueren-Calabuig, J.A.; Giraudon, C.; Galmarini, C.M.; Egly, J.M.; Gago, F. Temperature-induced melting of double-stranded DNA in the absence and presence of covalently bonded antitumour drugs: insight from molecular dynamics simulations. Nucleic Acids Res. 2011, 39, 8248–8257. [Google Scholar] [CrossRef]
- Feuerhahn, S.; Giraudon, C.; Martinez-Diez, M.; Bueren-Calabuig, J.A.; Galmarini, C.M.; Gago, F.; Egly, J.M. XPF-dependent DNA breaks and RNA polymerase II arrest induced by antitumor DNA interstrand crosslinking-mimetic alkaloids. Chem. Biol. 2011, 18, 988–999. [Google Scholar] [CrossRef]
- Friedman, D.; Hu, Z.; Kolb, E.A.; Gorfajn, B.; Scotto, K.W. Ecteinascidin-743 inhibits activated but not constitutive transcription. Cancer Res. 2002, 62, 3377–3381. [Google Scholar]
- Charytonowicz, E.; Terry, M.; Coakley, K.; Telis, L.; Remotti, F.; Cordon-Cardo, C.; Taub, R.N.; Matushansky, I. PPARgamma agonists enhance ET-743-induced adipogenic differentiation in a transgenic mouse model of myxoid round cell liposarcoma. J. Clin. Invest. 2012, 122, 886–898. [Google Scholar] [CrossRef]
- Grohar, P.J.; Griffin, L.B.; Yeung, C.; Chen, Q.R.; Pommier, Y.; Khanna, C.; Khan, J.; Helman, L.J. Ecteinascidin 743 interferes with the activity of EWS-FLI1 in Ewing sarcoma cells. Neoplasia 2011, 13, 145–153. [Google Scholar]
- Di Giandomenico, S.; Frapolli, R.; Bello, E.; Uboldi, S.; Licandro, S.A.; Marchini, S.; Beltrame, L.; Brich, S.; Mauro, V.; Tamborini, E.; et al. Mode of action of trabectedin in myxoid liposarcomas. Oncogene 2013, in press. [Google Scholar]
- Allavena, P.; Signorelli, M.; Chieppa, M.; Erba, E.; Bianchi, G.; Marchesi, F.; Olimpio, C.O.; Bonardi, C.; Garbi, A.; Lissoni, A.; et al. Anti-inflammatory properties of the novel antitumor agent yondelis (trabectedin): Inhibition of macrophage differentiation and cytokine production. Cancer Res. 2005, 65, 2964–2971. [Google Scholar] [CrossRef]
- Germano, G.; Frapolli, R.; Belgiovine, C.; Anselmo, A.; Pesce, S.; Liguori, M.; Erba, E.; Uboldi, S.; Zucchetti, M.; Pasqualini, F.; et al. Role of macrophage targeting in the antitumor activity of trabectedin. Cancer Cell 2013, 23, 249–262. [Google Scholar]
- Louneva, N.; Saitta, B.; Herrick, D.J.; Jimenez, S.A. Transcriptional inhibition of type I collagen gene expression in scleroderma fibroblasts by the antineoplastic drug ecteinascidin 743. J. Biol. Chem. 2003, 278, 40400–40407. [Google Scholar]
- Liguori, M.; Solinas, G.; Germano, G.; Mantovani, A.; Allavena, P. Tumor-associated macrophages as incessant builders and destroyers of the cancer stroma. Cancers 2011, 3, 3740–3761. [Google Scholar] [CrossRef]
- Murdoch, C.; Muthana, M.; Coffelt, S.B.; Lewis, C.E. The role of myeloid cells in the promotion of tumour angiogenesis. Nat. Rev. Cancer 2008, 8, 618–631. [Google Scholar] [CrossRef]
- Biswas, S.K.; Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: Cancer as a paradigm. Nat. Immunol. 2010, 11, 889–896. [Google Scholar] [CrossRef]
- Mantovani, A.; Sozzani, S.; Locati, M.; Allavena, P.; Sica, A. Macrophage polarization: Tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002, 23, 549–555. [Google Scholar]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef]
- Gordon, S.; Martinez, F.O. Alternative activation of macrophages: Mechanism and functions. Immunity 2010, 32, 593–604. [Google Scholar] [CrossRef]
- Qian, B.Z.; Pollard, J.W. Macrophage diversity enhances tumor progression and metastasis. Cell 2010, 141, 39–51. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- DeNardo, D.G.; Johansson, M.; Coussens, L.M. Immune cells as mediators of solid tumor metastasis. Cancer Metastas. Rev. 2008, 27, 11–18. [Google Scholar] [CrossRef]
- Movahedi, K.; Laoui, D.; Gysemans, C.; Baeten, M.; Stange, G.; Van den Bossche, J.; Mack, M.; Pipeleers, D.; In’t Veld, P.; De Baetselier, P.; et al. Different tumor microenvironments contain functionally distinct subsets of macrophages derived from Ly6C(high) monocytes. Cancer Res. 2010, 70, 5728–5739. [Google Scholar] [CrossRef]
- Pollard, J.W. Macrophages define the invasive microenvironment in breast cancer. J. Leukoc. Biol. 2008, 84, 623–630. [Google Scholar] [CrossRef]
- Mantovani, A.; Romero, P.; Palucka, A.K.; Marincola, F.M. Tumour immunity: effector response to tumour and role of the microenvironment. Lancet 2008, 371, 771–783. [Google Scholar] [CrossRef]
- Bingle, L.; Brown, N.J.; Lewis, C.E. The role of tumour-associated macrophages in tumour progression: Implications for new anticancer therapies. J. Pathol. 2002, 196, 254–265. [Google Scholar] [CrossRef]
- Clear, A.J.; Lee, A.M.; Calaminici, M.; Ramsay, A.G.; Morris, K.J.; Hallam, S.; Kelly, G.; Macdougall, F.; Lister, T.A.; Gribben, J.G. Increased angiogenic sprouting in poor prognosis FL is associated with elevated numbers of CD163+ macrophages within the immediate sprouting microenvironment. Blood 2010, 115, 5053–5056. [Google Scholar] [CrossRef]
- Heusinkveld, M.; van der Burg, S.H. Identification and manipulation of tumor associated macrophages in human cancers. J. Transl. Med. 2011, 9, 216:1–216:13. [Google Scholar]
- Leek, R.D.; Lewis, C.E.; Whitehouse, R.; Greenall, M.; Clarke, J.; Harris, A.L. Association of macrophage infiltration with angiogenesis and prognosis in invasive breast carcinoma. Cancer Res. 1996, 56, 4625–4629. [Google Scholar]
- Hagemann, T.; Wilson, J.; Burke, F.; Kulbe, H.; Li, N.F.; Pluddemann, A.; Charles, K.; Gordon, S.; Balkwill, F.R. Ovarian cancer cells polarize macrophages toward a tumor-associated phenotype. J. Immunol. 2006, 176, 5023–5032. [Google Scholar]
- Lee, C.H.; Espinosa, I.; Vrijaldenhoven, S.; Subramanian, S.; Montgomery, K.D.; Zhu, S.; Marinelli, R.J.; Peterse, J.L.; Poulin, N.; Nielsen, T.O.; et al. Prognostic significance of macrophage infiltration in leiomyosarcomas. Clin. Cancer Res. 2008, 14, 1423–1430. [Google Scholar] [CrossRef]
- Lissbrant, I.F.; Stattin, P.; Wikstrom, P.; Damber, J.E.; Egevad, L.; Bergh, A. Tumor associated macrophages in human prostate cancer: relation to clinicopathological variables and survival. Int. J. Oncol. 2000, 17, 445–451. [Google Scholar]
- Jensen, T.O.; Schmidt, H.; Moller, H.J.; Hoyer, M.; Maniecki, M.B.; Sjoegren, P.; Christensen, I.J.; Steiniche, T. Macrophage markers in serum and tumor have prognostic impact in American Joint Committee on Cancer stage I/II melanoma. J. Clin. Oncol. 2009, 27, 3330–3337. [Google Scholar] [CrossRef]
- van Dongen, M.; Savage, N.D.; Jordanova, E.S.; Briaire-de Bruijn, I.H.; Walburg, K.V.; Ottenhoff, T.H.; Hogendoorn, P.C.; van der Burg, S.H.; Gelderblom, H.; van Hall, T. Anti-inflammatory M2 type macrophages characterize metastasized and tyrosine kinase inhibitor-treated gastrointestinal stromal tumors. Int. J. Cancer 2010, 127, 899–909. [Google Scholar]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Nagai, T.; Tanaka, M.; Tsuneyoshi, Y.; Xu, B.; Michie, S.A.; Hasui, K.; Hirano, H.; Arita, K.; Matsuyama, T. Targeting tumor-associated macrophages in an experimental glioma model with a recombinant immunotoxin to folate receptor beta. Cancer Immunol. Immunother. 2009, 58, 1577–1586. [Google Scholar] [CrossRef]
- Germano, G.; Frapolli, R.; Simone, M.; Tavecchio, M.; Erba, E.; Pesce, S.; Pasqualini, F.; Grosso, F.; Sanfilippo, R.; Casali, P.G.; et al. Antitumor and anti-inflammatory effects of trabectedin on human myxoid liposarcoma cells. Cancer Res. 2010, 70, 2235–2244. [Google Scholar] [CrossRef]
- Munoz-Alonso, M.J.; Gonzalez-Santiago, L.; Martinez, T.; Losada, A.; Galmarini, C.M.; Munoz, A. The mechanism of action of plitidepsin. Curr. Opin. Investig. Drugs 2009, 10, 536–542. [Google Scholar]
- Suarez, Y.; Gonzalez-Santiago, L.; Zarich, N.; Davalos, A.; Aranda, J.F.; Alonso, M.A.; Lasuncion, M.A.; Rojas, J.M.; Munoz, A. Plitidepsin cellular binding and Rac1/JNK pathway activation depend on membrane cholesterol content. Mol. Pharmacol. 2006, 70, 1654–1663. [Google Scholar] [CrossRef]
- Garcia-Fernandez, L.F.; Losada, A.; Alcaide, V.; Alvarez, A.M.; Cuadrado, A.; Gonzalez, L.; Nakayama, K.; Nakayama, K.I.; Fernandez-Sousa, J.M.; Munoz, A.; et al. Aplidin induces the mitochondrial apoptotic pathway via oxidative stress-mediated JNK and p38 activation and protein kinase C delta. Oncogene 2002, 21, 7533–7544. [Google Scholar] [CrossRef]
- Gonzalez-Santiago, L.; Suarez, Y.; Zarich, N.; Munoz-Alonso, M.J.; Cuadrado, A.; Martinez, T.; Goya, L.; Iradi, A.; Saez-Tormo, G.; Maier, J.V.; et al. Aplidin induces JNK-dependent apoptosis in human breast cancer cells via alteration of glutathione homeostasis, Rac1 GTPase activation, and MKP-1 phosphatase downregulation. Cell Death Differ. 2006, 13, 1968–1981. [Google Scholar] [CrossRef]
- Cuadrado, A.; Garcia-Fernandez, L.F.; Gonzalez, L.; Suarez, Y.; Losada, A.; Alcaide, V.; Martinez, T.; Fernandez-Sousa, J.M.; Sanchez-Puelles, J.M.; Munoz, A. Aplidin induces apoptosis in human cancer cells via glutathione depletion and sustained activation of the epidermal growth factor receptor, Src, JNK, and p38 MAPK. J. Biol. Chem. 2003, 278, 241–250. [Google Scholar]
- Losada, A.; Martínez-Leal, J.F.; Bejarano, A.; Cuevas, C.; García-Fernández, L.F.; Galmarini, C.M. Aplidin triggers the activation of molecular components of the UPR as part of its pro-apoptotic program in tumor cells. In Proceedings of the AACR 104th Annual Meeting of the American Association for Cancer Research Washington, Washington, DC, USA, 6–10 April 2013. [CrossRef]
- Morande, P.E.; Zanetti, S.R.; Borge, M.; Nannini, P.; Jancic, C.; Bezares, R.F.; Bitsmans, A.; Gonzalez, M.; Rodriguez, A.L.; Galmarini, C.M.; et al. The cytotoxic activity of Aplidin in chronic lymphocytic leukemia (CLL) is mediated by a direct effect on leukemic cells and an indirect effect on monocyte-derived cells. Invest. New Drugs 2012, 30, 1830–1840. [Google Scholar] [CrossRef]
- Burger, J.A.; Tsukada, N. leukemia B cells from spontaneous apoptosis through stromal cell-derived factor-1. Blood 2000, 96, 2655–2663. [Google Scholar]
- Nishio, M.; Endo, T.; Tsukada, N.; Ohata, J.; Kitada, S.; Reed, J.C.; Zvaifler, N.J.; Kipps, T.J. Nurselike cells express BAFF and APRIL, which can promote survival of chronic lymphocytic leukemia cells via a paracrine pathway distinct from that of SDF-1alpha. Blood 2005, 106, 1012–1020. [Google Scholar] [CrossRef]
- Tsukada, N.; Burger, J.A.; Zvaifler, N.J.; Kipps, T.J. Distinctive features of “nurselike” cells that differentiate in the context of chronic lymphocytic leukemia. Blood 2002, 99, 1030–1037. [Google Scholar] [CrossRef]
- Baran, C.P.; Zeigler, M.M.; Tridandapani, S.; Marsh, C.B. The role of ROS and RNS in regulating life and death of blood monocytes. Curr. Pharm. Des. 2004, 10, 855–866. [Google Scholar] [CrossRef]
- Zurgil, N.; Solodeev, I.; Gilburd, B.; Shafran, Y.; Afrimzon, E.; Avtalion, R.; Shoenfeld, Y.; Deutsch, M. Monitoring the apoptotic process induced by oxidized low-density lipoprotein in Jurkat T-lymphoblast and U937 monocytic human cell lines. Cell Biochem. Biophys. 2004, 40, 97–113. [Google Scholar] [CrossRef]
- Sanfilippo, R.; Casali, P.G. The intriguing patterns of tumor response to trabectedin. Expert Rev. Anticancer Ther. 2013, 13, 21–24. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
