Isolation, Characterization, and Bioactivity Evaluation of 3-((6-Methylpyrazin-2-yl)methyl)-1H-indole, a New Alkaloid from a Deep-Sea-Derived Actinomycete Serinicoccus profundi sp. nov.
Abstract
:1. Introduction
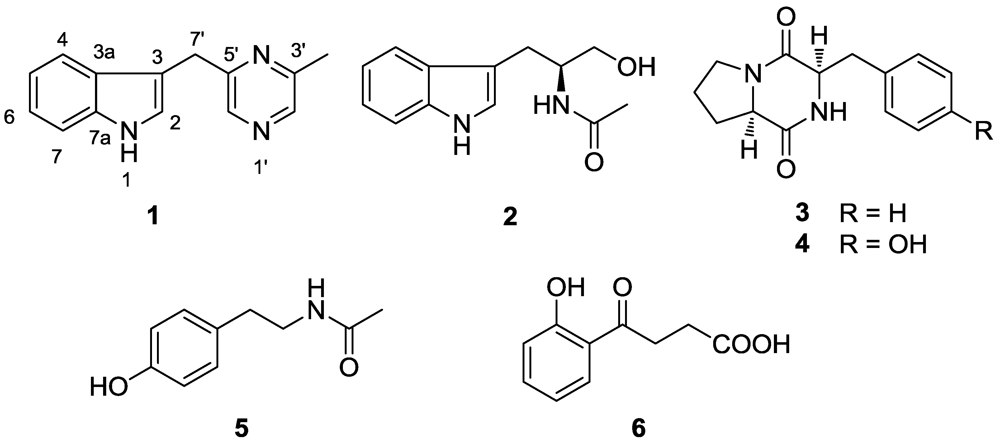
2. Results and Discussion
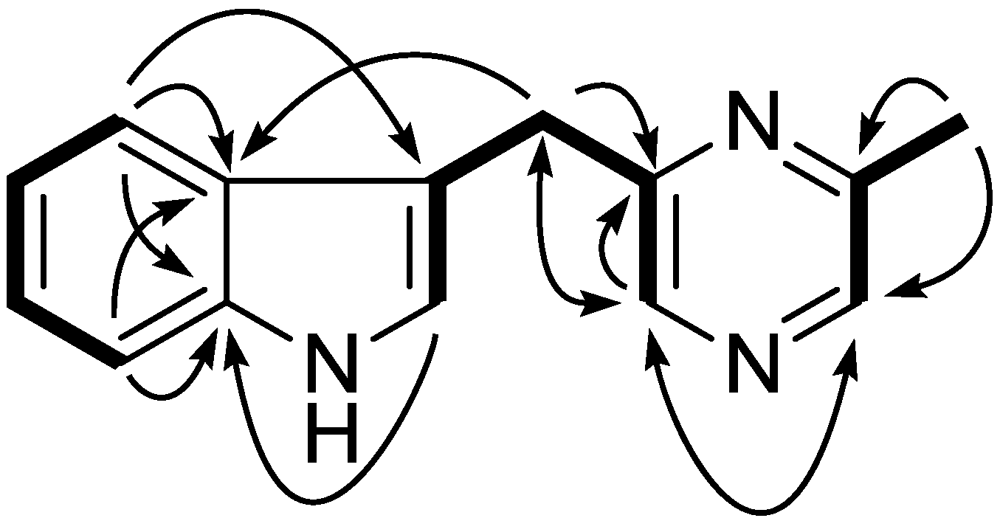
2.1. Structure Elucidation
| No. | δC | δH |
|---|---|---|
| 2 | 122.6 d | 7.08 m |
| 3 | 112.9 s | |
| 3a | 127.2 s | |
| 4 | 119.0 d | 7.55 (d, 8.0) |
| 5 | 119.6 d | 7.10 (dt, 7.6, 1.0) |
| 6 | 122.3 d | 7.20 (dt, 7.6, 1.1) |
| 7 | 111.2 d | 7.37 (d, 8.1) |
| 7a | 136.4 s | |
| 2′ | 141.8 d | 8.27 br s |
| 3′ | 152.7 s | |
| 5′ | 155.5 s | |
| 6′ | 141.4 d | 8.27 br s |
| 7′ | 32.1 t | 4.28 s |
| 8′ | 21.6 q | 2.56 s |
2.2. Bioactive Tests
3. Experimental Section
3.1. General Experimental Procedures
3.2. Microbial Source and Culture Conditions
3.3. Extraction and Isolation
3.4. Cytotoxic Assay
3.5. Antibacterial Activity
4. Conclusions
Acknowledgments
References
- Goodfellow, M.; Stach, J.E.; Brown, R.; Bonda, A.N.; Jones, A.L.; Mexson, J.; Fiedler, H.P.; Zucchi, T.D.; Bull, A.T. Verrucosispora maris sp. nov., a novel deep-sea actinomycete isolated from a marine sediment which produces abyssomicins. Antonie Van Leeuwenhoek 2012, 101, 185–193. [Google Scholar] [CrossRef]
- Tian, X.P.; Xu, Y.; Zhang, J.; Li, J.; Chen, Z.; Kim, C.J.; Li, W.J.; Zhang, C.S.; Zhang, S. Streptomyces oceani sp. nov., a new obligate marine actinomycete isolated from a deep-sea sample of seep authigenic carbonate nodule in South China Sea. Antonie Van Leeuwenhoek 2012, 102, 335–343. [Google Scholar] [CrossRef]
- Luo, Y.; Xiao, J.; Wang, Y.; Xu, J.; Xie, S.; Xu, J. Streptomyces indicus sp. nov., an actinomycete isolated from deep-sea sediment. Int. J. Syst. Evol. Microbiol. 2011, 61, 2712–2716. [Google Scholar] [CrossRef]
- Li, J.; Zhao, G.Z.; Long, L.J.; Wang, F.Z.; Tian, X.P.; Zhang, S.; Li, W.J. Rhodococcus nanhaiensis sp. nov., an actinobacterium isolated from marine sediment. Int. J. Syst. Evol. Microbiol. 2012, 62, 2517–2521. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, Z.; Li, S.; Yang, T.; Zhang, Q.; Ma, L.; Tian, X.; Zhang, H.; Huang, C.; Zhang, S.; et al. Spiroindimicins A–D: New bisindole alkaloids from a deep-sea-derived actinomycete. Org. Lett. 2012, 14, 3364–3367. [Google Scholar] [CrossRef]
- Huang, H.; Yang, T.; Ren, X.; Liu, J.; Song, Y.; Sun, A.; Ma, J.; Wang, B.; Zhang, Y.; Huang, C.; et al. Cytotoxic angucycline class glycosides from the deep sea actinomycete Streptomyces lusitanus SCSIO LR32. J. Nat. Prod. 2012, 75, 202–208. [Google Scholar] [CrossRef]
- Li, S.; Tian, X.; Niu, S.; Zhang, W.; Chen, Y.; Zhang, H.; Yang, X.; Zhang, W.; Li, W.; Zhang, S.; et al. Pseudonocardians A–C, new diazaanthraquinone derivatives from a deap-sea actinomycete Pseudonocardia sp. SCSIO 01299. Mar. Drugs 2011, 9, 1428–1439. [Google Scholar] [CrossRef]
- Abdel-Mageed, W.M.; Milne, B.F.; Wagner, M.; Schumacher, M.; Sandor, P.; Pathom-aree, W.; Goodfellow, M.; Bull, A.T.; Horikoshi, K.; Ebel, R.; et al. Dermacozines, a new phenazine family from deep-sea dermacocci isolated from a Mariana Trench sediment. Org. Biomol. Chem. 2010, 8, 2352–2362. [Google Scholar] [CrossRef]
- Sato, S.; Iwata, F.; Yamada, S.; Kawahara, H.; Katayama, M. Usabamycins A–C: New anthramycin-type analogues from a marine-derived actinomycete. Bioorg. Med. Chem. Lett. 2011, 21, 7099–7101. [Google Scholar] [CrossRef]
- Yi, H.; Schumann, P.; Sohn, K.; Chun, J. Serinicoccus marinus gen. nov., sp. nov., a novel actinomycete with L-ornithine and L-serine in the peptidoglycan. Int. J. Syst. Evol. Microbiol. 2004, 54, 1585–1859. [Google Scholar] [CrossRef]
- Traiwan, J.; Park, M.H.; Kim, W. Serinicoccus chungangensis sp. nov., isolated from tidal flat sediment, and emended description of the genus Serinicoccus. Int. J. Syst. Evol. Microbiol. 2011, 61, 1299–1303. [Google Scholar] [CrossRef]
- Xiao, J.; Luo, Y.; Xie, S.; Xu, J. Serinicoccus profundi sp. nov., an actinomycete isolated from deep-sea sediment, and emended description of the genus Serinicoccus. Int. J. Syst. Evol. Microbiol. 2011, 61, 16–19. [Google Scholar] [CrossRef]
- Li, Y.Q.; Huang, X.S.; Li, M.G.; Sattler, I.; Wen, M.L.; Grabley, S. A new compound isolated from a bioactive streptomycete. Chin. Chem. Lett. 2005, 16, 613–615. [Google Scholar]
- Adamczeski, M.; Reed, A.R.; Crews, P. New and known diketopiperazines from the Caribbean sponge, Calyx cf. podatypa. J. Nat. Prod. 1995, 58, 201–208. [Google Scholar] [CrossRef]
- Jayatilake, G.S.; Thornton, M.P.; Leonard, A.C.; Grimwade, J.E.; Baker, B.J. Metabolites from an Antarctic sponge-associated bacterium, Pseudomonas aeruginosa. J. Nat. Prod. 1996, 59, 293–296. [Google Scholar] [CrossRef]
- Yang, X.W.; Zeng, H.W.; Liu, X.H.; Li, S.M.; Xu, W.; Shen, Y.H.; Zhang, C.; Zhang, W.D. Anti-inflammatory and anti-tumour effects of Abies georgei extracts. J. Pharm. Pharmacol. 2008, 60, 937–941. [Google Scholar]
- Xiao, Y.; Li, S.; Niu, S.; Ma, L.; Zhang, G.; Zhang, H.; Zhang, G.; Ju, J.; Zhang, C. Characterization of tiacumicin B biosynthetic gene cluster affording diversified tiacumicin analogues and revealing a tailoring dihalogenase. J. Am. Chem. Soc. 2011, 133, 1092–1105. [Google Scholar] [CrossRef]
- Samples Availability: Available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, X.-W.; Zhang, G.-Y.; Ying, J.-X.; Yang, B.; Zhou, X.-F.; Steinmetz, A.; Liu, Y.-H.; Wang, N. Isolation, Characterization, and Bioactivity Evaluation of 3-((6-Methylpyrazin-2-yl)methyl)-1H-indole, a New Alkaloid from a Deep-Sea-Derived Actinomycete Serinicoccus profundi sp. nov. Mar. Drugs 2013, 11, 33-39. https://doi.org/10.3390/md11010033
Yang X-W, Zhang G-Y, Ying J-X, Yang B, Zhou X-F, Steinmetz A, Liu Y-H, Wang N. Isolation, Characterization, and Bioactivity Evaluation of 3-((6-Methylpyrazin-2-yl)methyl)-1H-indole, a New Alkaloid from a Deep-Sea-Derived Actinomycete Serinicoccus profundi sp. nov. Marine Drugs. 2013; 11(1):33-39. https://doi.org/10.3390/md11010033
Chicago/Turabian StyleYang, Xian-Wen, Gai-Yun Zhang, Jian-Xi Ying, Bing Yang, Xue-Feng Zhou, Andre Steinmetz, Yong-Hong Liu, and Ning Wang. 2013. "Isolation, Characterization, and Bioactivity Evaluation of 3-((6-Methylpyrazin-2-yl)methyl)-1H-indole, a New Alkaloid from a Deep-Sea-Derived Actinomycete Serinicoccus profundi sp. nov." Marine Drugs 11, no. 1: 33-39. https://doi.org/10.3390/md11010033
APA StyleYang, X.-W., Zhang, G.-Y., Ying, J.-X., Yang, B., Zhou, X.-F., Steinmetz, A., Liu, Y.-H., & Wang, N. (2013). Isolation, Characterization, and Bioactivity Evaluation of 3-((6-Methylpyrazin-2-yl)methyl)-1H-indole, a New Alkaloid from a Deep-Sea-Derived Actinomycete Serinicoccus profundi sp. nov. Marine Drugs, 11(1), 33-39. https://doi.org/10.3390/md11010033








