Dietary Carotenoids Regulate Astaxanthin Content of Copepods and Modulate Their Susceptibility to UV Light and Copper Toxicity
Abstract
:1. Introduction
2. Results and Discussion
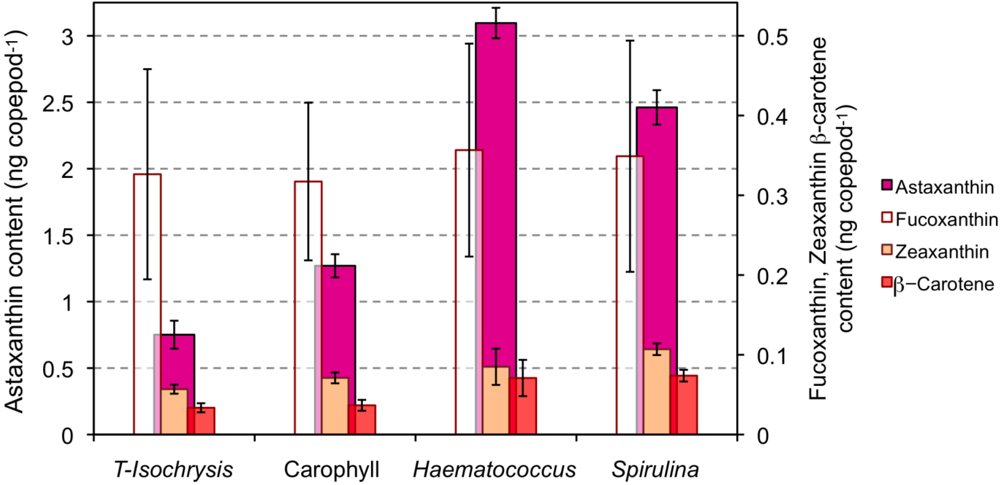
2.1. Effect of Diet on the Astaxanthin Content of Copepods
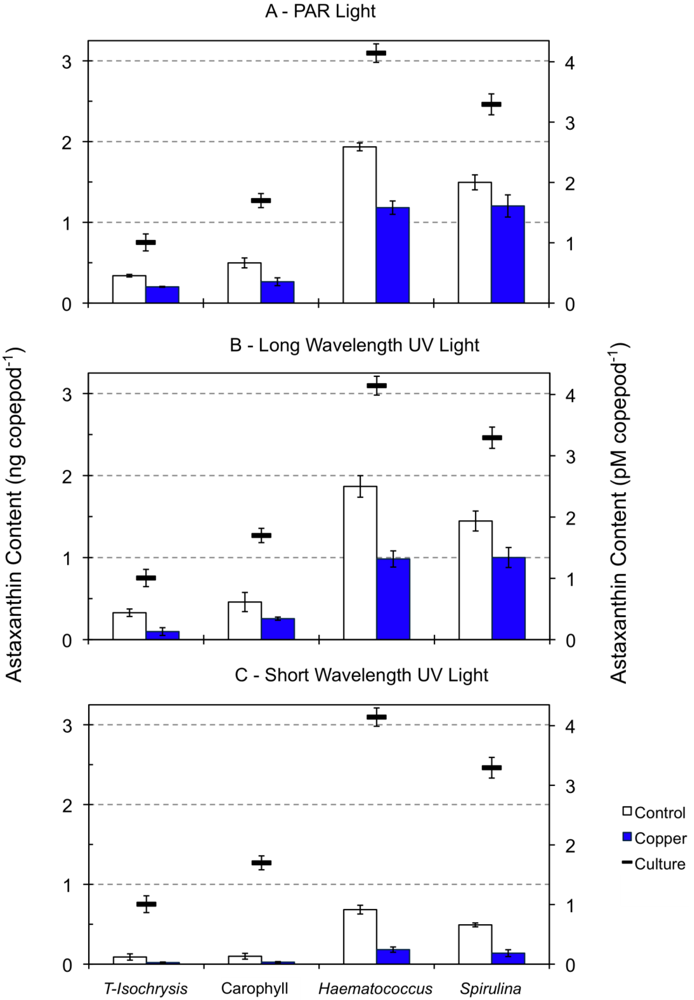
2.2. Effect of Light Type Exposure on the Astaxanthin Content of Copepods
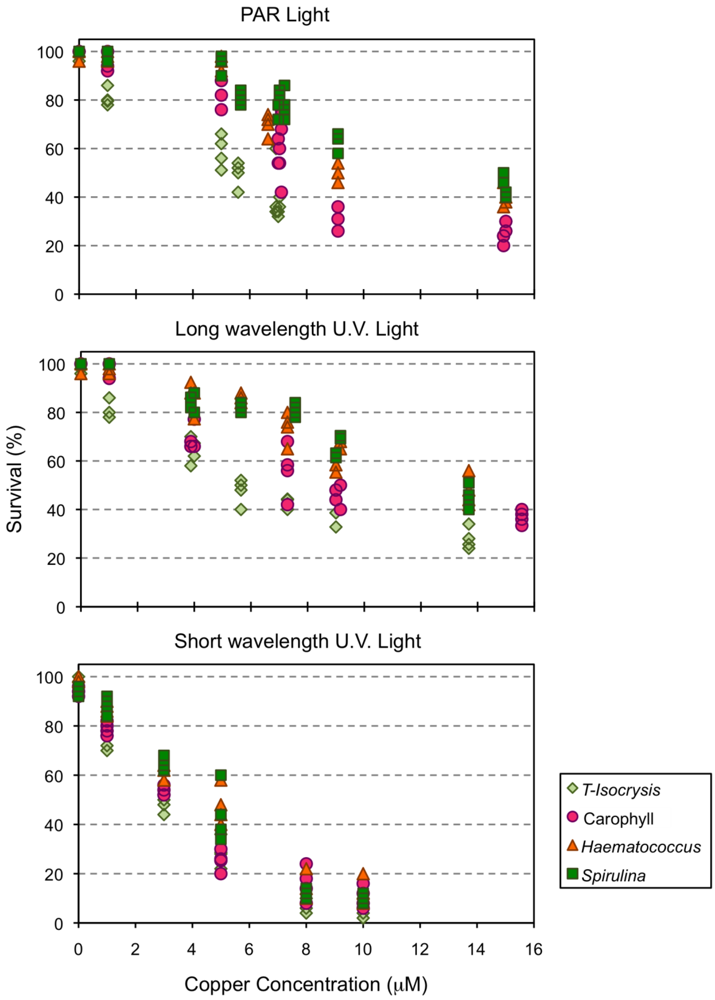
2.3. Effect of Copper Exposure on the Astaxanthin Content of Copepods and Their Survival
| Tested Effect | PAR (n = 24) | Long UV radiation (n = 26) | Short UV radiation (n = 24) |
|---|---|---|---|
| Diet (df = 3) | 179.3 | 162.6 | 246.1 |
| Copper (df = 1) | 51.8 | 83.0 | 432.7 |
| Diet × Copper (df = 3) | 7.8 * | 11.1 | 79.5 |
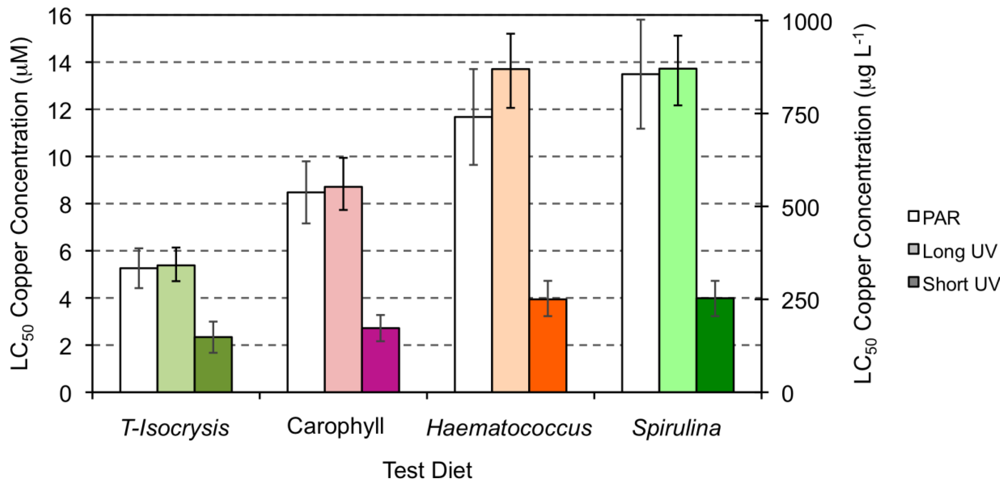
| Species | Stage | Food | Cu (μM) | Cu (μg L−1) | Reference |
|---|---|---|---|---|---|
| Amphiascoides atopus (under PAR light) | Adult | A = T-Isocrysis + enriched Torula yeast | 5.26(4.42–6.23) | 334.24(280.6–396.1) | Present study |
| A + Carophyll | 8.48(7.16–10.35) | 538.67(455.0–657.8) | |||
| A + Haematococcus | 11.67(9.64–15.66) | 741.83(612.7–995.3) | |||
| A + Spirulina | 13.49(11.17–15.90) | 857.19(710.3–1204.6) | |||
| A. atopus | Adult | T-Isocrysis | (Cd = 4.88 ± 0.75) | (Cd = 549 ± 84) | [63] |
| Nitocra spinipes | Adult | Unknown | 28.33 | 1800 | [70] |
| Tigriopus brevicornis | Adult | Field collected, unfed | 2.36 | 150 | [71] |
| Tigriopus californicus | Fish food (Wardley’s Basic Food Flakes) | 11.99 | 762 | [72] | |
| Tigriopus japonicus | Adult | Tetraselmis suecica | 61.37(Cd = 24.18) | 3900(Cd = 25,200) | [64] |
| Tigriopus japonicus | C5–C6 | Field collected, unfed | 16.11 | 1024 | [73] |
| Tigriopus japonicus | Adults | Enteromorpha spp. plus phytoplankton concentrate (Kent Marine Phytoplex, US) | 12.79 | 813 | [74] |
| Tisbe battagliai 1 | Adult | Artificial (Marine Invertebrate Diet™, Hawaiian Marine Imports Inc.) | 2.47 | 157 | [75] |
| Tisbe holothuriae 2 | Females with ovigerous bands | Unknown | 7.04(Cd = 8.15) | 447.3(Cd = 916.6) | [65] |
| Females with ovisacs | Unknown | 6.74(Cd = 7.76) | 428.2(Cd = 872.7) |
3. Experimental Section
3.1. Cultures
| Food | Carotenoid content(µg mg−1 DW) | Carotenoid Composition (weight%) | Astaxanthin Isomers |
|---|---|---|---|
| T-Isochrysis paste(Brine Shrimp Direct) | 3.18 ± 0.11 | 58%–Fucoxanthin 24%–Diadinoxanthin 3%–Diatoxanthin 15%–β-carotene | – |
| Enriched Torula yeast(Microfeast Plus® L-10) | 0.54 ± 0.07 | 87%–Astaxanthin 3%–Echinenone 2%–β-carotene | Unknown |
| Carophyll® Pink | 81.12 ± 1.09 | 100%–Astaxanthin | 18.75%–(R,R′) 18.75%–(S,S′) 37.50%–(R,S) 25%–Z-isomers [46] |
| Haematococcus meal(NatuRose™) | 17.86 ± 0.96 | 84%–Astaxanthin 2%–Canthaxanthin 7%–Lutein 2%-β-carotene | >99%-(S,S′) [47] |
| Spirulina meal | 3.36 ± 0.10 | 23%–Zeaxanthin 10%–Echinenone 5%–β-cryptoxanthin 54%–β-carotene | - |
3.2. Pigment Extraction and Analysis
3.3. 96-h Acute Toxicity Tests
3.4. Calculations and Statistics
4. Conclusions
Acknowledgements
References
- Blough, N.V.; Zepp, R.G. Reactive Oxygen Species in Natural Waters. In Active Oxygen in Chemistry; Foote, C.S., Ed.; Blackle Academic and Professional: Glasgow, UK, 1995; pp. 280–333. [Google Scholar]
- Whitehead, R.F.; de Mora, S.J.; Demers, S. Enhanced UV Radiation—A New Problem for the Marine environment; Cambridge University Press: Cambridge, UK, 2000; pp. 1–34. [Google Scholar]
- Asada, K. Radical Production and Scavenging in the Chloroplasts. In Photosynthesis and the Environment; Baker, N.R., Ed.; Kluwer Academic Publishers: Norwell, MA, USA, 1996; Volume 5, pp. 123–150. [Google Scholar]
- Nishiyama, Y.; Yamamoto, H.; Allakhverdiev, S.I.; Inaba, M.; Yokota, A.; Murata, N. Oxidative stress inhibits the repair of photodamage to the photosynthetic machinery. EMBO J. 2001, 20, 5587–5594. [Google Scholar]
- Winston, G.; Moore, M.; Kirchin, M.; Soverchia, C. Production of reactive oxygen species by hemocytes from the marine mussel, Mytilus edulis: Lysosomal localization and effect of xenobiotics. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1996, 113, 221–229. [Google Scholar] [CrossRef]
- Avery, E.L.; Dunstan, R.H.; Nell, J.A. The detection of pollutant impact in marine environments: Condition index, oxidative DNA damage, and their associations with metal bioaccumulation in the Sydney rock oyster Saccostrea commercialis. Arch. Environ. Contam. Toxicol. 1996, 31, 192–198. [Google Scholar]
- Sies, H. Strategies of antioxidant defense. Eur. J. Biochem. 1993, 215, 213–219. [Google Scholar]
- Palenik, B.; Price, N.M.; Morel, F.M.M. Potential effects of UV-B on the chemical environment of marine organisms: A review. Environ. Pollut. 1991, 70, 117–130. [Google Scholar]
- Lesser, M.P. Oxidative stress in marine environments: Biochemistry and physiological ecology. Annu. Rev. Physiol. 2006, 68, 253–278. [Google Scholar]
- Cadet, J.; Berger, M.; Douki, T.; Ravanat, J. Oxidative damage to DNA: Formation, measurement, and biological significance. In Reviews of Physiology, Biochemistry and Pharmacology; Blaustein, M.P., Greger, R., Grunicke, H., John, R., Mendell, L.M., Pette, D., Schultz, G., Schweiger, M., Eds.; Springer Verlag: Berlin, Germany; Volume 131, pp. 1–87.
- Bhandari, R.; Sharma, P.K. Effect of UV-B on photosynthesis, membrane lipids and MAAs in marine cyanobacterium, Phormidium corium (Agardh) Gomont. Indian J. Exp. Biol. 2006, 44, 330–335. [Google Scholar]
- Fucci, L.; Oliver, C.; Coon, M.; Stadtman, E. Inactivation of key metabolic enzymes by mixed function oxidation reactions: Possible implication in protein turnover and ageing. Proc. Natl. Acad. Sci. USA 1983, 80, 1521–1525. [Google Scholar] [CrossRef]
- Cabiscol, E.; Tamarit, J.; Ros, J. Oxidative stress in bacteria and protein damage by reactive oxygen species. Int. Microbiol. 2000, 3, 3–8. [Google Scholar]
- Flora, S.J.S.; Pachauri, V. Chelation in metal intoxication. Int. J. Environ. Res. Public Health 2010, 7, 2745–2788. [Google Scholar]
- Halliwell, B. Antioxidant defence mechanisms: From the beginning to the end (of the beginning). Free Radic. Res. 1999, 31, 261–272. [Google Scholar]
- Ndhlala, A.; Moyo, M.; van Staden, J. Natural antioxidants: Fascinating or mythical biomolecules? Molecules 2010, 15, 6905–6930. [Google Scholar] [CrossRef]
- Mimuro, M.; Katoh, T. Carotenoids in photosynthesis: Absorption, transfer and dissipation of light energy. Pure Appl. Chem. 1991, 63, 123–130. [Google Scholar]
- Demmig-Adams, B.; Gilmore, A.; Adams, W. Carotenoids 3: In vivo function of carotenoids in higher plants. FASEB J. 1996, 10, 403–412. [Google Scholar]
- Goodwin, T.W. The Biochemistry of the Carotenoids, Volume 1: Plants; Chapman and Hall: New York, NY, USA, 1980; p. 377. [Google Scholar]
- Yokoyama, A.; Sandmann, G.; Hoshino, T.; Adachi, K.; Sakai, M.; Shizuri, Y. Thermozeaxanthins, new carotenoid-glycoside-esters from thermophilic eubacterium Thermus thermophilus. Tetrahedron Lett. 1995, 36, 4901–4904. [Google Scholar]
- Antón, J.; Rosselló-Mora, R.; Rodríguez-Valera, F.; Amann, R. Extremely halophilic bacteria in crystallizer ponds from solar salterns. Appl. Environ. Microbiol. 2000, 66, 3052–3057. [Google Scholar]
- Petterson, A.; Lignell, A. Low astaxanthin levels in the Baltic salmon exhibiting the M74 syndrome. Am. Fish. Soc. Symp. 1998, 21, 26–30. [Google Scholar]
- Van Nieuwerburgh, L.; Wänstrand, I.; Liu, J.; Snoeijs, P. Astaxanthin production in marine pelagic copepods grazing on two different phytoplankton diets. J. Sea Res. 2005, 53, 147–160. [Google Scholar]
- Goodwin, T.W. The Biochemistry of the Carotenoids, 2nd ed; Chapman and Hall: London, UK, 1984; Volume 2. [Google Scholar]
- Matsuno, T. Aquatic animal carotenoids. Fish Sci. 2001, 67, 771–783. [Google Scholar]
- Harker, M.; Young, A.J. Inhibition of astaxanthin synthesis in the green alga, Haematococcus pluvialis. Eur. J. Phycol. 1995, 30, 179–187. [Google Scholar]
- Kobayashi, M.; Sakamoto, Y. Singlet oxygen quenching ability of astaxanthin esters from the green alga Haematococcus pluvialis. Biotechnol. Lett. 1999, 21, 265–269. [Google Scholar] [CrossRef]
- Bandaranayake, W.M.; Gentien, P. Carotenoids of Temora turbinata, Centropages furcatus, Undinula vulgaris and Euchaeta russelli. Comp. Biochem. Physiol. B 1982, 72, 409–414. [Google Scholar] [CrossRef]
- Terão, J. Antioxidant activity of beta-carotene-related carotenoids in solution. Lipids 1989, 24, 659–661. [Google Scholar] [CrossRef]
- Hairston, N.G., Jr. Photoprotection by carotenoid pigment in the copepod Diaptomus nevadensis. Proc. Natl. Acad. Sci. USA 1976, 73, 971–974. [Google Scholar] [CrossRef]
- Hairston, N.G., Jr. The adaptive significance of color polymorphism in two species of Diaptomus Copepoda. Limnol. Oceanogr. 1979, 24, 15–37. [Google Scholar] [CrossRef]
- Hansson, L.-A. Plasticity in pigmentation induced by conflicting threats from predation and UV radiation. Ecology 2004, 85, 1005–1016. [Google Scholar]
- Chalker-Scott, L. Survival and sex ratios of the intertidal copepod, Tigriopus californicus, following ultraviolet-B (290-320 nm) radiation exposure. Mar. Biol. 1995, 123, 799–804. [Google Scholar] [CrossRef]
- Karanas, J.J.; Van Dyke, H.; Worrest, R.C. Midultraviolet (UV-B) sensitivity of Acartia clausii Giesbrecht (Copepoda). Limnol. Oceanogr. 1979, 24, 1104–1116. [Google Scholar] [CrossRef]
- Williamson, C.E.; Zagarese, H.E.; Schulze, P.C.; Hargreaves, B.R.; Seva, J. The impact of short-term exposure to UV-B radiation on zooplankton communities in north temperate lakes. J. Plankton Res. 1994, 16, 205–218. [Google Scholar]
- Ringelberg, J. Aspects of red pigmentation in zooplankton, especially copepods. Am. Soc. Limnol. Oceanogr. Spec. Symp. 1980, 3, 91–97. [Google Scholar]
- Łotocka, M.; Styczyńska-Jurewicz, E.; Błędzki, L.A. Changes in carotenoid composition in different developmental stages of copepods: Pseudocalanus acuspes Giesbrecht and Acartia spp. J. Plankton Res. 2004, 26, 159–166. [Google Scholar] [CrossRef]
- Sigel, A.; Sigel, H. Interrelations between Free Radicals and Metal Ions in Life Processes. In Metal Ions in Biological Systems; Marcel Dekker: New York, NY, USA, 1999; Volume 36, p. 848. [Google Scholar]
- Woodall, A.A.; Britton, G.; Jackson, M.J. Carotenoids and protection of phospholipids in solution or in liposomes against oxidation by peroxyl radicals: Relationship between carotenoid structure and protective ability. Biochim. Biophys. Acta Gen. Subj. 1997, 1336, 575–586. [Google Scholar] [CrossRef]
- Jorgensen, K.; Skibsted, L.H. Carotenoid scavenging of radicals—Effect of carotenoid structure and oxygen oartial-pressure on antioxidative activity. Z. Lebensm. Unters. F 1993, 196, 423–429. [Google Scholar]
- Davenport, J.; Healy, A.; Casey, N.; Heffron, J.J.A. Diet-dependent UVAR and UVBR resistance in the high shore harpacticoid copepod Tigriopus brevicornis. Mar. Ecol. Prog. Ser. 2004, 276, 299–303. [Google Scholar] [CrossRef]
- Buffan-Dubau, E.; de Wit, R.; Castel, J. Feeding selectivity of the harpacticoid copepod Canuella perplexa in benthic muddy environments demonstrated by HPLC analyses of chlorin and carotenoid pigments. Mar. Ecol. Prog. Ser. 1996, 137, 71–82. [Google Scholar] [CrossRef]
- Andersson, M.; Nieuwerburgh, L.V.; Snoeijs, P. Pigment transfer from phytoplankton to zooplankton with emphasis on astaxanthin production in the Baltic Sea food web. Mar. Ecol. Prog. Ser. 2003, 254, 213–224. [Google Scholar]
- Rhodes, A.C.E. Dietary effects on carotenoid composition in the marine harpacticoid copepod Nitokra lacustris. J. Plankton Res. 2007, 29, i73–i83. [Google Scholar] [CrossRef]
- Lorenz, R.T. A Technical Review of Haematococcus Algae. NatuRoseTM Technical Bulletin #060. Cyanotech Corporation: Kailua-Kona, HI, USA , 1999; p. 12. [Google Scholar]
- Halldorsson, A.; Haraldsson, G. Fatty acid selectivity of microbial lipase and lipolytic enzymes from salmonid fish intestines toward astaxanthin diesters. J. Am. Oil Chem. Soc. 2004, 81, 347–353. [Google Scholar]
- Renstrøm, B.; Borch, G.; Skulberg, O.M.; Liaaen-Jensen, S. Optical purity of (3S,3′S)-astaxanthin from Haematococcus pluvialis. Phytochemistry 1981, 20, 2561–2564. [Google Scholar]
- Holeton, C.; Lindell, K.; Holmborn, T.; Hogfors, H.; Gorokhova, E. Decreased astaxanthin at high feeding rates in the calanoid copepod Acartia bifilosa. J. Plankton Res. 2009, 31, 661–668. [Google Scholar] [CrossRef]
- Lorenz, R.T. A Review of Spirulina and Haematococcus algae meal as a Carotenoid and Vitamin Supplement for Poultry. Spirulina Pacifica Technical Bulletin #053. Cyanotech Corporation: Kailua-Kona, HI, USA, 1999; p. 13. [Google Scholar]
- Kuo, H.-C.; Lee, T.-C.; Chichester, C.O.; Simpson, K.L. The carotenoids in the deep sea red crab, Geryon quinquedens. Comp. Biochem. Physiol. B Comp. Biochem. 1976, 54, 387–390. [Google Scholar] [CrossRef]
- Woodall, A.A.; Lee, S.W.-M.; Weesie, R.J.; Jackson, M.J.; Britton, G. Oxidation of carotenoids by free radicals: Relationship between structure and reactivity. Biochim. Biophys. Acta Gen. Subj. 1997, 1336, 33–42. [Google Scholar] [CrossRef]
- Conn, P.F.; Schalch, W.; Truscott, T.G. The singlet oxygen and carotenoid interaction. J. Photochem. Photobiol. B: Biol. 1991, 11, 41–47. [Google Scholar] [CrossRef]
- Mortensen, A.; Skibsted, L.H. Kinetics of parallel electron transfer from β-carotene to phenoxyl radical and adduct formation between phenoxyl radical and β-carotene. Free Radic. Res. 1996, 25, 515–523. [Google Scholar]
- Sommaruga, R. The role of solar UVR in the ecology of alpine lakes. J. Photochem. Photobiol. B Biol. 2001, 62, 35–42. [Google Scholar]
- Sommer, F.; Agurto, C.; Henriksen, P.; Kiørboe, T. Astaxanthin in the calanoid copepod Calanus helgolandicus: Dynamics of esterification and vertical distribution in the German Bight, North Sea. Mar. Ecol. Prog. Ser. 2006, 319, 167–173. [Google Scholar] [CrossRef]
- McNulty, H.; Jacob, R.F.; Mason, R.P. Biologic activity of carotenoids related to distinct membrane physicochemical interactions. Am. J. Cardiol. 2008, 101, S20–S29. [Google Scholar]
- Gruszecki, W.I.; Frank, H.A.; Young, A.J.; Britton, G.; Cogdell, R.J. The Photochemistry of Carotenoids; Kluwer Academic: Dordrecht, The Netherlands , 1999. [Google Scholar]
- Finkelstein, A. Water and nonelectrolyte permeability of lipid bilayer membranes. J. Gen. Physiol. 1976, 68, 127–135. [Google Scholar]
- Snel, M.M.E.; Marsh, D. Accessibility of spin-labeled phospholipids in anionic and zwitterionic bilayer membranes to paramagnetic relaxation agents. Continuous wave power saturation EPR studies. Biochim. Biophys. Acta Biomembr. 1993, 1150, 155–161. [Google Scholar] [CrossRef]
- Babu, T.S.; Marder, J.B.; Tripuranthakam, S.; Dixon, D.G.; Greenberg, B.M. Synergistic effects of a photooxidized polycyclic aromatic hydrocarbon and copper on photosynthesis and plant growth: Evidence that in vivo formation of reactive oxygen species is a mechanism of copper toxicity. Environ. Toxicol. Chem. 2001, 20, 1351–1358. [Google Scholar]
- van Kuijk, F.J.G.M.; Siems, W.G.; Sommerburg, O. Chapter 12: Oxidative Breakdown of Carotenoids and Biological Effects of Their Metabolites. In Handbook of Antioxidants; Packer, L., Cadenas, E., Eds.; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Bell, J.G.; McEvoy, J.; Tocher, D.R.; Sargent, J.R. Depletion of alpha-tocopherol and astaxanthin in Atlantic salmon (Salmo salar) affects autoxidative defense and fatty acid metabolism. J. Nutr. 2000, 130, 1800–1808. [Google Scholar]
- Fleeger, J.W.; Gust, K.A.; Marlborough, S.J.; Tita, G. Mixtures of metals and polynuclear aromatic hydrocarbons elicit complex, nonadditive toxicological interactions in meiobenthic copepods. Environ. Toxicol. Chem. 2007, 26, 1677–1685. [Google Scholar]
- Lee, K.W.; Raisuddin, S.; Hwang, D.S.; Park, H.G.; Lee, J.S. Acute toxicities of trace metals and common xenobiotics to the marine copepod Tigriopus japonicus: Evaluation of its use as a benchmark species for routine ecotoxicity tests in Western Pacific coastal regions. Environ. Toxicol. 2007, 22, 532–538. [Google Scholar] [CrossRef]
- Verriopoulos, G.; Moraïtou-Apostolopoulou, M. Differentiation of the sensitivity to copper and cadmium in different life stages of a copepod. Mar. Pollut. Bull. 1982, 113, 123–125. [Google Scholar]
- USEPA, . Methods for Measuring the Acute Toxicity of Effluents and Receiving Waters to Freshwater and Marine Organisms, 5th edEnvironmental Protection Agency: Washington, DC, USA, 2002; p. 266.
- ASTM E 2317-04. Standard Guide for Conducting Renewal Microplate-Based Life-Cycle Toxicity Tests with a Marine Meiobenthic Copepod; ASTM International: West Conshohochen, PA, USA, 2004; p. 15.
- Medina, M.H.; Morandi, B.; Correa, J.A. Copper effects in the copepod Tigriopus angulatus Lang, 1933: Natural broad tolerance allows maintenance of food webs in copper-enriched coastal areas. Mar. Freshw. Res. 2008, 59, 1061–1066. [Google Scholar] [CrossRef]
- Raisuddin, S.; Kwok, K.W.H.; Leung, K.M.Y.; Schlenk, D.; Lee, J.-S. The copepod Tigriopus: A promising marine model organism for ecotoxicology and environmental genomics. Aquat. Toxicol. 2007, 83, 161–173. [Google Scholar] [CrossRef]
- Bengtsson, B.E. Use of a harpacticoid copepod in toxicity tests. Mar. Pollut. Bull. 1978, 9, 238–241. [Google Scholar]
- Barkaa, S.; Pavillon, J.-F.; Amiard, J.-C. Influence of different essential and non-essential metals on MTLP levels in the copepod Tigriopus brevicornis. Comp. Biochem. Phys. C 2001, 128, 479–493. [Google Scholar]
- O’Brien, P.; Feldmen, H.; Grill, E.V.; Lewis, A.G. Copper tolerance of the life history stages of the splashpool copepod Tigriopus californicus (Copepoda, Harpacticoida). Mar. Ecol. Prog. Ser. 1988, 44, 59–64. [Google Scholar] [CrossRef]
- Kwok, K.W.H.; Leung, K.M.Y. 2005. Toxicity of antifouling biocides to the intertidal harpacticoid copepod Tigriopus japonicus (Crustacea, Copepoda): Effects of temperature and salinity. Mar. Pollut. Bull. 2005, 51, 830–837. [Google Scholar] [CrossRef]
- Kwok, K.W.; Leung, K.M.; Bao, V.W.; Lee, J.S. Copper toxicity in the marine copepod Tigropus japonicus: Low variability and high reproducibility of repeated acute and life-cycle tests. Mar. Pollut. Bull. 2008, 57, 632–636. [Google Scholar] [CrossRef]
- Diz, F.R.; Araújo, C.V.M.; Moreno-Garrido, I.; Hampel, M.; Blasco, J. Short-term toxicity tests on the harpacticoid copepod Tisbe battagliai: Lethal and reproductive endpoints. Ecotoxicol. Environ. Saf. 2009, 72, 1881–1886. [Google Scholar] [CrossRef]
- Lotufo, G.R.; Fleeger, J.W. Description of Amphiascoides atopus, a new species (Crustacea, Copepoda, Harpacticoida) from a mass culture system. Proc. Biol. Soc. Wash. 1995, 108, 117–124. [Google Scholar]
- Sun, B.; Fleeger, J.W. Sustained mass culture of Amphiascoides atopus a marine harpacticoid copepod in a recirculating system. Aquaculture 1995, 136, 313–321. [Google Scholar] [CrossRef]
- Bousquet, O.; Sellier, N.; Le Goffic, F. Characterization and purification of polyunsaturated fatty acids from microalgae by gas chromatography-mass spectrometry and counter current chromatography. Chromatographia 1994, 39, 40–44. [Google Scholar]
- Nanton, D.A.; Castell, J.D. Mass Culture of the Harpacticoid Copepod, Tisbe sp.; ICES CM Marine Fish Culture Committee: Murcia, Spain, 1997; pp. 1–10.
- Jacobs, P.B.; LeBoeuf, R.D.; McCommas, S.A.; Tauber, J.D. The cleavage of carotenoid esters by cholesterol esterase. Comp. Biochem. Physiol. B Comp. Biochem. 1982, 72, 157–160. [Google Scholar]
- Kraay, G.W.; Zapata, M.; Veldhuis, M.J.W. Separation of chlorophylls c1c2, and c3 of marine phytoplankton by reversed-phase C18 high performance liquid chromatography. J. Phycol. 1992, 28, 708–712. [Google Scholar]
- Hicks, G.R.F.; Coull, B.C. The ecology of marine meiobenthic harpacticoid copepods. Oceanogr. Mar. Biol. Ann. Rev. 1983, 21, 380–389. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Caramujo, M.-J.; De Carvalho, C.C.C.R.; Silva, S.J.; Carman, K.R. Dietary Carotenoids Regulate Astaxanthin Content of Copepods and Modulate Their Susceptibility to UV Light and Copper Toxicity. Mar. Drugs 2012, 10, 998-1018. https://doi.org/10.3390/md10050998
Caramujo M-J, De Carvalho CCCR, Silva SJ, Carman KR. Dietary Carotenoids Regulate Astaxanthin Content of Copepods and Modulate Their Susceptibility to UV Light and Copper Toxicity. Marine Drugs. 2012; 10(5):998-1018. https://doi.org/10.3390/md10050998
Chicago/Turabian StyleCaramujo, Maria-José, Carla C. C. R. De Carvalho, Soraya J. Silva, and Kevin R. Carman. 2012. "Dietary Carotenoids Regulate Astaxanthin Content of Copepods and Modulate Their Susceptibility to UV Light and Copper Toxicity" Marine Drugs 10, no. 5: 998-1018. https://doi.org/10.3390/md10050998
APA StyleCaramujo, M.-J., De Carvalho, C. C. C. R., Silva, S. J., & Carman, K. R. (2012). Dietary Carotenoids Regulate Astaxanthin Content of Copepods and Modulate Their Susceptibility to UV Light and Copper Toxicity. Marine Drugs, 10(5), 998-1018. https://doi.org/10.3390/md10050998






