Low Skeletal Muscle Index as a Predictor of Pathological Complete Response in HER-2 Positive and Triple-Negative Breast Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Data Collection
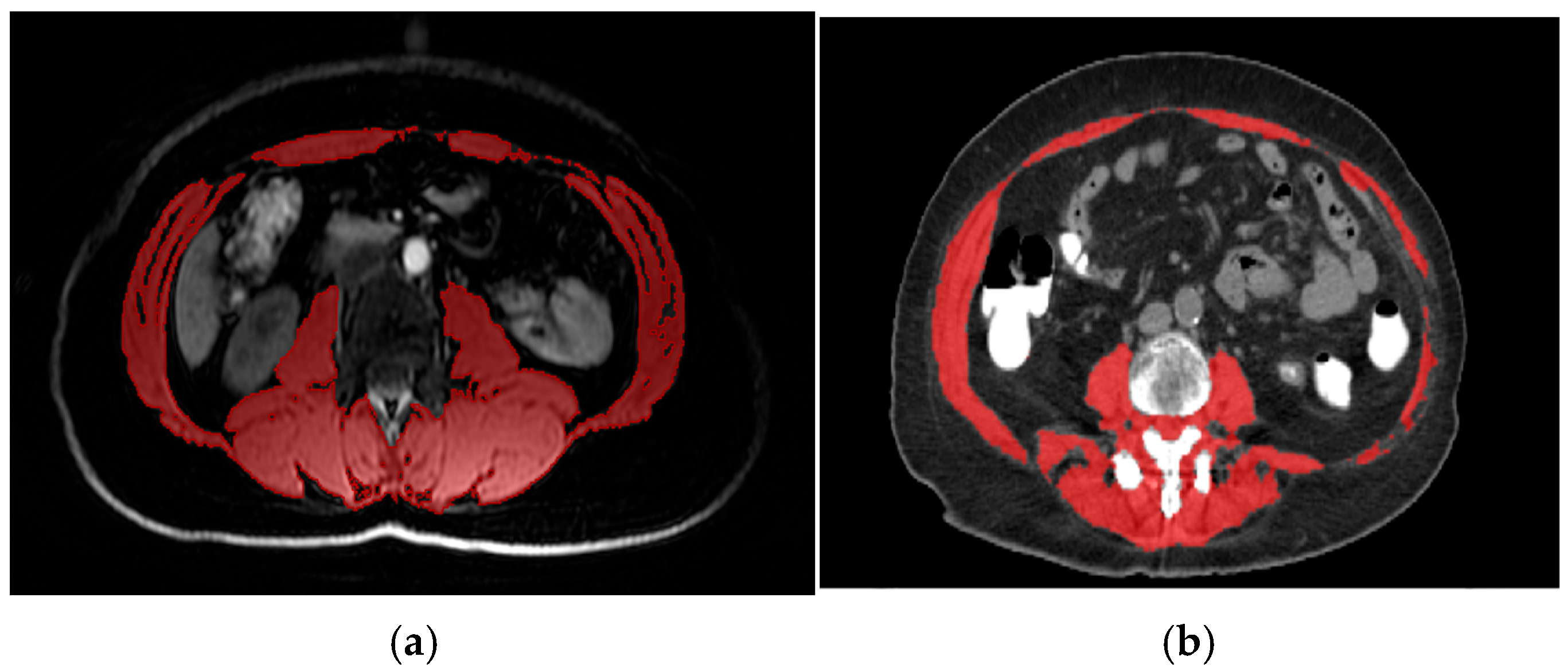
2.2. Assessment of Body Composition and Definition of Low SMI
2.3. Statistical Analysis
3. Results
3.1. Characteristics of Patients
3.2. Pathological Complete Response Outcomes
3.3. Treatment-Related Toxicity Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BCS | Breast-conserving surgery |
| BIA | Bioelectrical impedance analysis |
| BMI | Body mass index |
| CI | Confidence interval |
| cN stage | Clinical nodal stage |
| cTstage | Clinical tumor stage |
| CT | Computed tomography |
| DFS | Disease-free survival |
| DEXA | Dual-energy X-ray absorptiometry |
| HER-2 | Human epidermal growth factor receptor-2 |
| HR | Hormone receptor |
| HU | Hounsfield unit |
| L3 | Third lumbar vertebra |
| MRI | Magnetic resonance imaging |
| OR | Odds ratio |
| OS | Overall survival |
| pCR | Pathological complete response |
| PET | Positron Emission Tomography |
| PET-CT | Positron Emission Tomography/Computed Tomography |
| PET-MRI | Positron Emission Tomography/Magnetic resonance imaging |
| ROC | Receiver Operating Characteristic |
| SMA | Skeletal Muscle Area |
| SMI | Skeletal Muscle Index |
| TNBC | Triple-Negative Breast Cancer |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Harbeck, N.; Gnant, M. Breast cancer. Lancet 2017, 389, 1134–1150. [Google Scholar] [CrossRef]
- Denkert, C.; Von Minckwitz, G.; Darb-Esfahani, S.; Lederer, B.; Heppner, B.I.; Weber, K.E.; Budczies, J.; Huober, J.; Klauschen, F.; Furlanetto, J.; et al. Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: A pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol. 2018, 19, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Haque, W.; Verma, V.; Hatch, S.; Klimberg, V.S.; Butler, E.B.; Teh, B.S. Response rates and pathologic complete response by breast cancer molecular subtype following neoadjuvant chemotherapy. Breast Cancer Res. Treat. 2018, 170, 559–567. [Google Scholar] [CrossRef]
- Spring, L.M.; Fell, G.; Arfe, A.; Sharma, C.; Greenup, R.; Reynolds, K.L.; Smith, B.L.; Alexander, B.; Moy, B.; Isakoff, S.J.; et al. Pathologic complete response after neoadjuvant chemotherapy and impact on breast cancer recurrence and survival: A comprehensive meta-analysis. Clin. Cancer Res. 2020, 26, 2838–2848. [Google Scholar] [CrossRef]
- Guliyev, M.; Alan, Ö.; Günaltılı, M.; Safarov, S.; Fidan, M.C.; Şen, G.A.; Değerli, E.; Papila, B.; Demirci, N.S.; Papila, Ç. Obesity is an independent prognostic factor that reduced pathological complete response in operable breast cancer patients. Medicina 2024, 60, 1953. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Cesari, M.; Landi, F.; Vellas, B.; Bernabei, R.; Marzetti, E. Sarcopenia and physical frailty: Two sides of the same coin. Front. Aging Neurosci. 2014, 6, 192. [Google Scholar] [CrossRef] [PubMed]
- Plank, L.D. Dual-energy X-ray absorptiometry and body composition. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 305–309. [Google Scholar] [CrossRef]
- Hansen, R.D.; Williamson, D.A.; Finnegan, T.P.; Lloyd, B.D.; Grady, J.N.; Diamond, T.H.; Smith, E.U.; Stavrinos, T.M.; Thompson, M.W.; Gwinn, T.H.; et al. Estimation of thigh muscle cross-sectional area by dual-energy X-ray absorptiometry in frail elderly patients. Am. J. Clin. Nutr. 2007, 86, 952–958. [Google Scholar] [CrossRef]
- Prado, C.M.; Lieffers, J.R.; McCargar, L.J.; Reiman, T.; Sawyer, M.B.; Martin, L.; Baracos, V.E. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol. 2008, 9, 629–635. [Google Scholar] [CrossRef]
- Albano, D.; Pasinetti, N.; Dondi, F.; Giubbini, R.; Tucci, A.; Bertagna, F. Prognostic role of pre-treatment metabolic parameters and sarcopenia derived by 2-[18F]-FDG PET/CT in elderly mantle cell lymphoma. J. Clin. Med. 2022, 11, 1210. [Google Scholar] [CrossRef]
- Albano, D.; Camoni, L.; Rinaldi, R.; Tucci, A.; Zilioli, V.R.; Muzi, C.; Ravanelli, M.; Farina, D.; Coppola, A.; Camalori, M.; et al. Comparison between skeletal muscle and adipose tissue measurements with high-dose CT and low-dose attenuation correction CT of 18F-FDG PET/CT in elderly Hodgkin lymphoma patients: A two-centre validation. Br. J. Radiol. 2021, 94, 20200672. [Google Scholar] [CrossRef]
- Yuan, H.; Tan, X.; Sun, X.; He, L.; Li, D.; Jiang, L. Role of 18F-FDG PET/CT and sarcopenia in untreated non-small cell lung cancer with advanced stage. Jpn. J. Radiol. 2023, 41, 521–530. [Google Scholar] [CrossRef]
- Zwart, A.T.; Kok, L.M.C.; de Vries, J.; van Kester, M.S.; Dierckx, R.A.J.O.; de Bock, G.H.; van der Hoorn, A.; Halmos, G.B. Radiologically Defined Sarcopenia as a Biomarker for Frailty and Malnutrition in Head and Neck Skin Cancer Patients. J. Clin. Med. 2023, 12, 3445. [Google Scholar] [CrossRef]
- Orsaria, P.; Grasso, A.; Ippolito, E.; Pantano, F.; Sammarra, M.; Altomare, C.; Cagli, B.; Costa, F.; Perrone, G.; Soponaru, G.; et al. Clinical outcomes among major breast cancer subtypes after neoadjuvant chemotherapy: Impact on breast cancer recurrence and survival. Anticancer Res. 2021, 41, 2697–2709. [Google Scholar] [CrossRef]
- Zhao, B.; Zhao, H.; Zhao, J. Impact of hormone receptor status on the efficacy of HER2-targeted treatment. Endocr.-Relat. Cancer 2018, 25, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Boughey, J.C.; McCall, L.M.; Ballman, K.V.; Mittendorf, E.A.; Ahrendt, G.M.; Wilke, L.G.; Taback, B.; Leitch, A.M.; Flippo-Morton, T.; Hunt, K.K. Tumor biology correlates with rates of breast-conserving surgery and pathologic complete response after neoadjuvant chemotherapy for breast cancer: Findings from the ACOSOG Z1071 (Alliance) Prospective Multicenter Clinical Trial. Ann. Surg. 2014, 260, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.-X.; Qin, Q.-H.; Yang, W.-P.; Mo, Q.-G.; Wei, C.-Y. Prognostic value of Ki67 expression in HR-negative breast cancer before and after neoadjuvant chemotherapy. Int. J. Clin. Exp. Pathol. 2014, 7, 6862–6870. [Google Scholar] [PubMed]
- Wunderle, M.; Gass, P.; Häberle, L.; Flesch, V.M.; Rauh, C.; Bani, M.R.; Hack, C.C.; Schrauder, M.G.; Jud, S.M.; Emons, J.; et al. BRCA mutations and their influence on pathological complete response and prognosis in a clinical cohort of neoadjuvantly treated breast cancer patients. Breast Cancer Res. Treat. 2018, 171, 85–94. [Google Scholar] [CrossRef]
- Shachar, S.S.; Deal, A.M.; Weinberg, M.; Nyrop, K.A.; Williams, G.R.; Nishijima, T.F.; Benbow, J.M.; Muss, H.B. Skeletal muscle measures as predictors of toxicity, hospitalization, and survival in patients with metastatic breast cancer receiving taxane-based chemotherapy. Clin. Cancer Res. 2017, 23, 658–665. [Google Scholar] [CrossRef]
- Caan, B.J.; Feliciano, E.M.C.; Prado, C.M.; Alexeeff, S.; Kroenke, C.H.; Bradshaw, P.; Quesenberry, C.P.; Weltzien, E.K.; Castillo, A.L.; Olobatuyi, T.A.; et al. Association of muscle and adiposity measured by computed tomography with survival in patients with nonmetastatic breast cancer. JAMA Oncol. 2018, 4, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Bellieni, A.; Fusco, D.; Sanchez, A.M.; Franceschini, G.; Di Capua, B.; Allocca, E.; Di Stasio, E.; Marazzi, F.; Tagliaferri, L.; Masetti, R.; et al. Different impact of definitions of sarcopenia in defining frailty status in a population of older women with early breast cancer. J. Pers. Med. 2021, 11, 243. [Google Scholar] [CrossRef]
- Bilen, M.A.; Martini, D.J.; Liu, Y.; Shabto, J.M.; Brown, J.T.; Williams, M.; Khan, A.I.; Speak, A.; Lewis, C.; Collins, H.; et al. Combined Effect of Sarcopenia and Systemic Inflammation on Survival in Patients with Advanced Stage Cancer Treated with Immunotherapy. Oncologist 2020, 25, e528–e535. [Google Scholar] [CrossRef]
- Del Fabbro, E.; Parsons, H.; Warneke, C.L.; Pulivarthi, K.; Litton, J.K.; Dev, R.; Palla, S.L.; Brewster, A.; Bruera, E. The relationship between body composition and response to neoadjuvant chemotherapy in women with operable breast cancer. Oncologist 2012, 17, 1240–1245. [Google Scholar] [CrossRef]
- Ogston, K.N.; Miller, I.D.; Payne, S.; Hutcheon, A.W.; Sarkar, T.K.; Smith, I.; Schofield, A.; Heys, S.D. A new histological grading system to assess response of breast cancers to primary chemotherapy: Prognostic significance and survival. Breast 2003, 12, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.H.; Kim, M.Y.; Seo, Y.S.; Kim, S.G.; Lee, H.A.; Kim, T.H.; Jung, Y.K.; Kandemir, A.; Kim, J.H.; An, H.; et al. Clinical usefulness of psoas muscle thickness for the diagnosis of sarcopenia in patients with liver cirrhosis. Clin. Mol. Hepatol. 2018, 24, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Yip, C.; Dinkel, C.; Mahajan, A.; Siddique, M.; Cook, G.; Goh, V. Imaging body composition in cancer patients: Visceral obesity, sarcopenia and sarcopenic obesity may impact on clinical outcome. Insights Imaging 2015, 6, 489–497. [Google Scholar] [CrossRef]
- Bursac, Z.; Gauss, C.H.; Williams, D.K.; Hosmer, D.W. Purposeful selection of variables in logistic regression. Source Code Biol. Med. 2008, 3, 17. [Google Scholar] [CrossRef]
- Findlay, M.; White, K.; Lai, M.; Luo, D.; Bauer, J.D. The Association Between Computed Tomography-Defined Sarcopenia and Outcomes in Adult Patients Undergoing Radiotherapy of Curative Intent for Head and Neck Cancer: A Systematic Review. J. Acad. Nutr. Diet. 2020, 120, 1330–1347.e8. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.; Baracos, V.E.; McCargar, L.J.; Reiman, T.; Mourtzakis, M.; Tonkin, K.; Mackey, J.R.; Koski, S.; Pituskin, E.; Sawyer, M.B. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin. Cancer Res. 2009, 15, 2920–2926. [Google Scholar] [CrossRef]
- Roberto, M.; Barchiesi, G.; Resuli, B.; Verrico, M.; Speranza, I.; Cristofani, L.; Pediconi, F.; Tomao, F.; Botticelli, A.; Santini, D. Sarcopenia in Breast Cancer Patients: A Systematic Review and Meta-Analysis. Cancers 2024, 16, 596. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Lan, J.; Li, S.; Xu, G. Exploring the impact of sarcopenia on mortality in breast cancer patients: A comprehensive systematic review and meta-analysis. Breast Care 2024, 19, 316–328. [Google Scholar] [CrossRef]
- Simmons, L.O.; Cagney, D.; Hassan, F.; Lim, J.Y.; O’leary, D.P.; Liew, A.; Redmond, H.P.; Corrigan, M.; O’sullivan, M.; Kelly, L. Prevalence of sarcopenia and its impact on survival in breast cancer—A systematic review and meta-analysis. Mesentery Peritoneum 2019, 3, AB020. [Google Scholar] [CrossRef]
- Karaca, M.; Alemdar, M.S.; Karaca, Ö.D.; Kılar, Y.; Köker, G.; Sözel, H.; Yıldız, M.; Köker, G.Ö.; Arici, M.Ö. Sarcopenia’s Role in Neoadjuvant Chemotherapy Outcomes for Locally Advanced Breast Cancer: A Retrospective Analysis. Med. Sci. Monit. 2024, 30, e945240. [Google Scholar] [CrossRef]
- Sun, X.; Xu, J.; Chen, X.; Zhang, W.; Chen, W.; Zhu, C.; Sun, J.; Yang, X.; Wang, X.; Hu, Y.; et al. Sarcopenia in patients with normal body mass index is an independent predictor for postoperative complication and long-term survival in gastric cancer. Clin. Transl. Sci. 2020, 14, 137–146. [Google Scholar] [CrossRef]
- Du, L.; Liu, X.; Zhu, Q.; Zhu, K.; Li, P. Sarcopenia as a prognostic factor and multimodal interventions in breast cancer. Int. J. Gen. Med. 2024, 17, 6605–6616. [Google Scholar] [CrossRef]
- Zhang, X.; Dou, Q.; Zeng, Y.; Yang, Y.; Cheng, A.; Zhang, W. Sarcopenia as a predictor of mortality in women with breast cancer: A meta-analysis and systematic review. BMC Cancer 2020, 20, 172. [Google Scholar] [CrossRef] [PubMed]
- Isıklar, A.; Yilmaz, E.; Basaran, G. The Relationship Between Body Composition and Pathological Response to Neoadjuvant Chemotherapy in Breast Cancer Patients. Cureus 2024, 16, e61145. [Google Scholar] [CrossRef]
- Guo, J.; Meng, W.; Li, Q.; Zheng, Y.; Yin, H.; Liu, Y.; Zhao, S.; Ma, J. Pretreatment sarcopenia and MRI-based radiomics to predict the response of neoadjuvant chemotherapy in triple-negative breast cancer. Bioengineering 2024, 11, 663. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Hu, K.; Gao, J.; Shang, Y.; Mei, F.; Zhao, L.; Chen, F.; Ma, B. Prevalence and prognostic value of sarcopenic obesity in patients with cancer: A systematic review and meta-analysis. Nutrition 2022, 101, 111704. [Google Scholar] [CrossRef]
- Aleixo, G.F.P.; Williams, G.R.; Nyrop, K.A.; Muss, H.B.; Shachar, S.S. Muscle composition and outcomes in patients with breast cancer: Meta-analysis and systematic review. Breast Cancer Res. Treat. 2019, 177, 569–579. [Google Scholar] [CrossRef]
- Jang, M.K.; Park, S.; Raszewski, R.; Park, C.G.; Doorenbos, A.Z.; Kim, S. Prevalence and clinical implications of sarcopenia in breast cancer: A systematic review and meta-analysis. Support. Care Cancer 2024, 32, 328. [Google Scholar] [CrossRef]
- Matuszczak, M.; Kiljańczyk, A.; Marciniak, W.; Derkacz, R.; Stempa, K.; Baszuk, P.; Bryśkiewicz, M.; Cybulski, C.; Dębniak, T.; Gronwald, J.; et al. Antioxidant Properties of Zinc and Copper-Blood Zinc-to Copper-Ratio as a Marker of Cancer Risk BRCA1 Mutation Carriers. Antioxidants 2024, 13, 841. [Google Scholar] [CrossRef] [PubMed]
- Deluche, E.; Lachatre, D.; Di Palma, M.; Simon, H.; Tissot, V.; Vansteene, D.; Meingan, P.; Mohebi, A.; Lenczner, G.; Pigneur, F.; et al. Is sarcopenia a missed factor in the management of patients with metastatic breast cancer? Breast 2022, 61, 84–90. [Google Scholar] [CrossRef] [PubMed]
| Variables | All Patients n = 85 (%) | Normal SMI n = 50 (%) | Low SMI n = 35 (%) | p |
|---|---|---|---|---|
| Age (years) | 0.409 | |||
| Median (min–max) | 49 (24–77) | 49 (29–71) | 47 (24–77) | |
| BMI (kg/m2) | 0.003 | |||
| Median (min–max) | 28.3 (18.3–44.3) | 29.5 (20.0–44.3) | 25.8 (18.3–38.9) | |
| Menopausal Status, n (%) | 0.713 | |||
| Premenopausal | 49 (57.6) | 28 (56.0) | 21 (60.0) | |
| Postmenopausal | 36 (42.4) | 22 (44.0) | 14 (40.0) | |
| Histological type, n (%) | 0.918 | |||
| Invasive ductal carcinoma | 72 (84.7) | 43 (86.0) | 29 (82.9) | |
| Invasive lobular carcinoma | 2 (2.4) | 1 (2.0) | 1 (2.9) | |
| Others | 11 (12.9) | 6 (12.0) | 5 (14.3) | |
| Molecular subtype, n (%) | 0.682 | |||
| HR+, HER-2 + | 33 (38.8) | 21 (42.0) | 12 (34.3) | |
| HR−, HER-2 + | 16 (18.8) | 8 (16.0) | 8 (22.9) | |
| Triple negative | 36 (42.4) | 21 (42.0) | 15 (42.9) | |
| Histologic grade, n (%) | 0.814 | |||
| Grade 1–2 | 26 (30.6) | 16 (32.0) | 10 (28.6) | |
| Grade 3 | 59 (69.4) | 34 (68.0) | 25 (71.4) | |
| Ki-67 (%) | 0.635 | |||
| Median (min–max) | 45 (5–90) | 45 (5–90) | 40 (15–90) | |
| cT Stage, n (%) | 0.622 | |||
| T1 | 6 (7.1) | 4 (8.0) | 2 (5.7) | |
| T2 | 54 (63.5) | 33 (66.0) | 21 (60.0) | |
| T3 | 13 (15.3) | 8 (16.0) | 5 (14.3) | |
| T4 | 12 (14.1) | 5 (10.0) | 7 (20.0) | |
| cN Stage, n (%) | 0.160 | |||
| N0 | 17 (20.0) | 14 (28.0) | 3 (8.6) | |
| N1 | 33 (38.8) | 18 (36.0) | 15 (42.9) | |
| N2 | 20 (23.5) | 11 (22.0) | 9 (25.7) | |
| N3 | 15 (17.6) | 7 (14.0) | 8 (22.9) | |
| Clinical TNM Stage, n (%) | 0.320 | |||
| Stage II | 37 (43.5) | 24 (48.0) | 13 (37.1) | |
| Stage III | 48 (56.5) | 26 (52.0) | 22 (62.9) | |
| Radiological evaluation method, n (%) | 0.169 | |||
| PET-CT | 44 (51.8) | 29 (58.0) | 15 (42.9) | |
| PET-MRI | 41 (48.2) | 21 (42.0) | 20 (57.1) | |
| Surgery, n (%) | 0.804 | |||
| BCS | 28 (32.9) | 17 (34.0) | 11 (31.4) | |
| Mastectomy | 57 (67.1) | 33 (66.0) | 24 (68.6) | |
| Neoadjuvant treatment regimen, n (%) * | 0.236 | |||
| Anthracycline and taxane based with trastuzumab | 15 (30.6) | 7 (24.1) | 8 (40.0) | |
| Anthracycline and taxane based with trastuzumab and pertuzumab | 34 (69.4) | 22 (75.9) | 12 (60.0) | |
| Neoadjuvant treatment regimen, n (%) ** | 0.955 | |||
| Anthracycline and taxane based without platinum | 19 (52.8) | 11 (52.4) | 8 (53.3) | |
| Anthracycline and taxane based with platinum | 17 (47.2) | 10 (47.6) | 7 (46.7) |
| Variables | Pathologic Response | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|---|
| CR n = 45 (%) | Non-CR n = 40 (%) | OR (95% CI) | p | OR (95% CI) | p | ||
| Age (years) | <49 | 23 (56.1) | 18 (43.9) | Ref. | |||
| ≥49 | 22 (50.0) | 22 (50.0) | 0.78 (0.33–1.84) | 0.574 | |||
| Menopausal Status | Pre | 27 (55.1) | 22 (44.9) | Ref. | |||
| Post | 18 (50.0) | 18 (50.0) | 0.81 (0.34–1.93) | 0.642 | |||
| BMI (kg/m2) | <30 | 30 (53.6) | 26 (46.4) | Ref. | |||
| ≥30 | 15 (51.7) | 14 (48.3) | 0.93 (0.38–2.28) | 0.871 | |||
| HR status | Negative | 24 (46.2) | 28 (53.8) | Ref | |||
| Positive | 21 (63.6) | 12 (36.4) | 2.04 (0.83–4.99) | 0.118 | 1.29 (0.43–3.93) | 0.648 | |
| Grade | Grade 1–2 | 17 (65.4) | 9 (34.6) | Ref. | |||
| Grade 3 | 28 (47.5) | 31 (52.5) | 0.48 (0.18–1.24) | 0.130 | 0.57 (0.18–1.79) | 0.340 | |
| Ki 67 | <45 | 23 (60.5) | 15 (39.5) | Ref. | |||
| ≥45 | 22 (46.8) | 25 (53.2) | 0.57 (0.24–1.37) | 0.209 | 0.42 (0.14–1.30) | 0.132 | |
| cT Stage | T3–4 | 9 (36.0) | 16 (64.0) | Ref. | |||
| T1–2 | 36 (60.0) | 24 (40.0) | 2.66 (1.01–7.01) | 0.047 | 3.08 (0.95–10.07) | 0.062 | |
| cN Stage | N1–3 | 32 (47.1) | 36 (52.9) | Ref. | |||
| N0 | 13 (76.5) | 4 (23.5) | 3.66 (1.08–12.35) | 0.037 | 1.84 (0.38–8.95) | 0.450 | |
| Clinical Stage | Stage III | 21 (43.8) | 27 (56.2) | Ref. | |||
| Stage II | 24 (64.9) | 13 (35.1) | 2.37 (0.98–5.74) | 0.055 | 1.06 (0.28–3.97) | 0.936 | |
| Low SMI | Yes | 11 (31.4) | 24 (68.6) | Ref. | |||
| No | 34 (68.0) | 16 (32.0) | 4.64 (1.83–11.73) | 0.001 | 5.17 (1.74–15.40) | 0.003 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Günaltılı, M.; Guliyev, M.; Fidan, M.C.; Birsin, Z.; Çerme, E.; Aliyev, V.; Abbasov, H.; Cebeci, S.; Jeral, S.; Alan, Ö.; et al. Low Skeletal Muscle Index as a Predictor of Pathological Complete Response in HER-2 Positive and Triple-Negative Breast Cancer. Medicina 2025, 61, 1508. https://doi.org/10.3390/medicina61091508
Günaltılı M, Guliyev M, Fidan MC, Birsin Z, Çerme E, Aliyev V, Abbasov H, Cebeci S, Jeral S, Alan Ö, et al. Low Skeletal Muscle Index as a Predictor of Pathological Complete Response in HER-2 Positive and Triple-Negative Breast Cancer. Medicina. 2025; 61(9):1508. https://doi.org/10.3390/medicina61091508
Chicago/Turabian StyleGünaltılı, Murat, Murad Guliyev, Mehmet Cem Fidan, Zeliha Birsin, Emir Çerme, Vali Aliyev, Hamza Abbasov, Selin Cebeci, Seda Jeral, Özkan Alan, and et al. 2025. "Low Skeletal Muscle Index as a Predictor of Pathological Complete Response in HER-2 Positive and Triple-Negative Breast Cancer" Medicina 61, no. 9: 1508. https://doi.org/10.3390/medicina61091508
APA StyleGünaltılı, M., Guliyev, M., Fidan, M. C., Birsin, Z., Çerme, E., Aliyev, V., Abbasov, H., Cebeci, S., Jeral, S., Alan, Ö., Demirci, N. S., Papila, Ç., Şahin, O. E., Bıyıkoğlu, S. E., Öztürk, T., & Papila, B. (2025). Low Skeletal Muscle Index as a Predictor of Pathological Complete Response in HER-2 Positive and Triple-Negative Breast Cancer. Medicina, 61(9), 1508. https://doi.org/10.3390/medicina61091508





