Recovery and Recurrence in Bell’s Palsy: A Propensity Score-Matched Comparative Study Across ENT, Pain Medicine, and Traditional Korean Medicine
Abstract
1. Introduction
Korean Healthcare Context
2. Materials and Methods
2.1. Study Design and Setting
Diagnostic Criteria
2.2. Study Population
2.3. Treatment Group Classification
2.4. Data Collection
2.5. Outcomes
2.6. Propensity Score Matching and Quality Control
2.7. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Treatment Outcomes
3.3. Predictors of Complete Recovery
3.4. Recurrence Risk Factors
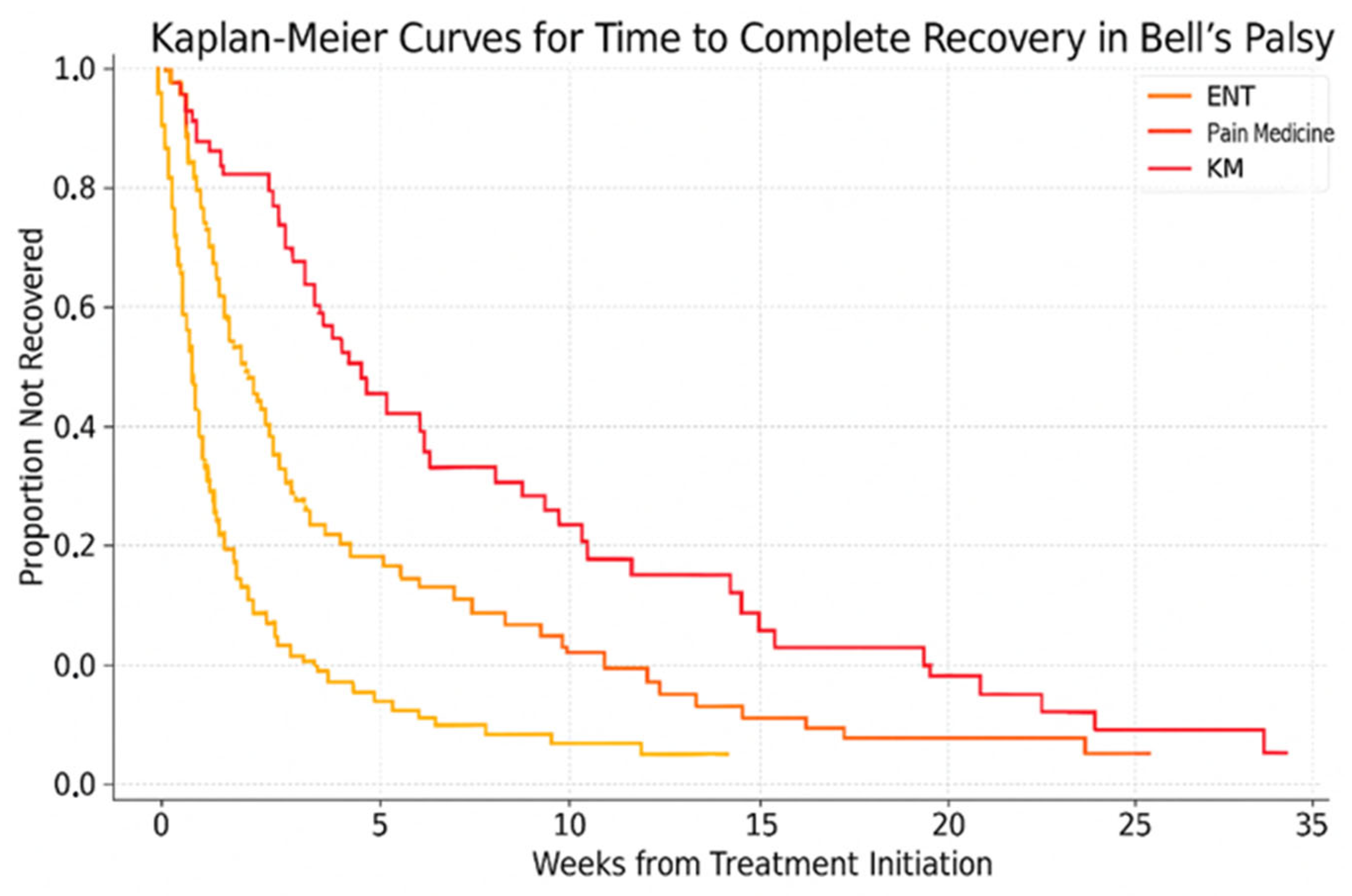
3.5. Time to Complete Recovery by Department
3.6. Subgroup Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Definition |
| ENT | Ear, Nose, and Throat |
| KM | Traditional Korean Medicine |
| HB | House–Brackmann |
| PSM | Propensity Score Matching |
| OR | Odds Ratio |
| CI | Confidence Interval |
| EMR | Electronic Medical Record |
| ICD-10 | International Classification of Diseases, 10th Revision |
References
- Eviston, T.J.; Croxson, G.R.; Kennedy, P.G.; Hadlock, T.; Krishnan, A.V. Bell’s palsy: Aetiology, clinical features and multidisciplinary care. J. Neurol. Neurosurg. Psychiatry 2015, 86, 1356–1361. [Google Scholar] [PubMed]
- Baugh, R.F.; Basura, G.J.; Ishii, L.E.; Schwartz, S.R.; Drumheller, C.M.; Burkholder, R.; Deckard, N.A.; Dawson, C.; Driscoll, C.; Gillespie, M.B.; et al. Clinical practice guideline: Bell’s palsy. Otolaryngol. Head Neck Surg. 2013, 149, S1–S27. [Google Scholar] [PubMed]
- Peitersen, E. Bell’s palsy: The spontaneous course of 2,500 peripheral facial nerve palsies of different etiologies. Acta Otolaryngol. Suppl. 2002, 122, 4–30. [Google Scholar]
- Cuenca-Martínez, F.; Zapardiel-Sánchez, E.; Carrasco-González, E.; La Touche, R.; Suso-Martí, L. Assessing anxiety, depression and quality of life in patients with peripheral facial palsy: A systematic review. PeerJ 2020, 8, e10449. [Google Scholar] [PubMed]
- Jeong, J.; Chung, J.H.; Ryu, S.; Lee, J.D.; Kim, J.; Lee, H.Y.; Song, C.I.; Cho, Y.S.; Lee, S.A.; Jun, B. Monthly Variation in Bell’s Palsy Based on Population Data of Korea. Audiol. Neurotol. 2024, 29, 290–296. [Google Scholar]
- Kumar, S.; Marlapudi, S.K.; Biradar, K. Unraveling Bell’s Palsy: Exploring HSV’s Role and Facial Therapy’s Impact on Aberrant Reinnervation and Synkinesis. Indian J. Otolaryngol. Head Neck Surg. 2024, 76, 764–769. [Google Scholar] [PubMed]
- Gronseth, G.S.; Paduga, R. Evidence-based guideline update: Steroids and antivirals for Bell palsy: Report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology 2012, 79, 2209–2213. [Google Scholar] [PubMed]
- de Almeida, J.R.; Guyatt, G.H.; Sud, S.; Dorion, J.; Hill, M.D.; Kolber, M.R.; Lea, J.; Reg, S.L.; Somogyi, B.K.; Westerberg, B.D.; et al. Management of Bell palsy: Clinical practice guideline. Can. Med. Assoc. J. 2014, 186, 917–922. [Google Scholar]
- Li, P.; Qiu, T.; Qin, C. Efficacy of acupuncture for Bell’s palsy: A systematic review and meta-analysis of randomized controlled trials. PLoS ONE 2015, 10, e0121880. [Google Scholar]
- Yang, L.-S.; Zhou, D.-F.; Zheng, S.-Z.; Zhao, B.-M.; Li, H.-G.; Chen, Q.-Q.; Zhong, Y.; Yang, H.-Z.; Zhang, K.; Tang, C.-Z. Early intervention with acupuncture improves the outcome of patients with Bell’s palsy: A propensity score-matching analysis. Neurol. 2022, 13, 943453. [Google Scholar]
- Jeong, J.; Yoon, S.R.; Lim, H.; Choi, H.S. Distribution of medical service use for facial palsy between medicine and traditional Korean medicine based on population-based data of Korea. J. Korean Med. Sci. 2022, 37, e119. [Google Scholar] [PubMed]
- Jeong, J.; Yoon, S.R.; Lim, H.; Oh, J.; Choi, H.S. Risk factors for Bell’s palsy based on the Korean national health insurance service national sample cohort data. Sci. Rep. 2021, 11, 23387. [Google Scholar]
- Paolino, E.; Granieri, E.; Tola, M.; Panarelli, M.; Carreras, M. Predisposing factors in Bell’s palsy: A case-control study. J. Neurol. 1985, 232, 363–365. [Google Scholar] [PubMed]
- Kim, D.; Jung, B.; Cho, M.-U.; Song, S.-B.; Chung, S.H.; Park, T.-Y.; Ha, I.-H. Analysis of medical services provided to patients with peripheral facial palsy in Korea: A descriptive, cross-sectional study of the health insurance review and assessment service national patient sample database. BMC Heal. Serv. Res. 2021, 21, 1–11. [Google Scholar]
- Hohman, M.H.; Hadlock, T.A. Etiology, diagnosis, and management of facial palsy: 2000 patients at a facial nerve center. Laryngoscope 2014, 124, E283–E293. [Google Scholar] [PubMed]
- House, J.W.; Brackmann, D.E. Facial nerve grading system. Otolaryngol. Head Neck Surg. 1985, 93, 146–147. [Google Scholar] [PubMed]
- Alkureishi, M.A.; Lee, W.W.; Lyons, M.; Press, V.G.; Imam, S.; Nkansah-Amankra, A.; Werner, D.; Arora, V.M. Impact of electronic medical record use on the patient–doctor relationship and communication: A systematic review. J. Gen. Intern. Med. 2016, 31, 548–560. [Google Scholar] [PubMed]
- Austin, P.C. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar. Behav. Res. 2011, 46, 399–424. [Google Scholar]
- Li, P.; Stuart, E.A.; Allison, D.B. Multiple imputation: A flexible tool for handling missing data. JAMA 2015, 314, 1966–1967. [Google Scholar] [PubMed]
- Walker, N.R.; Mistry, R.K.; Mazzoni, T. Facial nerve palsy.StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Rothwell, P.M. External validity of randomised controlled trials: “To whom do the results of this trial apply?”. Lancet 2005, 365, 82–93. [Google Scholar] [PubMed]
| Group | ENT (n = 200) | Pain Medicine (n = 200) | KM (n = 200) | p-Value | |
|---|---|---|---|---|---|
| Variables | |||||
| Age, years | 42.1 ± 12.4 | 46.7 ± 14.1 | 44.5 ± 13.9 | 0.068 | |
| Age ≥ 65 years | 38 (19.0) | 59 (29.5) | 45 (22.5) | 0.081 | |
| Female, n (%) | 108 (54.0) | 122 (61.0) | 116 (58.0) | 0.143 | |
| Hypertension | 55 (27.5) | 68 (34.0) | 60 (30.0) | 0.192 | |
| Diabetes | 51 (25.5) | 56 (28.0) | 47 (23.5) | 0.327 | |
| Hyperlipidemia | 36 (18.0) | 50 (25.0) | 42 (21.0) | 0.097 | |
| Obesity (BMI > 30) | 38 (19.0) | 45 (22.5) | 40 (20.0) | 0.317 | |
| Pregnancy, n (%) | 6 (3.0) | 4 (2.0) | 7 (3.5) | 0.725 | |
| Autoimmune disease | 13 (6.5) | 18 (9.0) | 15 (7.5) | 0.443 | |
| Hypothyroidism | 19 (9.5) | 23 (11.5) | 21 (10.5) | 0.690 | |
| Initial HB Grade IV | 54 (27.0) | 66 (33.0) | 58 (29.0) | 0.215 | |
| Group | ENT (n = 200) | Pain Medicine (n = 200) | KM (n = 200) | p-Value | Effect Size | |
|---|---|---|---|---|---|---|
| Variables | ||||||
| Complete Recovery (HB 1, 6 mo) | 175 (87.5) | 148 (74.0) | 139 (69.5) | <0.001 | Phi = 0.18 | |
| Time to Recovery, weeks | 4 [3–7] | 6 [4–9] | 7 [5–9] | <0.001 | Cohen’s d = 0.65 (ENT vs. KM) | |
| Synkinesis | 12 (6.0) | 17 (8.5) | 23 (11.5) | 0.027 | Phi = 0.10 | |
| Patient Satisfaction | 165 (82.5) | 156 (78.0) | 169 (84.5) | 0.049 | Phi = 0.08 | |
| Recurrence | 7 (3.5) | 9 (4.5) | 8 (4.0) | 0.856 | Phi = 0.02 | |
| OR | p-Value | 95% CI | |
|---|---|---|---|
| Age < 40 years | 1.28 | 0.072 | 0.96–1.70 |
| Early Acupuncture | 0.76 | 0.076 | 0.57–1.02 |
| ENT Department | 1.75 | <0.001 | 1.29–2.37 |
| Pain Medicine Department | 1.10 | 0.550 | 0.81–1.49 |
| Early Corticosteroid Treatment | 1.95 | <0.001 | 1.42–2.68 |
| Lower Initial HB Grade (I–III vs. IV) | 1.58 | 0.002 | 1.18–2.11 |
| Absence of Comorbidities | 1.40 | 0.022 | 1.05–1.86 |
| Early Signs of Recovery | 1.70 | <0.001 | 1.26–2.29 |
| OR | p-Value | 95% CI | |
|---|---|---|---|
| Age ≥ 65 years | 1.12 | 0.992 | 0.87–1.45 |
| Female (vs. Male) | 1.75 | 0.345 | 0.83–3.70 |
| Hypertension | 4.40 | 0.006 | 1.62–11.95 |
| Diabetes | 1.40 | 0.021 | 1.05–1.87 |
| Hyperlipidemia | 8.20 | 0.025 | 1.52–44.80 |
| Obesity (BMI > 30) | 1.20 | 0.301 | 0.85–1.69 |
| Hypothyroidism | 1.37 | 0.043 | 1.00–1.88 |
| Autoimmune Disease | 1.47 | 0.022 | 1.05–2.05 |
| Initial HB Grade IV (vs. I–III) | 1.71 | <0.001 | 1.24–2.35 |
| History of Recurrence | 2.53 | <0.001 | 1.71–3.75 |
| Subgroup | Variable | ENT | Pain Medicine | KM | p-Value |
|---|---|---|---|---|---|
| Initial HB Grade IV (n = 178) | Complete Recovery, n (%) | 48/54 (90.0) | 49/66 (74.0) | 38/58 (65.0) | <0.01 |
| Age < 40 years (n = 204) | Time to Recovery, weeks (IQR) | 3 (2–5) | 5 (3–7) | 6 (4–8) | <0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, J.; Park, E.; Lee, J.; Lee, C. Recovery and Recurrence in Bell’s Palsy: A Propensity Score-Matched Comparative Study Across ENT, Pain Medicine, and Traditional Korean Medicine. Medicina 2025, 61, 1239. https://doi.org/10.3390/medicina61071239
Chung J, Park E, Lee J, Lee C. Recovery and Recurrence in Bell’s Palsy: A Propensity Score-Matched Comparative Study Across ENT, Pain Medicine, and Traditional Korean Medicine. Medicina. 2025; 61(7):1239. https://doi.org/10.3390/medicina61071239
Chicago/Turabian StyleChung, Jaeyoon, Eunsung Park, Jin Lee, and Cheol Lee. 2025. "Recovery and Recurrence in Bell’s Palsy: A Propensity Score-Matched Comparative Study Across ENT, Pain Medicine, and Traditional Korean Medicine" Medicina 61, no. 7: 1239. https://doi.org/10.3390/medicina61071239
APA StyleChung, J., Park, E., Lee, J., & Lee, C. (2025). Recovery and Recurrence in Bell’s Palsy: A Propensity Score-Matched Comparative Study Across ENT, Pain Medicine, and Traditional Korean Medicine. Medicina, 61(7), 1239. https://doi.org/10.3390/medicina61071239







