Challenges in the Diagnosis and Individualized Treatment of Cervical Cancer
Abstract
1. Introduction
2. Pathogenesis and Risk Factors
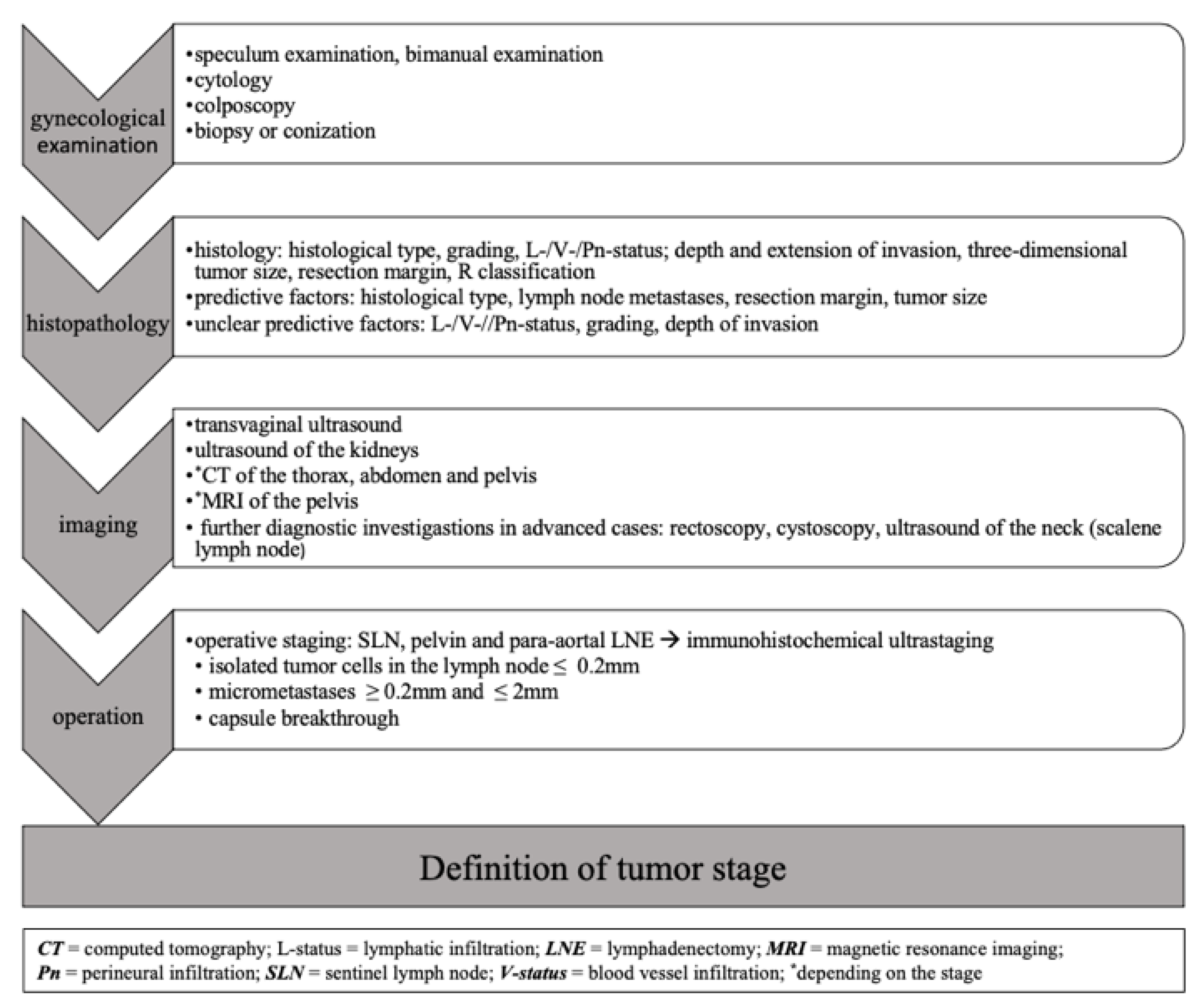
3. Diagnostic Investigation and Prevention
4. Surgical Treatment
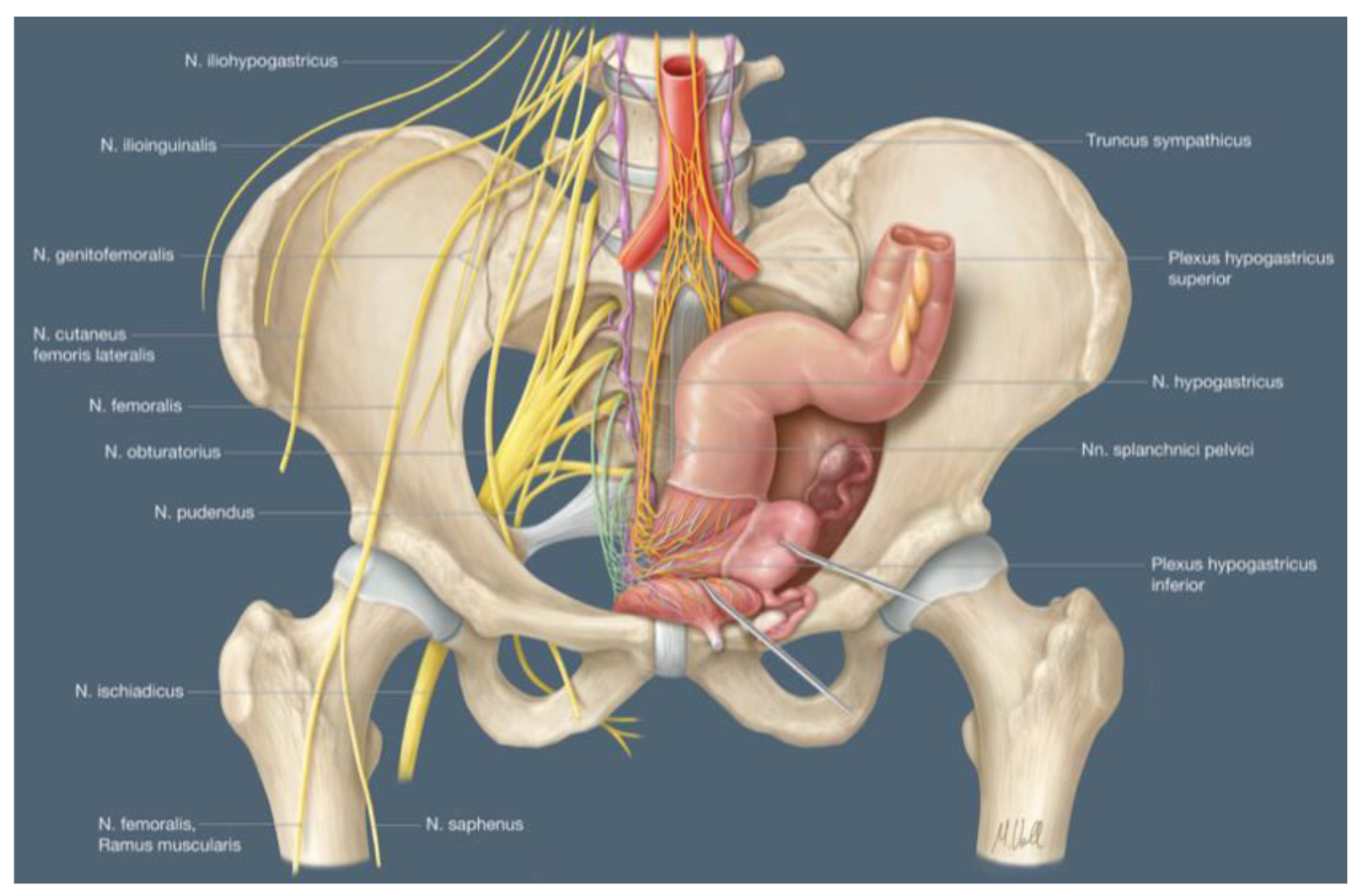
4.1. Nerve-Sparing Radical Hysterectomy
4.2. Fertility Preservation
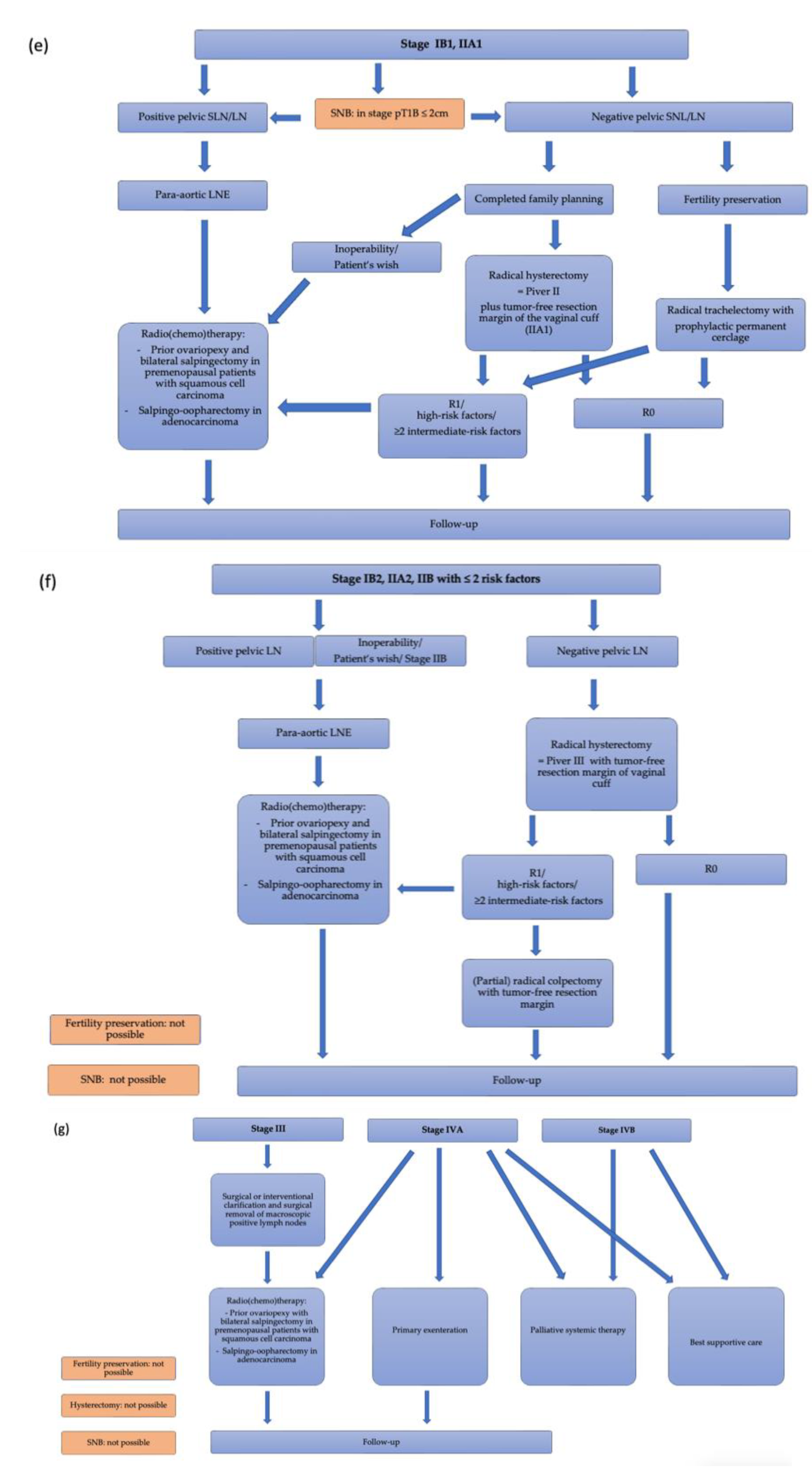
4.3. Stage-Specific Therapy Guides
5. Radio(Chemo)Therapy
6. Medical Therapy
Neoadjuvant Chemotherapy
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACIS | adenocarcinoma in situ |
| ADC | antibody–drug conjugate |
| Anti-CTLA4 | cytotoxic T-lymphocyte antigen 4 antibody |
| CIN | cervical intraepithelial neoplasm |
| CDC | Centers for Disease Control and Prevention |
| CPS | combined positive score |
| CT | computed tomography |
| DFS | disease-free survival |
| ERBT | pelvic external beam radiotherapy |
| ESGO | European Society of Gynaecological Oncology |
| ESRO | European Society for Radiotherapy and Oncology |
| ESP | European Society of Pathology |
| FDA | Food and Drug Administration |
| FIGO | Fédération Internationale de Gynécologie et d’Obstétrique |
| HIV | human immune deficiency virus |
| HPV | human papilloma virus |
| HN | hypogastric nerve |
| HSIL | high-grade squamous intraepithelial lesion |
| ICG | indocyanine green |
| IGABIT | image-guided adaptive brachytherapy |
| IHO | inferior hypogastric plexus |
| ITC | isolated tumor cells |
| LACC | Laparoscopic Approach to Cervical Cancer |
| LARVH | laparo-assisted vaginal hysterectomy |
| LMIC | low- and middle-income countries |
| LNE | lymphadenectomy |
| LSIL | low-grade squamous intraepithelial lesion |
| MRI | magnetic resonance imaging |
| NACT | neoadjuvant chemotherapy |
| NCCN | National Comprehensive Cancer Network |
| PAP | Papanicolaou |
| PD/PD-L1 | programmed cell death/programmed cell death ligand 1 |
| PSN | pelvic splanchnic nerve |
| RACC | Robot-assisted Approach to Cervical Cancer |
| RCT | radiochemotherapy |
| ROCC | Robotic versus Open Radical Hysterectomy for Cervical Cancer |
| SLN | sentinel lymph node |
| SNB | sentinel node biopsy |
| STIKO | Standing Commission on Vaccination |
| TCR | T cell receptor |
| VEGF | vascular endothelial-derived growth factor |
| WHO | World Health Organization |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF): S3-Leitlinie Diagnostik, Therapie und Nachsorge der Patientin mit Zervixkarzinom, Langversion, 2.2. 2022; AWMF-Registernummer: 032/033OL. Available online: https://www.leitlinienprogramm-onkologie.de/leitlinien/zervixkarzinom/ (accessed on 17 August 2022).
- Chrysostomou, A.C.; Stylianou, D.C.; Constantinidou, A.; Kostrikis, L.G. Cervical Cancer Screening Programs in Europe: The Transition Towards HPV Vaccination and Population-Based HPV Testing. Viruses 2018, 10, 729. [Google Scholar] [CrossRef] [PubMed]
- Raspollini, M.R.; Lax, S.F.; McCluggage, W.G. The central role of the pathologist in the management of patients with cervical cancer: ESGO/ESTRO/ESP guidelines. Virchows Arch. 2018, 473, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Schiffman, M.; Wentzensen, N.; Wacholder, S.; Kinney, W.; Gage, J.C.; Castle, P.E. Human papillomavirus testing in the prevention of cervical cancer. J. Natl. Cancer Inst. 2011, 103, 368–383. [Google Scholar] [CrossRef] [PubMed]
- Kopp, S.A.; Turk, D.E. Human Papillomavirus Vaccinations: Provider Education to Enhance Vaccine Uptake. Clin. Pediatr. 2023, onlinefirst. [Google Scholar] [CrossRef] [PubMed]
- Serrano, B.; Ibáñez, R.; Robles, C.; Peremiquel-Trillas, P.; de Sanjosé, S.; Bruni, L. Worldwide use of HPV self-sampling for cervical cancer screening. Prev. Med. 2022, 154, 106900. [Google Scholar] [CrossRef]
- Gizaw, M.; Teka, B.; Ruddies, F.; Abebe, T.; Kaufmann, A.M.; Worku, A.; Wienke, A.; Jemal, A.; Addissie, A.; Kantelhardt, E.J. Uptake of Cervical Cancer Screening in Ethiopia by Self-Sampling HPV DNA Compared to Visual Inspection with Acetic Acid: A Cluster Randomized Trial. Cancer Prev. Res. 2019, 12, 609–616. [Google Scholar] [CrossRef]
- Wang, R.; Pan, W.; Jin, L.; Huang, W.; Li, Y.; Wu, D.; Gao, C.; Ma, D.; Liao, S. Human papillomavirus vaccine against cervical cancer: Opportunity and challenge. Cancer Lett. 2020, 471, 88–102. [Google Scholar] [CrossRef]
- Karimi-Zarchi, M.; Allahqoli, L.; Nehmati, A.; Kashi, A.M.; Taghipour-Zahir, S.; Alkatout, I. Can the prophylactic quadrivalent HPV vaccine be used as a therapeutic agent in women with CIN? A randomized trial. BMC Public Health 2020, 20, 274. [Google Scholar] [CrossRef]
- Brisson, M.; Kim, J.J.; Canfell, K.; Drolet, M.; Gingras, G.; Burger, E.A.; Martin, D.; Simms, K.T.; Bénard, É.; Boily, M.C.; et al. Impact of HPV vaccination and cervical screening on cervical cancer elimination: A comparative modelling analysis in 78 low-income and lower-middle-income countries. Lancet 2020, 395, 575–590. [Google Scholar] [CrossRef]
- Hillemanns, P.; Kampers, J.; Hachenberg, J.; Jentschke, M. Vaccination against human papillomavirus. Internist 2021, 62, 816–826. [Google Scholar] [CrossRef]
- Thomeer, M.G.; Gerestein, C.; Spronk, S.; van Doorn, H.C.; van der Ham, E.; Hunink, M.G. Clinical examination versus magnetic resonance imaging in the pretreatment staging of cervical carcinoma: Systematic review and meta-analysis. Eur. Radiol. 2013, 23, 2005–2018. [Google Scholar] [CrossRef]
- Cibula, D.; Pötter, R.; Planchamp, F.; Avall-Lundqvist, E.; Fischerova, D.; Haie Meder, C.; Köhler, C.; Landoni, F.; Lax, S.; Lindegaard, J.C.; et al. The European Society of Gynaecological Oncology/European Society for Radiotherapy and Oncology/European Society of Pathology Guidelines for the Management of Patients with Cervical Cancer. Int. J. Gynecol. Cancer 2018, 28, 641–655. [Google Scholar] [CrossRef] [PubMed]
- Selman, T.J.; Mann, C.; Zamora, J.; Appleyard, T.L.; Khan, K. Diagnostic accuracy of tests for lymph node status in primary cervical cancer: A systematic review and meta-analysis. CMAJ 2008, 178, 855–862. [Google Scholar] [CrossRef] [PubMed]
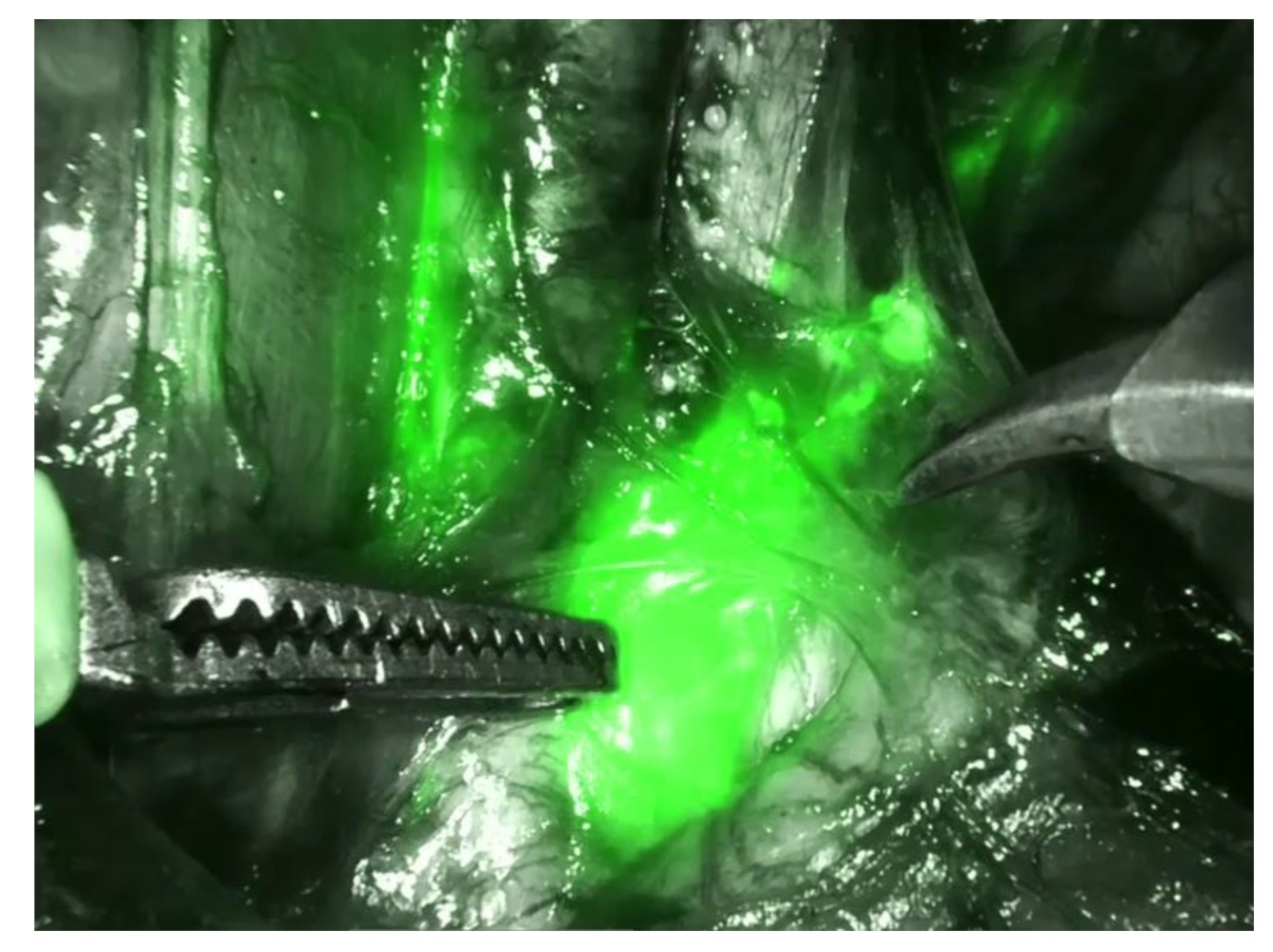
- Balaya, V.; Guani, B.; Morice, P.; Querleu, D.; Fourchotte, V.; Leblanc, E.; Daraï, E.; Baron, M.; Marret, H.; Levêque, J.; et al. Long-term oncological safety of sentinel lymph node biopsy in early-stage cervical cancer: A post-hoc analysis of SENTICOL I and SENTICOL II cohorts. Gynecol. Oncol. 2022, 164, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Lecuru, F.R.; McCormack, M.; Hillemanns, P.; Anota, A.; Leitao, M.; Mathevet, P.; Zweemer, R.; Fujiwara, K.; Zanagnolo, V.; Zahl Eriksson, A.G.; et al. SENTICOL III: An international validation study of sentinel node biopsy in early cervical cancer. A GINECO, ENGOT, GCIG and multicenter study. Int. J. Gynecol. Cancer 2019, 29, 829–834. [Google Scholar] [CrossRef]
- Cibula, D.; Dusek, J.; Jarkovsky, J.; Dundr, P.; Querleu, D.; van der Zee, A.; Kucukmetin, A.; Kocian, R. A prospective multicenter trial on sentinel lymph node biopsy in patients with early-stage cervical cancer (SENTIX). Int. J. Gynecol. Cancer 2019, 29, 212–215. [Google Scholar] [CrossRef]
- Cibula, D.; Kocian, R.; Plaikner, A.; Jarkovsky, J.; Klat, J.; Zapardiel, I.; Pilka, R.; Torne, A.; Sehnal, B.; Ostojich, M.; et al. Sentinel lymph node mapping and intraoperative assessment in a prospective, international, multicentre, observational trial of patients with cervical cancer: The SENTIX trial. Eur. J. Cancer 2020, 137, 69–80. [Google Scholar] [CrossRef]
- Tu, H.; Huang, H.; Xian, B.; Li, J.; Wang, P.; Zhao, W.; Chen, X.; Xie, X.; Wang, C.; Kong, B.; et al. Sentinel lymph node biopsy versus pelvic lymphadenectomy in early-stage cervical cancer: A multi-center randomized trial (PHENIX/CSEM 010). Int. J. Gynecol. Cancer 2020, 30, 1829–1833. [Google Scholar] [CrossRef]
- Ramirez, P.T.; Frumovitz, M.; Pareja, R.; López, A.; Vieira, M.d.A.; Ribeiro, R. Phase III randomized trial of laparoscopic or robotic versus abdominal radical hysterectomy in patients with early-stage cervical cancer: LACC Trial. Gynecol. Oncol. 2018, 149, 245. [Google Scholar] [CrossRef]
- Melamed, A.; Ramirez, P.T. Changing treatment landscape for early cervical cancer: Outcomes reported with minimally invasive surgery compared with an open approach. Curr. Opin. Obstet. Gynecol 2020, 32, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Bixel, K.L.; Leitao, M.M.; Chase, D.M.; Quick, A.; Lim, P.C.; Eskander, R.N.; Gotlieb, W.H.; LoCoco, S.; Martino, M.A.; McCormick, C.; et al. ROCC/GOG-3043: A randomized non-inferiority trial of robotic versus open radical hysterectomy for early-stage cervical cancer. J. Clin. Oncol. 2022, 40, TPS5605. [Google Scholar] [CrossRef]
- Ronsini, C.; Köhler, C.; de Franciscis, P.; La Verde, M.; Mosca, L.; Solazzo, M.C.; Colacurci, N. Laparo-assisted vaginal radical hysterectomy as a safe option for minimal invasive surgery in early stage cervical cancer: A systematic review and meta-analysis. Gynecol. Oncol. 2022, 166, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Alfonzo, E.; Wallin, E.; Ekdahl, L.; Staf, C.; Rådestad, A.F.; Reynisson, P.; Stålberg, K.; Falconer, H.; Persson, J.; Dahm-Kähler, P. No survival difference between robotic and open radical hysterectomy for women with early-stage cervical cancer: Results from a nationwide population-based cohort study. Eur. J. Cancer 2019, 116, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Fusegi, A.; Kanao, H.; Ishizuka, N.; Nomura, H.; Tanaka, Y.; Omi, M.; Aoki, Y.; Kurita, T.; Yunokawa, M.; Omatsu, K.; et al. Oncologic Outcomes of Laparoscopic Radical Hysterectomy Using the No-Look No-Touch Technique for Early Stage Cervical Cancer: A Propensity Score-Adjusted Analysis. Cancers 2021, 13, 6097. [Google Scholar] [CrossRef]
- Casarin, J.; Bogani, G.; Papadia, A.; Ditto, A.; Pinelli, C.; Garzon, S.; Donadello, N.; Laganà, A.S.; Cromi, A.; Mueller, M.; et al. Preoperative Conization and Risk of Recurrence in Patients Undergoing Laparoscopic Radical Hysterectomy for Early Stage Cervical Cancer: A Multicenter Study. J. Minim. Invasive Gynecol. 2021, 28, 117–123. [Google Scholar] [CrossRef]
- Chacon, E.; Manzour, N.; Zanagnolo, V.; Querleu, D.; Núñez-Córdoba, J.M.; Martin-Calvo, N.; Căpîlna, M.E.; Fagotti, A.; Kucukmetin, A.; Mom, C.; et al. SUCCOR cone study: Conization before radical hysterectomy. Int. J. Gynecol. Cancer 2022, 32, 117–124. [Google Scholar] [CrossRef]
- Li, J.; Gong, X.; Li, P.; Xiao, L.; Chang, X.; Ouyang, X.; Tang, J. Application of Da Vinci robotic surgery system in cervical cancer: A single institution experience of 557 cases. Asian J. Surg. 2022, 45, 707–711. [Google Scholar] [CrossRef]
- Sekhon, R.; Naithani, A.; Makkar, P.; Pratima, R.; Sharma, P.; Rawal, S.; Goyal, Y.; Mitra, S.; Sharma, A.; Mehta, A. Robotic radical hysterectomy versus open radical hysterectomy for cervical cancer: A single-centre experience from India. J. Robot. Surg. 2022, 16, 935–941. [Google Scholar] [CrossRef]
- Uwins, C.; Patel, H.; Prakash Bhandoria, G.; Butler-Manuel, S.; Tailor, A.; Ellis, P.; Chatterjee, J. Laparoscopic and Robotic Surgery for Endometrial and Cervical Cancer. Clin. Oncol. 2021, 33, e372–e382. [Google Scholar] [CrossRef]
- Trifanescu, O.G.; Gales, L.N.; Serbanescu, G.L.; Zgura, A.F.; Iliescu, L.; Mehedintu, C.; Anghel, R.M. Long-term oncological outcome in patients with cervical cancer after 3 trimodality treatment (radiotherapy, platinum-based chemotherapy, and robotic surgery). Medicine 2021, 100, e25271. [Google Scholar] [CrossRef]
- Leitao, M.M.J.; Kreaden, U.S.; Laudone, V.; Park, B.J.; Pappou, E.P.; Davis, J.W.; Rice, D.C.; Chang, G.J.; Rossi, E.C.; Hebert, A.E.; et al. The RECOURSE Study: Long-term Oncologic Outcomes Associated With Robotically Assisted Minimally Invasive Procedures for Endometrial, Cervical, Colorectal, Lung, or Prostate Cancer: A Systematic Review and Meta-analysis. Ann. Surg. 2023, 277, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Falconer, H.; Palsdottir, K.; Stalberg, K.; Dahm-Kähler, P.; Ottander, U.; Lundin, E.S.; Wijk, L.; Kimmig, R.; Jensen, P.T.; Zahl Eriksson, A.G.; et al. Robot-assisted approach to cervical cancer (RACC): An international multi-center, open-label randomized controlled trial. Int. J. Gynecol. Cancer 2019, 29, 1072–1076. [Google Scholar] [CrossRef] [PubMed]
- Piver, M.S.; Rutledge, F.; Smith, J.P. Five classes of extended hysterectomy for women with cervical cancer. Obstet. Gynecol. 1974, 44, 265–272. [Google Scholar] [CrossRef]
- Sakuragi, N.; Murakami, G.; Konno, Y.; Kaneuchi, M.; Watari, H. Nerve-sparing radical hysterectomy in the precision surgery for cervical cancer. J. Gynecol. Oncol. 2020, 31, e49. [Google Scholar] [CrossRef]
- Kietpeerakool, C.; Aue-Aungkul, A.; Galaal, K.; Ngamjarus, C.; Lumbiganon, P. Nerve-sparing radical hysterectomy compared to standard radical hysterectomy for women with early stage cervical cancer (stage Ia2 to IIa). Cochrane Database Syst. Rev. 2019, 2, Cd012828. [Google Scholar] [CrossRef]
- Muallem, M.Z.; Armbrust, R.; Neymeyer, J.; Miranda, A.; Muallem, J. Nerve Sparing Radical Hysterectomy: Short-Term Oncologic, Surgical, and Functional Outcomes. Cancers 2020, 12, 483. [Google Scholar] [CrossRef]
- Muallem, M.Z.; Jöns, T.; Seidel, N.; Sehouli, J.; Diab, Y.; Querleu, D. A Concise Paradigm on Radical Hysterectomy: The Comprehensive Anatomy of Parametrium, Paracolpium and the Pelvic Autonomic Nerve System and Its Surgical Implication. Cancers 2020, 12, 1839. [Google Scholar] [CrossRef]
- Muallem, M.Z. A New Anatomic and Staging-Oriented Classification of Radical Hysterectomy. Cancers 2021, 13, 3326. [Google Scholar] [CrossRef]
- Cibula, D.; Velechovska, P.; Sláma, J.; Fischerova, D.; Pinkavova, I.; Pavlista, D.; Dundr, P.; Hill, M.; Freitag, P.; Zikan, M. Late morbidity following nerve-sparing radical hysterectomy. Gynecol. Oncol. 2010, 116, 506–511. [Google Scholar] [CrossRef]
- Plotti, F.; Ficarola, F.; Messina, G.; Terranova, C.; Montera, R.; Guzzo, F.; de Cicco Nardone, C.; Rossini, G.; Schirò, T.; Gatti, A.; et al. Tailoring parametrectomy for early cervical cancer (Stage IA-IIA FIGO): A review on surgical, oncologic outcome and sexual function. Minerva Ginecol. 2021, 73, 149–159. [Google Scholar] [CrossRef]
- Yamamoto, A.; Kamoi, S.; Ikeda, M.; Yamada, T.; Yoneyama, K.; Takeshita, T. Effectiveness and Long-term Outcomes of Nerve-Sparing Radical Hysterectomy for Cervical Cancer. J. Nippon. Med. Sch. 2021, 88, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Alkatout, I.; Wedel, T.; Pape, J.; Possover, M.; Dhanawat, J. Review: Pelvic nerves - from anatomy and physiology to clinical applications. Transl. Neurosci. 2021, 12, 362–378. [Google Scholar] [CrossRef] [PubMed]
- Abu-Rustum, N.R.; Yashar, C.M.; Bean, S.; Bradley, K.; Campos, S.M.; Chon, H.S.; Chu, C.; Cohn, D.; Crispens, M.A.; Damast, S.; et al. NCCN Guidelines Insights: Cervical Cancer, Version 1.2020: Featured Updates to the NCCN Guidelines. J. Natl. Compr. Cancer Netw. 2020, 18, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.J.; Abu-Rustum, N.R.; Bean, S.; Bradley, K.; Campos, S.M.; Cho, K.R.; Chon, H.S.; Chu, C.; Clark, R.; Cohn, D.; et al. Cervical Cancer, Version 3.2019, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2019, 17, 64–84. [Google Scholar] [CrossRef] [PubMed]
- Nezhat, F.; Erfani, H.; Nezhat, C. A systematic review of the reproductive and oncologic outcomes of fertility-sparing surgery for early-stage cervical cancer. J. Turk.-Ger. Gynecol. Assoc. 2022, 23, 287–313. [Google Scholar] [CrossRef]
- Chu, R.; Zhang, Y.; Qiao, X.; Xie, L.; Chen, W.; Zhao, Y.; Xu, Y.; Yuan, Z.; Liu, X.; Yin, A.; et al. Risk Stratification of Early-Stage Cervical Cancer with Intermediate-Risk Factors: Model Development and Validation Based on Machine Learning Algorithm. Oncologist 2021, 26, e2217–e2226. [Google Scholar] [CrossRef]
- Shapira-Frommer, R.; Alexandre, J.; Monk, B.; Fehm, T.N.; Colombo, N.; Caceres, M.V.; Hasegawa, K.; Dubot, C.; Li, J.J.; Stein, K.; et al. KEYNOTE-826: A phase 3, randomized, double-blind, placebo-controlled study of pembrolizumab plus chemotherapy for first-line treatment of persistent, recurrent, or metastatic cervical cancer. J. Clin. Oncol. 2019, 37, TPS5595. [Google Scholar] [CrossRef]
- Tewari, K.S.; Sill, M.W.; Penson, R.T.; Huang, H.; Ramondetta, L.M.; Landrum, L.M.; Oaknin, A.; Reid, T.J.; Leitao, M.M.; Michael, H.E.; et al. Bevacizumab for advanced cervical cancer: Final overall survival and adverse event analysis of a randomised, controlled, open-label, phase 3 trial (Gynecologic Oncology Group 240). Lancet 2017, 390, 1654–1663. [Google Scholar] [CrossRef]
- Sedlis, A.; Bundy, B.N.; Rotman, M.Z.; Lentz, S.S.; Muderspach, L.I.; Zaino, R.J. A randomized trial of pelvic radiation therapy versus no further therapy in selected patients with stage IB carcinoma of the cervix after radical hysterectomy and pelvic lymphadenectomy: A Gynecologic Oncology Group Study. Gynecol. Oncol. 1999, 73, 177–183. [Google Scholar] [CrossRef]
- Kitagawa, R.; Katsumata, N.; Shibata, T.; Kamura, T.; Kasamatsu, T.; Nakanishi, T.; Nishimura, S.; Ushijima, K.; Takano, M.; Satoh, T.; et al. Paclitaxel Plus Carboplatin Versus Paclitaxel Plus Cisplatin in Metastatic or Recurrent Cervical Cancer: The Open-Label Randomized Phase III Trial JCOG0505. J. Clin. Oncol. 2015, 33, 2129–2135. [Google Scholar] [CrossRef] [PubMed]
- Redondo, A.; Colombo, N.; McCormack, M.; Dreosti, L.; Nogueira-Rodrigues, A.; Scambia, G.; Lorusso, D.; Joly, F.; Schenker, M.; Ruff, P.; et al. Primary results from CECILIA, a global single-arm phase II study evaluating bevacizumab, carboplatin and paclitaxel for advanced cervical cancer. Gynecol. Oncol. 2020, 159, 142–149. [Google Scholar] [CrossRef]
- Chung, H.C.; Ros, W.; Delord, J.P.; Perets, R.; Italiano, A.; Shapira-Frommer, R.; Manzuk, L.; Piha-Paul, S.A.; Xu, L.; Zeigenfuss, S.; et al. Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Cervical Cancer: Results From the Phase II KEYNOTE-158 Study. J. Clin. Oncol. 2019, 37, 1470–1478. [Google Scholar] [CrossRef]
- Colombo, N.; Dubot, C.; Lorusso, D.; Caceres, M.V.; Hasegawa, K.; Shapira-Frommer, R.; Tewari, K.S.; Salman, P.; Hoyos Usta, E.; Yañez, E.; et al. Pembrolizumab for Persistent, Recurrent, or Metastatic Cervical Cancer. N. Engl. J. Med. 2021, 385, 1856–1867. [Google Scholar] [CrossRef] [PubMed]
- Lorusso, D.; Colombo, N.; Coleman, R.L.; Randall, L.M.; Duska, L.R.; Xiang, Y.; Hasegawa, K.; Rodrigues, A.N.; Cibula, D.; Mirza, M.R.; et al. ENGOT-cx11/KEYNOTE-A18: A phase III, randomized, double-blind study of pembrolizumab with chemoradiotherapy in patients with high-risk locally advanced cervical cancer. J. Clin. Oncol. 2020, 38, TPS6096. [Google Scholar] [CrossRef]
- Naumann, R.W.; Hollebecque, A.; Meyer, T.; Devlin, M.-J.; Oaknin, A.; Kerger, J.; López-Picazo, J.M.; Machiels, J.-P.; Delord, J.-P.; Evans, T.R.J.; et al. Safety and Efficacy of Nivolumab Monotherapy in Recurrent or Metastatic Cervical, Vaginal, or Vulvar Carcinoma: Results From the Phase I/II CheckMate 358 Trial. J. Clin. Oncol. 2019, 37, 2825–2834. [Google Scholar] [CrossRef]
- Tewari, K.S.; Monk, B.J.; Vergote, I.; Miller, A.; de Melo, A.C.; Kim, H.-S.; Kim, Y.M.; Lisyanskaya, A.; Samouëlian, V.; Lorusso, D.; et al. Survival with Cemiplimab in Recurrent Cervical Cancer. N. Engl. J. Med. 2022, 386, 544–555. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.L.; Lorusso, D.; Gennigens, C.; González-Martín, A.; Randall, L.; Cibula, D.; Lund, B.; Woelber, L.; Pignata, S.; Forget, F.; et al. Efficacy and safety of tisotumab vedotin in previously treated recurrent or metastatic cervical cancer (innovaTV 204/GOG-3023/ENGOT-cx6): A multicentre, open-label, single-arm, phase 2 study. Lancet Oncol. 2021, 22, 609–619. [Google Scholar] [CrossRef]
- Dass, S.A.; Selva Rajan, R.; Tye, G.J.; Balakrishnan, V. The potential applications of T cell receptor (TCR)-like antibody in cervical cancer immunotherapy. Hum. Vaccines Immunother. 2021, 17, 2981–2994. [Google Scholar] [CrossRef]
- Doran, S.L.; Stevanović, S.; Adhikary, S.; Gartner, J.J.; Jia, L.; Kwong, M.L.M.; Faquin, W.C.; Hewitt, S.M.; Sherry, R.M.; Yang, J.C.; et al. T-Cell Receptor Gene Therapy for Human Papillomavirus-Associated Epithelial Cancers: A First-in-Human, Phase I/II Study. J. Clin. Oncol. 2019, 37, 2759–2768. [Google Scholar] [CrossRef]
- Rydzewska, L.; Tierney, J.; Vale, C.L.; Symonds, P.R. Neoadjuvant chemotherapy plus surgery versus surgery for cervical cancer. Cochrane Database Syst. Rev. 2012, 12, Cd007406. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.H.; Wang, X.X.; Zhu, J.S.; Gao, L. Neo-adjuvant chemotherapy plus surgery versus surgery alone for cervical cancer: Meta-analysis of randomized controlled trials. J. Obstet. Gynaecol. Res. 2016, 42, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Sardi, J.E.; Katsumata, N.; Ryu, H.S.; Nam, J.H.; Chung, H.H.; Park, N.H.; Song, Y.S.; Behtash, N.; Kamura, T.; et al. Efficacy of neoadjuvant chemotherapy in patients with FIGO stage IB1 to IIA cervical cancer: An international collaborative meta-analysis. Eur. J. Surg. Oncol. 2013, 39, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Cosio, A.G.S. Neoadjuvant Chemotherapy in Locally Advanced Cervical Cancer: Review of the Literature and Perspectives of Clinical Research. Anticancer. Res. 2020, 40, 4819–4828. [Google Scholar] [CrossRef]
- Gupta, S.; Maheshwari, A.; Parab, P.; Mahantshetty, U.; Hawaldar, R.; Sastri Chopra, S.; Kerkar, R.; Engineer, R.; Tongaonkar, H.; Ghosh, J.; et al. Neoadjuvant Chemotherapy Followed by Radical Surgery Versus Concomitant Chemotherapy and Radiotherapy in Patients With Stage IB2, IIA, or IIB Squamous Cervical Cancer: A Randomized Controlled Trial. J. Clin. Oncol. 2018, 36, 1548–1555. [Google Scholar] [CrossRef]
- Miriyala, R.; Mahantshetty, U.; Maheshwari, A.; Gupta, S. Neoadjuvant chemotherapy followed by surgery in cervical cancer: Past, present and future. Int. J. Gynecol. Cancer 2022, 32, 260–265. [Google Scholar] [CrossRef]
- Kenter, G.; Greggi, S.; Vergote, I.; Katsaros, D.; Kobierski, J.; Massuger, L.; van Doorn, H.C.; Landoni, F.; Van Der Velden, J.; Reed, N.S.; et al. Results from neoadjuvant chemotherapy followed by surgery compared to chemoradiation for stage Ib2-IIb cervical cancer, EORTC 55994. J. Clin. Oncol. 2019, 37, 5503. [Google Scholar] [CrossRef]
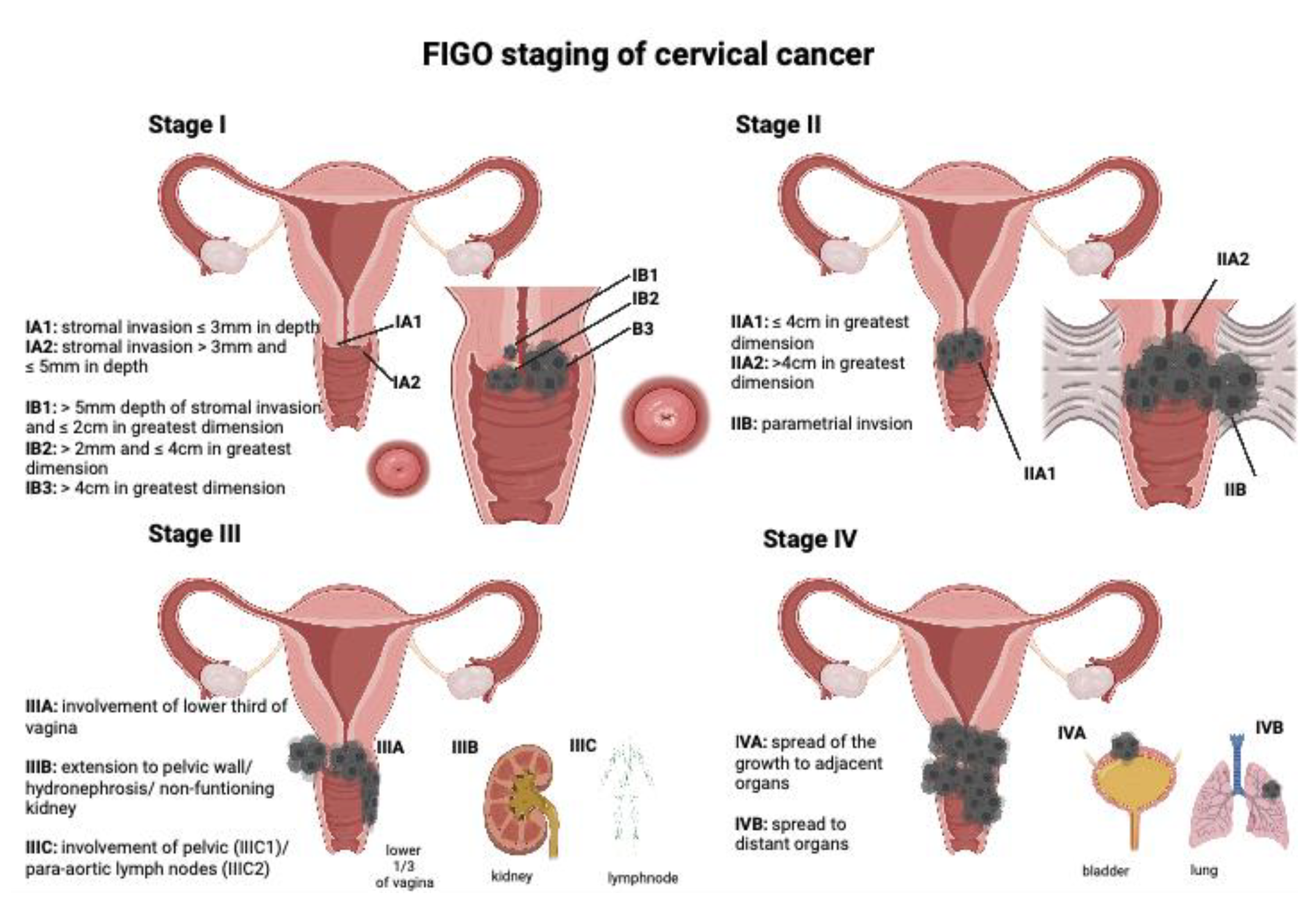



| Stage | |||
|---|---|---|---|
| I | Carcinoma strictly confined to the cervix | ||
| IA | Invasive carcinoma with maximum depth of invasion ≤ 5 mm | ||
| IA1 | Stromal invasion ≤ 3 mm in depth | ||
| IA2 | Stromal invasion > 3 mm and ≤5 mm in depth | ||
| IB | Deepest invasion > 5 mm; lesion limited to cervix uteri with size measured according to maximum tumor diameter | ||
| IB1 | >5 mm depth of stromal invasion and ≤2 cm in greatest dimension | ||
| IB2 | >2 cm and ≤4 cm in greatest dimension | ||
| IB3 | >4 cm in greatest dimension | ||
| II | Invasion beyond the uterus, but no extension into the lower third of the vagina or to the pelvic wall | ||
| IIA | Involvement limited to the upper two thirds of the vagina without parametrial invasion | ||
| IIA1 | ≤4 cm in greatest dimension | ||
| IIA2 | >4 cm in greatest dimension | ||
| IIB | Parametrial invasion but not to the pelvic wall | ||
| III | Involvement of the lower third of the vagina and/or extension to the pelvic wall and/or causes hydronephrosis or non-functioning kidney and/or involvement of pelvic and/or para-aortic lymph nodes | ||
| IIIA | Involvement of the lower third of the vagina, with no extension to the pelvic wall | ||
| IIIB | Extension to the pelvic wall and/or hydronephrosis or non-functioning kidney (unless known to be due to another cause) | ||
| IIIC | Involvement of pelvic and/or para-aortic lymph nodes (including micrometastases), irrespective of tumor size and extent | ||
| IIIC1 | Pelvic lymph node metastases only | ||
| IIIC2 | Para-aortic lymph node metastases | ||
| IV | Extension beyond the true pelvis or involvement (biopsy proven) of the mucosa of the bladder or rectum | ||
| IVA | Spread of the growth to adjacent organs | ||
| IVB | Spread to distant organs |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schubert, M.; Bauerschlag, D.O.; Muallem, M.Z.; Maass, N.; Alkatout, I. Challenges in the Diagnosis and Individualized Treatment of Cervical Cancer. Medicina 2023, 59, 925. https://doi.org/10.3390/medicina59050925
Schubert M, Bauerschlag DO, Muallem MZ, Maass N, Alkatout I. Challenges in the Diagnosis and Individualized Treatment of Cervical Cancer. Medicina. 2023; 59(5):925. https://doi.org/10.3390/medicina59050925
Chicago/Turabian StyleSchubert, Melanie, Dirk Olaf Bauerschlag, Mustafa Zelal Muallem, Nicolai Maass, and Ibrahim Alkatout. 2023. "Challenges in the Diagnosis and Individualized Treatment of Cervical Cancer" Medicina 59, no. 5: 925. https://doi.org/10.3390/medicina59050925
APA StyleSchubert, M., Bauerschlag, D. O., Muallem, M. Z., Maass, N., & Alkatout, I. (2023). Challenges in the Diagnosis and Individualized Treatment of Cervical Cancer. Medicina, 59(5), 925. https://doi.org/10.3390/medicina59050925





