Towards Regenerative Audiology: Immune Modulation of Adipose-Derived Mesenchymal Cells Preconditioned with Citric Acid-Coated Antioxidant-Functionalized Magnetic Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. MNP Characterization
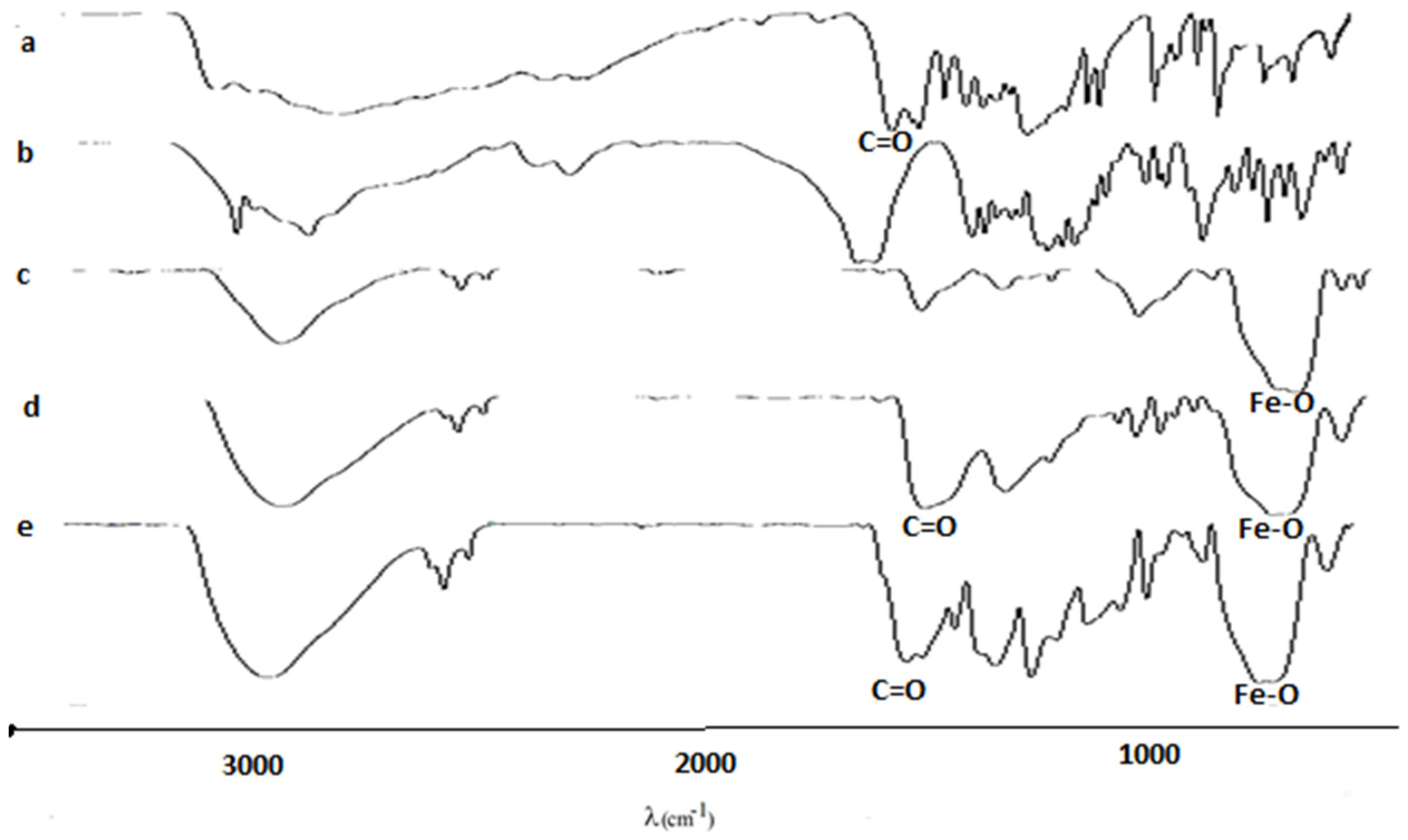
3.1.1. FTIR
3.1.2. DLS for the Uncoated MNPs
3.1.3. STEM
3.1.4. EDX
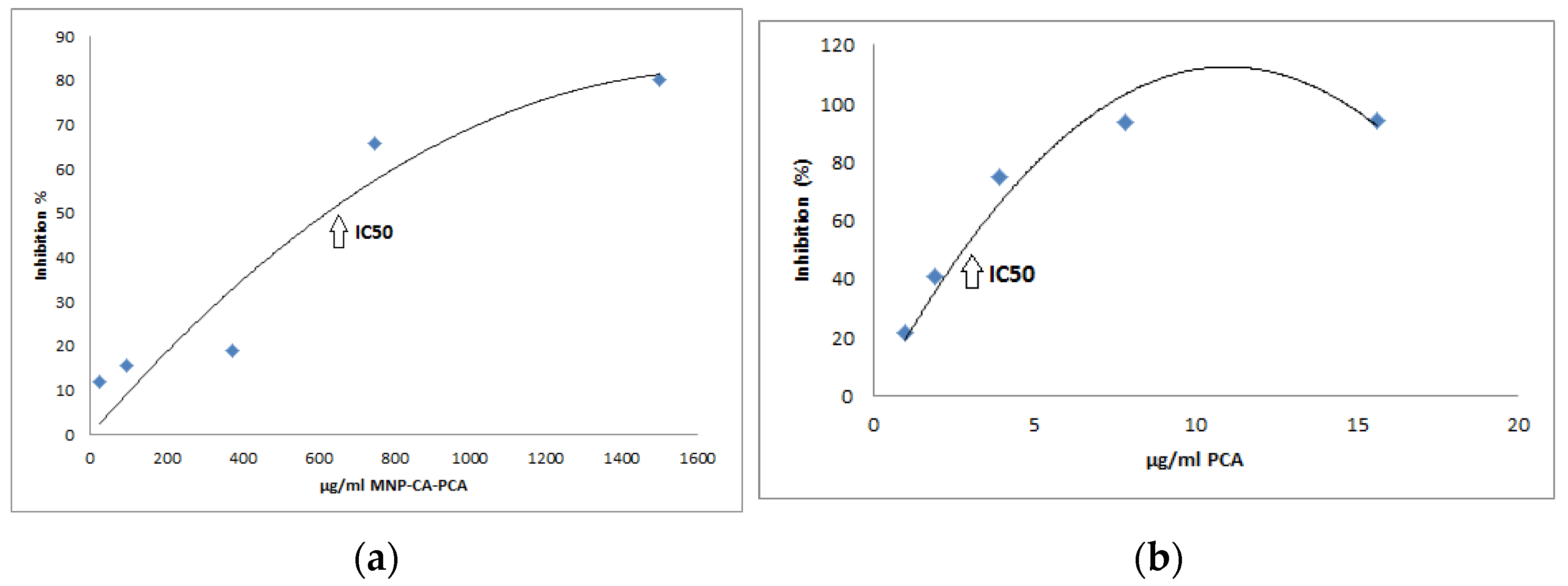
3.1.5. DPPH
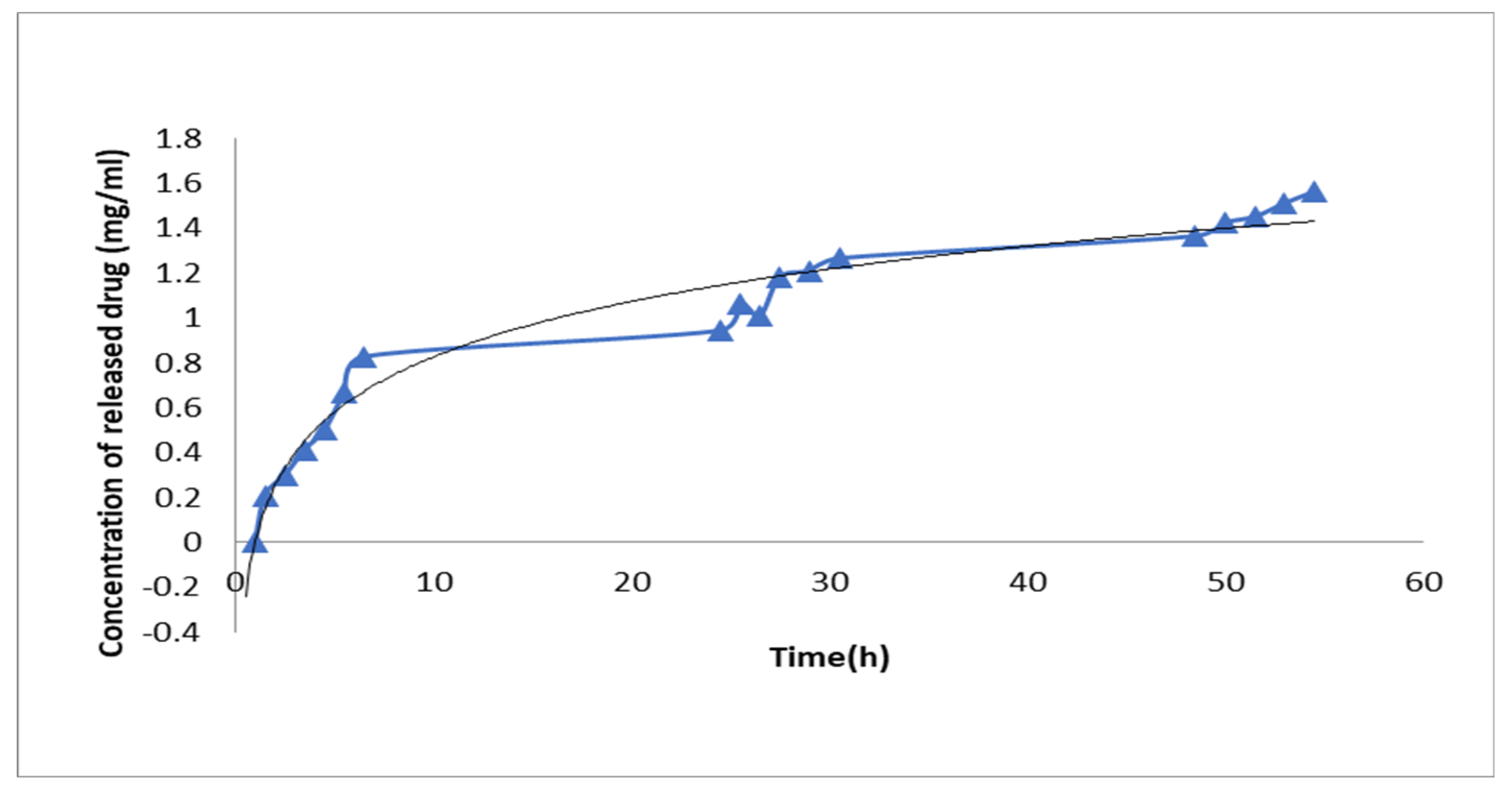
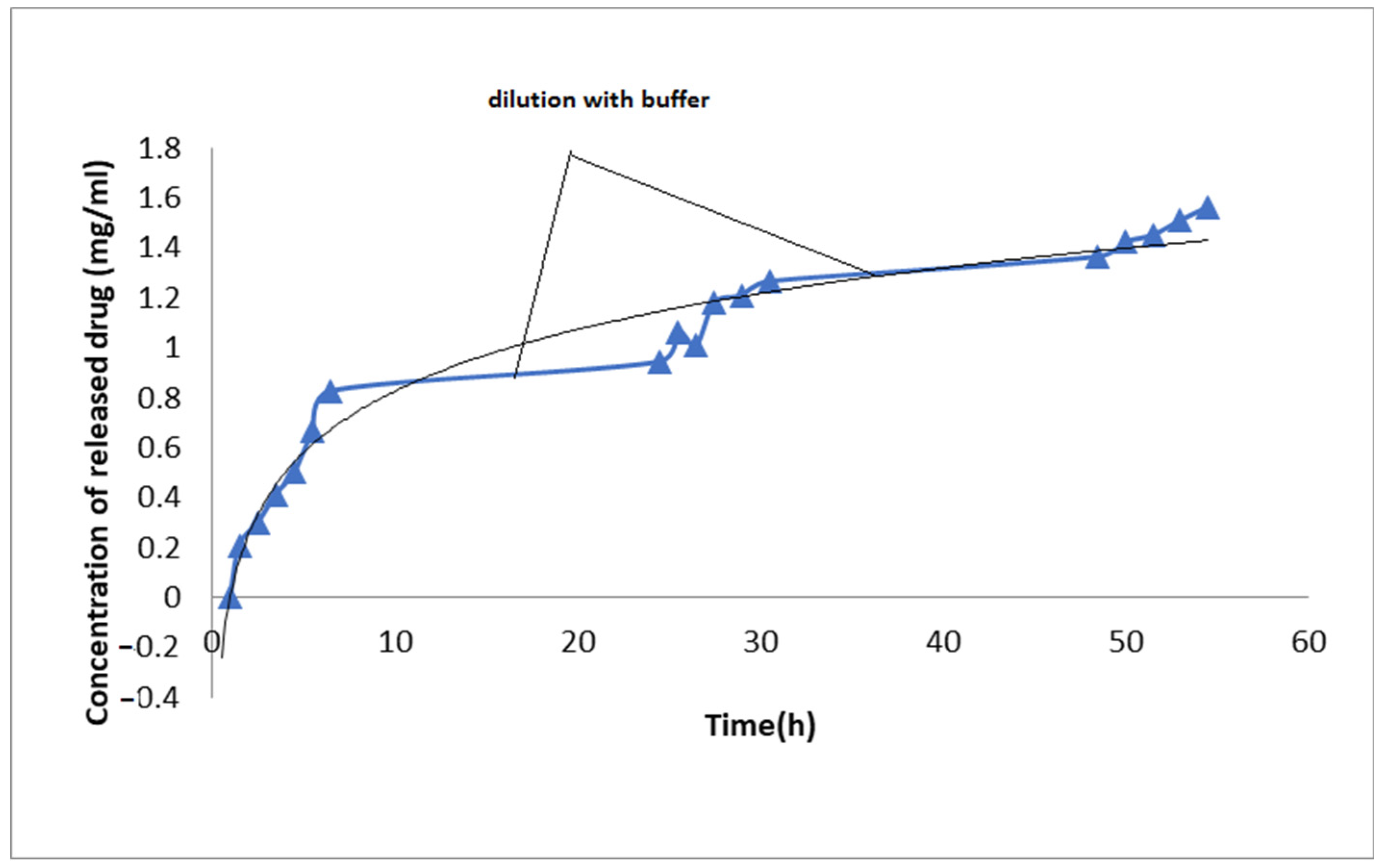
3.2. Drug Release
3.2.1. In Vitro MNP Cytotoxicity/ADSC Viability
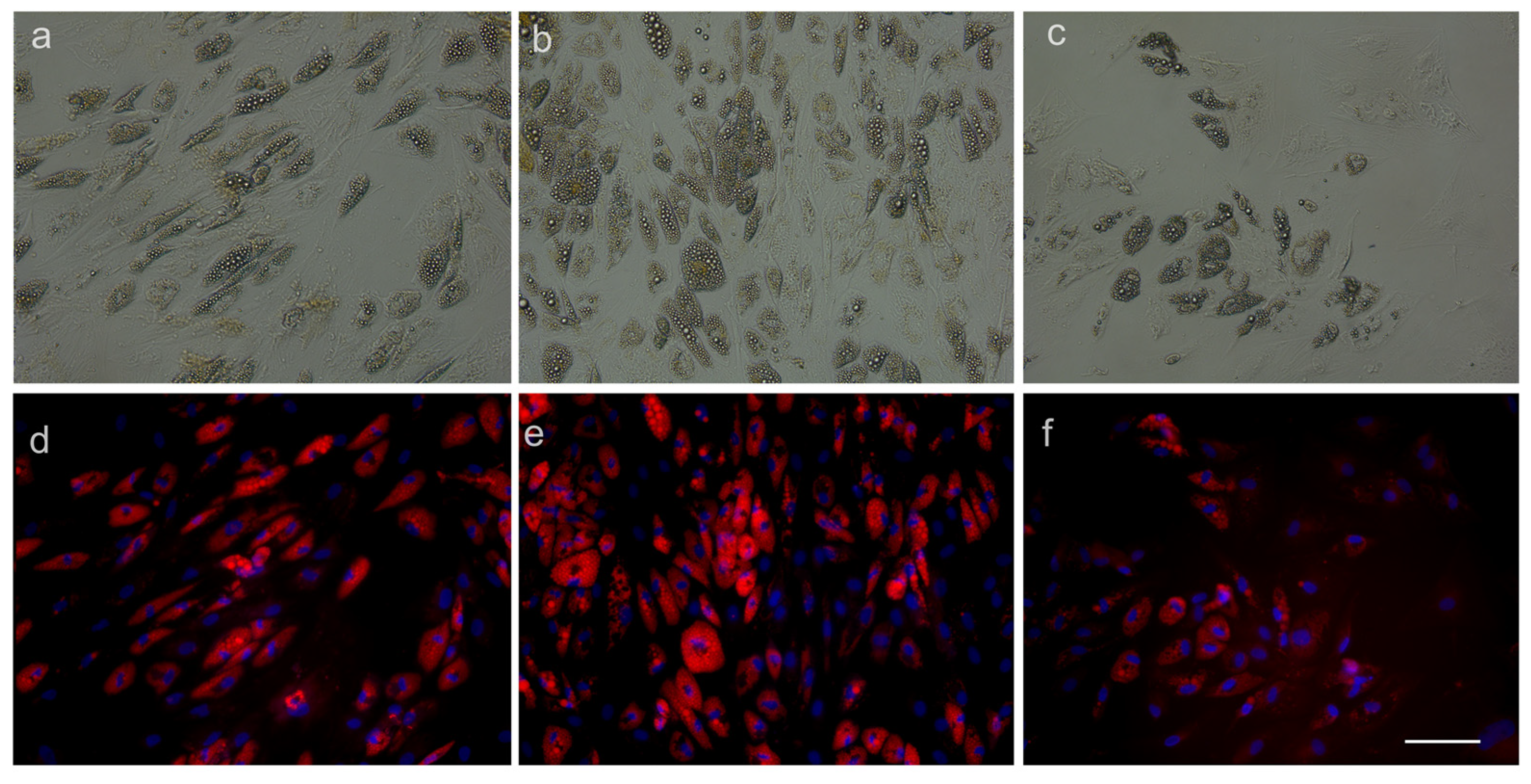
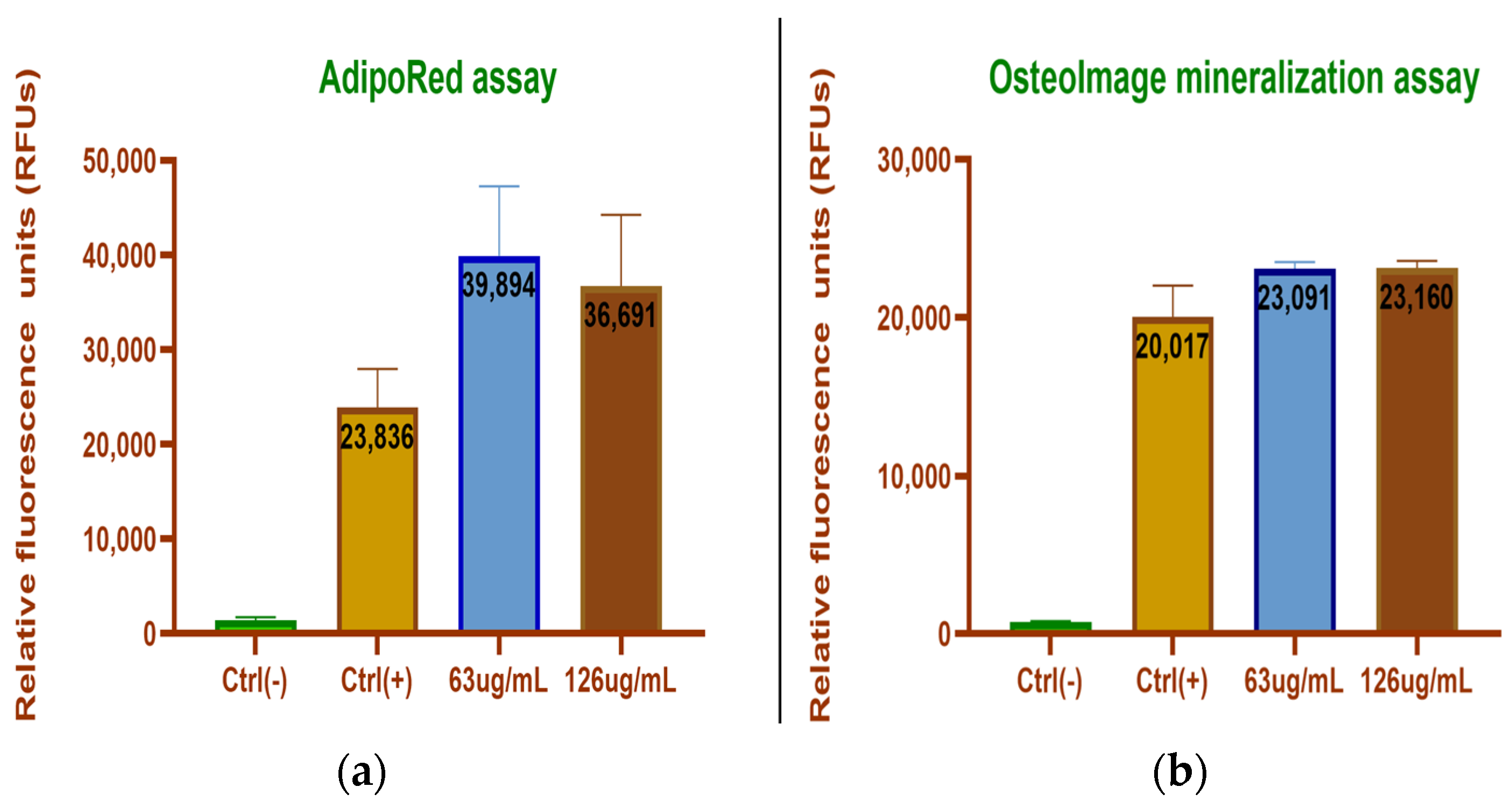
3.2.2. ADSC Differentiation
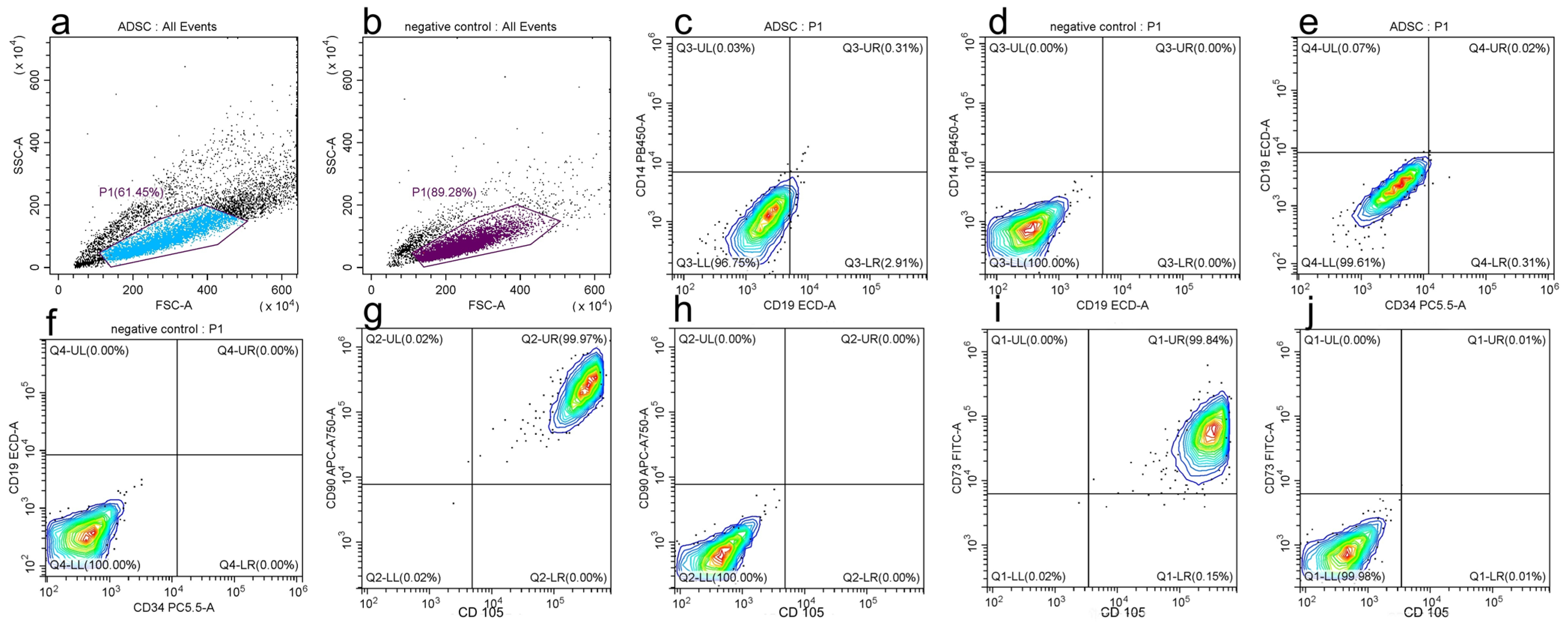
3.2.3. Mesenchymal Stem-Cell-Specific Surface Markers
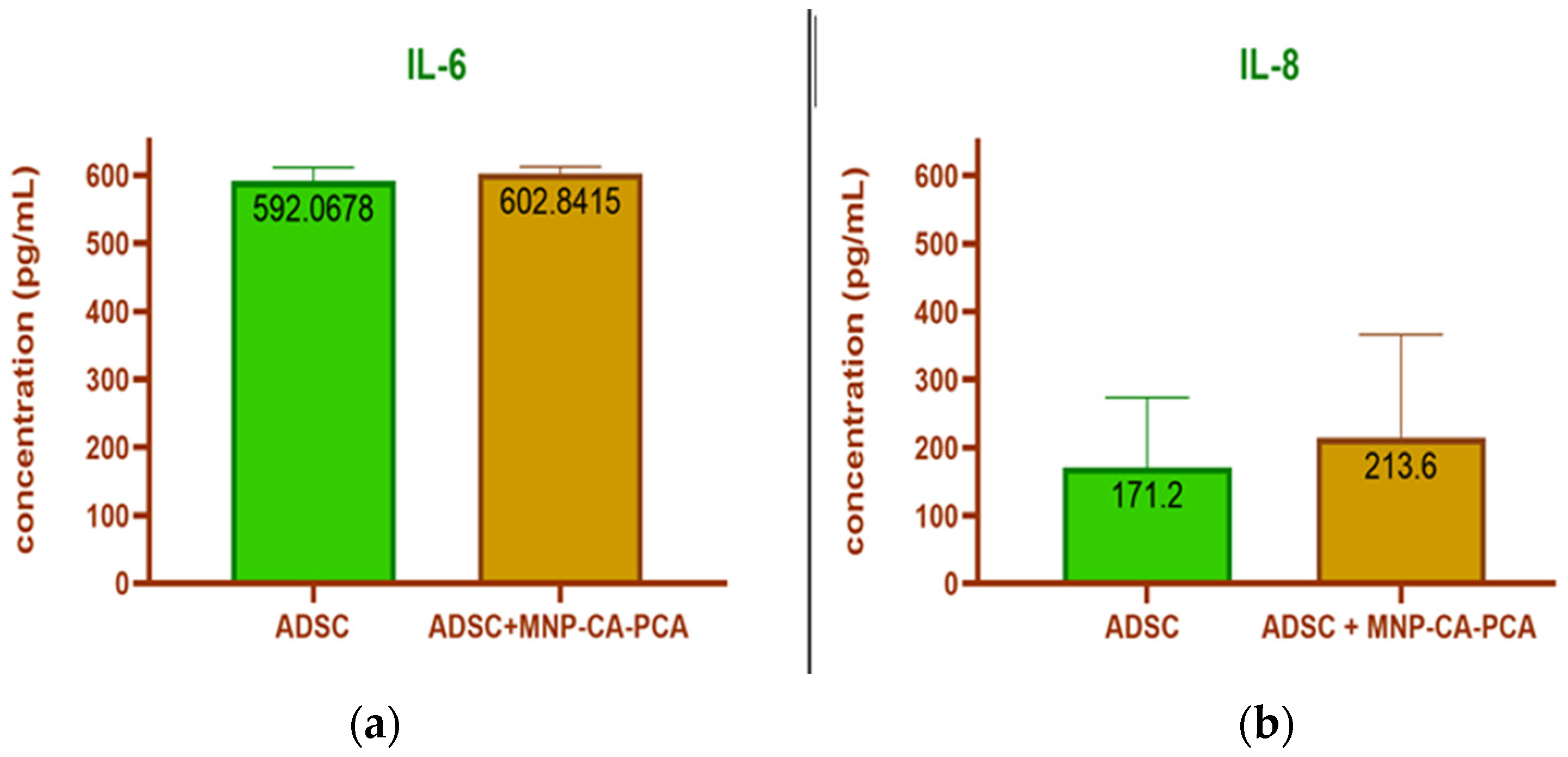
3.2.4. Cytokine Release
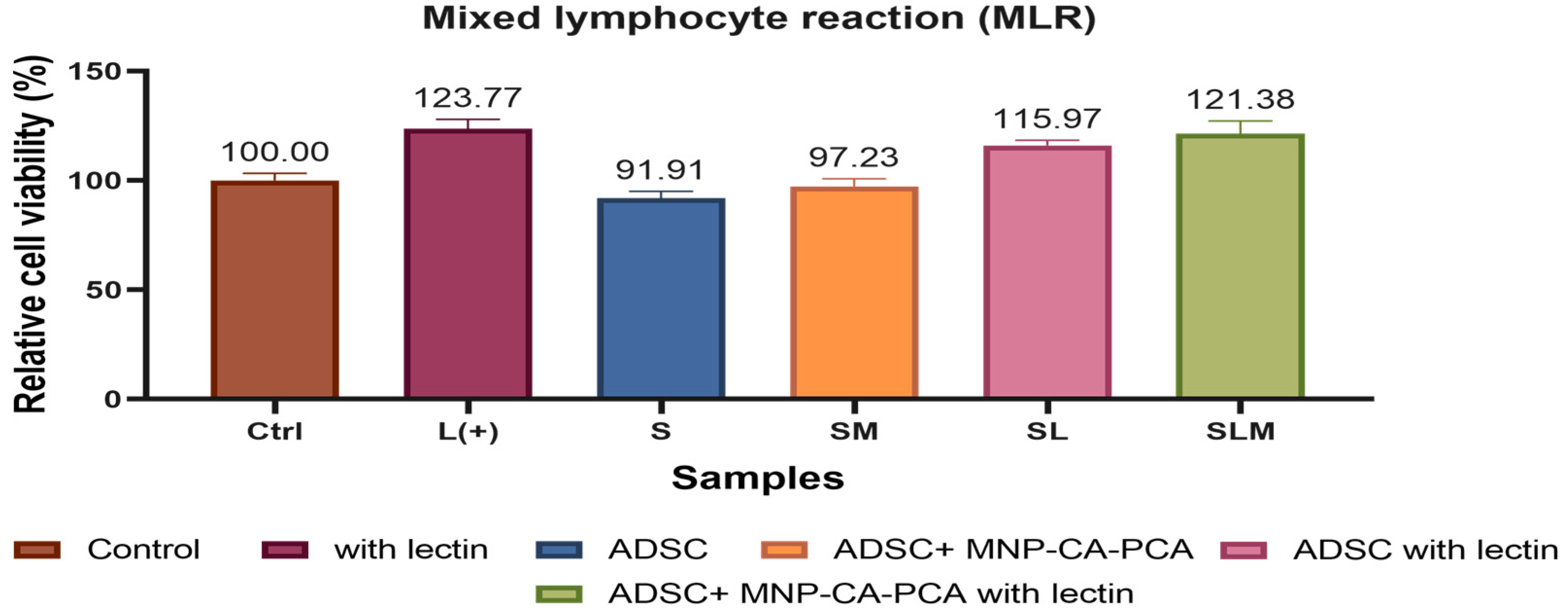
3.2.5. MLR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Daar, A.S.; Greenwood, H.L. A proposed definition of regenerative medicine. J. Tissue Eng. Regen. Med. 2007, 1, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; McDonald, C. Stem Cell Therapies in Clinical Trials: Progress and Challenges. Cell Stem Cell 2015, 17, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xiang, S.; Li, E.N.; Fritch, M.R.; Alexander, P.G.; Lin, H.; Tuan, R.S. Tissue Engineering for Musculoskeletal Regeneration and Disease Modeling. Organotypic Model. Drug Dev. 2020, 265, 235–268. [Google Scholar] [CrossRef]
- Dzobo, K.; Thomford, N.E.; Senthebane, D.A.; Shipanga, H.; Rowe, A.; Dandara, C.; Pillay, M.; Motaung, K.S.C.M. Advances in regenerative medicine and tissue engineering: Innovation and transformation of medicine. Stem Cells Int. 2018, 2018, 2495848. [Google Scholar] [CrossRef] [PubMed]
- Oshima, K.; Shin, K.; Diensthuber, M.; Peng, A.W.; Ricci, A.J.; Heller, S. Mechanosensitive hair cell-like cells from embryonic and in-ducedpluripotent stem cells. Cell 2010, 141, 704–716. [Google Scholar] [CrossRef]
- Reyes, J.H.; O’Shea, K.S.; Wys, N.L.; Velkey, J.M.; Prieskorn, D.M.; Wesolowski, K.; Miller, J.M.; Altschuler, R.A. Glutamatergic neuronal differentiation of mouse embryonic stem cells after transient expression of neurogenin 1 and treatmentwith BDNF and GDNF: In vitro and in vivo studies. J. Neurosci. 2008, 28, 12622–12631. [Google Scholar] [CrossRef]
- Erdö, F.; Bührle, C.; Blunk, J.; Hoehn, M.; Xia, Y.; Fleischmann, B.; Föcking, M.; Küstermann, E.; Kolossov, E.; Hescheler, J.; et al. Hostdependenttumorigenesis of embryonic stem cell trans-plantation inexperimental stroke. J. Cereb. Blood Flow Metab. 2003, 23, 780–785. [Google Scholar] [CrossRef]
- Labusca, L. Adipose tissue in bone regeneration—Stem cell source and beyond. World J. Stem Cells 2022, 14, 372–392. [Google Scholar] [CrossRef]
- Lin, Z.; Perez, P.; Sun, Z.; Liu, J.-J.; Shin, J.H.; Hyrc, K.L.; Samways, D.; Egan, T.; Holley, M.C.; Bao, J. Reprogramming of Single-Cell–Derived Mesenchymal Stem Cells into Hair Cell-like Cells. Otol. Neurotol. 2012, 33, 1648–1655. [Google Scholar] [CrossRef]
- Chen, Y.; Hou, S. Application of magnetic nanoparticles in cell therapy. Stem Cell Res. Ther. 2022, 13, 135. [Google Scholar] [CrossRef]
- Hauser, A.K.; Wydra, R.J.; Stocke, N.A.; Anderson, K.W.; Hilt, J.Z. Magnetic nanoparticles and nanocomposites for remote controlled therapies. J. Control. Release 2015, 219, 76–94. [Google Scholar] [CrossRef] [PubMed]
- Billings, C.; Langley, M.; Warrington, G.; Mashali, F.; Johnson, J. Magnetic Particle Imaging: Current and Future Applications, Magnetic Nanoparticle Synthesis Methods and Safety Measures. Int. J. Mol. Sci. 2021, 22, 7651. [Google Scholar] [CrossRef] [PubMed]
- Labusca, L.S.; Herea, D.-D.; Radu, E.; Danceanu, C.; Chiriac, H.; Lupu, N. Human Adipose Derived Stem Cells and Osteoblasts Interaction with Fe–Cr–Nb–B Magnetic Nanoparticles. J. Nanosci. Nanotechnol. 2018, 18, 5143–5153. [Google Scholar] [CrossRef]
- Liao, N.; Shi, Y.; Wang, Y.; Liao, F.; Zhao, B.; Zheng, Y.; Zeng, Y.; Liu, X.; Liu, J. Antioxidant preconditioning improves therapeutic outcomes of adipose tissue-derived mesenchymal stem cells through enhancing intrahepatic engraftment efficiency in a mouse liver fibrosis model. Stem Cell Res. Ther. 2020, 11, 237. [Google Scholar] [CrossRef]
- Abu-Shahba, N.; Mahmoud, M.; Abdel-Rasheed, M.; Darwish, Y.; AbdelKhaliq, A.; Mohammed, E.; ElHefnawi, M.; Azmy, O. Immunomodulatory and Antioxidative potentials of adipose-derived Mesenchymal stem cells isolated from breast versus abdominal tissue: A comparative study. Cell Regen. 2020, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; He, Y.; Luo, C.; Feng, B.; Ran, F.; Xu, H.; Ci, Z.; Xu, R.; Han, L.; Zhang, D. New progress in the pharmacology of protocatechuic acid: A compound ingested in daily foods and herbs frequently and heavily. Pharmacol. Res. 2020, 161, 105109. [Google Scholar] [CrossRef] [PubMed]
- Salihu, R.; Razak, S.I.A.; Zawawi, N.A.; Kadir, M.R.A.; Ismail, N.I.; Jusoh, N.; Mohamad, M.R.; Nayan, N.H.M. Citric acid: A green cross-linker of biomaterials for biomedical applications. Eur. Polym. J. 2021, 146, 110271. [Google Scholar] [CrossRef]
- Li, L.; Mak, K.Y.; Leung, C.W.; Chan, K.Y.; Chan, W.K.; Zhong, W.; Pong, P.W.T. Effect of synthesis conditions on the prop-erties of citric acid coated iron oxide nanoparticles. Microelectron. Eng. 2013, 110, 329–334. [Google Scholar] [CrossRef]
- Herzig, M.C.; Delavan, C.P.; Jensen, K.J.; Cantu, C.; Montgomery, R.K.; Christy, B.A.; Cap, A.P.; Bynum, J.A. A streamlined proliferation assay using mixed lymphocytes for evaluation of human mesenchymal stem cell immunomodulation activity. J. Immunol. Methods 2020, 488, 112915. [Google Scholar] [CrossRef]
- Abnave, P.; Ghigo, E. Role of the immune system in regeneration and its dynamic interplay with adult stem cells. Semin. Cell Dev. Biol. 2019, 87, 160–168. [Google Scholar] [CrossRef]
- Hoang, D.M.; Pham, P.T.; Bach, T.Q.; Ngo, A.T.L.; Nguyen, Q.T.; Phan, T.T.K.; Nguyen, G.H.; Le, P.T.T.; Hoang, V.T.; Forsyth, N.R.; et al. Stem cell-based therapy for human diseases. Signal Transduct. Target. Ther. 2022, 7, 272. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Ding, Y.; Mu, Y.; Xu, X.; Kong, W.; Chai, R.; Chen, X. Stem Cell-Based Therapies in Hearing Loss. Front. Cell Dev. Biol. 2021, 9, 730042. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Schaudinn, C.; Blume-Peytavi, U.; Vogt, A.; Rancan, F. Effects of Adipose-Derived Stem Cells and Their Conditioned Medium in a Human Ex Vivo Wound Model. Cells 2022, 11, 1198. [Google Scholar] [CrossRef]
- Yoo, T.; Du, X.; Zhou, B. The paracrine effect of mesenchymal human stem cells restored hearing in β-tubulin induced autoimmune sensorineural hearing loss. Hear. Res. 2015, 330, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Lotina, A.; Portela, R.; Baeza, P.; Alcolea-Rodriguez, V.; Villarroel, M.; Ávila, P. Zeta potential as a tool for functional materials development. Catal. Today, 2022, in press. [CrossRef]
- Liu, S.; Yu, B.; Wang, S.; Shen, Y.; Cong, H. Preparation, surface functionalization and application of Fe3O4 magnetic nanoparticles. Adv. Colloid Interface Sci. 2020, 281, 102165. [Google Scholar] [CrossRef] [PubMed]
- Śliwka, L.; Wiktorska, K.; Suchocki, P.; Milczarek, M.; Mielczarek, S.; Lubelska, K.; Cierpiał, T.; Łyżwa, P.; Kiełbasiński, P.; Jaromin, A.; et al. The Comparison of MTT and CVS Assays for the Assessment of Anticancer Agent Interactions. PLoS ONE 2016, 11, e0155772. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, V.F.; Francesko, A.; Ribeiro, C.; Bañobre-López, M.; Martins, P.; Lanceros-Mendez, S. Advances in Magnetic Nanoparticles for Biomedical Applications. Adv. Healthc. Mater. 2018, 7, 1700845. [Google Scholar] [CrossRef]
- Chu, D.-T.; Nguyen Thi Phuong, T.; Tien, N.L.B.; Tran, D.K.; Minh, L.B.; Thanh, V.V.; Gia Anh, P.; Pham, V.H.; Thi Nga, V. Adipose Tissue Stem Cells for Therapy: An Update on the Progress of Isolation, Culture, Storage, and Clinical Application. J. Clin. Med. 2019, 8, 917. [Google Scholar] [CrossRef] [PubMed]
- Kolosnjaj-Tabi, J.; Wilhelm, C.; Clément, O.; Gazeau, F. Cell labeling with magnetic nanoparticles: Opportunity for magnetic cell imaging and cell manipulation. J. Nanobiotechnol. 2013, 11 (Suppl. S1), S7. [Google Scholar] [CrossRef]
- Volatron, J.; Carn, F.; Kolosnjaj-Tabi, J.; Javed, Y.; Vuong, Q.L.; Gossuin, Y.; Ménager, C.; Luciani, N.; Charron, G.; Hémadi, M.; et al. Ferritin Protein Regulates the Degradation of Iron Oxide Nanoparticles. Small 2016, 13, 1602030. [Google Scholar] [CrossRef]
- Van de Walle, A.; Sangnier, A.P.; Abou-Hassan, A.; Curcio, A.; Hémadi, M.; Menguy, N.; Lalatonne, Y.; Luciani, N.; Wilhelm, C. Biosynthesis of magnetic nanoparticles from nano-degradation products revealed in human stem cells. Proc. Natl. Acad. Sci. USA 2019, 116, 4044–4053. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, E.M.; Le Blanc, K.; Dominici, M.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Deans, R.J.; Krause, D.S.; Keating, A.; International Society for Cellular, T. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy 2005, 7, 393–395. [Google Scholar] [CrossRef]
- Zhou, S.; Yin, T.; Zou, Q.; Zhang, K.; Gao, G.; Shapter, J.G.; Huang, P.; Fu, Q. Labeling adipose derived stem cell sheet by ultrasmall super-paramagnetic Fe3O4 nanoparticles and magnetic resonance tracking in vivo. Sci. Rep. 2017, 7, srep42793. [Google Scholar] [CrossRef]
- Zhang, H.; Li, S.; Liu, Y.; Yu, Y.; Lin, S.; Wang, Q.; Miao, L.; Wei, H.; Sun, W. Fe3O4@GO magnetic nanocomposites protect mesenchymal stem cells and promote osteogenic differentiation of rat bone marrow mesenchymal stem cells. Biomater. Sci. 2020, 8, 5984–5993. [Google Scholar] [CrossRef]
- Di Wu, D.; Chang, X.; Tian, J.; Kang, L.; Wu, Y.; Liu, J.; Wu, X.; Huang, Y.; Gao, B.; Wang, H.; et al. Bone mesenchymal stem cells stimulation by magnetic nanoparticles and a static magnetic field: Release of exosomal miR-1260a improves osteogenesis and angiogenesis. J. Nanobiotechnol. 2021, 19, 209. [Google Scholar] [CrossRef]
- Fan, J.; Tan, Y.; Jie, L.; Wu, X.; Yu, R.; Zhang, M. Biological activity and magnetic resonance imaging of superparamagnetic iron oxide nanoparticles-labeled adipose-derived stem cells. Stem Cell Res. Ther. 2013, 4, 44. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Cao, J.; Qi, J.; Liu, Z.; Liu, G.; Deng, W. Regulation of Stem Cell Differentiation by Inorganic Nanomaterials: Recent Advances in Regenerative Medicine. Front. Bioeng. Biotechnol. 2021, 9, 898. [Google Scholar] [CrossRef]
- Kostura, L.; Kraitchman, D.L.; Mackay, A.M.; Pittenger, M.F.; Bulte, J.W.M. Feridex labeling of mesenchymal stem cells inhibits chondrogenesis but not adipogenesis or osteogenesis. NMR Biomed. 2004, 17, 513–517. [Google Scholar] [CrossRef]
- Uciechowski, P.; Dempke, W.C. Interleukin-6: A Masterplayer in the Cytokine Network. Oncology 2020, 98, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Nara, H.; Watanabe, R. Anti-Inflammatory Effect of Muscle-Derived Interleukin-6 and Its Involvement in Lipid Metabolism. Int. J. Mol. Sci. 2021, 22, 9889. [Google Scholar] [CrossRef]
- Cui, L.; Yin, S.; Liu, W.; Li, N.; Zhang, W.; Cao, Y. Expanded Adipose-Derived Stem Cells Suppress Mixed Lymphocyte Reaction by Secretion of Prostaglandin E2. Tissue Eng. 2007, 13, 1185–1195. [Google Scholar] [CrossRef]
- Fiori, A.; Uhlig, S.; Klüter, H.; Bieback, K. Human Adipose Tissue-Derived Mesenchymal Stromal Cells Inhibit CD4+ T Cell Proliferation and Induce Regulatory T Cells as Well as CD127 Expression on CD4+CD25+ T Cells. Cells 2021, 10, 58. [Google Scholar] [CrossRef]
- Gheorghe, D.C.; Epure, V.; Oprea, D.; Zamfir-Chiru-Anton, A. Persistent Stapedial Artery, Oval Window Atresia and Congenital Stapes Agenesis—Case Report. Medicina 2023, 59, 461. [Google Scholar] [CrossRef]
- Baldari, S.; Di Rocco, G.; Piccoli, M.; Pozzobon, M.; Muraca, M.; Toietta, G. Challenges and Strategies for Improving the Regenerative Effects of Mesenchymal Stromal Cell-Based Therapies. Int. J. Mol. Sci. 2017, 18, 2087. [Google Scholar] [CrossRef]
- Xia, Y.; Li, Y.; Wu, X.; Zhang, Q.; Chen, S.; Ma, X.; Yu, M. Ironing Out the Details: How Iron Orchestrates Macrophage Polarization. Front. Immunol. 2021, 12, 669566. [Google Scholar] [CrossRef]




| Sample | Hydrodynamic Diameter (nm) | Zeta Potential (mV) |
|---|---|---|
| MNP | 325.6 | −2.88 |
| MNP-CA | 478 | −17.25 |
| MNP-CA-PCA | 397.9 | −20.24 |
| Title 1 | Title 2 | Title 3 |
| entry 1 | data | data |
| entry 2 | data | data |
| Sample | Fe% | O% | C% |
|---|---|---|---|
| MNP | 84.9 | 15.1 | 0 |
| MNP-CA | 95.8 | 0.3 | 3.9 |
| MNP-CA-PCA | 67.1 | 24.4 | 8.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cumpata, A.J.; Peptanariu, D.; Lungoci, A.-L.; Labusca, L.; Pinteala, M.; Radulescu, L. Towards Regenerative Audiology: Immune Modulation of Adipose-Derived Mesenchymal Cells Preconditioned with Citric Acid-Coated Antioxidant-Functionalized Magnetic Nanoparticles. Medicina 2023, 59, 587. https://doi.org/10.3390/medicina59030587
Cumpata AJ, Peptanariu D, Lungoci A-L, Labusca L, Pinteala M, Radulescu L. Towards Regenerative Audiology: Immune Modulation of Adipose-Derived Mesenchymal Cells Preconditioned with Citric Acid-Coated Antioxidant-Functionalized Magnetic Nanoparticles. Medicina. 2023; 59(3):587. https://doi.org/10.3390/medicina59030587
Chicago/Turabian StyleCumpata, Adeline Josephine, Dragos Peptanariu, Ana-Lacramioara Lungoci, Luminita Labusca, Mariana Pinteala, and Luminita Radulescu. 2023. "Towards Regenerative Audiology: Immune Modulation of Adipose-Derived Mesenchymal Cells Preconditioned with Citric Acid-Coated Antioxidant-Functionalized Magnetic Nanoparticles" Medicina 59, no. 3: 587. https://doi.org/10.3390/medicina59030587
APA StyleCumpata, A. J., Peptanariu, D., Lungoci, A.-L., Labusca, L., Pinteala, M., & Radulescu, L. (2023). Towards Regenerative Audiology: Immune Modulation of Adipose-Derived Mesenchymal Cells Preconditioned with Citric Acid-Coated Antioxidant-Functionalized Magnetic Nanoparticles. Medicina, 59(3), 587. https://doi.org/10.3390/medicina59030587







