A Diagnostic Insight of Dental Pulp Testing Methods in Pediatric Dentistry
Abstract
1. Introduction
2. Aspects of Dental Pulp Physiology in Children
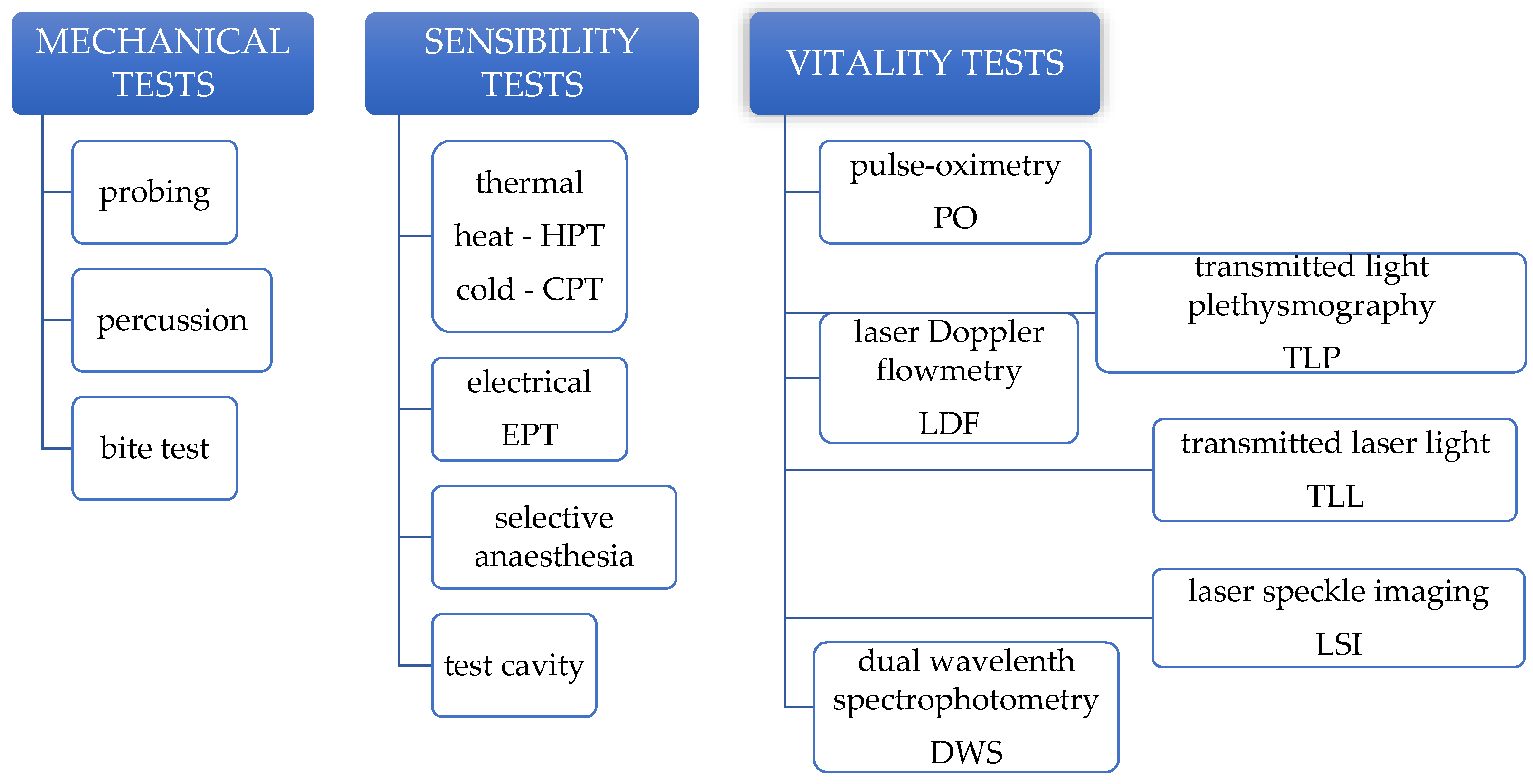
3. Mechanical Pulp Tests
4. Sensibility Pulp Tests
4.1. Thermal Pulp Testing
4.2. Electric Pulp Testing
4.3. Selective Anaesthesia and Test Cavity
4.4. Reliability of Sensibility Pulp Tests in Children
5. Vitality Pulp Tests
5.1. Laser Doppler Flowmetry
5.2. Transmitted Laser Light
5.3. Laser Speckle Imaging
5.4. Pulse-Oximetry
5.5. Transmitted Light Photoplethysmography
5.6. Dual Wavelength Spectrophotometry
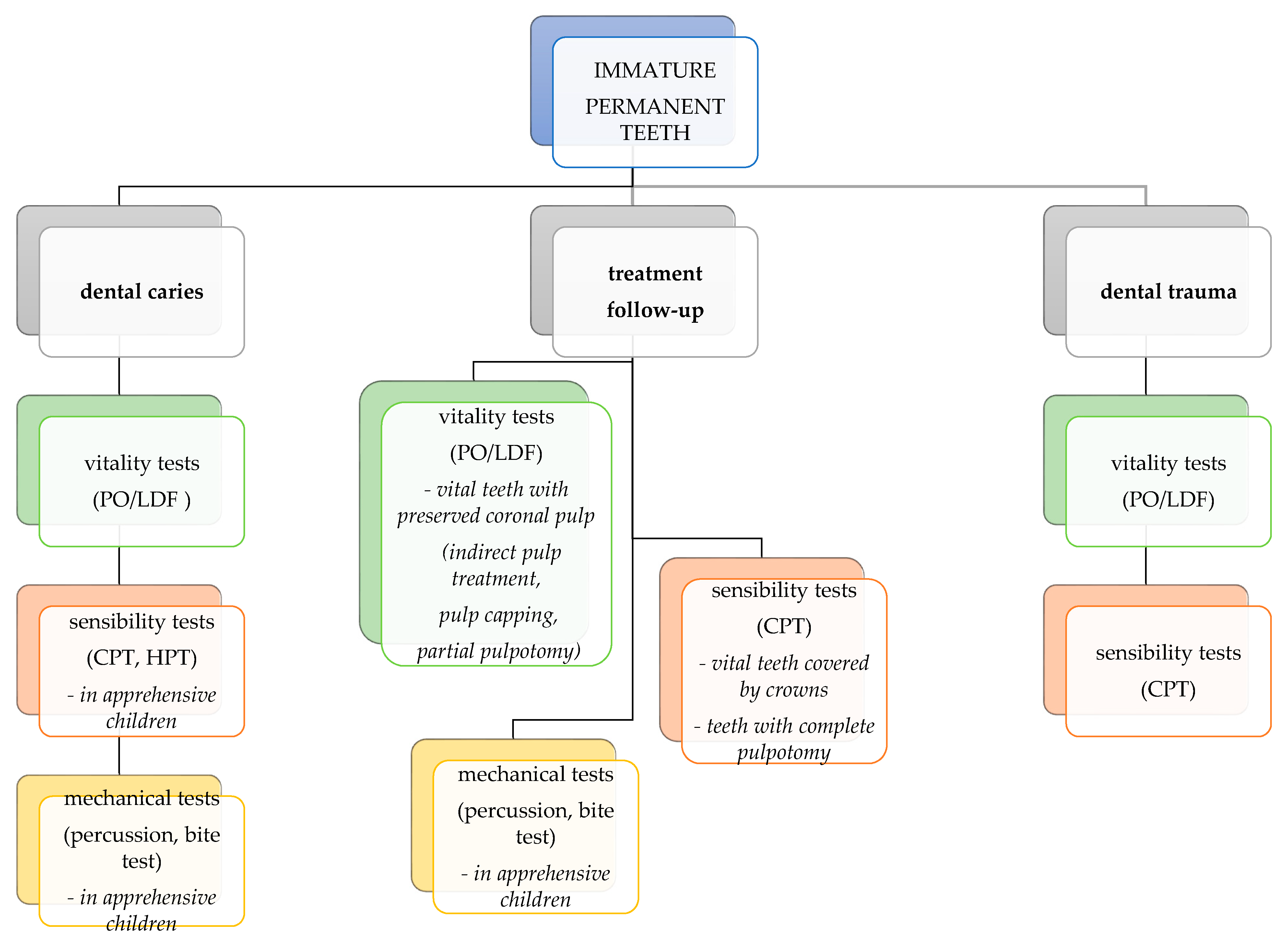
6. Pulp Testing of Traumatized Teeth
7. Comparative Studies
8. Guideline for Using Pulp Tests in Pediatric Clinical Practice
9. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Vargas, K.G.; Fuks, A.B.; Petretz, B. Pulpotomy Techniques: Cervical (Traditional) and Partial. In Pediatric Endodontics; Fuks, A., Petretz, B., Eds.; Springer International Publishing: Cham, Switzerland, 2016; p. 52. [Google Scholar]
- Salgar, A.; Singh, S.; Podar, R.; Kulkarni, G.; Babel, S. Determining predictability and accuracy of thermal and electrical dental pulp tests: An in vivo study. J. Conserv. Dent. 2017, 20, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.; Abbott, P.V. Dental Pulp Testing: A Review. Int. J. Dent. 2009, 12, 365785. [Google Scholar] [CrossRef] [PubMed]
- Vaghela, D.; Sinha, A. Pulse oximetry and laser doppler flowmetry for diagnosis of pulpal vitality. J. Interdiscip. Dent. 2011, 1, 14. [Google Scholar] [CrossRef]
- Chandler, N.P.; Pitt Ford, T.R.; Monteith, B.D. Effect of restorations on pulpal blood flow in molars measured by laser Doppler flowmetry. Int. Endod. J. 2010, 43, 41–46. [Google Scholar] [CrossRef]
- Alghaithy, R.A.; Qualtrough, A.J.E. Pulp sensibility and vitality tests for diagnosing pulpal health in permanent teeth: A critical review. Int. Endod. J. 2017, 50, 135–142. [Google Scholar] [CrossRef]
- Mejàre, I.A.; Axelsson, S.; Davidson, T.; Frisk, F.; Hakeberg, M.; Kvist, T.; Norlund, A.; Petersson, A.; Portenier, I.; Sandberg, H.; et al. Diagnosis of the condition of the dental pulp: A systematic review. Int. Endod. J. 2012, 45, 597–613. [Google Scholar] [CrossRef]
- Guelmann, M. Clinical Pulpal Diagnosis. In Pediatric Endodontics; Fuks, A.B., Peretz, B., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 17–27. [Google Scholar]
- Balevi, B. Cold pulp testing is the simplest and most accurate of all dental pulp sensibility tests. Evid.-Based Dent. 2019, 20, 22–23. [Google Scholar] [CrossRef]
- Mainkar, A.; Kim, S.G. Diagnostic Accuracy of 5 Dental Pulp Tests: A Systematic Review and Meta-analysis. J. Endod. 2018, 44, 694–702. [Google Scholar] [CrossRef]
- Fuks, A.B.; Peretz, B. Pediatric Endodontics: Past and Present Perspectives and Future Directions. In Pediatric Endodontics; Fuks, A., Peretz, B., Eds.; Springer International Publishing: Cham, Switzerland, 2016; p. 2. [Google Scholar]
- Waterhouse, P.; Whitworth, J.; Camp, J.; Fuks, A. Pediatric Endodontics. In Cohen’s Pathways of the Pulp; Hargreaves, K.M., Cohen, S., Berman, L.H., Eds.; Mosby Elsevier: St. Loius, MO, USA, 2011; pp. 809–818. [Google Scholar]
- Egan, C.A.; Hector, M.P.; Bishop, M.A. On the pulpal nerve supply in primary human teeth: Evidence for the innervation of primary dentine. Int. J. Paediatr. Dent. 1999, 9, 57–66. [Google Scholar] [CrossRef]
- Egan, C.A.; Bishop, M.A.; Hector, M.P. An immunohistochemical study of the pulpal nerve supply in primary human teeth: Evidence for the innervation of deciduous dentine. J. Anat. 1996, 188, 623–631. [Google Scholar]
- Khademi, M.; Shekaari, M.A.; Parizi, M.T.; Poureslami, H. Comparison of nerve fibers in the deciduous first and second molar teeth: An in vitro study. Eur. Arch. Paediatr. Dent. 2021, 22, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, J.; Day, P.; Duggal, M.; Morgan, C.; Rodd, H. Pulpal status of human primary teeth with physiological root resorption. Int. J. Paediatr. Dent. 2009, 19, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Day, P.F.; Flores, M.T.; O’Connell, A.C.; Abbott, P.V.; Tsilingaridis, G.; Fouad, A.F.; Cohenca, N.; Lauridsen, E.; Bourguignon, C.; Hicks, L.; et al. International Association of Dental Traumatology guidelines for the management of traumatic dental injuries: 3. Injuries in the primary dentition. Dent. Traumatol. 2020, 36, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Fuks, A.; Guelmann, M.; Kupietzky, A. Current developments in pulp therapy for primary teeth. Endod. Top. 2012, 23, 50–72. [Google Scholar] [CrossRef]
- Fuks, A.; Kupietzky, A.; Guelmann, M. Pulp Therapy for the Primary Dentition. In Pediatric Dentistry-Infancy through Adolescence; Nowak, A.J., Christensen, J.R., Mabry, T.R., Townsend, J., Wells, M.H., Eds.; Elsevier, Inc.: Philadelphia, PA, USA, 2019; p. 1030. [Google Scholar]
- Lin, J.; Chandler, N.P. Electric pulp testing: A review. Int. Endod. J. 2008, 41, 365–374. [Google Scholar] [CrossRef]
- Ricucci, D.; Loghin, S.; Siqueira, J.F. Correlation between clinical and histologic pulp diagnoses. J. Endod. 2014, 40, 1932–1939. [Google Scholar] [CrossRef]
- Peters, D.D.; Baumgartner, J.C.; Lorton, L. Adult pulpal diagnosis. I. Evaluation of the positive and negative responses to cold and electrical pulp tests. J. Endod. 1994, 20, 506–511. [Google Scholar] [CrossRef]
- Samuel, S.; Thomas, A.; Singh, N. A comparative study of pulse oximetry with the conventional pulp testing methods to assess vitality in immature and mature permanent maxillary incisors. CHRISMED J. Health Res. 2014, 1, 235. [Google Scholar] [CrossRef]
- Ghouth, N.; Duggal, M. The use of dental pulp tests in children with dental trauma. A national survey of the British Society of Paediatric Ddentistry’s members. Braz. Dent. J. 2019, 226, 2017–2212. [Google Scholar] [CrossRef]
- Hazard, M.L.; Wicker, C.; Qian, F.; Williamson, A.E.; Teixeira, F.B. Accuracy of cold sensibility testing on teeth with full-coverage restorations: A clinical study. Int. Endod. J. 2021, 54, 1008–1015. [Google Scholar] [CrossRef]
- Fuss, Z.; Trowbridge, H.; Bender, I.B.; Rickoff, B.; Sorin, S. Assessment of reliability of electrical and thermal pulp testing agents. J. Endod. 1986, 12, 301–305. [Google Scholar] [CrossRef]
- Abd-Elmeguid, A.; Yu, D.C. Dental Pulp Neurophysiology: Part 2. Current Diagnostic Tests to Assess Pulp Vitality. J. Can. Dent. Assoc. 2009, 75, 139–143. [Google Scholar] [PubMed]
- Gopakumar, R.; Gopakumar, M. Diagnostic Aids in Pediatric Dentistry. Int. J. Clin. Pediatr. Dent. 2011, 4, 1–7. [Google Scholar] [CrossRef]
- Sui, H.; Lv, Y.; Xiao, M.; Zhou, L.; Qiao, F.; Zheng, J.; Sun, C.; Fu, J.; Chen, Y.; Liu, Y.; et al. Relationship between the difference in electric pulp test values and the diagnostic type of pulpitis. BMC Oral Health 2021, 21, 339. [Google Scholar] [CrossRef]
- Dean, J.A.; Sanders, B.J. Treatment of Deep Caries, Vital Pulp Exposure, and Nonvital Teeth. In McDonald and Avery’s Dentistry for the Child and Adolescent; Dean, J.A., Jones, J.E., Vinson, L.A.W., Sanders, B.J., Yepes, J.F., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2021; pp. 266–285. [Google Scholar]
- Bender, I.B. Pulpal pain diagnosis—A review. J. Endod. 2000, 26, 175–179. [Google Scholar] [CrossRef]
- Blicher, B.; Pryles, R. The Use of Selective Anesthesia in Endodontic Diagnosis. Compend. Contin. Educ. Dent. 2021, 49, 498–502. [Google Scholar]
- Jafarzadeh, H.; Abbott, P.V. Review of pulp sensibility tests. Part II: Electric pulp tests and test cavities. Int. Endod. J. 2010, 43, 945–958. [Google Scholar] [CrossRef]
- Hori, A.; Poureslami, H.R.; Parirokh, M.; Mirzazadeh, A.; Abbott, P. The ability of pulp sensibility tests to evaluate the pulp status in primary teeth. Int. J. Paediatr. Dent. 2011, 21, 441–445. [Google Scholar] [CrossRef]
- Moheb, D.; El Dokky, N. Diagnostic Accuracy of Electrical Pulp Testing Compared to the Thermal in Primary Teeth. Egypt. Dent. J. 2018, 64, 3031–3037. [Google Scholar] [CrossRef][Green Version]
- Nagarathna, C.; Shakuntala, B.S.; Jaiganesh, I. Efficiency and reliability of thermal and electrical tests to evaluate pulp status in primary teeth with assessment of anxiety levels in children. J. Clin. Pediatr. Dent. 2015, 39, 447–451. [Google Scholar] [CrossRef]
- Asfour, M.A.M.; Millar, B.J.; Smith, P.B. An assessment of the reliability of pulp testing deciduous teeth. Int. J. Paediatr. Dent. 1996, 6, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Dean, J.A.; Sanders, B.J. Vital Pulp Exposure, 11th ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Camp, J.H. Diagnosis Dilemmas in Vital Pulp Therapy: Treatment for the Toothache Is Changing, Especially in Young, Immature Teeth. Pediatr. Dent. 2008, 30, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Krastl, G.; Weiger, R.; Filippi, A.; Van Waes, H.; Ebeleseder, K.; Ree, M.; Connert, T.; Widbiller, M.; Tjäderhane, L.; Dummer, P.M.H.; et al. European Society of Endodontology position statement: Endodontic management of traumatized permanent teeth. Int. Endod. J. 2021, 54, 1473–1481. [Google Scholar] [CrossRef]
- Holan, G.; Topf, J.; Fuks, A.B. Effect of root canal infection and treatment of traumatized primary incisors on their permanent successors. Dent. Traumatol. 1992, 8, 12–15. [Google Scholar] [CrossRef]
- Chen, E.; Abbott, P.V. Evaluation of accuracy, reliability, and repeatability of five dental pulp tests. J. Endod. 2011, 37, 1619–1623. [Google Scholar] [CrossRef]
- Motoki, O.; Fergus, D.H.; Yusuke, T.; Nanako, K.; Matsumoto, S.; Mikako, H. Partial Pulpotomy to Successfully Treat a Caries-Induced Pulpal Micro-Abscess: A Case Report. Front. Dent. Med. 2021, 2, 678632. [Google Scholar] [CrossRef]
- Santos, J.M.; Pereira, J.F.; Marques, A.; Sequeira, D.B.; Friedman, S. Vital pulp therapy in permanent mature posterior teeth with symptomatic irreversible pulpitis: A systematic review of treatment outcomes. Medicina 2021, 57, 573. [Google Scholar] [CrossRef]
- Sharma, A.; Madan, M.; Shahi, P.; Sood, P.; Shahi, N. Comparative Study of Pulp Vitality in Primary and Young Permanent Molars in Human Children with Pulse Oximeter and Electric Pulp Tester. Int. J. Clin. Pediatr. Dent. 2015, 8, 94–98. [Google Scholar] [CrossRef]
- Samraj, R.; Indira, R.; Srinivasan, M.R.; Kumar, A. Recent advances in pulp vitality testing. Endodontology 2003, 15, 14–18. [Google Scholar]
- Sasano, T.; Onodera, D.; Hashimoto, K.; Iikubo, M.; Satoh-Kuriwada, S.; Shoji, N.; Miyahara, T. Possible application of transmitted laser light for the assessment of human pulp vitality. Part 2. Increased laser power for enhanced detection of pulpal blood flow. Dent. Traumatol. 2005, 21, 37–41. [Google Scholar] [CrossRef]
- Tomer, A.K.; Ajaz Raina, A.; Bin Ayub, F.; Bhatt, M. Recent advances in pulp vitality testing: A review. Int. J. Appl. Dent. Sci. 2019, 5, 8–12. [Google Scholar]
- Polat, S.; Er, K.; Akpinar, K.E.; Polat, N.T. The sources of laser Doppler blood-flow signals recorded from vital and root canal treated teeth. Arch. Oral Biol. 2004, 49, 53–57. [Google Scholar] [CrossRef]
- Matthews, B.; Vongsavan, N. Advantages and limitations of laser Doppler flow meters. Int. Endod. J. 1993, 26, 9–10. [Google Scholar] [CrossRef]
- Miron, M.-I.; Dodenciu, D.; Sarbescu, P.; Canjau, S.; Ardelean, L.; Rusu, L.C.; Todea, C. Condensation Silicones and Light-curing Resin Used within a Laser Doppler Pulp Vitality Testing Method. Mater. Plast. 2012, 49, 171–175. [Google Scholar]
- Roebuck, E.M.; Evans, D.J.P.; Stirrups, D.; Strang, R. The effect of wavelength, bandwidth, and probe design and position on assessing the vitality of anterior teeth with laser Doppler flowmetry. Int. J. Paediatr. Dent. 2000, 10, 213–220. [Google Scholar] [CrossRef]
- Lobo, E.; Nguyen, S.; Pogrel, M. The Effect of Exercise on Pulpal and Gingival Blood Flow in Physically Active and Inactive Subjects as Assessed by Laser Doppler. Open Dent. J. 2012, 6, 56–60. [Google Scholar] [CrossRef][Green Version]
- Belcheva, A.; Shindova, M.; Hanna, R. Efficacy of Laser Doppler Flowmetry, as a Diagnostic Tool in Assessing Pulp Vitality of Traumatised Teeth: A Split Mouth Clinical Study. J. Pers. Med. 2021, 11, 801. [Google Scholar] [CrossRef]
- Lee, H.N.; Yan, D.Y.; Huang, C.Y.; Chen, S.C.; Pan, C.Y.; Jeng, J.H.; Chen, Y.K.; Chuang, F.H. Laser Doppler for Accurate Diagnosis of Oehler’s Type III Dens Invaginatus: A Case Report. Appl. Sci. 2021, 11, 3848. [Google Scholar] [CrossRef]
- Ghouth, N.; Duggal, M.S.; Kang, J.; Nazzal, H. A Diagnostic Accuracy Study of Laser Doppler Flowmetry for the Assessment of Pulpal Status in Children’s Permanent Incisor Teeth. J. Endod. 2019, 45, 543–548. [Google Scholar] [CrossRef]
- Komatsu, H.; Ikawa, M.; Mayanagi, H. Age-related changes of pulpal blood flow in primary teeth measured by laser Doppler blood flowmetry. Pediatr. Dent. J. 2007, 17, 27–31. [Google Scholar] [CrossRef][Green Version]
- Karayilmaz, H.; Kirzioǧlu, Z. Evaluation of pulpal blood flow changes in primary molars with physiological root resorption by laser doppler flowmetry and pulse oximetry. J. Clin. Pediatr. Dent. 2011, 36, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Stoianovici, C.; Wilder-Smith, P.; Choi, B. Assessment of pulpal vitality using laser speckle imaging. Lasers Surg. Med. 2011, 43, 833–837. [Google Scholar] [CrossRef] [PubMed]
- Regan, C.; Yang, B.Y.; Mayzel, K.C.; Ramirez-San-Juan, J.C.; Wilder-Smith, P.; Choi, B. Fiber-based laser speckle imaging for the detection of pulsatile flow. Lasers Surg. Med. 2015, 47, 520–525. [Google Scholar] [CrossRef]
- Xu, F.; Xie, C.; Zhang, Y.; Shi, G.; Shi, J.; Xu, X.; Zhao, Y.; Zhu, Y.; He, X. Vertically Polarized Laser Speckle Contrast Imaging to Monitor Blood Flow in Pulp. J. Mod. Opt. 2021, 68, 1075–1082. [Google Scholar] [CrossRef]
- Kakanur, M. Evaluation of pulp vitality in Primary teeth using Invasive and Noninvasive Techniques. Evaluation of pulp vitality in Primary teeth using Invasive and Noninvasive Techniques. RGUHS J. Dent. Sci. 2012, 4, 43–46. [Google Scholar]
- Bargrizan, M.; Ashari, M.A.; Ahmadi, M.; Ramezani, J. The use of pulse oximetry in evaluation of pulp vitality in immature permanent teeth. Dent. Traumatol. 2016, 32, 43–47. [Google Scholar] [CrossRef]
- Grabliauskienė, Ž.; Zamaliauskienė, R.; Lodienė, G. Pulp vitality testing with a developed universal pulse oximeter probe holder. Medicina 2021, 57, 101. [Google Scholar] [CrossRef]
- Janani, K.; Ajitha, P.; Sandhya, R.; Subbaiyan, H.; Jose, J. Efficiency of new custom-made pulse oximeter sensor holder in assessment of actual pulp status. J. Fam. Med. Prim. Care 2020, 9, 3333–3337. [Google Scholar] [CrossRef]
- Sharma, D.S.; Mishra, S.; Banda, N.R.; Vaswani, S. In vivo evaluation of customized pulse oximeter and sensitivity pulp tests for assessment of pulp vitality. J. Clin. Pediatr. Dent. 2019, 43, 11–15. [Google Scholar] [CrossRef]
- Almudever-Garcia, A.; Forner, L.; Sanz, J.L.; Llena, C.; Rodríguez-Lozano, F.J.; Guerrero-Gironés, J.; Melo, M. Pulse oximetry as a diagnostic tool to determine pulp vitality: A systematic review. Appl. Sci. 2021, 11, 2747. [Google Scholar] [CrossRef]
- Curt, G. Pulse oximetry evaluation of vitality in primary and immature permanent teeth. Pediatr. Dent. 1999, 21, 125–127. [Google Scholar]
- Schmitt, J.M.; Webber, R.L.; Walker, E.C. Optical Determination of Dental Pulp Vitality. IEEE Trans. Biomed. Eng. 1991, 38, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Anusha, B.; Madhusudhana, K.; Chinni, S.K.; Paramesh, Y. Assessment of pulp oxygen saturation levels by pulse oximetry for pulpal diseases -a diagnostic study. J. Clin. Diagn. Res. 2017, 11, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Farias, T.; Lima, R.; Luize, S.; Kelly, T.; Silva, L.; Jo, E.; Lima, T.F.R.; dos Santos, S.L.; da Silva Fidalgo, T.K.; Silva, E.J.N.L. Vitality Tests for Pulp Diagnosis of Traumatized Teeth: A Systematic Review. J. Endod. 2021, 45, 490–499. [Google Scholar] [CrossRef]
- Miwa, Z.; Ikawa, M.; Iijima, H.; Saito, M.; Takagi, Y. Pulpal blood flow in vital and nonvital young permanent teeth measured by transmitted-light photoplethysmography: A pilot study. Pediatr. Dent. 2002, 24, 594–598. [Google Scholar]
- Knörzer, S.; Hiller, K.A.; Brandt, M.; Niklas, A.; Putzger, J.; Monkman, G.J.; Danilov, S.N.; Ganichev, S.D.; Schulz, I.; Schmalz, G. Detection of pulsed blood flow through a molar pulp chamber and surrounding tissue in vitro. Clin. Oral Investig. 2019, 23, 1121–1132. [Google Scholar] [CrossRef]
- Mythri, H.; Arun, A.; Chachapan, D. Pulp vitality tests—An overview on comparison of sensitivity and vitality. Indian J. Oral Sci. 2015, 6, 41. [Google Scholar] [CrossRef]
- Bastos, J.V.; Goulart, E.M.A.; de Souza Côrtes, M.I. Pulpal response to sensibility tests after traumatic dental injuries in permanent teeth. Dent. Traumatol. 2014, 30, 188–192. [Google Scholar] [CrossRef]
- Andreasen, F.M. Transient apical breakdown and its relation to color and sensibility changes after luxation injuries to teeth. Dent. Traumatol. 1986, 2, 9–19. [Google Scholar] [CrossRef]
- Caldeira, C.L.; Barletta, F.B.; Ilha, M.C.; Abrão, C.V.; Gavini, G. Pulse oximetry: A useful test for evaluating pulp vitality in traumatized teeth. Dent. Traumatol. 2016, 32, 385–389. [Google Scholar] [CrossRef]
- Rodrigues, E.M.; Gomes-Cornélio, A.L.; Soares-Costa, A.; Salles, L.P.; Velayutham, M.; Rossa-Junior, C.; Guerreiro-Tanomaru, J.M.; Tanomaru-Filho, M. An assessment of the overexpression of BMP-2 in transfected human osteoblast cells stimulated by mineral trioxide aggregate and Biodentine. Int. Endod. J. 2017, 50, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Janani, K.; Palanivelu, A.; Sandhya, R. Diagnostic accuracy of dental pulse oximeter with customized sensor holder, thermal test and electric pulp test for the evaluation of pulp vitality: An in vivo study. Braz. Dent. Sci. 2020, 23, 8. [Google Scholar] [CrossRef]
- Karayilmaz, H.; Kirzioǧlu, Z. Comparison of the reliability of laser Doppler flowmetry, pulse oximetry and electric pulp tester in assessing the pulp vitality of human teeth. J. Oral Rehabil. 2011, 38, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Gopikrishna, V.; Tinagupta, K.; Kandaswamy, D. Comparison of Electrical, Thermal, and Pulse Oximetry Methods for Assessing Pulp Vitality in Recently Traumatized Teeth. J. Endod. 2007, 33, 531–535. [Google Scholar] [CrossRef]
- Ghouth, N.; Duggal, M.S.; BaniHani, A.; Nazzal, H. The diagnostic accuracy of laser Doppler flowmetry in assessing pulp blood flow in permanent teeth: A systematic review. Dent. Traumatol. 2018, 34, 311–319. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Igna, A.; Mircioagă, D.; Boariu, M.; Stratul, Ș.-I. A Diagnostic Insight of Dental Pulp Testing Methods in Pediatric Dentistry. Medicina 2022, 58, 665. https://doi.org/10.3390/medicina58050665
Igna A, Mircioagă D, Boariu M, Stratul Ș-I. A Diagnostic Insight of Dental Pulp Testing Methods in Pediatric Dentistry. Medicina. 2022; 58(5):665. https://doi.org/10.3390/medicina58050665
Chicago/Turabian StyleIgna, Andreea, Doina Mircioagă, Marius Boariu, and Ștefan-Ioan Stratul. 2022. "A Diagnostic Insight of Dental Pulp Testing Methods in Pediatric Dentistry" Medicina 58, no. 5: 665. https://doi.org/10.3390/medicina58050665
APA StyleIgna, A., Mircioagă, D., Boariu, M., & Stratul, Ș.-I. (2022). A Diagnostic Insight of Dental Pulp Testing Methods in Pediatric Dentistry. Medicina, 58(5), 665. https://doi.org/10.3390/medicina58050665





