Photobiomodulation Therapy in the Treatment of Oral Mucositis—A Case Report
Abstract
:1. Introduction
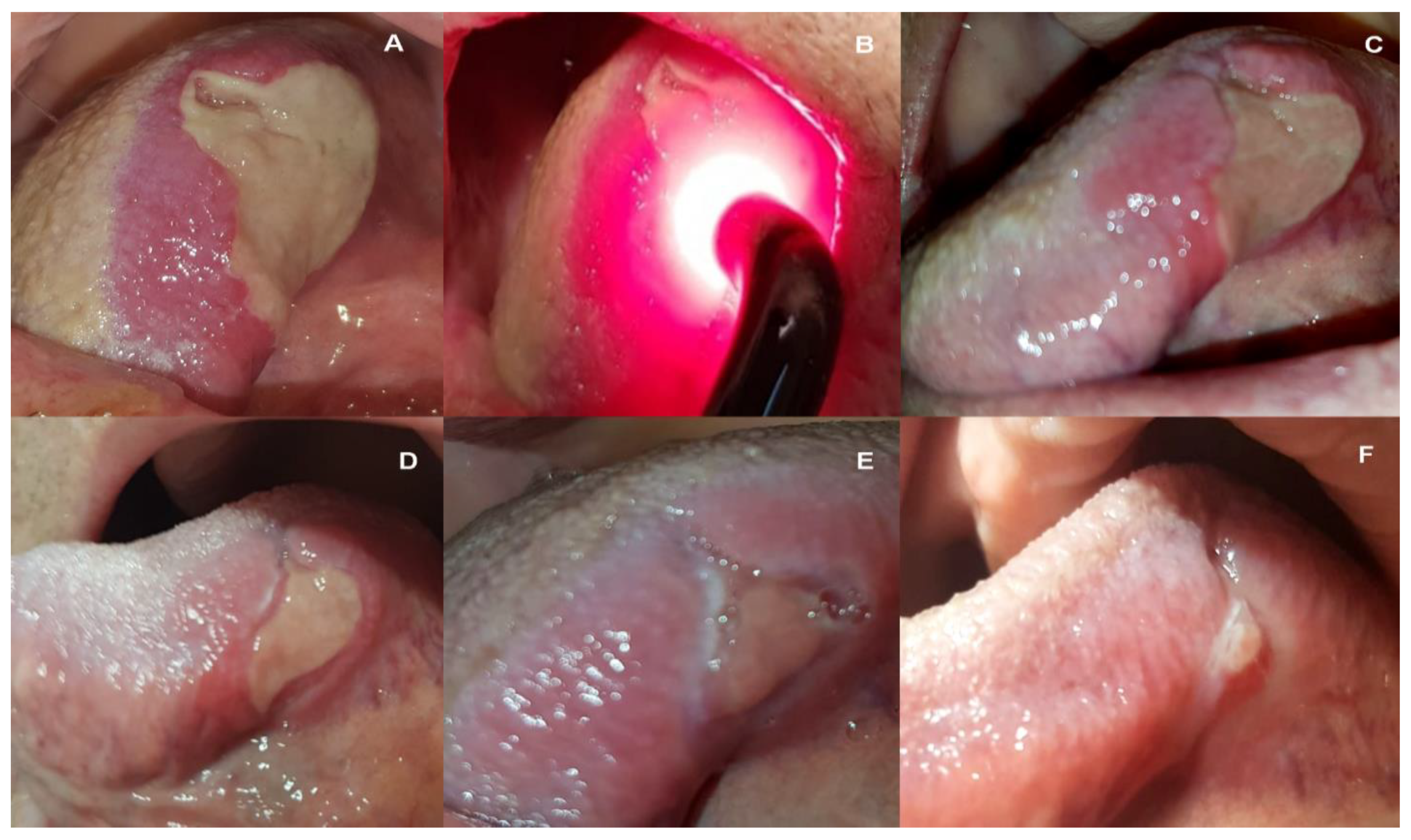
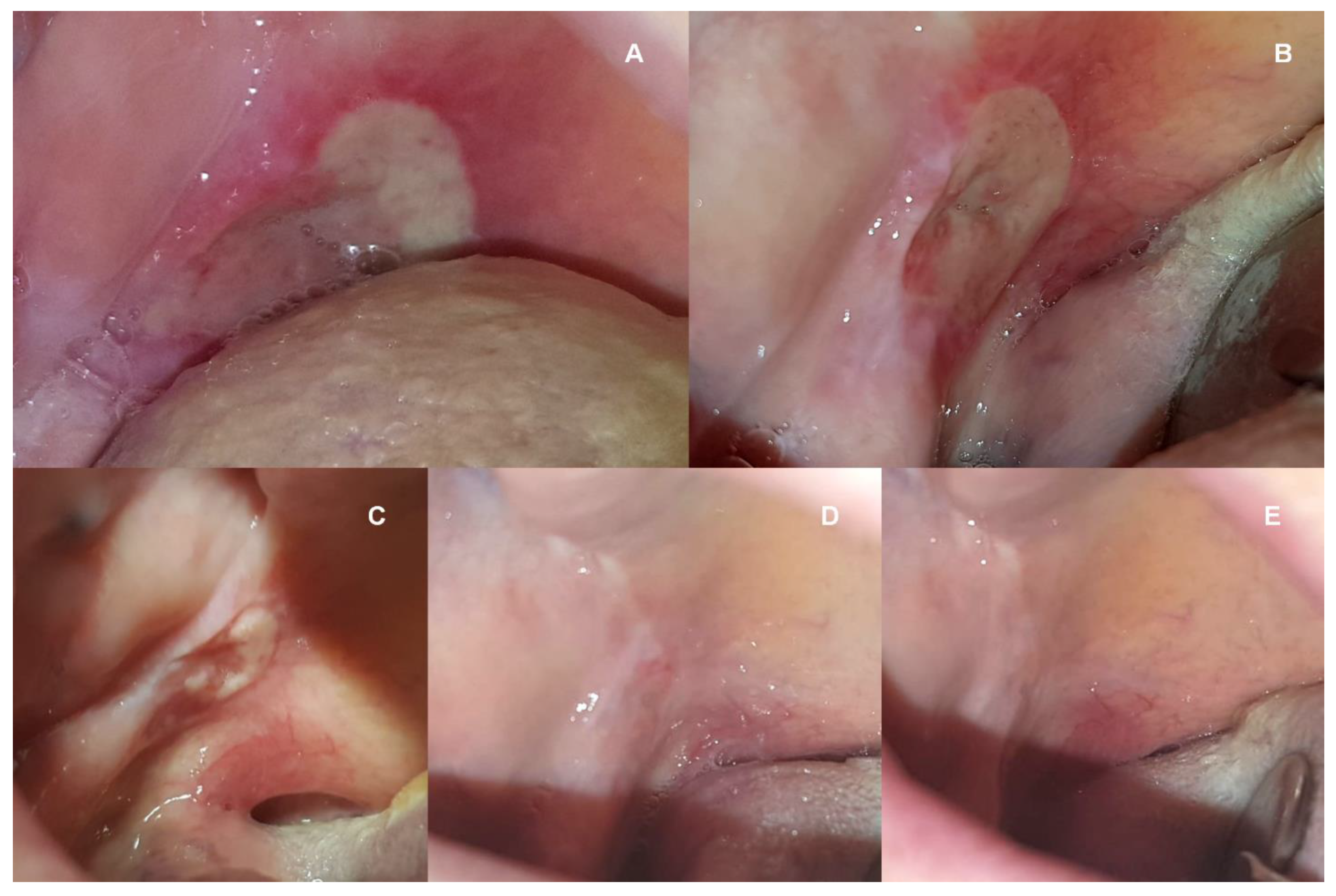
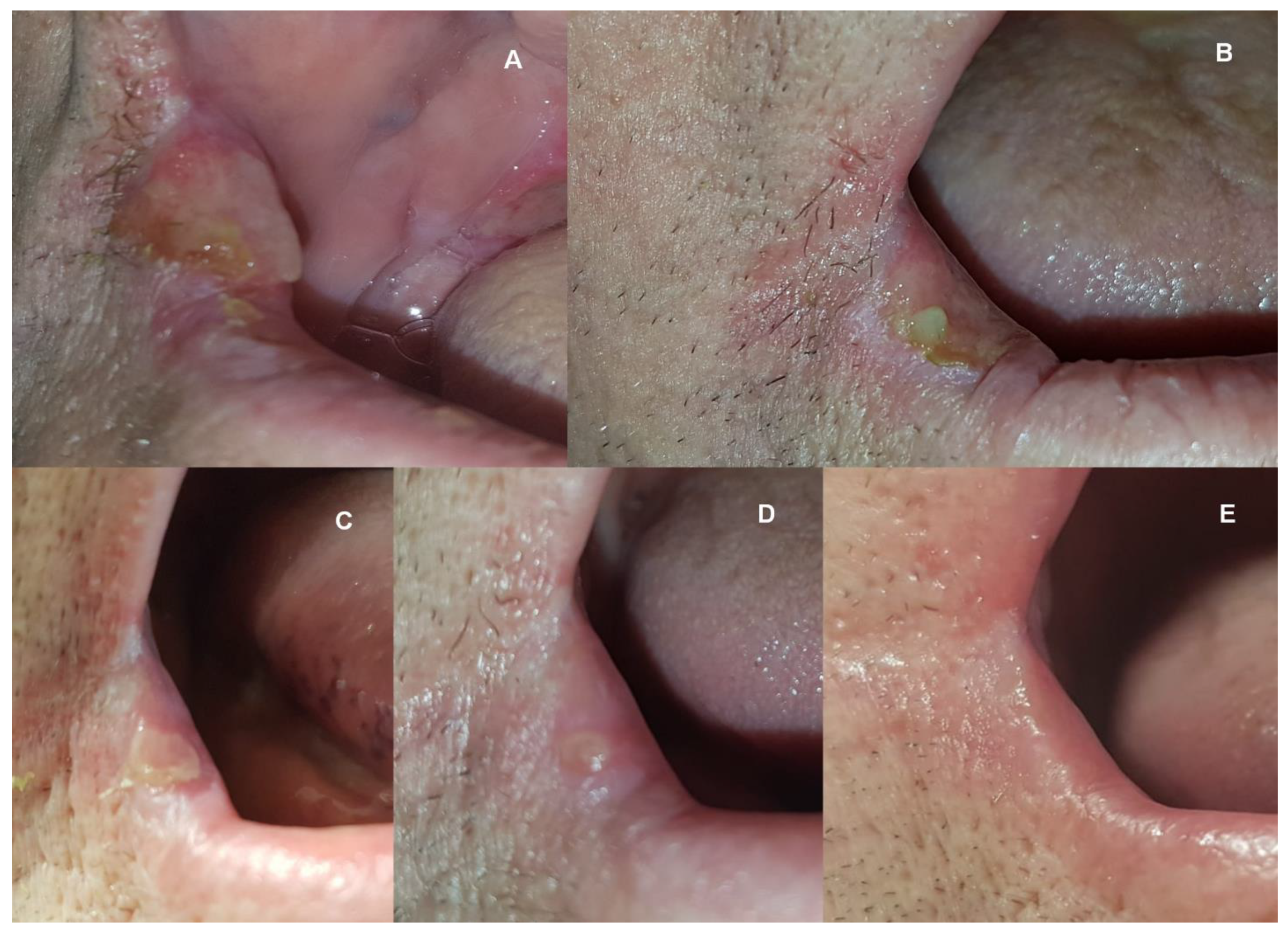
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
References
- Sonis, S.T.; Elting, L.S.; Keefe, D.; Peterson, D.E.; Schubert, M.; Hauer-Jensen, M.; Bekele, B.N.; Raber-Durlacher, J.; Donnelly, J.P.; Rubenstein, E.B. Mucositis Study Section of the Multinational Association for Supportive Care in Cancer; International Society for Oral Oncology. Perspectives on cancer therapy-induced mucosal injury: Pathogenesis, measurement, epidemiology, and consequences for patients. Cancer 2004, 100, 1995–2025. [Google Scholar] [CrossRef]
- Zecha, J.A.; Raber-Durlacher, J.E.; Nair, R.G.; Epstein, J.B.; Sonis, S.T.; Elad, S.; Hamblin, M.R.; Barasch, A.; Migliorati, C.A.; Milstein, D.M.; et al. Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: Part 1: Mechanisms of action, dosimetric, and safety considerations. Support Care Cancer 2016, 24, 2781–2792. [Google Scholar] [CrossRef] [Green Version]
- Melton, C. Chemotherapy-Induced and Radiotherapy-Induced Oral Mucositis. 2017. Cancer Therapy Advisor. Available online: https://www.cancertherapyadvisor.com/home/cancer-topics/supportive-care/side-effect-management/chemotherapy-induced-and-radiotherapy-induced-oral-mucositis/4/ (accessed on 1 May 2017).
- Zecha, J.A.; Raber-Durlacher, J.E.; Nair, R.G.; Epstein, J.B.; Elad, S.; Hamblin, M.R.; Barasch, A.; Migliorati, C.A.; Milstein, D.M.; Genot, M.T.; et al. Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: Part 2: Proposed applications and treatment protocols. Support Care Cancer 2016, 24, 2793–2805. [Google Scholar] [CrossRef] [Green Version]
- Lalla, R.V.; Bowen, J.; Barasch, A.; Elting, L.; Epstein, J.; Keefe, D.M.; McGuire, D.B.; Migliorati, C.; Nicolatou-Galitis, O.; Peterson, D.E.; et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 2014, 120, 1453–1461. [Google Scholar] [CrossRef] [Green Version]
- Elad, S.; Cheng, K.K.F.; Lalla, R.V.; Yarom, N.; Hong, C.; Logan, R.M.; Bowen, J.; Gibson, R.; Saunders, D.P.; Zadik, Y.; et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 2020, 126, 4423–4431. [Google Scholar] [CrossRef]
- Correa, M.E.P.; Cheng, K.K.F.; Chiang, K.; Kandwal, A.; Loprinzi, C.L.; Mori, T.; Potting, C.; Rouleau, T.; Toro, J.J.; Ranna, V.; et al. Systematic review of oral cryotherapy for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer 2020, 28, 2449–2456. [Google Scholar] [CrossRef]
- Peterson, D.E.; Ohrn, K.; Bowen, J.; Fliedner, M.; Lees, J.; Loprinzi, C.; Mori, T.; Osaguona, A.; Weikel, D.S.; Elad, S.; et al. Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO). A systematic review of oral cryotherapy for management of oral mucositis caused by cancer therapy. Support Care Cancer 2013, 21, 327–332. [Google Scholar] [CrossRef] [Green Version]
- Grzech-Leśniak, K.; Bencharit, S.; Dalal, N.; Mroczka, K.; Deeb, J.G. In-Vitro Examination of the Use of Er: YAG Laser to Retrieve Lithium Disilicate Crowns from Titanium Implant Abutments. J. Prosthodont. 2019, 28, 672–676. [Google Scholar] [CrossRef]
- Golob Deeb, J.; Smith, J.; Belvin, B.R.; Lewis, J.; Grzech-Leśniak, K. Er: YAG Laser Irradiation Reduces Microbial Viability When Used in Combination with Irrigation with Sodium Hypochlorite, Chlorhexidine, and Hydrogen Peroxide. Microorganisms 2019, 7, 612. [Google Scholar] [CrossRef] [Green Version]
- Deeb, J.G.; Bencharit, S.; Dalal, N.; Abdulmajeed, A.; Grzech-Leśniak, K. Using Er: YAG laser to remove lithium disilicate crowns from zirconia implant abutments: An in vitro study. PLoS ONE 2019, 14, e0223924. [Google Scholar] [CrossRef] [Green Version]
- Deeb, J.G.; Grzech-Leśniak, K.; Weaver, C.; Matys, J.; Bencharit, S. Retrieval of Glass Fiber Post Using Er: YAG Laser and Conventional Endodontic Ultrasonic Method: An In Vitro Study. J. Prosthodont. 2019, 28, 1024–1028. [Google Scholar] [CrossRef]
- Grzech-Leśniak, K.; Bencharit, S.; Skrjanc, L.; Kanduti, D.; Matys, J.; Deeb, J.G. Utilization of Er: YAG Laser in Retrieving and Reusing of Lithium Disilicate and Zirconia Monolithic Crowns in Natural Teeth: An In Vitro Study. Appl. Sci. 2020, 10, 4357. [Google Scholar] [CrossRef]
- Arnabat-Dominguez, J.; Vecchio, A.D.; Todea, C.; Grzech-Leśniak, K.; Vescovi, P.; Romeo, U.; Nammour, S. Laser dentistry in daily practice during the COVID-19 pandemic: Benefits, risks, and recommendations for safe treatments. Adv. Clin. Exp. Med. 2021, 30, 119–125. [Google Scholar] [CrossRef]
- Marín-Conde, F.; Castellanos-Cosano, L.; Pachón-Ibañez, J.; Serrera-Figallo, M.A.; Gutiérrez-Pérez, J.L.; Torres-Lagares, D. Photobiomodulation with low-level laser therapy reduces oral mucositis caused by head and neck radio-chemotherapy: Prospective randomized controlled trial. Int. J. Oral Maxillofac. Surg. 2019, 48, 917–923. [Google Scholar] [CrossRef]
- Bensadoun, R.J. Photobiomodulation or low-level laser therapy in the management of cancer therapy-induced mucositis, dermatitis, and lymphedema. Curr. Opin. Oncol. 2018, 30, 226–232. [Google Scholar] [CrossRef]
- de Lima, V.H.S.; de Oliveira-Neto, O.B.; da Hora Sales, P.H.; da Silva Torres, T.; de Lima, F.J.C. Effectiveness of low-level laser therapy for oral mucositis prevention in patients undergoing chemoradiotherapy for the treatment of head and neck cancer: A systematic review and meta-analysis. Oral. Oncol. 2020, 102, 104524. [Google Scholar] [CrossRef]
- Gobbo, M.; Verzegnassi, F.; Ronfani, L.; Zanon, D.; Melchionda, F.; Bagattoni, S.; Majorana, A.; Bardellini, E.; Mura, R.; Piras, A.; et al. A multicenter randomized, double-blind controlled trial to evaluate the efficacy of laser therapy for the treatment of severe oral mucositis induced by chemotherapy in children: LaMPO RCT. Pediatr. Blood Cancer 2018, 65, e27098. [Google Scholar] [CrossRef] [Green Version]
- Mobadder, M.E.; Farhat, F.; El Mobadder, W.; Nammour, S. Photobiomodulation Therapy in the Treatment of Oral Mucositis, Dysphagia, Oral Dryness, Taste Alteration, and Burning Mouth Sensation Due to Cancer Therapy: A Case Series. Int. J. Environ. Res. Public Health 2019, 16, 4505. [Google Scholar] [CrossRef] [Green Version]
- Dompe, C.; Moncrieff, L.; Matys, J.; Grzech-Leśniak, K.; Kocherova, I.; Bryja, A.; Bruska, M.; Dominiak, M.; Mozdziak, P.; Skiba, T.H.I.; et al. Photobiomodulation-Underlying Mechanism and Clinical Applications. J. Clin. Med. 2020, 9, 1724. [Google Scholar] [CrossRef]
- Kocherova, I.; Bryja, A.; Błochowiak, K.; Kaczmarek, M.; Stefańska, K.; Matys, J.; Grzech-Leśniak, K.; Dominiak, M.; Mozdziak, P.; Kempisty, B.; et al. Photobiomodulation with Red and Near-Infrared Light Improves Viability and Modulates Expression of Mesenchymal and Apoptotic-Related Markers in Human Gingival Fibroblasts. Materials 2021, 14, 3427. [Google Scholar] [CrossRef]
- Mobadder, M.E.; Farhat, F.; Mobadder, W.E.; Nammour, S. Photobiomodulation Therapy in the Treatment of Oral Mucositis, Dysgeusia and Oral Dryness as Side-Effects of Head and Neck Radiotherapy in a Cancer Patient: A Case Report. Dent. J. 2018, 6, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oberoi, S.; Zamperlini-Netto, G.; Beyene, J.; Treister, N.S.; Sung, L. Effect of prophylactic low-level laser therapy on oral mucositis: A systematic review and meta-analysis. PLoS ONE 2014, 9, e107418. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Zhang, B.; Shen, N.; Wu, N.; Sun, J. A systematic review and meta-analysis of the effect of low-level laser therapy (LLLT) on chemotherapy-induced oral mucositis in pediatric and young patients. Eur. J. Pediatr. 2018, 177, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Migliorati, C.; Hewson, I.; Lalla, R.V.; Antunes, H.S.; Estilo, C.L.; Hodgson, B.; Lopes, N.N.F.; Schubert, M.M.; Bowen, J.; Elad, S. Systematic review of laser and other light therapy for the management of oral mucositis in cancer patients. Support Care Cancer 2013, 21, 333–341. [Google Scholar] [CrossRef] [Green Version]
- Martins, A.F.L.; Nogueira, T.E.; Morais, M.O.; Oton-Leite, A.F.; Valadares, M.C.; Batista, A.C.; Freitas, N.M.A.; Leles, C.R.; Mendonça, E.F. Effect of photobiomodulation on the severity of oral mucositis and molecular changes in head and neck cancer patients undergoing radiotherapy: A study protocol for a cost-effectiveness randomized clinical trial. Trials 2019, 20, 97. [Google Scholar] [CrossRef]
- Patini, R.; Staderini, E.; Camodeca, A.; Guglielmi, F.; Gallenzi, P. Case Reports in Pediatric Dentistry Journals: A Systematic Review about Their Effect on Impact Factor and Future Investigations. Dent. J. 2019, 7, 103. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jabłoński, P.; Musiał, M.; Wiench, R.; Stefanik, N.; Olchowy, C.; Matys, J.; Skaba, D.; Grzech-Leśniak, K. Photobiomodulation Therapy in the Treatment of Oral Mucositis—A Case Report. Medicina 2022, 58, 618. https://doi.org/10.3390/medicina58050618
Jabłoński P, Musiał M, Wiench R, Stefanik N, Olchowy C, Matys J, Skaba D, Grzech-Leśniak K. Photobiomodulation Therapy in the Treatment of Oral Mucositis—A Case Report. Medicina. 2022; 58(5):618. https://doi.org/10.3390/medicina58050618
Chicago/Turabian StyleJabłoński, Przemysław, Mikołaj Musiał, Rafał Wiench, Natalia Stefanik, Cyprian Olchowy, Jacek Matys, Dariusz Skaba, and Kinga Grzech-Leśniak. 2022. "Photobiomodulation Therapy in the Treatment of Oral Mucositis—A Case Report" Medicina 58, no. 5: 618. https://doi.org/10.3390/medicina58050618
APA StyleJabłoński, P., Musiał, M., Wiench, R., Stefanik, N., Olchowy, C., Matys, J., Skaba, D., & Grzech-Leśniak, K. (2022). Photobiomodulation Therapy in the Treatment of Oral Mucositis—A Case Report. Medicina, 58(5), 618. https://doi.org/10.3390/medicina58050618







