Odontogenic Effect of Icariin on the Human Dental Pulp Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection, Cell Isolation, and Cell Culture
2.2. Icariin Treatment
2.3. Cytotoxicity Assay
2.4. Odontogenic Differentiation
2.5. Scratch Migration Assay
2.6. Alizarin Red S (ARS) Staining Assay and Alkaline Phosphatase (ALP) Staining Assay
2.7. RNA Isolation and Gene Expression Analysis by Quantitative Real-Time Polymerase Chain Reaction
2.8. Western Blot Analysis
2.9. Statistical Analysis
3. Results
3.1. Effects of Icariin on Cell Viability and Migration of HDPCs
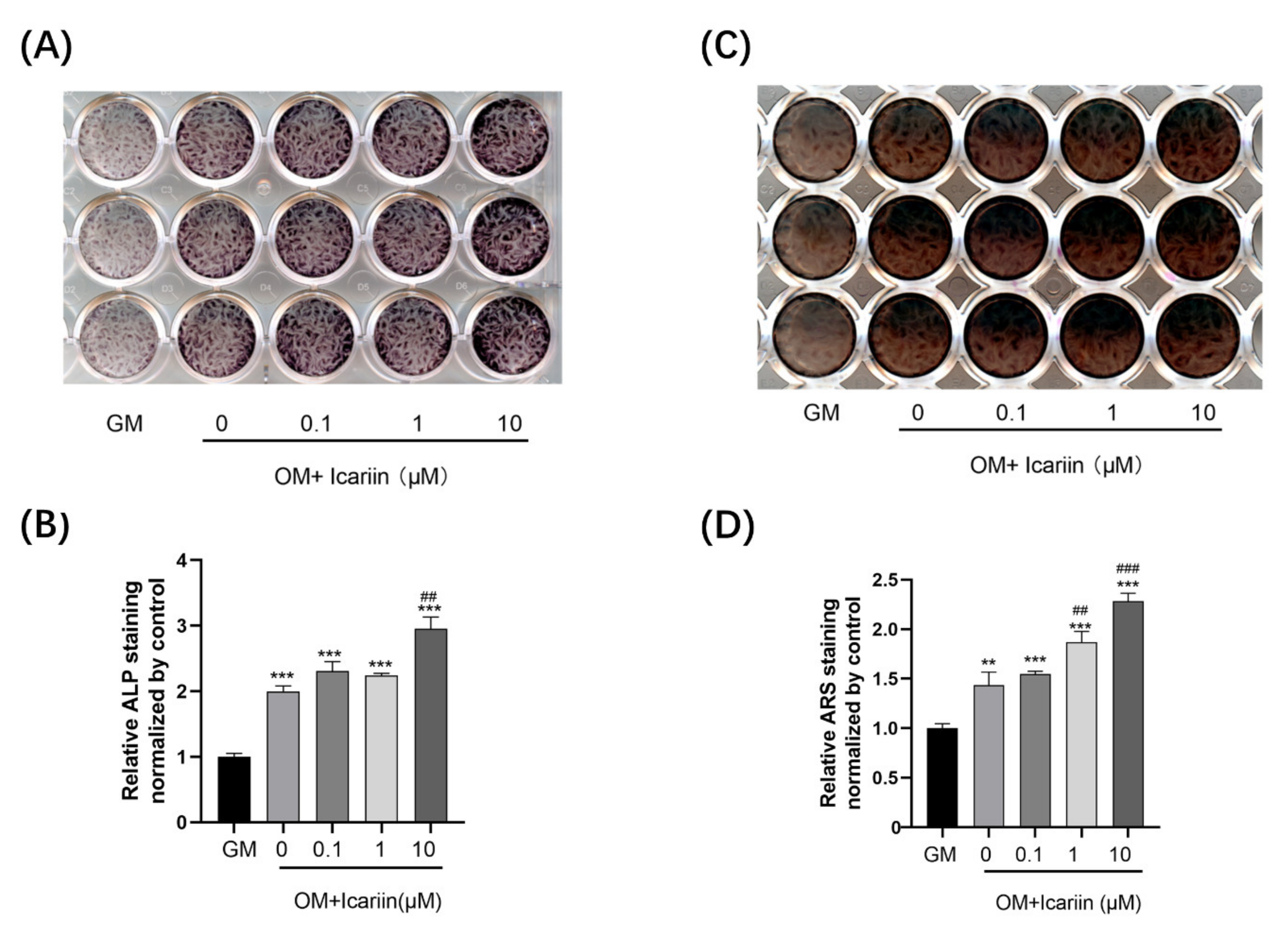
3.2. Icariin Accelerated ALP Activity and Mineralization Ability of HDPCs
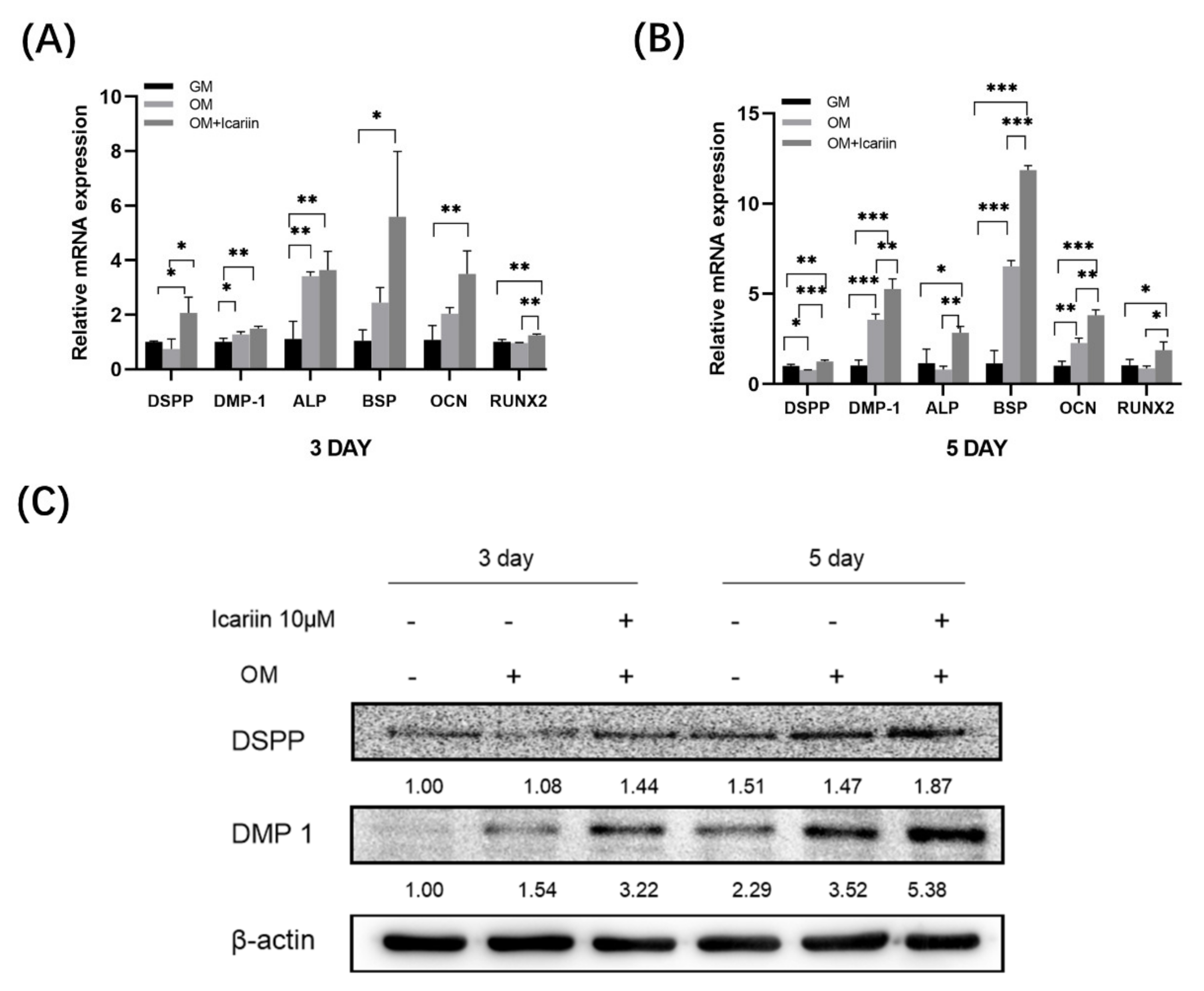
3.3. Effect of Icariin on Gene Expression and Protein Expression of Odontogenic Markers in HDPCs
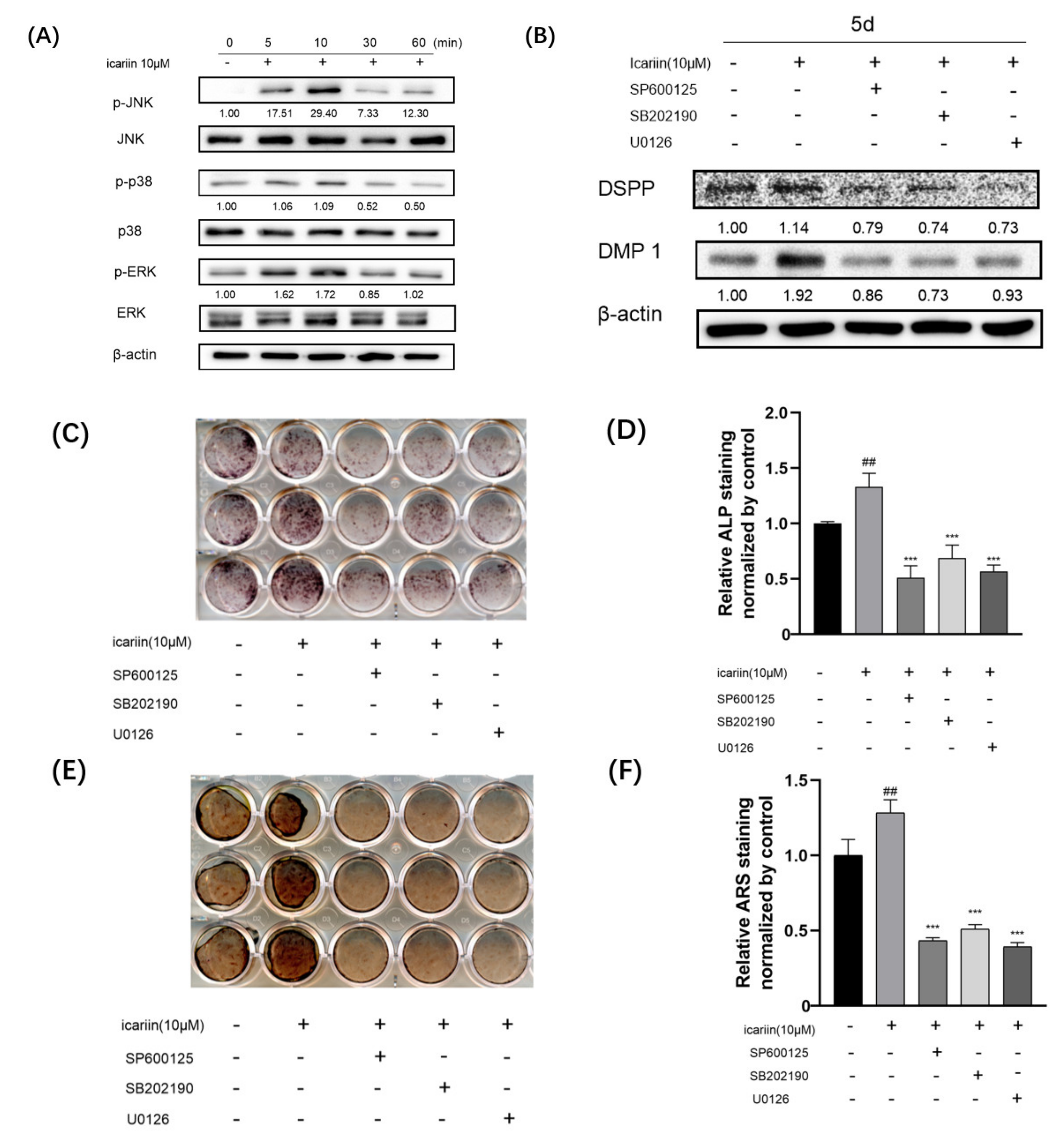
3.4. Icariin-Mediated Activation of Mitogen-Activated Protein Kinase Signaling during Odontogenic Differentiation of HDPCs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yu, C.; Abbott, P.V. An overview of the dental pulp: Its functions and responses to injury. Aust. Dent. J. 2007, 52 (Suppl. S1), S4–S16. [Google Scholar] [CrossRef] [PubMed]
- Farges, J.C.; Alliot-Licht, B.; Renard, E.; Ducret, M.; Gaudin, A.; Smith, A.J.; Cooper, P.R. Dental Pulp Defence and Repair Mechanisms in Dental Caries. Mediat. Inflamm. 2015, 2015, 230251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neves, V.C.M.; Yianni, V.; Sharpe, P.T. Macrophage modulation of dental pulp stem cell activity during tertiary dentinogenesis. Sci. Rep. 2020, 10, 20216. [Google Scholar] [CrossRef] [PubMed]
- Komabayashi, T.; Zhu, Q.; Eberhart, R.; Imai, Y. Current status of direct pulp-capping materials for permanent teeth. Dent. Mater. J. 2016, 35, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Bjørndal, L.; Simon, S.; Tomson, P.L.; Duncan, H.F. Management of deep caries and the exposed pulp. Int. Endod. J. 2019, 52, 949–973. [Google Scholar] [CrossRef]
- Cushley, S.; Duncan, H.F.; Lappin, M.J.; Chua, P.; Elamin, A.D.; Clarke, M.; El-Karim, I.A. Efficacy of direct pulp capping for management of cariously exposed pulps in permanent teeth: A systematic review and meta-analysis. Int. Endod. J. 2021, 54, 556–571. [Google Scholar] [CrossRef]
- Paula, A.; Carrilho, E.; Laranjo, M.; Abrantes, A.M.; Casalta-Lopes, J.; Botelho, M.F.; Marto, C.M.; Ferreira, M.M. Direct Pulp Capping: Which is the Most Effective Biomaterial? A Retrospective Clinical Study. Materials 2019, 12, 3382. [Google Scholar] [CrossRef] [Green Version]
- Sabbagh, J.; Ghassibe-Sabbagh, M.; Fayyad-Kazan, M.; Al-Nemer, F.; Fahed, J.C.; Berberi, A.; Badran, B. Differences in osteogenic and odontogenic differentiation potential of DPSCs and SHED. J. Dent. 2020, 101, 103413. [Google Scholar] [CrossRef]
- Ching, H.S.; Luddin, N.; Rahman, I.A.; Ponnuraj, K.T. Expression of Odontogenic and Osteogenic Markers in DPSCs and SHED: A Review. Curr. Stem Cell Res. Ther. 2017, 12, 71–79. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, D.; Yang, D.; Zhen, W.; Zhang, J.; Peng, S. The effect of icariin on bone metabolism and its potential clinical application. Osteoporos. Int. 2018, 29, 535–544. [Google Scholar] [CrossRef]
- Jiao, F.; Tang, W.; Huang, H.; Zhang, Z.; Liu, D.; Zhang, H.; Ren, H. Icariin Promotes the Migration of BMSCs In Vitro and In Vivo via the MAPK Signaling Pathway. Stem Cells Int. 2018, 2018, 2562105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.; Xia, L.; Zhou, Y.; Xu, Y.; Jiang, X. Icariin induces osteogenic differentiation of bone mesenchymal stem cells in a MAPK-dependent manner. Cell Prolif. 2015, 48, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Lee, K.Y.; Park, B. Icariin abrogates osteoclast formation through the regulation of the RANKL-mediated TRAF6/NF-κB/ERK signaling pathway in Raw264.7 cells. Phytomedicine 2018, 51, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, L.; Tang, Y.; Yang, J.; Jin, Y.; Ma, C. Icariin promotes osteogenic differentiation by suppressing Notch signaling. Eur. J. Pharmacol. 2019, 865, 172794. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.J.; Liu, X.Y.; Jing, D.B. Icariin induces the growth, migration and osteoblastic differentiation of human periodontal ligament fibroblasts by inhibiting Toll-like receptor 4 and NF-κB p65 phosphorylation. Mol. Med. Rep. 2018, 18, 3325–3331. [Google Scholar] [CrossRef] [Green Version]
- Pei, Z.; Zhang, F.; Niu, Z.; Shi, S. Effect of icariin on cell proliferation and the expression of bone resorption/formation-related markers in human periodontal ligament cells. Mol. Med. Rep. 2013, 8, 1499–1504. [Google Scholar] [CrossRef] [Green Version]
- Svandova, E.; Peterkova, R.; Matalova, E.; Lesot, H. Formation and Developmental Specification of the Odontogenic and Osteogenic Mesenchymes. Front. Cell Dev. Biol. 2020, 8, 640. [Google Scholar] [CrossRef]
- Rathinam, E.; Govindarajan, S.; Rajasekharan, S.; Declercq, H.; Elewaut, D.; De Coster, P.; Martens, L.; Leybaert, L. The calcium dynamics of human dental pulp stem cells stimulated with tricalcium silicate-based cements determine their differentiation and mineralization outcome. Sci. Rep. 2021, 11, 645. [Google Scholar] [CrossRef]
- An, S. The emerging role of extracellular Ca2+ in osteo/odontogenic differentiation and the involvement of intracellular Ca (2+) signaling: From osteoblastic cells to dental pulp cells and odontoblasts. J. Cell Physiol. 2019, 234, 2169–2193. [Google Scholar] [CrossRef]
- Seang, S.; Pavasant, P.; Everts, V.; Limjeerajarus, C.N. Prostacyclin Analog Promotes Human Dental Pulp Cell Migration via a Matrix Metalloproteinase 9-related Pathway. J. Endod. 2019, 45, 873–881. [Google Scholar] [CrossRef]
- Son, J.W.; Choi, S.H.; Jang, J.H.; Koh, J.T.; Oh, W.M.; Hwang, Y.C.; Lee, B.N. Irisin promotes odontogenic differentiation and angiogenic potential in human dental pulp cells. Int. Endod. J. 2021, 54, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Zu, Y.; Mu, Y.; Li, Q.; Zhang, S.T.; Yan, H.J. Icariin alleviates osteoarthritis by inhibiting NLRP3-mediated pyroptosis. J. Orthop. Surg. Res. 2019, 14, 307. [Google Scholar] [CrossRef] [PubMed]
- Chung, B.H.; Kim, J.D.; Kim, C.K.; Kim, J.H.; Won, M.H.; Lee, H.S.; Dong, M.S.; Ha, K.S.; Kwon, Y.G.; Kim, Y.M. Icariin stimulates angiogenesis by activating the MEK/ERK- and PI3K/Akt/eNOS-dependent signal pathways in human endothelial cells. Biochem. Biophys. Res. Commun. 2008, 376, 404–408. [Google Scholar] [CrossRef]
- Fan, J.-J.; Cao, L.-G.; Wu, T.; Wang, D.-X.; Jin, D.; Jiang, S.; Zhang, Z.-Y.; Bi, L.; Pei, G.-X. The dose-effect of icariin on the proliferation and osteogenic differentiation of human bone mesenchymal stem cells. Molecules 2011, 16, 10123–10133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cui, D.; Xiao, J.; Zhou, Y.; Zhou, X.; Liu, Y.; Peng, Y.; Yu, Y.; Li, H.; Zhou, X.; Yuan, Q.; et al. Epiregulin enhances odontoblastic differentiation of dental pulp stem cells via activating MAPK signalling pathway. Cell Prolif. 2019, 52, e12680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giraud, T.; Jeanneau, C.; Rombouts, C.; Bakhtiar, H.; Laurent, P.; About, I. Pulp capping materials modulate the balance between inflammation and regeneration. Dent. Mater. 2019, 35, 24–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKee, M.D.; Addison, W.N.; Kaartinen, M.T. Hierarchies of extracellular matrix and mineral organization in bone of the craniofacial complex and skeleton. Cells Tissues Organs 2005, 181, 176–188. [Google Scholar] [CrossRef]
- Fisher, L.W. DMP1 and DSPP: Evidence for duplication and convergent evolution of two SIBLING proteins. Cells Tissues Organs 2011, 194, 113–118. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Yuan, S.; Sun, J.; Gong, Y.; Liu, S.; Guo, R.; He, W.; Kang, P.; Li, R. Inhibitory effect of the TSG-6 on the BMP-4/Smad signaling pathway and odonto/osteogenic differentiation of dental pulp stem cells. Biomed. Pharmacother. 2020, 128, 110266. [Google Scholar] [CrossRef]
- Ye, L.; MacDougall, M.; Zhang, S.; Xie, Y.; Zhang, J.; Li, Z.; Lu, Y.; Mishina, Y.; Feng, J.Q. Deletion of dentin matrix protein-1 leads to a partial failure of maturation of predentin into dentin, hypomineralization, and expanded cavities of pulp and root canal during postnatal tooth development. J. Biol. Chem. 2004, 279, 19141–19148. [Google Scholar] [CrossRef] [Green Version]
- Thyagarajan, T.; Sreenath, T.; Cho, A.; Wright, J.T.; Kulkarni, A.B. Reduced expression of dentin sialophosphoprotein is associated with dysplastic dentin in mice overexpressing transforming growth factor-beta 1 in teeth. J. Biol. Chem. 2001, 276, 11016–11020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, X.; Ling, J.; Wu, L.; Liu, L.; Xiao, Y. Expression of mineralization markers in dental pulp cells. J. Endod. 2007, 33, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Gluhak-Heinrich, J.; Wang, Y.H.; Wu, Y.M.; Chuang, H.H.; Chen, L.; Yuan, J.H.; Dong, J.; Gay, I.; MacDougall, M. Runx2, osx, and dspp in tooth development. J. Dent. Res. 2009, 88, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Papagerakis, P.; Berdal, A.; Mesbah, M.; Peuchmaur, M.; Malaval, L.; Nydegger, J.; Simmer, J.; Macdougall, M. Investigation of osteocalcin, osteonectin, and dentin sialophosphoprotein in developing human teeth. Bone 2002, 30, 377–385. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, H.T. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res. 2002, 12, 9–18. [Google Scholar] [CrossRef]
- Hu, X.; Zhong, Y.; Kong, Y.; Chen, Y.; Feng, J.; Zheng, J. Lineage-specific exosomes promote the odontogenic differentiation of human dental pulp stem cells (DPSCs) through TGFβ1/smads signaling pathway via transfer of microRNAs. Stem Cell Res. Ther. 2019, 10, 170. [Google Scholar] [CrossRef] [Green Version]
- Ngo, V.A.; Jung, J.Y.; Koh, J.T.; Oh, W.M.; Hwang, Y.C.; Lee, B.N. Leptin Induces Odontogenic Differentiation and Angiogenesis in Human Dental Pulp Cells via Activation of the Mitogen-activated Protein Kinase Signaling Pathway. J. Endod. 2018, 44, 585–591. [Google Scholar] [CrossRef]
- Han, J.W.; Lee, B.N.; Kim, S.M.; Koh, J.T.; Min, K.S.; Hwang, Y.C. Odontogenic Potential of Parathyroid Hormone-related Protein (107–111) Alone or in Combination with Mineral Trioxide Aggregate in Human Dental Pulp Cells. J. Endod. 2017, 43, 2054–2060. [Google Scholar] [CrossRef]

| Genes | Primer Sequences (5′-3′) |
|---|---|
| DMP 1 | Forward: TGG TCC CAG CAG TGA GTC CA |
| Reverse: TGT GTG CGA GCT GTC CTC CT | |
| DSPP | Forward: GGG AAT ATT GAG GGC TGG AA |
| Reverse: TCA TTG TGA CCT GCA TCG CC | |
| RUNX2 | Forward: CCA GAT GGG ACT GTG GTT AC |
| Reverse: ACT TGG TGC AGA GTT CAG GG | |
| ALP | Forward: CGG GCA CCA TGA AGG AAA |
| Reverse: GGC CAG ACC AAA GAT AGA GTT | |
| OCN | Forward: CTC ACA CTC CTC GCC GTA TT |
| Reverse: GCT CCC AGC CAT TGA TAC AG | |
| BSP | Forward: AGC GAA GCA GAA GTG GAT GAA |
| Reverse: CTG CAT TGG CTC CAG TGA CA | |
| GAPDH | Forward: CAT CAC CAT CTT CCA GGA G |
| Reverse: AGG CTG TTG TCA TAC TTC TC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, G.; Yang, Y.; Min, K.-S.; Lee, B.-N.; Hwang, Y.-C. Odontogenic Effect of Icariin on the Human Dental Pulp Cells. Medicina 2022, 58, 434. https://doi.org/10.3390/medicina58030434
Liu G, Yang Y, Min K-S, Lee B-N, Hwang Y-C. Odontogenic Effect of Icariin on the Human Dental Pulp Cells. Medicina. 2022; 58(3):434. https://doi.org/10.3390/medicina58030434
Chicago/Turabian StyleLiu, Guo, Ying Yang, Kyung-San Min, Bin-Na Lee, and Yun-Chan Hwang. 2022. "Odontogenic Effect of Icariin on the Human Dental Pulp Cells" Medicina 58, no. 3: 434. https://doi.org/10.3390/medicina58030434
APA StyleLiu, G., Yang, Y., Min, K.-S., Lee, B.-N., & Hwang, Y.-C. (2022). Odontogenic Effect of Icariin on the Human Dental Pulp Cells. Medicina, 58(3), 434. https://doi.org/10.3390/medicina58030434






