Long-Term Follow-Up of Survivors of Extracorporeal Life Support Therapy for Cardiogenic Shock: Are They Really Survivors?
Abstract
:1. Introduction
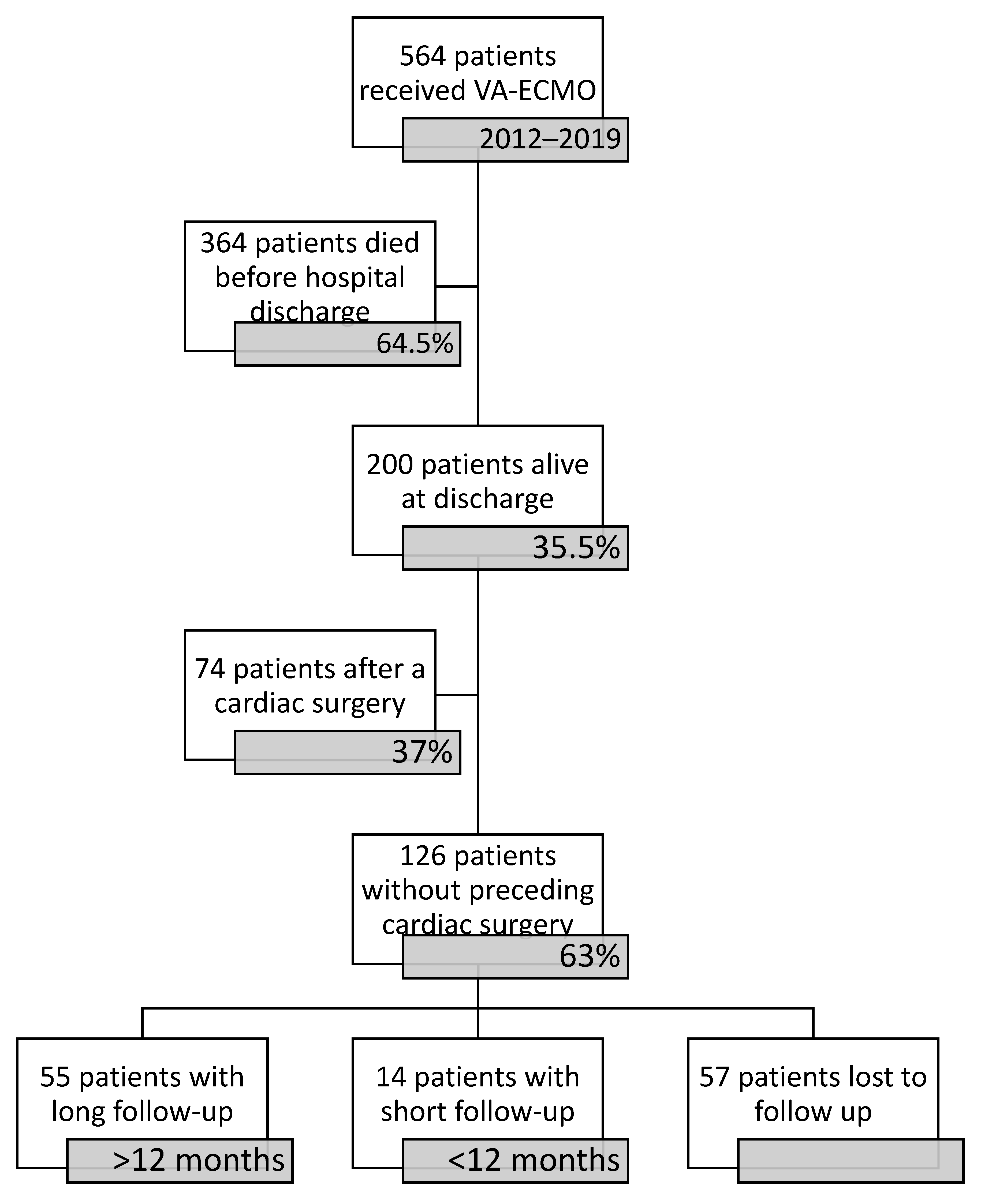
2. Materials and Methods
2.1. Patient Selection
2.2. Statistical Analysis
2.3. Ethical Approval
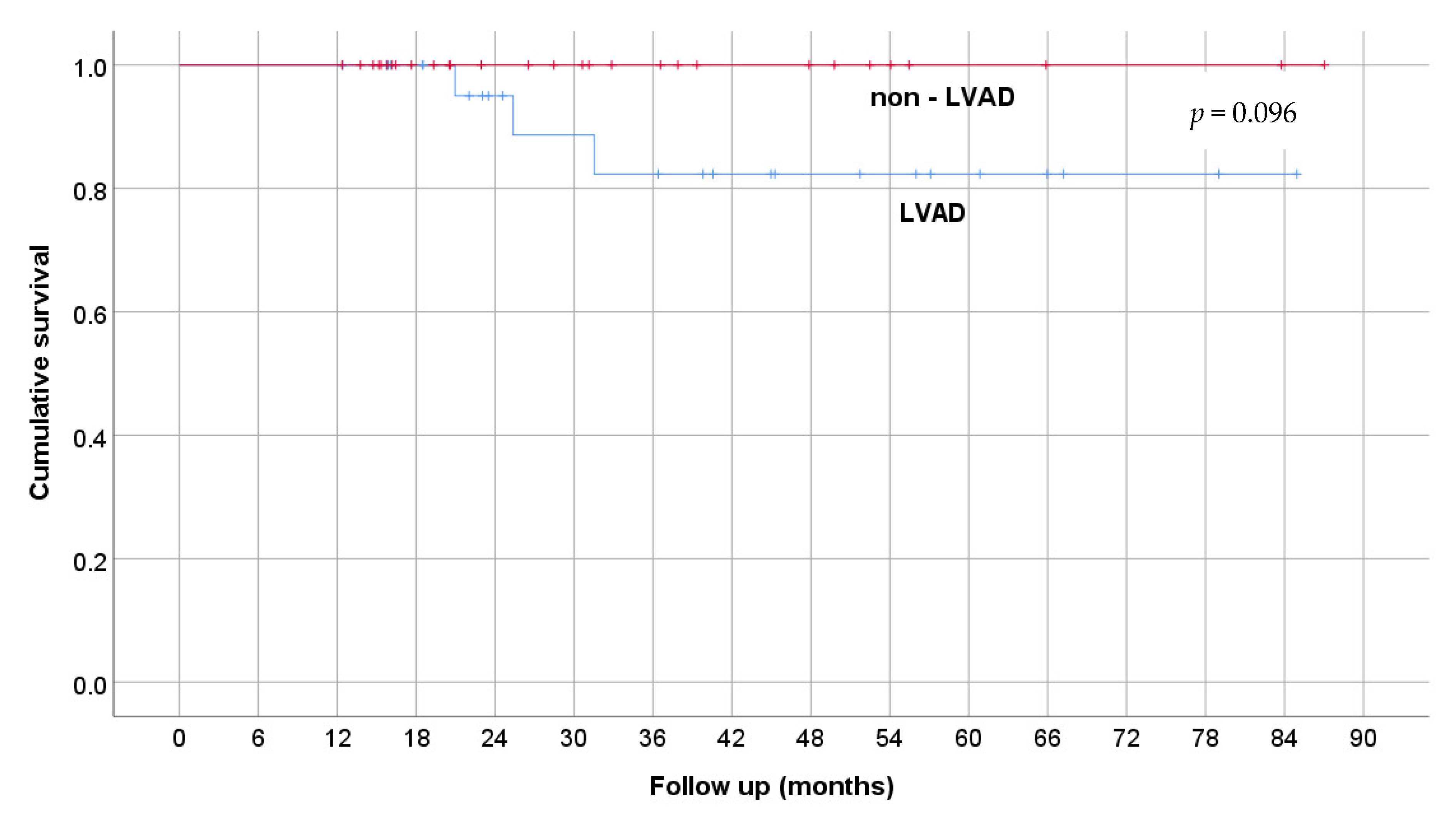
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- El Sibai, R.; Bachir, R.; El Sayed, M. Outcomes in Cardiogenic Shock Patients with Extracorporeal Membrane Oxygenation Use: A Matched Cohort Study in Hospitals across the United States. Biomed Res. Int. 2018, 2018, 2428648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fux, T.; Holm, M.; Corbascio, M.; Lund, L.H.; van der Linden, J. VA-ECMO Support in Nonsurgical Patients With Refractory Car-diogenic Shock: Pre-Implant Outcome Predictors. Artif. Organs 2019, 43, 132–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakaramakkil, M.J.; Sivathasan, C. ECMO and Short-term Support for Cardiogenic Shock in Heart Failure. Curr. Cardiol. Rep. 2018, 20, 87. [Google Scholar] [CrossRef] [PubMed]
- Boeken, U.; Assmann, A.; Klotz, S.; Born, F.; Rieth, A.; Schmid, C. (Eds.) Mechanische Unterstützung im Akuten Kreislaufversagen, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Conrad, S.A.; Broman, L.M.; Taccone, F.S.; Lorusso, R.; Malfertheiner, M.V.; Pappalardo, F.; Di Nardo, M.; Belliato, M.; Grazioli, L.; Barbaro, R.P.; et al. The Extracorporeal Life Support Organization Maastricht Treaty for Nomenclature in Extracorporeal Life Support. A Position Paper of the Extracorporeal Life Support Organization. Am. J. Respir. Crit. Care Med. 2018, 198, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Burrell, A.; Roberts, L.; Bailey, M.; Sheldrake, J.; Rycus, P.T.; Hodgson, C.; Scheinkestel, C.; Cooper, D.J.; Thiagarajan, R.R.; et al. Predicting survival after ECMO for refractory cardiogenic shock: The survival after veno-arterial-ECMO (SAVE)-score. Eur. Heart J. 2015, 36, 2246–2256. [Google Scholar] [CrossRef] [PubMed]
- Thiagarajan, R.R.; Barbaro, R.P.; Rycus, P.T.; Mcmullan, D.M.; Conrad, S.A.; Fortenberry, J.D.; Paden, M.L. Extracorporeal Life Support Organization Registry International Report 2016. ASAIO J. 2017, 63, 60–67. [Google Scholar] [CrossRef] [PubMed]
- El Sibai, R.; Bachir, R.; El Sayed, M. ECMO use and mortality in adult patients with cardiogenic shock: A retrospective observational study in U.S. hospitals. BMC Emerg. Med. 2018, 18, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blumenstein, J.; Leick, J.; Liebetrau, C.; Kempfert, J.; Gaede, L.; Groß, S.; Krug, M.; Berkowitsch, A.; Nef, H.; Rolf, A.; et al. Extracorporeal life support in cardiovascular patients with observed refractory in-hospital cardiac arrest is associated with favourable short and long-term outcomes: A propensi-ty-matched analysis. Eur. Heart J. Acute Cardiovasc. Care 2016, 5, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Muller, G.; Flecher, E.; Lebreton, G.; Luyt, C.-E.; Trouillet, J.-L.; Bréchot, N.; Schmidt, M.; Mastroianni, C.; Chastre, J.; Leprince, P.; et al. The ENCOURAGE mortality risk score and analysis of long-term outcomes after VA-ECMO for acute myocardial infarction with cardiogenic shock. Intensive Care Med. 2016, 42, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.; Khalpey, Z.; Smith, R.; Burkhoff, D.; Kociol, R.D. Venoarterial Extracorporeal Membrane Oxygenation for Cardiogenic Shock and Cardiac Arrest. Circ. Heart Fail. 2018, 11, e004905. [Google Scholar] [CrossRef] [PubMed]
- Berger, R.; Hamdoun, H.; Sandoval Boburg, R.; Radwan, M.; Acharya, M.; Waeschle, R.M.; Schlensak, C.; Popov, A.; Risteski, P. Quality of Life Following Urgent LVAD Implantation for ECMO Therapy in Cardiogenic Shock: A Long-Term Follow-Up. Medicina 2021, 57, 747. [Google Scholar] [CrossRef] [PubMed]
- Truby, L.; Mundy, L.; Kalesan, B.; Kirtane, A.; Colombo, P.C.; Takeda, K.; Fukuhara, S.; Naka, Y.; Takayama, H. Contemporary Outcomes of Venoarterial Extracorporeal Membrane Oxygenation for Refractory Cardiogenic Shock at a Large Tertiary Care Center. ASAIO J. 2015, 61, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Burrell, A.J.C.; Pellegrino, V.A.; Wolfe, R.; Wong, W.K.; Cooper, D.J.; Kaye, D.M.; Pilcher, D.V. Long-term survival of adults with cardi-ogenic shock after venoarterial extracorporeal membrane oxygenation. J. Crit. Care 2015, 30, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Ørbo, M.C.; Karlsen, S.F.; Pedersen, E.P.; Hermansen, S.E.; Rønning, P.B.; Nergaard, K.A.; Naesheim, T.; Myrmel, T. Health-related quality of life after extracorporeal membrane oxygenation: A single centre’s experience. ESC Heart Fail. 2019, 6, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Camboni, D.; Philipp, A.; Rottenkolber, V.; Zerdzitzki, M.; Holzamer, A.; Floerchinger, B.; Lunz, D.; Mueller, T.; Schmid, C.; Diez, C. Long-term survival and quality of life after extracorporeal life support: A 10-year report. Eur. J. Cardiothorac. Surg. 2017, 52, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Beurtheret, S.; Mordant, P.; Paoletti, X.; Marijon, E.; Celermajer, D.S.; Léger, P.; Pavie, A.; Combes, A.; Leprince, P. Emergency circulatory support in refractory cardiogenic shock patients in remote institutions: A pilot study (the cardiac-RESCUE program). Eur. Heart J. 2013, 34, 112–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Age (Years) Male | 58.2 ± 12.9 42 (76.4) |
|---|---|
| Diagnoses associated with cardiogenic shock: | |
| Acute myocardial infarction | 31 (56.4) |
| Myocarditis | 4 (7.3) |
| Pulmonary embolism | 2 (3.6) |
| Refractory ventricular VT/VF | 19 (34.5) |
| Sepsis | 2 (3.6) |
| Chronic heart failure | 22 (40) |
| Complication of cardiac intervention | 2 (3.6) |
| Other indications | 2 (3.6) |
| Myocardial function before implantation: | |
| Left heart failure | 35 (63.6) |
| Right heart failure | 13 (23.6) |
| LV ejection fraction (%) | 17.7 ± 10.1 |
| Pre-ECLS cardiac arrest | 34 (61.8) |
| CPR time (min) | 28.5 ± 22.8 |
| Implantation under CPR | 8 (14.4) |
| Pre-ECLS IABP/Impella® | 2 (3.6) |
| Implantation out of centre | 13 (23.6) |
| Duration of support (days) | 10.9 ± 7.7 |
| ICU stay (days) | 38.2 ± 29.9 |
| Hospital stay (days) | 49.9 ± 30.5 |
| Complications while on support: | |
| Bleeding requiring invasive intervention | 21 (38.2) |
| NOMI | 3 (5.5) |
| Stroke | 2 (3.6) |
| LV Thrombus | 2 (3.6) |
| Limb ischaemia | 1 (1.8) |
| Management: | |
| LVAD-implantation | 26 (47.3) |
| Vascular surgery to treat complications | 17 (30.9) |
| PCI | 11 (20) |
| Other cardiac surgery | 7 (12.7) |
| Impella® | 7 (12.7) |
| Temporary RVAD | 5 (9.1) |
| Pacemaker | 4 (7.3) |
| IABP | 4 (7.3) |
| Catheter ablation | 2 (3.6) |
| VV-ECMO | 2 (3.6) |
| Re-VA-ECMO | 1 (1.8) |
| Follow-up duration (months) | 36 ± 20.9 |
| Deaths | 3 (5.5) |
| Survival to death (months) | 26 ± 5.3 |
| Renal failure | 11 (20) |
| Wound infection | 10 (18.2) |
| Cerebrovascular accident | 6 (10.9) |
| New episode of cardiogenic shock | 1 (1.8) |
| Myocardial infarction | 1 (1.8) |
| Pulmonary embolism | 1 (1.8) |
| Hypoxic neurologic injury | 1 (1.8) |
| Depression | 1 (1.8) |
| Implantation of pacemaker | 4 (7.3) |
| PCI | 2 (3.6) |
| MitraClip® | 2 (3.6) |
| Catheter ablation | 1 (1.8) |
| Heart transplantation | 1 (1.8) |
| Other cardiac surgery | 1 (1.8) |
| LVAD-explantation | 4 (7.3) |
| Time on LVAD (months) | 18.6 ± 11.5 |
| LVAD, n = 26 | Non-LVAD, n = 29 | p Value | |
|---|---|---|---|
| Diagnosis: | |||
| Acute myocardial infarction | 14 | 17 | 0.372 |
| Myocarditis | 3 | 1 | 0.253 |
| Refractory ventricular VT/VF | 8 | 11 | 0.581 |
| Chronic heart failure | 18 | 4 | 0.001 * |
| CPR: | |||
| Pre-ECMO cardiac arrest | 10 (38.5) | 16 (55.2) | 0.01 * |
| CPR time (min) | 10 (1–60) | 25 (1–75) | 0.122 |
| Follow up after VA-ECMO implantation: | |||
| Time on VA-ECMO (days) | 13 ± 8.1 | 9.7 ± 6.3 | 0.096 |
| ICU stay (days) | 42.7 (28.4) | 38.25 (29.9) | 0.523 |
| Hospital stay (days) | 60.4 ± 38.5 | 45.24 ± 29.3 | 0.105 |
| Follow-up (months) | 38.4 ± 21.2 | 33.8 ± 20.7 | 0.281 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berger, R.; Nemeth, A.; Sandoval Boburg, R.; Vöhringer, L.; Lausberg, H.F.; Acharya, M.; Schlensak, C.; Popov, A.-F. Long-Term Follow-Up of Survivors of Extracorporeal Life Support Therapy for Cardiogenic Shock: Are They Really Survivors? Medicina 2022, 58, 427. https://doi.org/10.3390/medicina58030427
Berger R, Nemeth A, Sandoval Boburg R, Vöhringer L, Lausberg HF, Acharya M, Schlensak C, Popov A-F. Long-Term Follow-Up of Survivors of Extracorporeal Life Support Therapy for Cardiogenic Shock: Are They Really Survivors? Medicina. 2022; 58(3):427. https://doi.org/10.3390/medicina58030427
Chicago/Turabian StyleBerger, Rafal, Attila Nemeth, Rodrigo Sandoval Boburg, Louise Vöhringer, Henning Florian Lausberg, Metesh Acharya, Christian Schlensak, and Aron-Frederik Popov. 2022. "Long-Term Follow-Up of Survivors of Extracorporeal Life Support Therapy for Cardiogenic Shock: Are They Really Survivors?" Medicina 58, no. 3: 427. https://doi.org/10.3390/medicina58030427
APA StyleBerger, R., Nemeth, A., Sandoval Boburg, R., Vöhringer, L., Lausberg, H. F., Acharya, M., Schlensak, C., & Popov, A.-F. (2022). Long-Term Follow-Up of Survivors of Extracorporeal Life Support Therapy for Cardiogenic Shock: Are They Really Survivors? Medicina, 58(3), 427. https://doi.org/10.3390/medicina58030427







