Informed Consent for Endoscopic Biliary Drainage: Time for a New Paradigm
Abstract
:1. Introduction
2. From General Principles to Our Starting Point
3. Mechanisms of Action
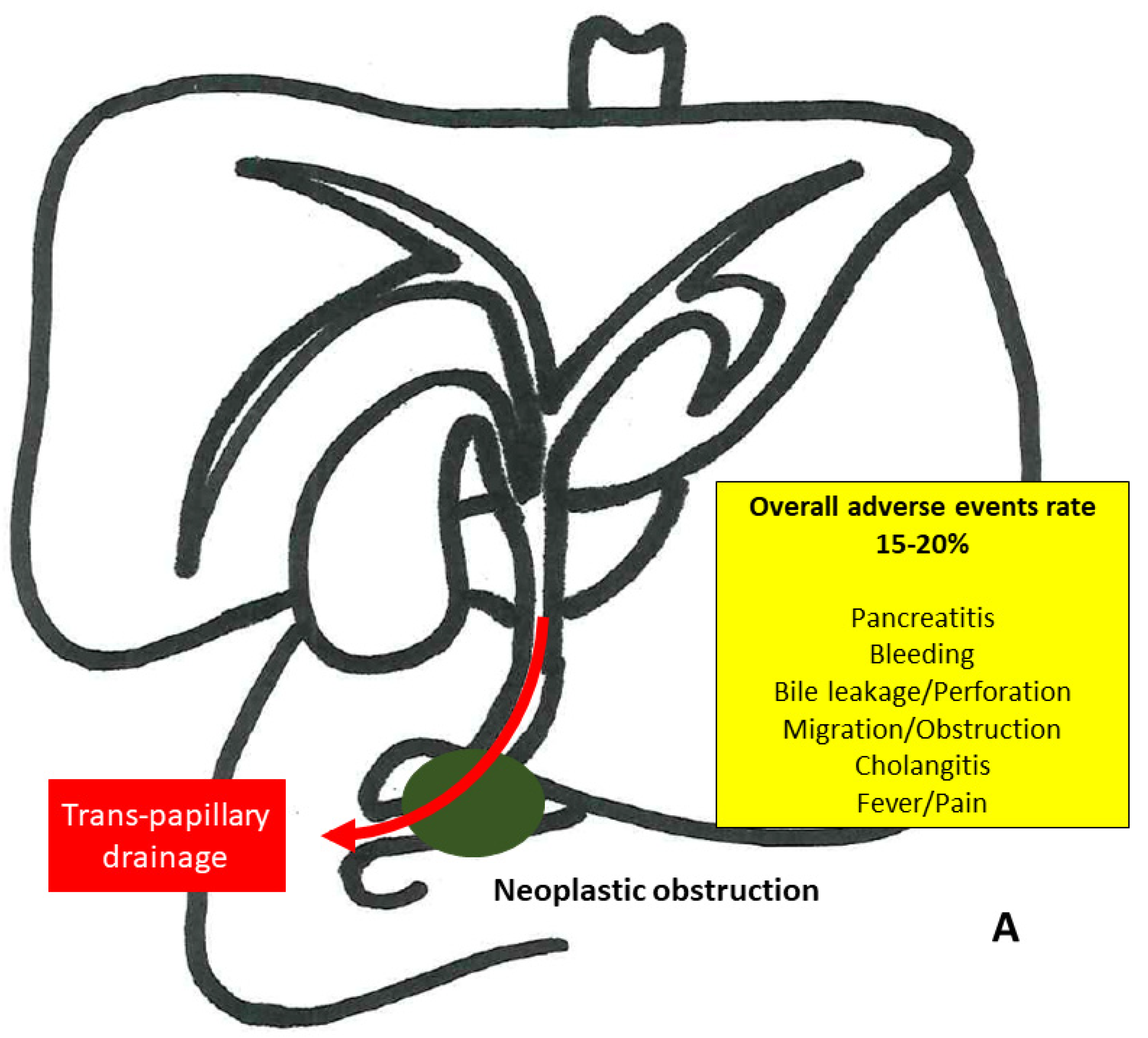
3.1. Endoscopic Retrograde Cholangiopancreatography (ERCP)
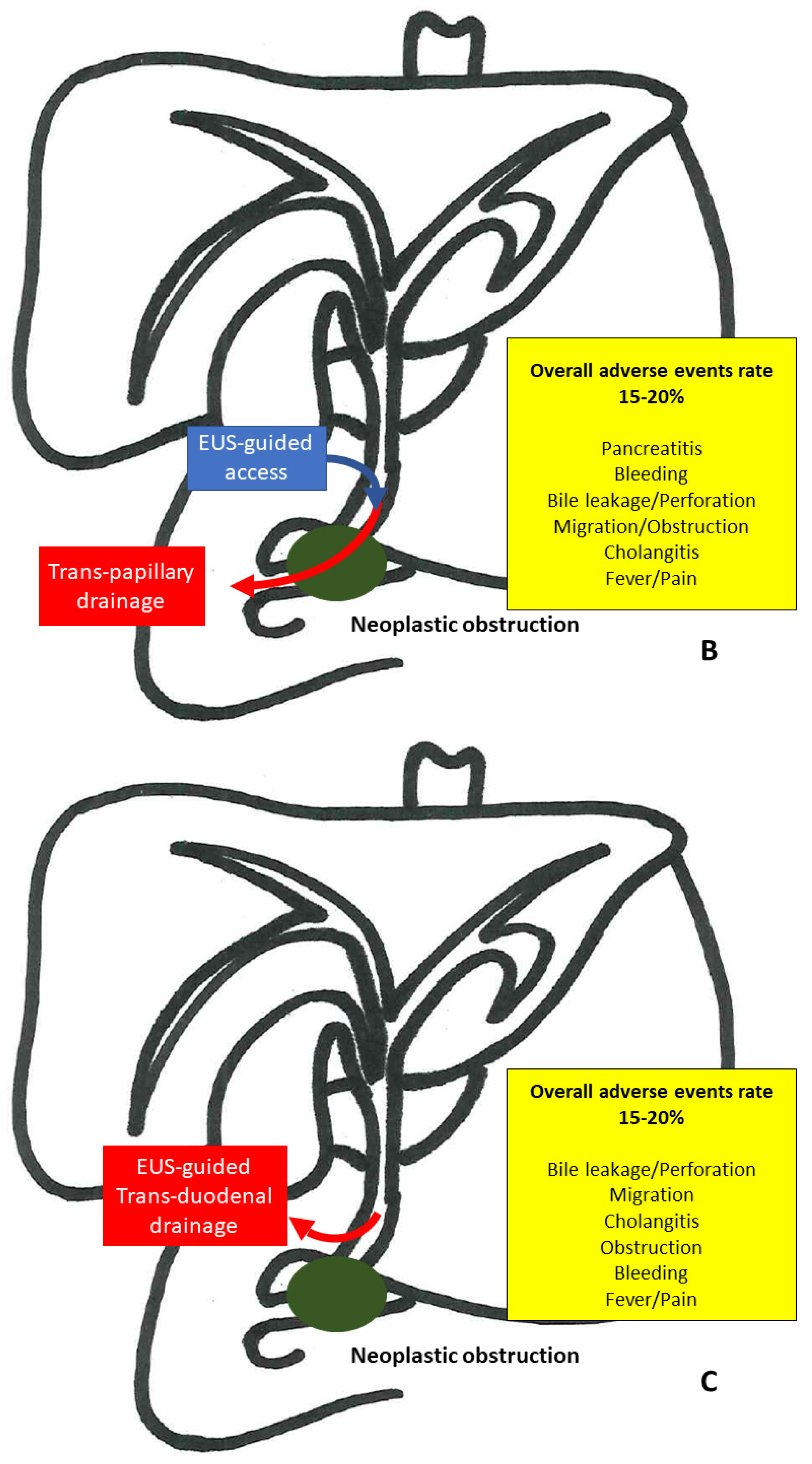
3.2. EUS-Guided Rendezvous
3.3. EUS-Guided Choledochoduodenostomy
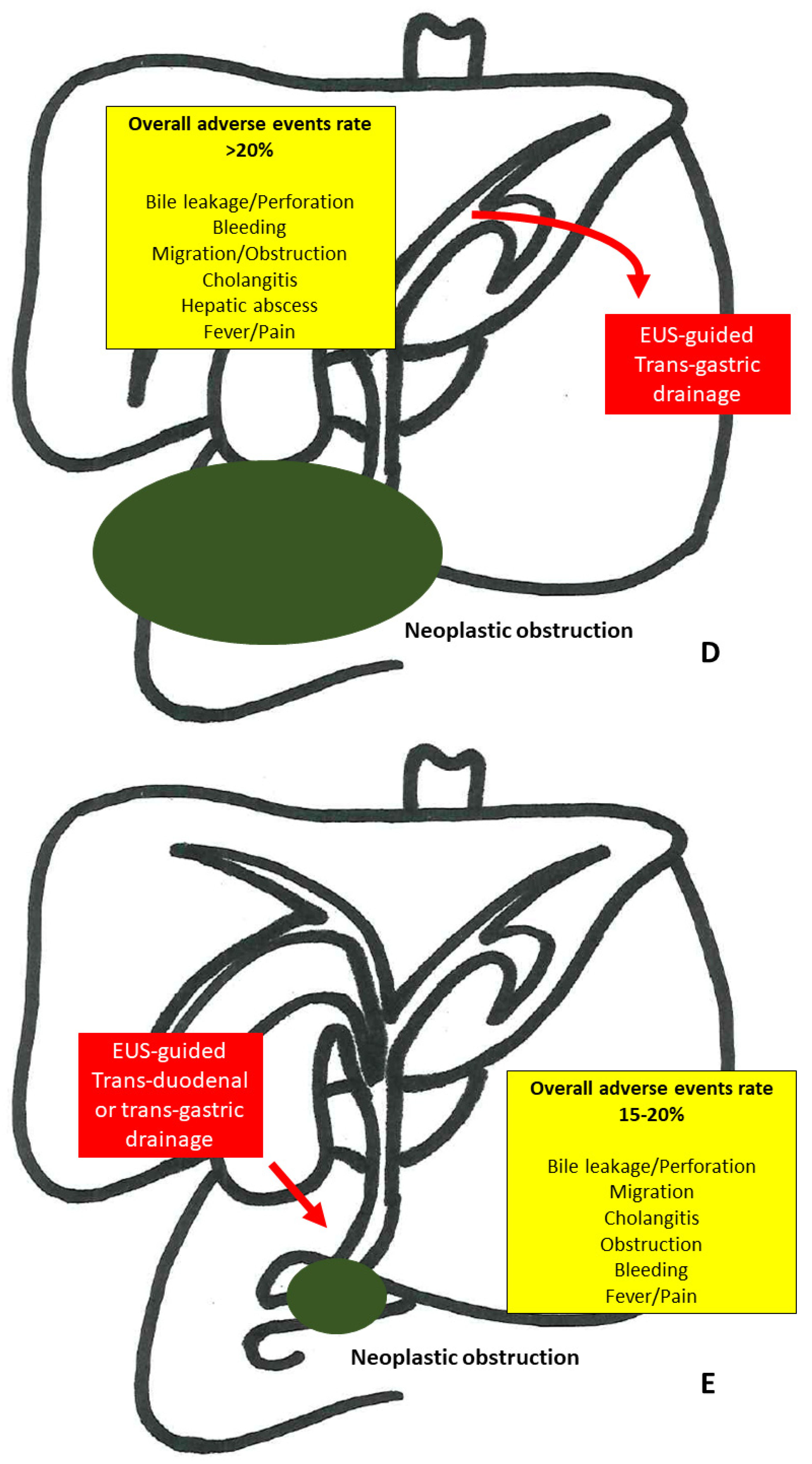
3.4. EUS-Guided Hepaticogastrostomy
3.5. EUS-Guided Gallbladder Drainage
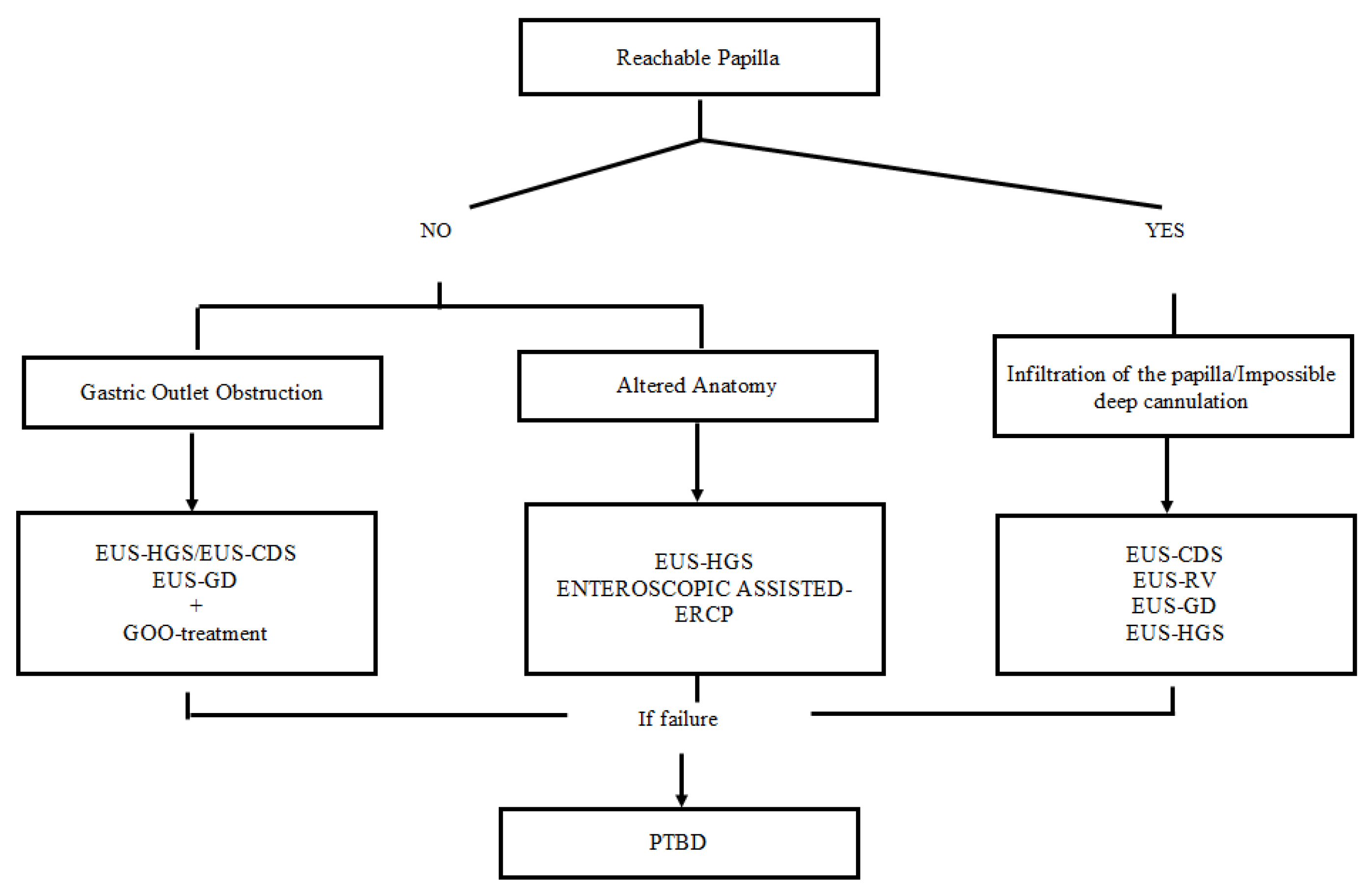
4. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Enochsson, L.; Swahn, F.; Arnelo, U.; Nilsson, M.; Löhr, M.; Persson, G. Nationwide, population-based data from 11,074 ERCP procedures from the Swedish Registry for Gallstone Surgery and ERCP. Gastrointest. Endosc. 2010, 72, 1175–1184.e1. [Google Scholar] [CrossRef] [PubMed]
- DeBenedet, A.T.; Elmunzer, B.J.; McCarthy, S.T.; Elta, G.H.; Schoenfeld, P.S. Intraprocedural quality in endoscopic retrograde cholangiopancreatography: A meta-analysis. Am. J. Gastroenterol. 2013, 108, 1696–1704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, C.; Nietert, P.J.; Cotton, P.B.; Lackland, D.T.; Romagnuolo, J. Predicting native papilla biliary cannulation success using a multinational Endoscopic Retrograde Cholangiopancreatography (ERCP) Quality Network. BMC Gastroenterol. 2013, 13, 147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balik, E.; Eren, T.; Keskin, M.; Ziyade, S.; Bulut, T.; Buyukuncu, Y.; Yamaner, S. Parameters That May Be Used for Predicting Failure during Endoscopic Retrograde Cholangiopancreatography. J. Oncol. 2013, 2013, 201681. [Google Scholar] [CrossRef]
- Fugazza, A.; Troncone, E.; Amato, A.; Tarantino, I.; Iannone, A.; Donato, G.; D’Amico, F.; Mogavero, G.; Amata, M.; Fabbri, C.; et al. Difficult biliary cannulation in patients with distal malignant biliary obstruction: An underestimated problem? Dig. Liver Dis. 2021, 4. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Kung, J.; Liu, Y.; Tse, A.; Datta, A.; Singh, I.; Eysselein, V.E.; Reicher, S. Use of double wireguided technique and transpancreatic papillary septotomy in difficult ERCP: 4-year experience. Endosc. Int. Open 2016, 4, E1107–E1110. [Google Scholar]
- Fugazza, A.; Anderloni, A.; Paduano, D.; Badalamenti, M.; Maselli, R.; Carrara, S.; Gabbiadini, R.; Colombo, M.; Spadaccini, M.; Cappello, A.; et al. Underwater cap-assisted endoscopic retrograde cholangiopancreatography in patients with surgically altered anatomy: A pilot study. Endoscopy 2021, 53, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Nennstiel, S.; Weber, A.; Frick, G.; Haller, B.; Meining, A.; Schmid, R.M.; Neu, B. Drainage-related Complications in Percutaneous Transhepatic Biliary Drainage: An Analysis Over 10 Years. J. Clin. Gastroenterol. 2015, 49, 764–770. [Google Scholar] [CrossRef]
- Baniya, R.; Upadhaya, S.; Madala, S.; Subedi, S.C.; Shaik Mohammed, T.; Bachuwa, G. Endoscopic ultrasound-guided biliary drainage versus percutaneous transhepatic biliary drainage after failed endoscopic retrograde cholangiopancreatography: A meta-analysis. Clin. Exp. Gastroenterol. 2017, 10, 67–74. [Google Scholar] [CrossRef] [Green Version]
- Giovannini, M.; Moutardier, V.; Pesenti, C.; Bories, E.; Lelong, B.; Delpero, J.R. Endoscopic ultrasoundguided bilioduodenal anastomosis: A new technique for biliary drainage. Endoscopy 2001, 33, 898–900. [Google Scholar] [CrossRef]
- Bapaye, A.; Dubale, N.; Aher, A. Comparison of endosonography-guided vs. percutaneous biliary stenting when papilla is inaccessible for ERCP. United Eur. Gastroenterol. J. 2013, 1, 285–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khashab, M.A.; Valeshabad, A.K.; Afghani, E.; Singh, V.K.; Kumbhari, V.; Messallam, A.; Saxena, P.; El Zein, M.; Lennon, A.M.; Canto, M.I.; et al. A comparative evaluation of EUS-guided biliary drainage and percutaneous drainage in patients with distal malignant biliary obstruction and failed ERCP. Dig. Dis. Sci. 2015, 60, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Choi, J.H.; Park, H.; Song, T.J.; Kim, D.U.; Paik, W.H.; Hwangbo, Y.; Lee, S.S.; Seo, D.W.; Lee, S.K.; et al. Similar Efficacies of Endoscopic Ultrasound-guided Transmural and Percutaneous Drainage for Malignant Distal Biliary Obstruction. Clin. Gastroenterol. Hepatol. 2016, 14, 1011–1019.e3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moole, H.; Bechtold, M.L.; Forcione, D.; Puli, S.R. A meta-analysis and systematic review: Success of endoscopic ultrasound guided biliary stenting in patients with inoperable malignant biliary strictures and a failed ERCP. Medicine 2017, 96, e5154. [Google Scholar] [CrossRef] [PubMed]
- Sharaiha, R.Z.; Khan, M.A.; Kamal, F.; Tyberg, A.; Tombazzi, C.R.; Ali, B.; Tombazzi, C.; Kahaleh, M. Efficacy and safety of EUS-guided biliary drainage in comparison with percutaneous biliary drainage when ERCP fails: A systematic review and meta-analysis. Gastrointest. Endosc. 2017, 85, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Vila, J.J.; Fernández-Urién, I.; Carrascosa, J. EUS and ERCP: A rationale categorization of a productive partnership. Endosc. Ultrasound 2021, 10, 25–32. [Google Scholar] [CrossRef]
- Aagaard, L.; Kristensen, K. Off-label and unlicensed prescribing in Europe: Implications for patients’ informed consent and liability. Int. J. Clin. Pharm. 2018, 40, 509–512. [Google Scholar] [CrossRef]
- James, J.T.; Eakins, D.J.; Scully, R.R. Informed consent, shared-decision making and a reasonable patient’s wishes based on a cross-sectional, national survey in the USA using a hypothetical scenario. BMJ Open 2019, 9, e028957. [Google Scholar] [CrossRef] [Green Version]
- Kinnersley, P.; Phillips, K.; Savage, K.; Kelly, M.J.; Farrell, E.; Morgan, B.; Whistance, R.; Lewis, V.; Mann, M.K.; Stephens, B.L.; et al. Interventions to promote informed consent for patients undergoing surgical and other invasive healthcare procedures. Cochrane Database Syst. Rev. 2013, 7, CD009445. [Google Scholar] [CrossRef]
- Hanson, M.; Pitt, D. Informed consent for surgery: Risk discussion and documentation. Can. J. Surg. 2017, 60, 69–70. [Google Scholar] [CrossRef]
- Parzeller, M.; Wenk, M.; Zedler, B.; Rothschild, M. Aufklärung und Einwilligung bei ärztlichen Eingriffen. Dtsch Arztebl Int. 2007, 104, 576. [Google Scholar]
- Binda, C.; Spadaccini, M.; Cugia, L.; Anderloni, A. EUS and ERCP partnership. Endosc. Ultrasound 2021. ahead of print. [Google Scholar]
- Dumonceau, J.M.; Tringali, A.; Papanikolaou, I.S.; Blero, D.; Mangiavillano, B.; Schmidt, A.; Vanbiervliet, G.; Costamagna, G.; Devière, J.; García-Cano, J.; et al. Endoscopic biliary stenting: Indications, choice of stents, and results: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline—Updated October 2017. Endoscopy 2018, 50, 910–930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Merwe, S.W.; van Wanrooij, R.L.J.; Bronswijk, M.; Everett, S.; Lakhtakia, S.; Rimbas, M.; Hucl, T.; Kunda, R.; Badaoui, A.; Law, R.; et al. Therapeutic endoscopic ultrasound: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2022, 54, 185–205. [Google Scholar] [CrossRef] [PubMed]
- Vila, J.J.; Kutz, M.; Goñi, S.; Ostiz, M.; Amorena, E.; Prieto, C.; Rodriguez, C.; Fernandez-Urien, I.; Jimenez, F.J. Endoscopic and anesthetic feasibility of EUS and ERCP combined in a single session versus two different sessions. World J. Gastrointest. Endosc. 2011, 3, 57. [Google Scholar] [CrossRef]
- Gornals, J.B.; Moreno, R.; Castellote, J.; Loras, C.; Barranco, R.; Catala, I.; Xiol, X.; Fabregat, J.; Corbella, X. Single session endosonography and endoscopic retrograde cholangiopancreatography for biliopancreatic diseases is feasible, effective and cost beneficial. Dig. Liver Dis. 2013, 45, 578–583. [Google Scholar] [CrossRef]
- Chu, Y.L.; Wang, X.F.; Gao, X.Z.; Qiao, X.-L.; Liu, F.; Yu, S.-Y.; Zhang, J. Endoscopic ultrasonography in tandem with endoscopic retrograde cholangiopancreatography in the management of suspected distal obstructive jaundice. Eur. J. Gastroenterol. Hepatol. 2013, 25, 455. [Google Scholar] [CrossRef]
- Kawakubo, K.; Kawakami, H.; Kuwatani, M.; Haba, S.; Kudo, T.; Abe, Y.; Kawahata, S.; Onodera, M.; Ehira, N.; Yamato, H.; et al. Safety and utility of single session endoscopic ultrasonography and endoscopic retrograde cholangiopancreatography for the evaluation of pancreatobiliary diseases. Gut Liver 2014, 8, 329–332. [Google Scholar] [CrossRef] [Green Version]
- Custer, R.L.; Scarcella, J.A.; Stewart, B.R. The Modified Delphi Technique—A Rotational Modification. J. Career Tech. Educ. 1999, 15. Available online: https://eric.ed.gov/?id=EJ590767 (accessed on 13 February 2022). [CrossRef]
- Fung, B.M.; Pitea, T.C.; Tabibian, J.H. Difficult Biliary Cannulation in Endoscopic Retrograde Cholangiopancreatography: An Overview of Advanced Techniques. Eur. Med. J. Hepatol. 2021, 1, 73–82. [Google Scholar] [CrossRef]
- Mallery, S.; Matlock, J.; Freeman, M.L. EUS-guided rendezvous drainage of obstructed biliary and pancreatic ducts: Report of 6 cases. Gastrointest. Endosc. 2004, 59, 100–107. [Google Scholar] [CrossRef]
- Matsubara, S.; Nakagawa, K.; Suda, K.; Otsuka, T.; Isayama, H.; Nakai, Y.; Oka, M.; Nagoshi, S. A Proposed Algorithm for Endoscopic Ultrasound-Guided Rendezvous Technique in Failed Biliary Cannulation. J. Clin. Med. 2020, 9, 3879. [Google Scholar] [CrossRef] [PubMed]
- Artifon, E.L.A.; Visconti, T.A.C.; Brunaldi, V.O. Choledochoduodenostomy: Outcomes and limitations. Endosc. Ultrasound 2019, 8, S72–S78. [Google Scholar] [CrossRef] [PubMed]
- Khashab, M.A.; Messallam, A.A.; Penas, I.; Nakai, Y.; Modayil, R.J.; De la Serna, C.; Hara, K.; El Zein, M.; Stavropoulos, S.N.; Perez-Miranda, M.; et al. International multicenter comparative trial of transluminal EUS-guided biliary drainage via hepatogastrostomy vs. choledochoduodenostomy approaches. Endosc. Int. Open 2016, 4, E175–E181. [Google Scholar] [CrossRef] [Green Version]
- Mussetto, A.; Fugazza, A.; Fuccio, L.; Triossi, O.; Repici, A.; Anderloni, A. Current uses and outcomes of lumen-apposing metal stents. Ann. Gastroenterol. 2018, 31, 535–540. [Google Scholar] [CrossRef]
- Binmoeller, K.F.; Shah, J. A novel lumen-apposing stent for transluminal drainage of nonadherent extraintestinal fluid collections. Endoscopy 2011, 43, 337–342. [Google Scholar] [CrossRef]
- Jacques, J.; Privat, J.; Pinard, F.; Fumex, F.; Valats, J.C.; Chaoui, A.; Cholet, F.; Godard, B.; Grandval, P.; Legros, R.; et al. Endoscopic ultrasound-guided choledochoduodenostomy with electrocautery-enhanced lumen-apposing stents: A retrospective analysis. Endoscopy 2019, 51, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Sobani, Z.A.; Ling, C.; Rustagi, T. Endoscopic Ultrasound-Guided Gallbladder Drainage. Dig. Dis. Sci. 2021, 66, 2154–2161. [Google Scholar] [CrossRef]
- Dollhopf, M.; Larghi, A.; Will, U.; Rimbaş, M.; Anderloni, A.; Sanchez-Yague, A.; Teoh, A.Y.B.; Kunda, R. EUS-guided gallbladder drainage in patients with acute cholecystitis and high surgical risk using an electrocautery-enhanced lumen-apposing metal stent device. Gastrointest. Endosc. 2017, 86, 636–643. [Google Scholar] [CrossRef]
- Lisotti, A.; Napoleon, B.; Fabbri, C.; Anderloni, A.; Linguerri, R.; Bacchilega, I.; Fusaroli, P. Treatment of acute cholecystitis in high-risk surgical patients. Systematic review of the literature according to the levels of evidence. Minerva Gastroenterol. 2021. [Google Scholar] [CrossRef]
- Auriemma, F.; Fugazza, A.; Colombo, M.; Spadaccini, M.; Repici, A.; Anderloni, A. Safety issues in endoscopy ultrasound-guided interventions using lumen apposing metal stents: A review of the literature. Minerva Gastroenterol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Cotton, P.B.; Eisen, G.M.; Aabakken, L.; Baron, T.H.; Hutter, M.M.; Jacobson, B.C.; Mergener, K.; Nemcek, A., Jr.; Petersen, B.T.; Petrini, J.L.; et al. A lexicon for endoscopic adverse events: Report of an ASGE workshop. Gastrointest. Endosc. 2010, 71, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zhu, J.; Xing, L.; Wang, Y.; Jin, Z.; Li, Z. Assessment of efficacy and safety of EUS-guided biliary drainage: A systematic review. Gastrointest. Endosc. 2016, 83, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Khashab, M.; Jovani, M.; Ichkhanian, Y.; Vosoughi, K. EUS-guided biliary drainage for postsurgical anatomy. Endosc. Ultrasound 2019, 8 (Suppl. S1), S57–S66. [Google Scholar] [CrossRef]
- Bang, J.Y.; Navaneethan, U.; Hasan, M.; Hawes, R.; Varadarajulu, S. Stent placement by EUS or ERCP for primary biliary decompression in pancreatic cancer: A randomized trial (with videos). Gastrointest. Endosc. 2018, 88, 9–17. [Google Scholar] [CrossRef] [Green Version]
- Isayama, H.; Nakai, Y.; Kawakami, H.; Ishiwatari, H.; Kitano, M.; Ito, Y.; Yasuda, I.; Kato, H.; Matsubara, S.; Irisawa, A.; et al. Prospective multicenter study of primary EUS guided choledochoduodenostomy using a covered metal stent. Endosc. Ultrasound 2019, 8, 111. [Google Scholar] [CrossRef]
- Kawakubo, K.; Kawakami, H.; Kuwatani, M.; Kubota, Y.; Kawahata, S.; Kubo, K.; Sakamoto, N. Endoscopic ultrasound guided choledochoduodenostomy vs. transpapillary stenting for distal biliary obstruction. Endoscopy 2016, 48, 164–169. [Google Scholar] [CrossRef]
- Park, J.K.; Woo, Y.S.; Noh, D.H.; Yang, J.-I.; Bae, S.Y.; Yun, H.S.; Lee, J.K.; Lee, K.T.; Lee, K.H. Efficacy of EUS guided and ERCP guided biliary drainage for malignant biliary obstruction: Prospective randomized controlled study. Gastrointest. Endosc. 2018, 88, 277–282. [Google Scholar] [CrossRef]
- Paik, W.H.; Lee, T.H.; Park, D.H.; Choi, J.H.; Kim, S.O.; Jang, S.; Uk, K.D.; Hyun, S.J.; Jun, S.T.; Kim, M.H. EUS guided biliary drainage versus ERCP for the primary palliation of malignant biliary obstruction: A multicenter randomized clinical trial. Am. J. Gastroenterol. 2018, 113, 987–997. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spadaccini, M.; Binda, C.; Fugazza, A.; Repici, A.; Tarantino, I.; Fabbri, C.; Cugia, L.; Anderloni, A.; on behalf of the Interventional Endoscopy & Ultra Sound (I-EUS) Group. Informed Consent for Endoscopic Biliary Drainage: Time for a New Paradigm. Medicina 2022, 58, 331. https://doi.org/10.3390/medicina58030331
Spadaccini M, Binda C, Fugazza A, Repici A, Tarantino I, Fabbri C, Cugia L, Anderloni A, on behalf of the Interventional Endoscopy & Ultra Sound (I-EUS) Group. Informed Consent for Endoscopic Biliary Drainage: Time for a New Paradigm. Medicina. 2022; 58(3):331. https://doi.org/10.3390/medicina58030331
Chicago/Turabian StyleSpadaccini, Marco, Cecilia Binda, Alessandro Fugazza, Alessandro Repici, Ilaria Tarantino, Carlo Fabbri, Luigi Cugia, Andrea Anderloni, and on behalf of the Interventional Endoscopy & Ultra Sound (I-EUS) Group. 2022. "Informed Consent for Endoscopic Biliary Drainage: Time for a New Paradigm" Medicina 58, no. 3: 331. https://doi.org/10.3390/medicina58030331
APA StyleSpadaccini, M., Binda, C., Fugazza, A., Repici, A., Tarantino, I., Fabbri, C., Cugia, L., Anderloni, A., & on behalf of the Interventional Endoscopy & Ultra Sound (I-EUS) Group. (2022). Informed Consent for Endoscopic Biliary Drainage: Time for a New Paradigm. Medicina, 58(3), 331. https://doi.org/10.3390/medicina58030331







