Enterococcus durans Cardiac Implantable Electronic Device Lead Infection and Review of Enterococcus durans Endocarditis Cases
Abstract
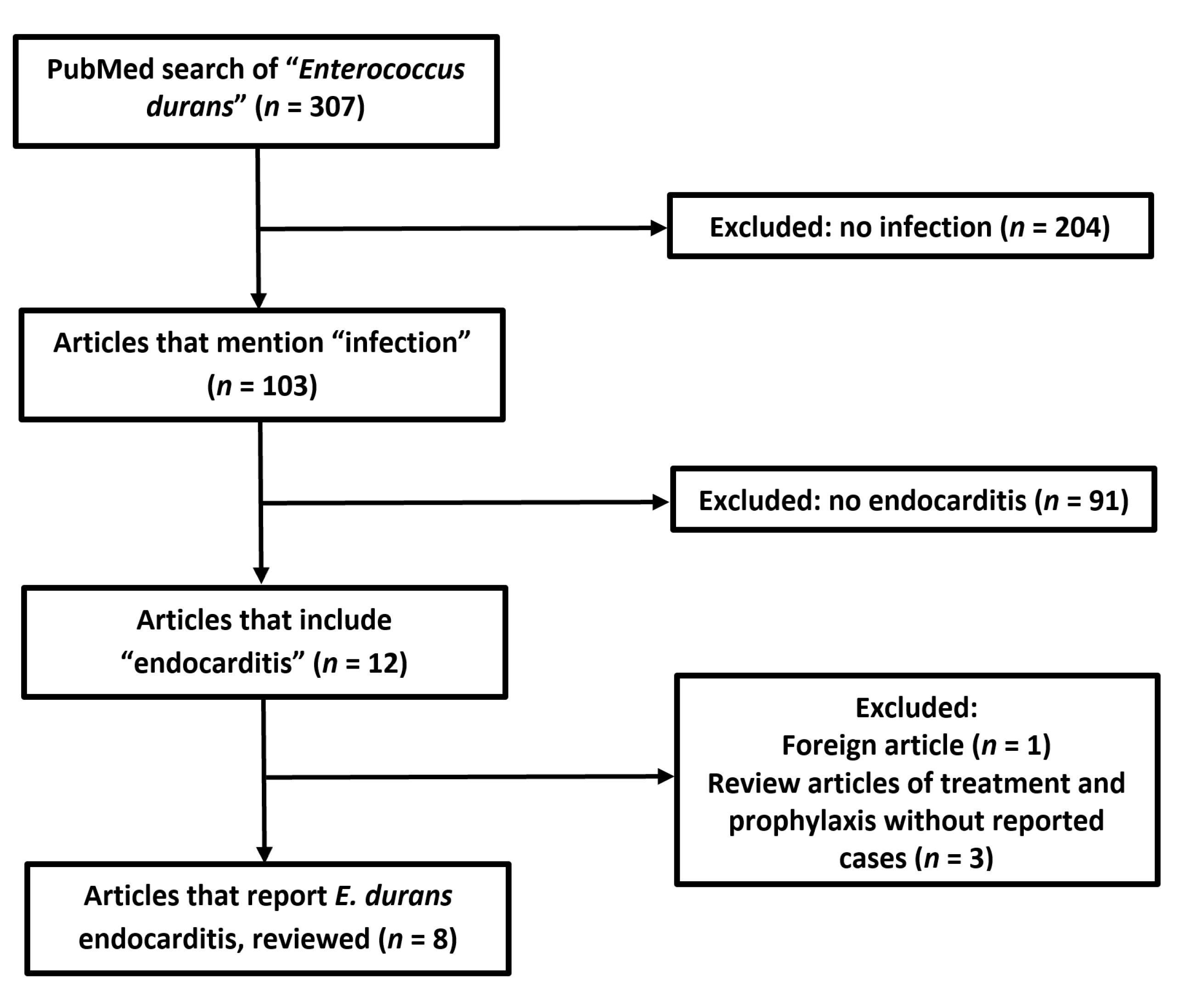
:1. Introduction
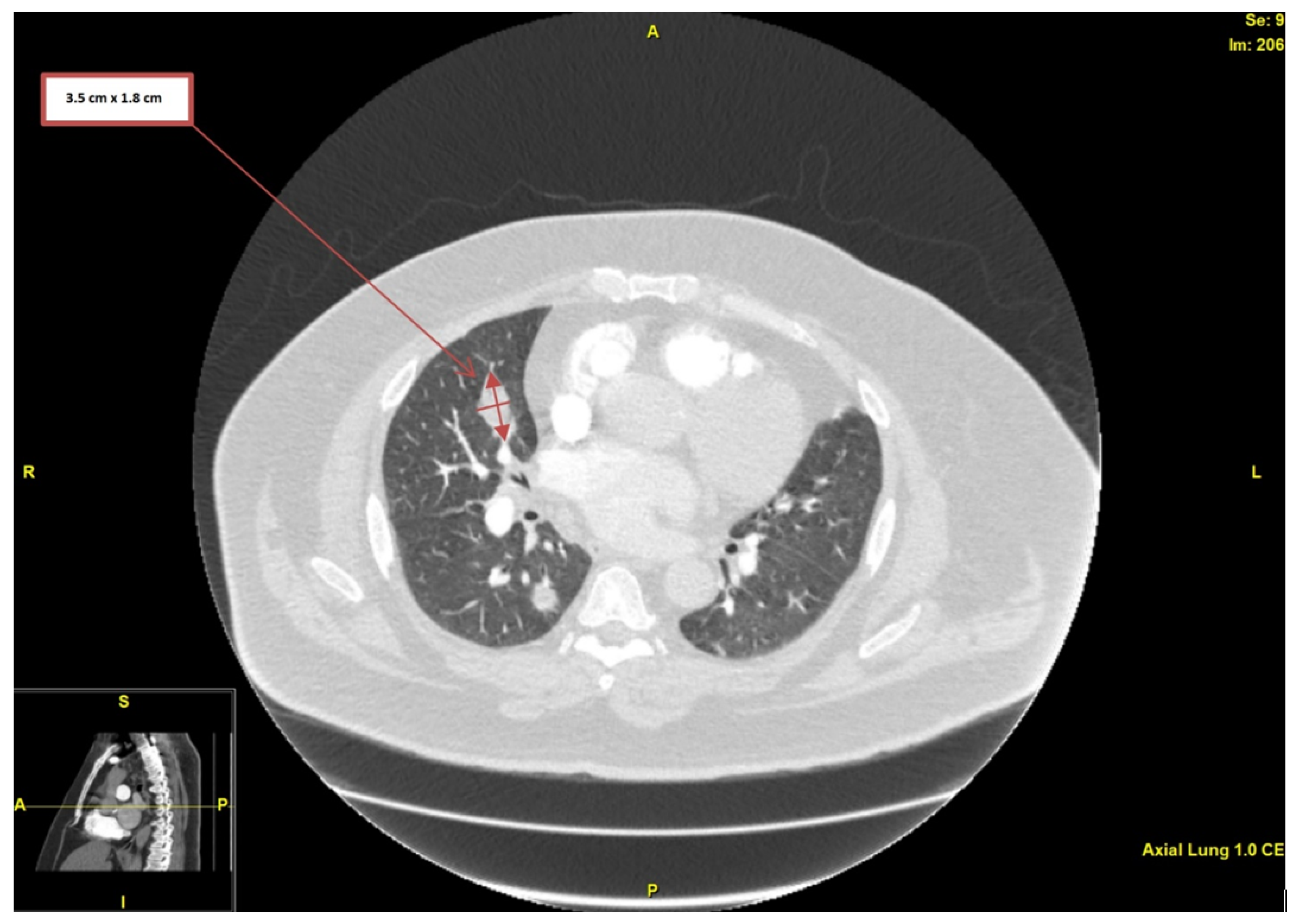
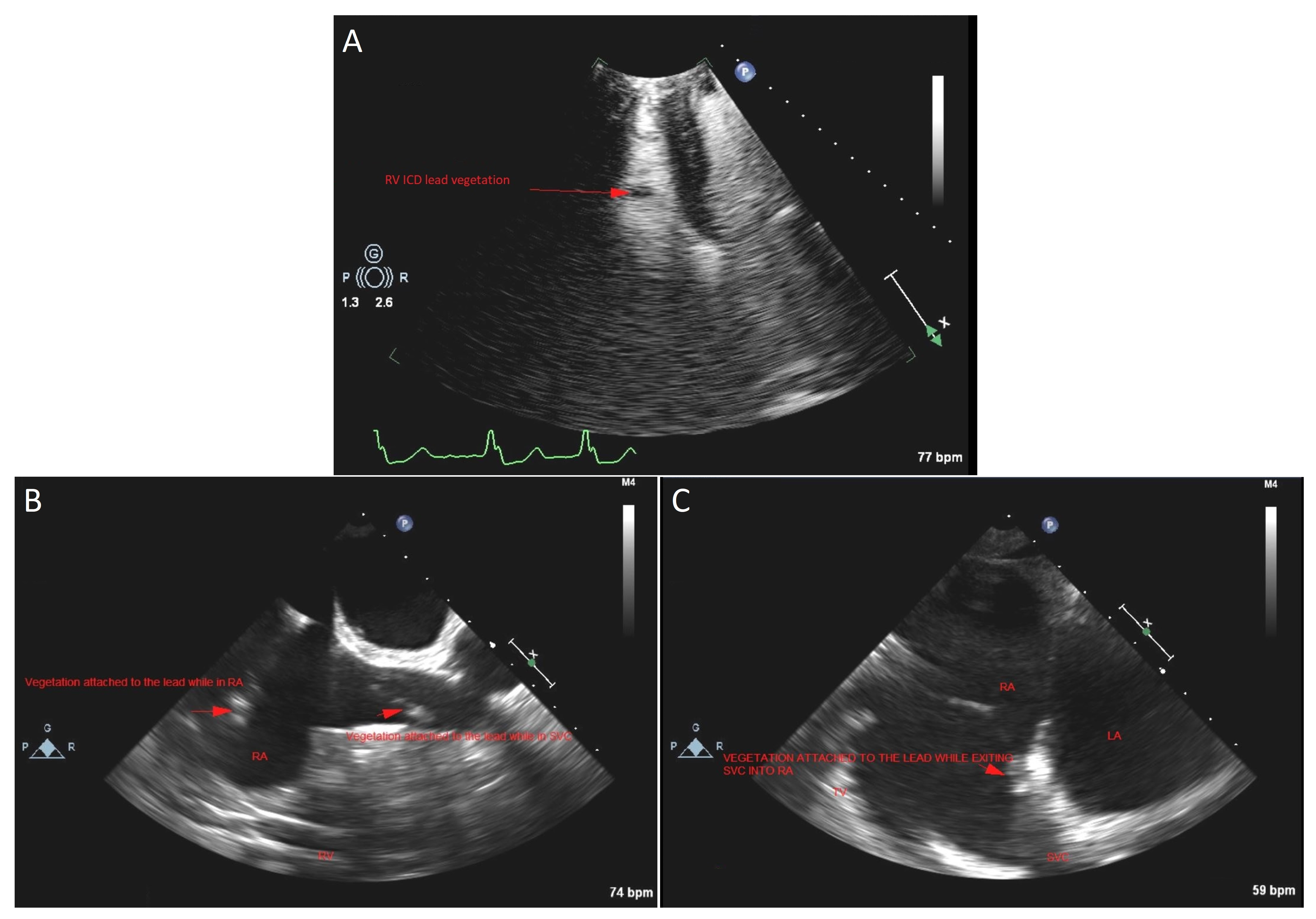
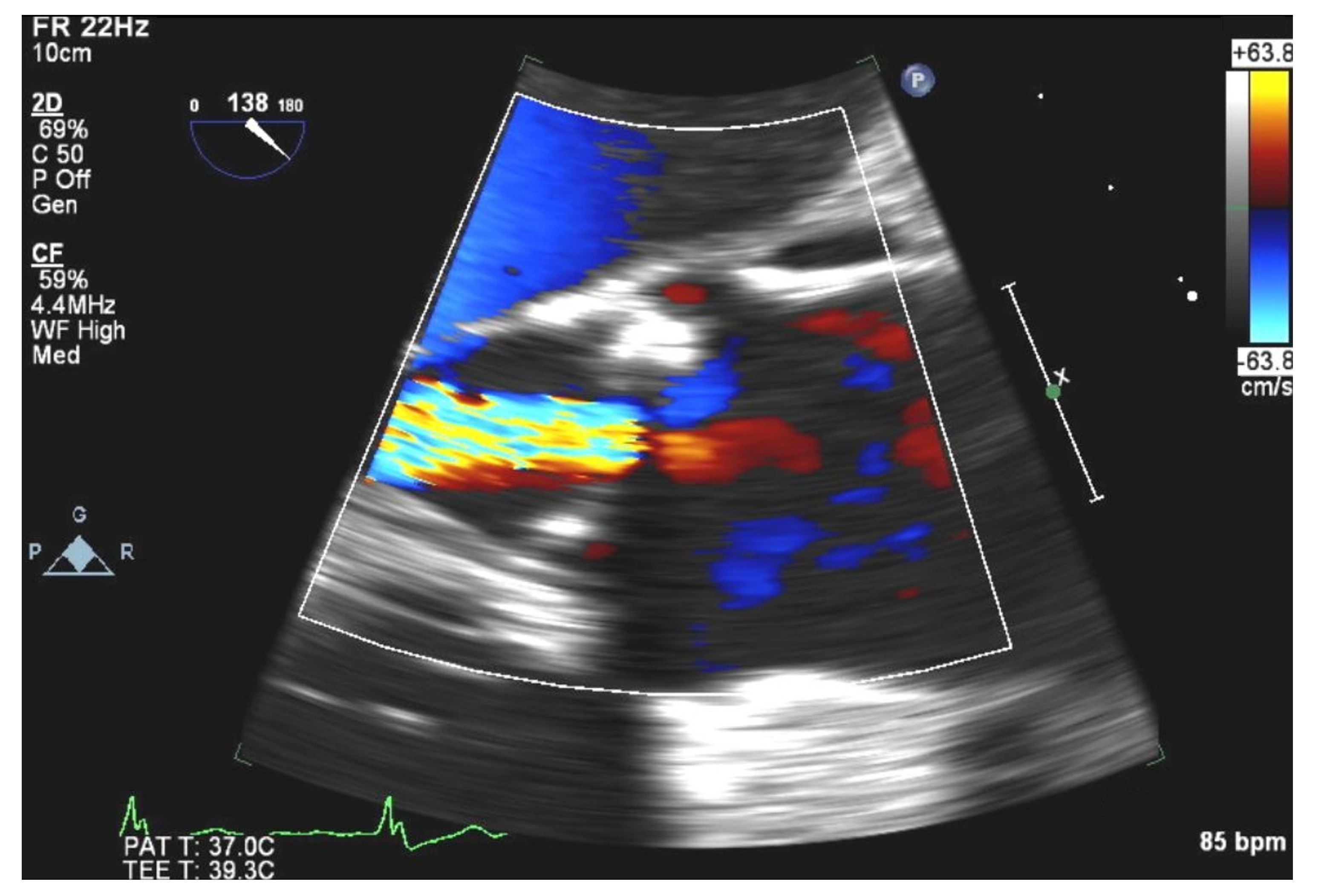
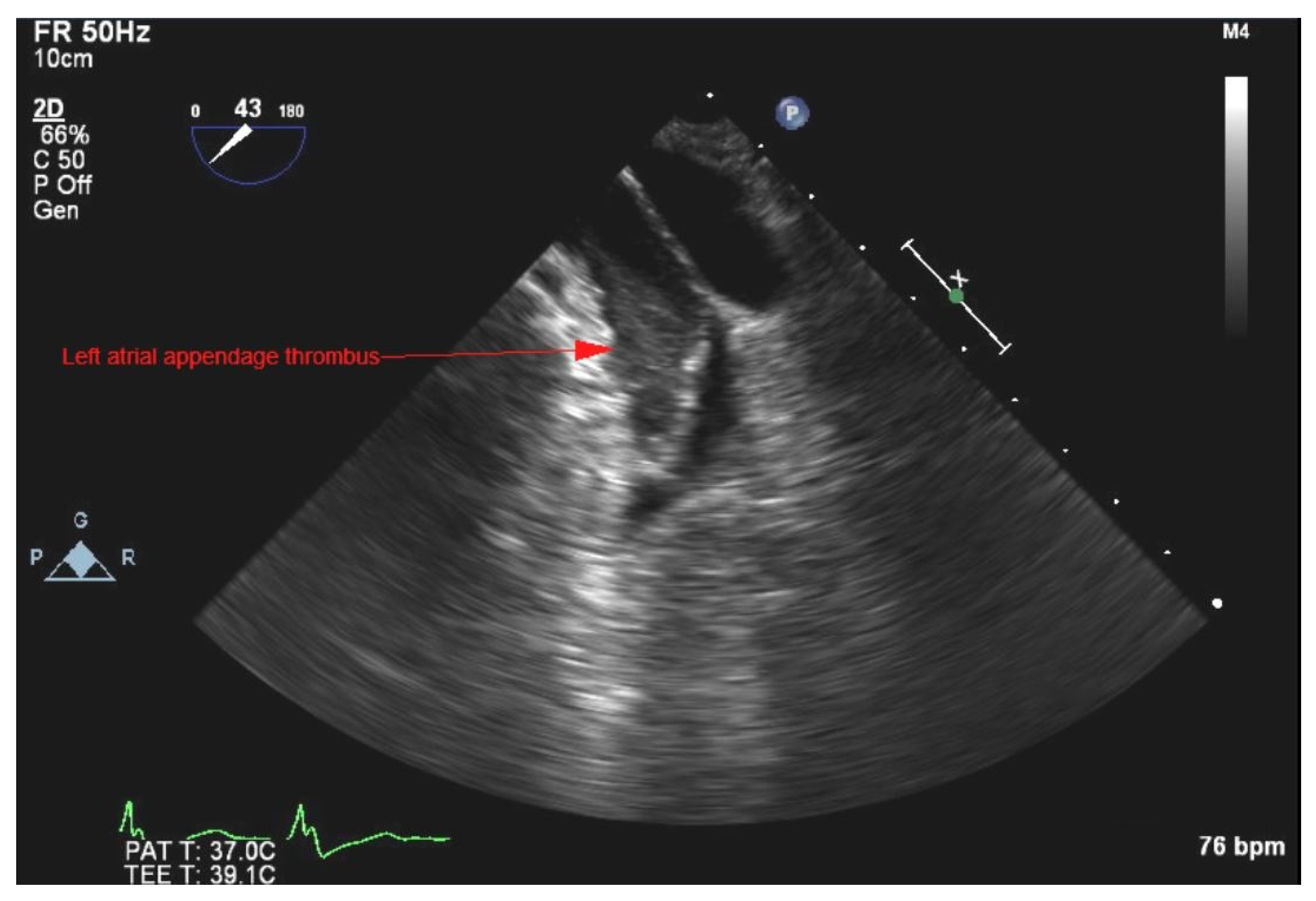
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Fiore, E.; Van Tyne, D.; Gilmore, M.S. Pathogenicity of enterococci. Microbiol. Spectr. 2019, 7, 4–7. [Google Scholar] [CrossRef]
- Vijayakrishnan, R.; Rapose, A. Fatal Enterococcus durans aortic valve endocarditis: A case report and review of the literature. BMJ Case Rep. 2012, 2012, bcr0220125855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sohail, M.R.; Uslan, D.Z.; Khan, A.H.; Friedman, P.A.; Hayes, D.L.; Wilson, W.R.; Steckelberg, J.M.; Stoner, S.; Baddour, L.M. Management and outcome of permanent pacemaker and implantable cardioverter-defibrillator infections. J. Am. Coll. Cardiol. 2007, 49, 1851–1859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blomström-Lundqvist, C.; Traykov, V.; Erba, P.A.; Burri, H.; Nielsen, J.C.; Bongiorni, M.G.; Poole, J.; Boriani, G.; Costa, R.; Deharo, J.C.; et al. European Heart Rhythm Association (EHRA) international consensus document on how to prevent, diagnose, and treat cardiac implantable electronic device infections-endorsed by the Heart Rhythm Society (HRS), the Asia Pacific Heart Rhythm Society (APHRS), the Latin American Heart Rhythm Society (LAHRS), International Society for Cardiovascular Infectious Diseases (ISCVID) and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur. Eur. Pacing Arrhythm. Card. Electrophysiol. J. Work. Groups Card. Pacing Arrhythm. Card. Cell. Electrophysiol. Eur. Soc. Cardiol. 2020, 22, 515–549. [Google Scholar] [CrossRef]
- Urien, J.M.; Camus, C.; Leclercq, C.; Dejoies, L.; Mabo, P.; Martins, R.; Boukthir, S.; Bénézit, F.; Behar, N.; Revest, M.; et al. The emergence of Staphylococcus aureus as the primary cause of cardiac device-related infective endocarditis. Infection 2021, 49, 999–1006. [Google Scholar] [CrossRef]
- Polyzos, K.A.; Konstantelias, A.A.; Falagas, M.E. Risk factors for cardiac implantable electronic device infection: A systematic review and meta-analysis. Eur. Eur. Pacing Arrhythm. Card. Electrophysiol. J. Work. Groups Card. Pacing Arrhythm. Card. Cell. Electrophysiol. Eur. Soc. Cardiol. 2015, 17, 767–777. [Google Scholar] [CrossRef]
- DeSimone, D.C.; Sohail, M.R.; Mulpuru, S.K. Contemporary management of cardiac implantable electronic device infection. Heart Br. Card. Soc. 2019, 105, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Hussein, A.A.; Baghdy, Y.; Wazni, O.M.; Brunner, M.P.; Kabbach, G.; Shao, M.; Gordon, S.; Saliba, W.I.; Wilkoff, B.L.; Tarakji, K.G. Microbiology of cardiac implantable electronic device infections. JACC Clin. Electrophysiol. 2016, 2, 498–505. [Google Scholar] [CrossRef]
- Nagpal, A.; Baddour, L.M.; Sohail, M.R. Microbiology and pathogenesis of cardiovascular implantable electronic device infections. Circ. Arrhythmia Electrophysiol. 2012, 5, 433–441. [Google Scholar] [CrossRef] [Green Version]
- Comerlato, C.B.; Resende, M.C.; Caierão, J.; d’Azevedo, P.A. Presence of virulence factors in Enterococcus faecalis and Enterococcus faecium susceptible and resistant to vancomycin. Mem. Inst. Oswaldo Cruz 2013, 108, 590–595. [Google Scholar] [CrossRef] [Green Version]
- Tan, C.K.; Lai, C.C.; Wang, J.Y.; Lin, S.H.; Liao, C.H.; Huang, Y.T.; Wang, C.Y.; Lin, H.I.; Hsueh, P.R. Bacteremia caused by non-faecalis and non-faecium enterococcus species at a Medical center in Taiwan, 2000 to 2008. J. Infect. 2010, 61, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Al Shehri, M.; Zarak, M.S.; Sarwari, A.R. Late prosthetic valve infective endocarditis by Enterococcus durans. J. Glob. Infect. Dis. 2020, 12, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Sunbul, M.; Cerik, H.O.; Cerik, B.; Gulel, O.; Leblebicioglu, H. Enterococcus durans endocarditis. Am. J. Ther. 2018, 25, e663–e665. [Google Scholar] [CrossRef] [PubMed]
- Zala, A.; Collins, N. Enterococcus durans prosthetic valve endocarditis: A previously unreported clinical entity. Heart Lung Circ. 2016, 25, e133–e136. [Google Scholar] [CrossRef]
- Fallavollita, L.; Di Gioacchino, L.; Balestrini, F. Bioprosthetic aortic valve endocarditis in association with Enterococcus durans. Tex. Heart Inst. J. 2016, 43, 165–167. [Google Scholar] [CrossRef] [Green Version]
- Kenzaka, T.; Takamura, N.; Kumabe, A.; Takeda, K. A case of subacute infective endocarditis and blood access infection caused by Enterococcus durans. BMC Infect. Dis. 2013, 13, 594. [Google Scholar] [CrossRef] [Green Version]
- Stepanović, S.; Jovanović, M.; Lavadinović, L.; Stošović, B.; Pelemiš, M. Enterococcus durans endocarditis in a patient with transposition of the great vessels. J. Med. Microbiol. 2004, 53, 259–261. [Google Scholar] [CrossRef] [Green Version]
- Tripodi, M.F.; Locatelli, A.; Adinolfi, L.E.; Andreana, A.; Utili, R. Successful treatment with ampicillin and fluoroquinolones of human endocarditis due to high-level gentamicin-resistant enterococci. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 1998, 17, 734–736. [Google Scholar] [CrossRef]
- Connelly, M.S.; Liu, P.P.; Williams, W.G.; Webb, G.D.; Robertson, P.; McLaughlin, P.R. Congenitally corrected transposition of the great arteries in the adult: Functional status and complications. J. Am. Coll. Cardiol. 1996, 27, 1238–1243. [Google Scholar] [CrossRef] [Green Version]
- Fiuza, C.; Salcedo, M.; Clemente, G.; Tellado, J.M. In Vivo neutrophil dysfunction in cirrhotic patients with advanced liver disease. J. Infect. Dis. 2000, 182, 526–533. [Google Scholar] [CrossRef] [Green Version]
- Rohacek, M.; Baddour, L.M. Cardiovascular implantable electronic device infections: Associated risk factors and prevention. Swiss Med. Wkly. 2015, 145, w14157. [Google Scholar] [CrossRef] [PubMed]
- Manning, W.J.; Weintraub, R.M.; Waksmonski, C.A.; Haering, J.M.; Rooney, P.S.; Maslow, A.D.; Johnson, R.G.; Douglas, P.S. Accuracy of transesophageal echocardiography for identifying left atrial thrombi. A prospective, intraoperative study. Ann. Intern. Med. 1995, 123, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Durmaz, E.; Karpuz, M.H.; Bilgehan, K.; Ikitimur, B.; Ozmen, E.; Ebren, C.; Polat, F.; Koca, D.; Tokdil, K.O.; Kandemirli, S.G.; et al. Left atrial thrombus in patients with atrial fibrillation and under oral anticoagulant therapy; 3-D transesophageal echocardiographic study. Int. J. Cardiovasc. Imaging 2020, 36, 1097–1103. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, E.; Wise, E.; Hubbard, B.; Girard, S.; Kong, B.; Moudgal, V. Tucked away: An infected thrombus. Am. J. Med. 2016, 129, 576–579. [Google Scholar] [CrossRef] [Green Version]
- Miller, W.R.; Munita, J.M.; Arias, C.A. Mechanisms of antibiotic resistance in enterococci. Expert Rev. Anti-Infect. Ther. 2014, 12, 1221–1236. [Google Scholar] [CrossRef]
- Baddour, L.M.; Wilson, W.R.; Bayer, A.S.; Fowler, V.G., Jr.; Tleyjeh, I.M.; Rybak, M.J.; Barsic, B.; Lockhart, P.B.; Gewitz, M.H.; Levison, M.E.; et al. Infective endocarditis in adults: Diagnosis, antimicrobial therapy, and management of complications: A scientific statement for healthcare professionals from the american heart association. Circulation 2015, 132, 1435–1486. [Google Scholar] [CrossRef]
- Habib, G.; Erba, P.A.; Iung, B.; Donal, E.; Cosyns, B.; Laroche, C.; Popescu, B.A.; Prendergast, B.; Tornos, P.; Sadeghpour, A.; et al. Clinical presentation, aetiology and outcome of infective endocarditis. Results of the ESC-EORP EURO-ENDO (European infective endocarditis) registry: A prospective cohort study. Eur. Heart J. 2019, 40, 3222–3232. [Google Scholar] [CrossRef] [Green Version]
- McDonald, J.R.; Olaison, L.; Anderson, D.J.; Hoen, B.; Miro, J.M.; Eykyn, S.; Abrutyn, E.; Fowler, V.G., Jr.; Habib, G.; Selton-Suty, C.; et al. Enterococcal endocarditis: 107 cases from the international collaboration on endocarditis merged database. Am. J. Med. 2005, 118, 759–766. [Google Scholar] [CrossRef]
| Reference | Age/Sex | Valve (Anatomy) | Native or Artificial | Preceding Symptoms (Duration) | Complication (If Present) | Treatment (Abx Only or Abx + Surgery) | Length of Treatment | Outcome |
|---|---|---|---|---|---|---|---|---|
| Al Shehri et al. (2020) [12] | 56 F | Mitral | Mechanical | 12 weeks | Splenic infarcts | Abx only | 6 weeks | Exitus |
| Sunbul et al. (2018) [13] | 71 M | Mitral, anterior leaflet | Native | 2 weeks | Septic emboli to brain and legs | Abx + Surgery | 69 days | Exitus |
| Zala et al. (2016) [14] | 61 M | Aortic | Bioprosthetic | 8 weeks | AMI, septic embolus to left ophthalmic artery, bioprosthesis dysfunction | Abx + Surgery (re-do aortic valve replacement) | 18 weeks | Recovered |
| Fallavollita et al. (2016) [15] | 74 M | Aortic | Bioprosthetic | 8 weeks | None reported | Abx only | 6 weeks | Recovered |
| Kenzaka et al. (2013) [16] | 83 M | Aortic | Native | 16 weeks | Right brachial artery infective aneurysm (hemodialysis access fistula) with thrombus | Abx only | 6 weeks | Recovered |
| Vijayayakrishnan et al. (2012) [2] | 61 M | Aortic | Native | 4 weeks | Aortic sinus abscesses | Abx only | 6 weeks | Exitus |
| Stepanovic et al. (2004) [17] | 44 M | Tricuspid | Native | 12 weeks | None reported | Abx only | 7.5 weeks | Recovered |
| Tripodi et al. (1998) [18] | 74 M | Mitral | Native | 1 week | None reported | Abx only | 6 weeks | Recovered |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radovanovic, M.; Jevtic, D.; Barsoum, M.K.; Patel, J.; Dumic, I. Enterococcus durans Cardiac Implantable Electronic Device Lead Infection and Review of Enterococcus durans Endocarditis Cases. Medicina 2022, 58, 307. https://doi.org/10.3390/medicina58020307
Radovanovic M, Jevtic D, Barsoum MK, Patel J, Dumic I. Enterococcus durans Cardiac Implantable Electronic Device Lead Infection and Review of Enterococcus durans Endocarditis Cases. Medicina. 2022; 58(2):307. https://doi.org/10.3390/medicina58020307
Chicago/Turabian StyleRadovanovic, Milan, Djordje Jevtic, Michel K. Barsoum, Janki Patel, and Igor Dumic. 2022. "Enterococcus durans Cardiac Implantable Electronic Device Lead Infection and Review of Enterococcus durans Endocarditis Cases" Medicina 58, no. 2: 307. https://doi.org/10.3390/medicina58020307
APA StyleRadovanovic, M., Jevtic, D., Barsoum, M. K., Patel, J., & Dumic, I. (2022). Enterococcus durans Cardiac Implantable Electronic Device Lead Infection and Review of Enterococcus durans Endocarditis Cases. Medicina, 58(2), 307. https://doi.org/10.3390/medicina58020307









