Clinical Value of EZH2 in Hepatocellular Carcinoma and Its Potential for Target Therapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. The Cancer Genome Atlas (TCGA) Data Analysis
2.2. Cell Culture and siRNA Transfection
2.3. Quantitative Real-Time PCR Analysis (RT-qPCR)
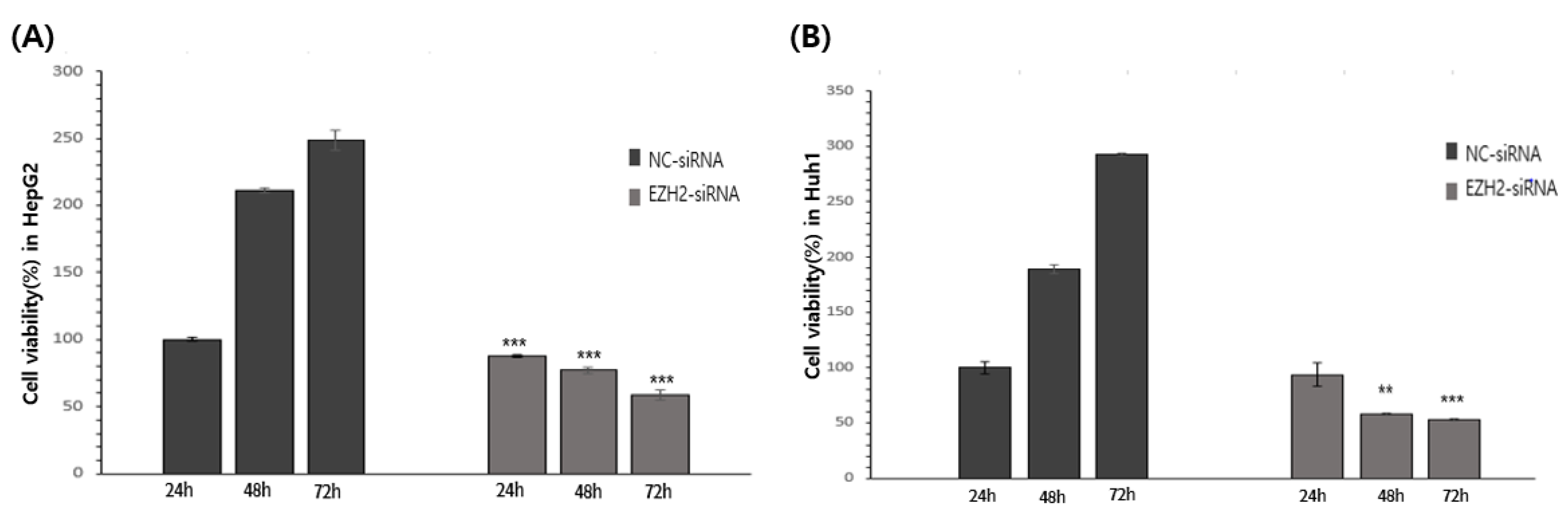
2.4. Cell Viability Assay
2.5. Wound Healing Assay
2.6. Statistical Analysis
3. Results
3.1. Clinical Characteristics of EZH2 Expression in Hepatocellular Carcinoma
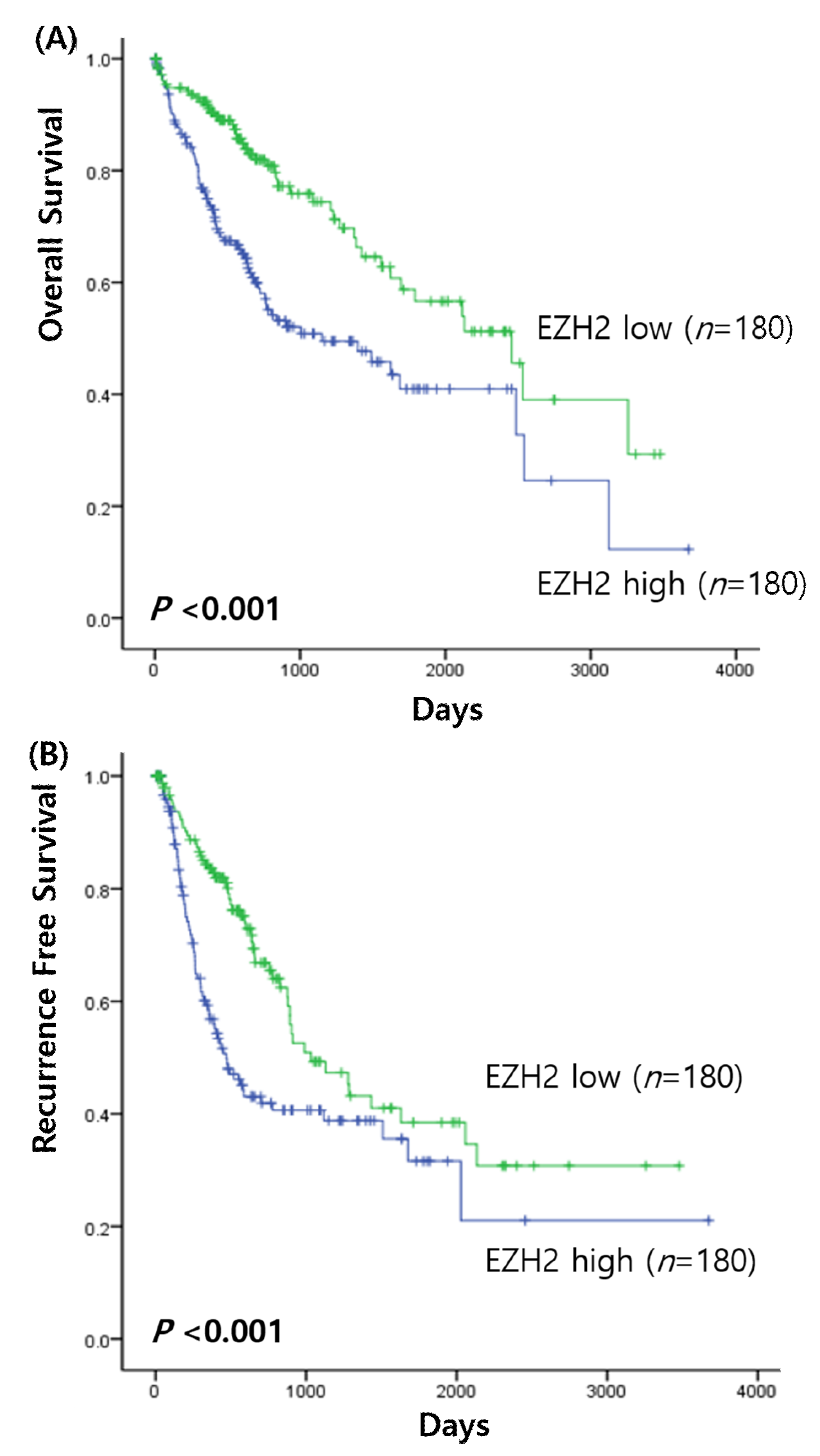
3.2. Prognostic Value of EZH2 mRNA Expressions in HCC
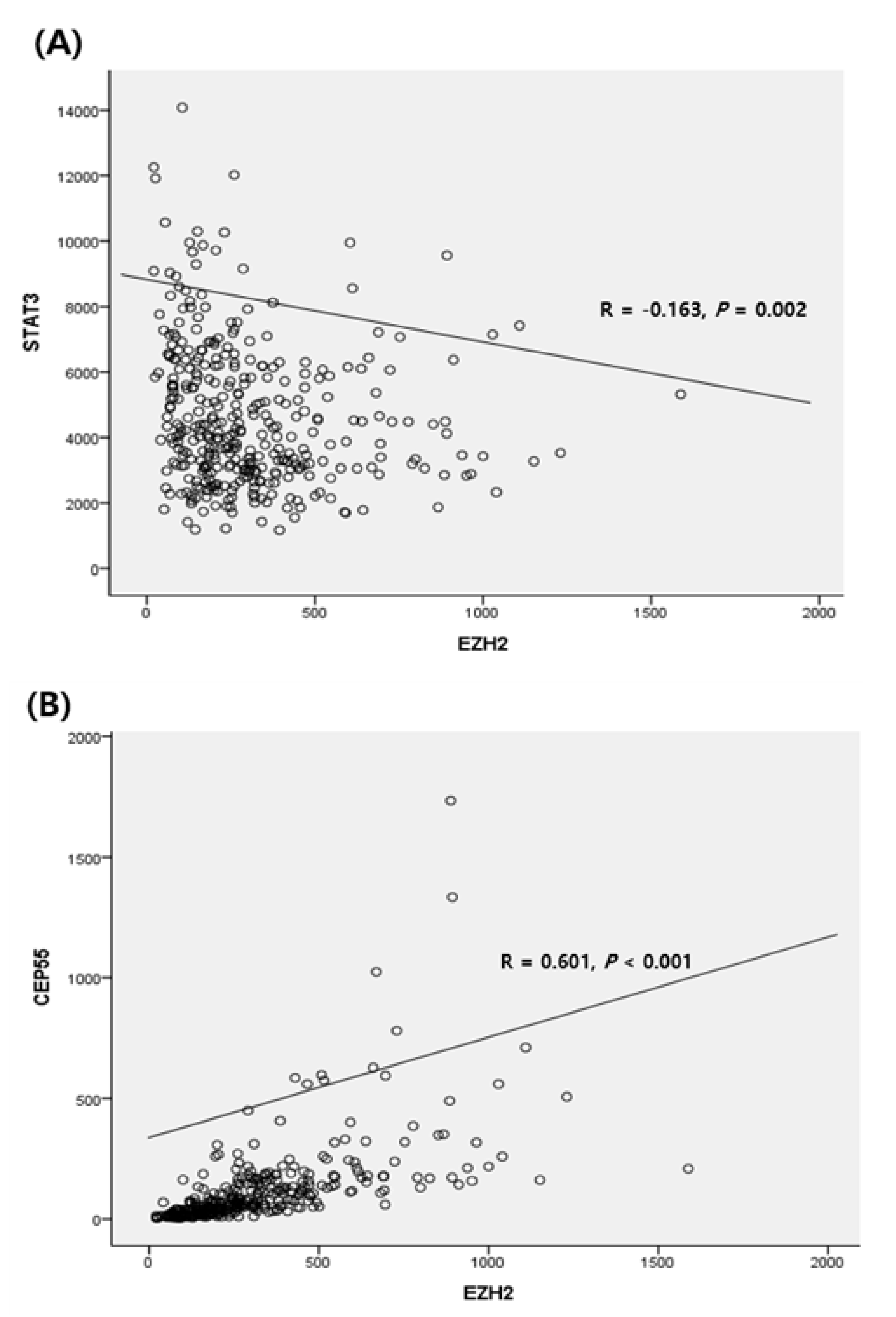
3.3. Correlation with Downstream Genes Related to EZH2
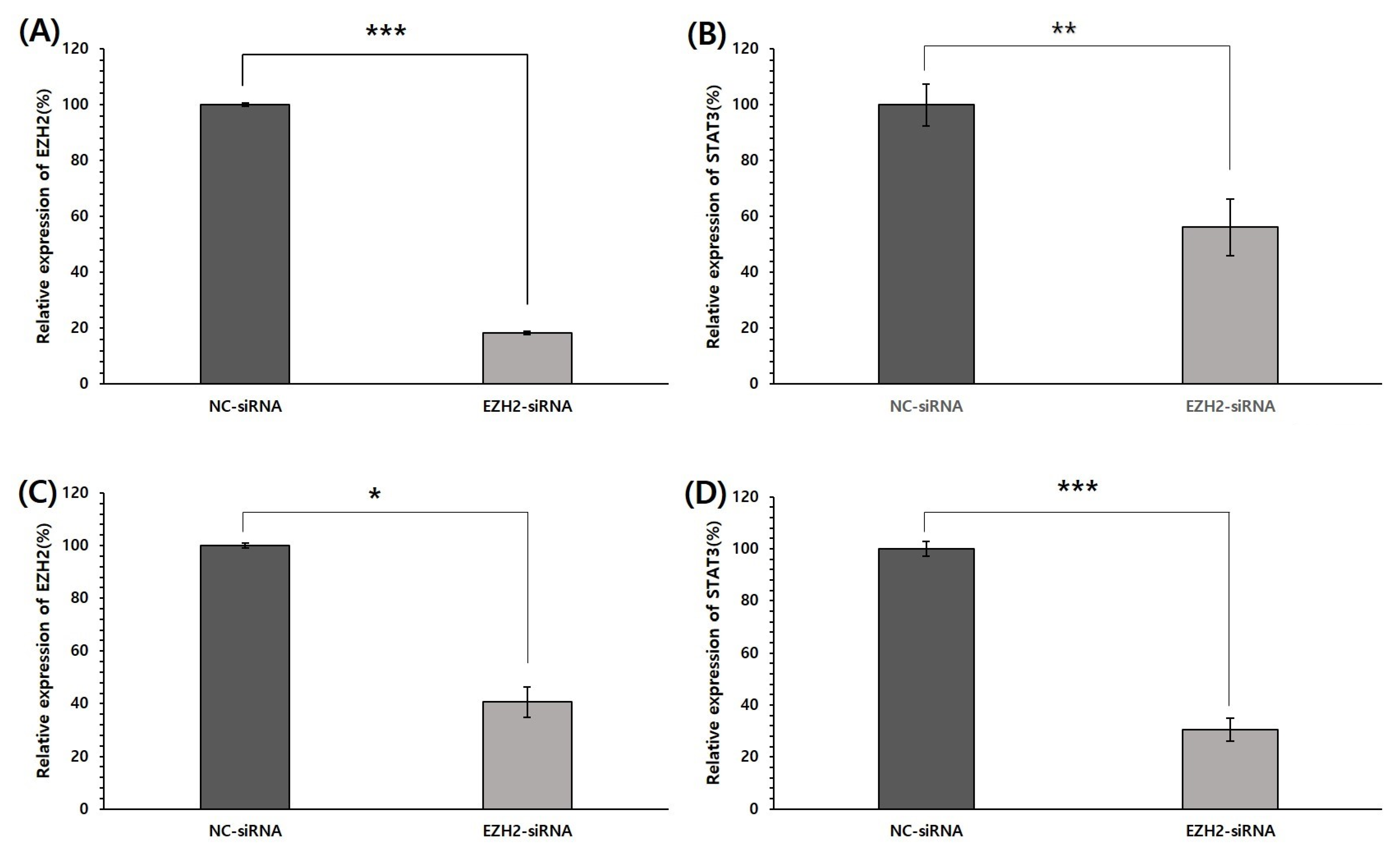
3.4. Effect of EZH2 Silencing on STAT3 mRNA Expression
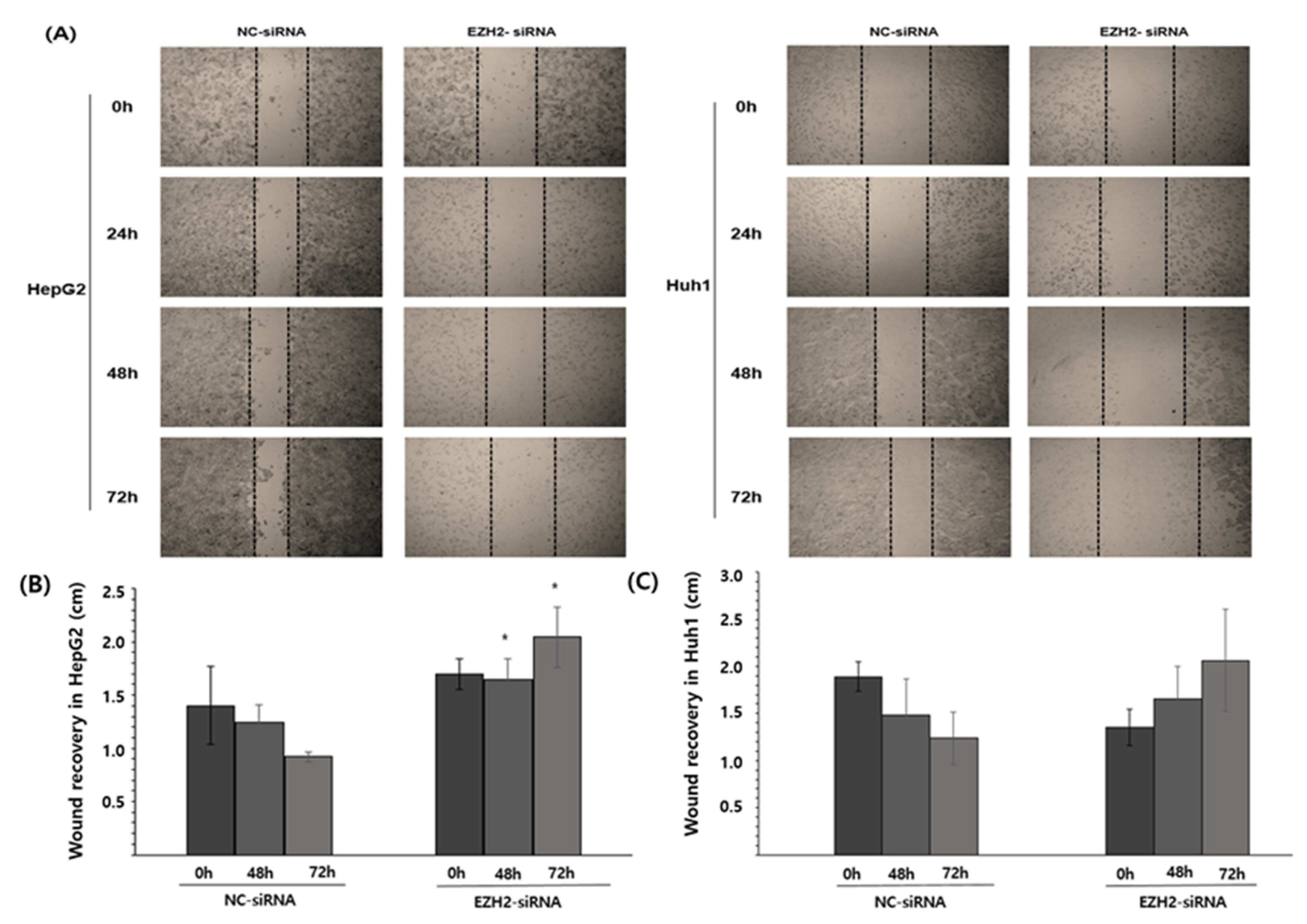
3.5. EZH2 Knockdown Reduces HCC Cell Viability and Cell Recovery
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rawla, P.; Sunkara, T.; Muralidharan, P.; Raj, J.P. Update in global trends and aetiology of hepatocellular carcinoma. Contemp. Oncol. 2018, 22, 141–150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, J.J.H.; Thng, D.K.H.; Lim, J.J.; Toh, T.B. JAK/STAT signaling in hepatocellular carcinoma. Hepat. Oncol. 2020, 7, HEP18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belghiti, J.; Kianmanesh, R. Surgical treatment of hepatocellular carcinoma. HPB 2005, 7, 42–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Xu, J. Identification of Potentially Therapeutic Target Genes of Hepatocellular Carcinoma. Int. J. Environ. Res. 2020, 17, 1053. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.F.; Zhang, M.F.; Tian, Q.H.; Fu, J.; Yang, X.; Zhang, C.Z.; Yang, H. SPAG5 interacts with CEP55 and exerts oncogenic activities via PI3K/AKT pathway in hepatocellular carcinoma. Mol. Cancer 2018, 17, 117. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Tan, X.; Cen, H. EZH2 is a negative prognostic biomarker associated with immunosuppression in hepatocellular carcinoma. PLoS ONE 2020, 15, e0242191. [Google Scholar] [CrossRef] [PubMed]
- Duan, R.; Du, W.; Guo, W. EZH2: A novel target for cancer treatment. J. Hematol. Oncol. 2020, 13, 104. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. The Cancer Genome Atlas Program. Available online: https://tcga-data.nci.nih.gov/tcga/ (accessed on 8 March 2021).
- Lu, Y.; Zhu, M.; Li, W.; Lin, B.; Dong, X.; Chen, Y.; Xie, X.; Guo, J.; Li, M. Alpha fetoprotein plays a critical role in promoting metastasis of hepatocellular carcinoma cells. J. Cell. Mol. Med. 2016, 20, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Au, S.L.; Wong, C.C.; Lee, J.M.; Fan, D.N.; Tsang, F.H.; Ng, I.O.; Wong, C.M. Enhancer of zeste homolog 2 epigenetically silences multiple tumor suppressor microRNAs to promote liver cancer metastasis. Hepatology 2012, 56, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Nienstedt, J.C.; Schroeder, C.; Clauditz, T.; Simon, R.; Sauter, G.; Muenscher, A.; Blessmann, M.; Hanken, H.; Pflug, C. EZH2 overexpression in head and neck cancer is related to lymph node metastasis. J. Oral. Pathol. Med. 2018, 47, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; He, Y.; Zhang, Z.; Wang, W. Upregulation of CEP55 Predicts Dismal Prognosis in Patients with Liver Cancer. Biomed. Res. Int. 2020, 2020, 4139320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.; Gao, J.; Li, D.; Yin, Y. CEP55 Promotes Cell Motility via JAK2⁻STAT3⁻MMPs Cascade in Hepatocellular Carcinoma. Cells 2018, 7, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, S.; Wu, D.; Pan, Y.; Liu, H.; Shao, Z.; Wang, M. Correlation between EZH2 and CEP55 and lung adenocarcinoma prognosis. Pathol. Res. Pract. 2019, 215, 292–301. [Google Scholar] [CrossRef]
- Pan, Y.M.; Wang, C.G.; Zhu, M.; Xing, R.; Cui, J.T.; Li, W.M.; Yu, D.D.; Wang, S.B.; Zhu, W.; Ye, Y.J.; et al. STAT3 signaling drives EZH2 transcriptional activation and mediates poor prognosis in gastric cancer. Mol. Cancer 2016, 15, 79. [Google Scholar] [CrossRef] [Green Version]
| Name | Primer (5′ to 3′) |
|---|---|
| EZH2 | Forward: GACCTCTGTCTTACTTGTGGAGC |
| Reverse: CGTCAGATGGTGCCAGCAATAG | |
| STAT3 | Forward: GCTTTTGTCAGCGATGGAGT |
| Reverse: ATTTGTTGACGGGTCTGAAGTT | |
| GAPDH | Forward: GAAAGGTGAAGGTCGGAGTC |
| Reverse: GTTGAGGTCAATGAAGGGGTC |
| Cancer Type | p |
|---|---|
| Bladder urothelial carcinoma | 0.726 |
| Breast invasive carcinoma | 0.402 |
| Cervical squamous cell carcinoma | 0.293 |
| Colon adenocarcinoma | 0.333 |
| Esophageal carcinoma | 0.837 |
| Glioblastoma multiforme | 0.994 |
| Head and neck squamous cell carcinoma | 0.183 |
| Kidney renal clear cell carcinoma | <0.001 * |
| Kidney renal papillary cell carcinoma | 0.00794 * |
| Acute myeloid leukemia | 0.378 |
| Brain lower grade glioma | 0.00212 * |
| Liver hepatocellular carcinoma | <0.001 * |
| Lung adenocarcinoma | 0.476 |
| Lung squamous cell carcinoma | 0.0946 |
| Ovarian serous cystadenocarcinoma | 0.526 |
| Pancreatic adenocarcinoma | 0.145 |
| Rectum adenocarcinoma | 0.959 |
| Sarcoma | 0.415 |
| Skin cutaneous melanoma | 0.182 |
| Stomach adenocarcinoma | 0.0874 |
| Uterine corpus endometrial carcinoma | 0.144 |
| EZH2 Expression | |||
|---|---|---|---|
| High (%, n) | Low (%, n) | p Value | |
| Age | 0.027 * | ||
| <65 | 56.0 (98) | 44.0 (77) | |
| ≥65 | 44.3 (82) | 55.7 (103) | |
| Sex | 0.176 | ||
| Male | 47.5 (116) | 52.5 (128) | |
| Female | 55.2 (64) | 44.8 (52) | |
| T stage | 0.386 | ||
| T1 | 51.4 (90) | 48.6 (85) | |
| T2 | 43.0 (40) | 57.0 (53) | |
| T3 | 56.0 (42) | 44.0 (33) | |
| T4 | 50 (7) | 50 (7) | |
| N stage | 0.045 * | ||
| N0 | 49.4 (117) | 50.6 (120) | |
| N1 | 100 (4) | 0 (0) | |
| M stage | 0.614 | ||
| M0 | 51.4 (129) | 48.6 (122) | |
| M1 | 40.0 (2) | 60.0 (3) | |
| AFP | <0.001 * | ||
| <20 ng/mL | 34.9 (51) | 65.1 (95) | |
| ≥20 ng/mL | 65.9 (85) | 34.1 (44) | |
| Child–Pugh class | 0.543 | ||
| A | 44.9 (97) | 55.1 (119) | |
| B | 45.0 (9) | 55.0 (11) | |
| C | 100 (1) | 0 (0) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bae, A.-N.; Jung, S.-J.; Lee, J.-H.; Lee, H.; Park, S.G. Clinical Value of EZH2 in Hepatocellular Carcinoma and Its Potential for Target Therapy. Medicina 2022, 58, 155. https://doi.org/10.3390/medicina58020155
Bae A-N, Jung S-J, Lee J-H, Lee H, Park SG. Clinical Value of EZH2 in Hepatocellular Carcinoma and Its Potential for Target Therapy. Medicina. 2022; 58(2):155. https://doi.org/10.3390/medicina58020155
Chicago/Turabian StyleBae, An-Na, Soo-Jung Jung, Jae-Ho Lee, Hyunsu Lee, and Seung Gyu Park. 2022. "Clinical Value of EZH2 in Hepatocellular Carcinoma and Its Potential for Target Therapy" Medicina 58, no. 2: 155. https://doi.org/10.3390/medicina58020155
APA StyleBae, A.-N., Jung, S.-J., Lee, J.-H., Lee, H., & Park, S. G. (2022). Clinical Value of EZH2 in Hepatocellular Carcinoma and Its Potential for Target Therapy. Medicina, 58(2), 155. https://doi.org/10.3390/medicina58020155







