Utility of Measuring Circulating Bio-Adrenomedullin and Proenkephalin for 30-Day Mortality Risk Prediction in Patients with COVID-19 and Non-COVID-19 Interstitial Pneumonia in the Emergency Department
Abstract
1. Introduction
2. Materials and Methods
- -
- Age ≥ 18 years;
- -
- First-presentation symptoms such as dyspnea, fever, and dry cough;
- -
- High-Resolution Computed Tomography (HRCT) scan findings of suspicious images for interstitial pneumonia;
- -
- Adhesion to informed consent.
- -
- Age < 18 years;
- -
- Refusal of consent;
- -
- Dyspnea from a non-infectious cause;
- -
- Symptom onset more than 3 days before the enrollment.
2.1. Study Design
2.2. Biomarkers Analysis
2.3. Statistical Analysis
3. Results
3.1. Population Characteristics
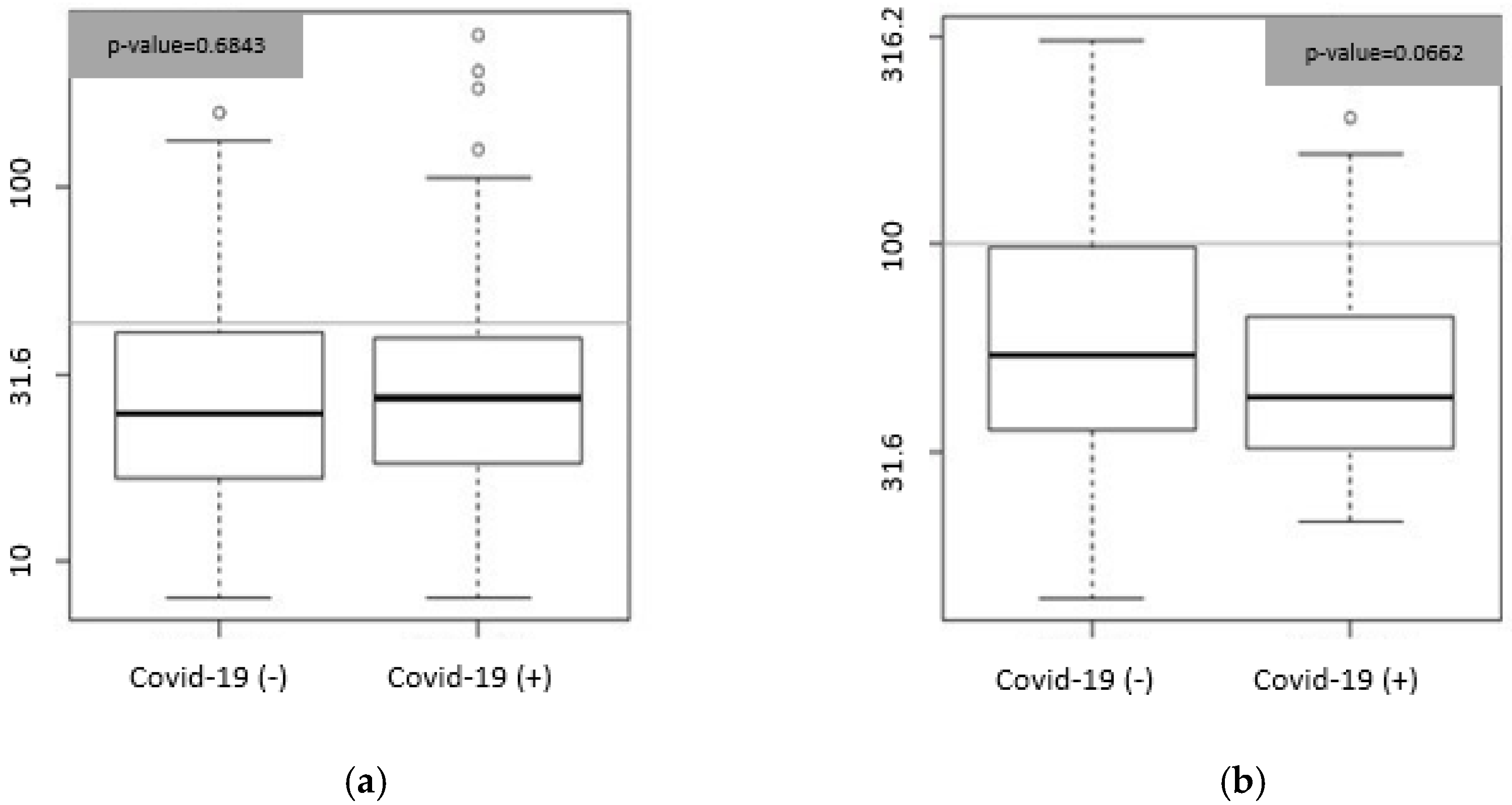
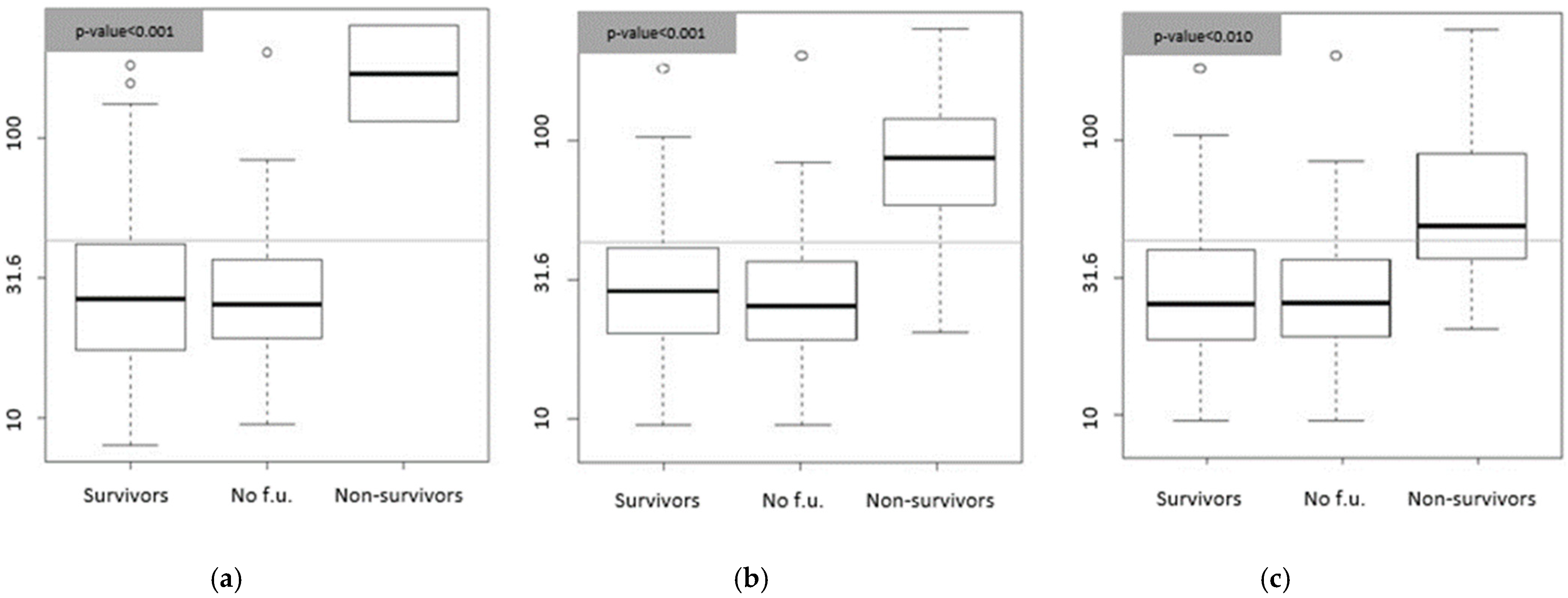
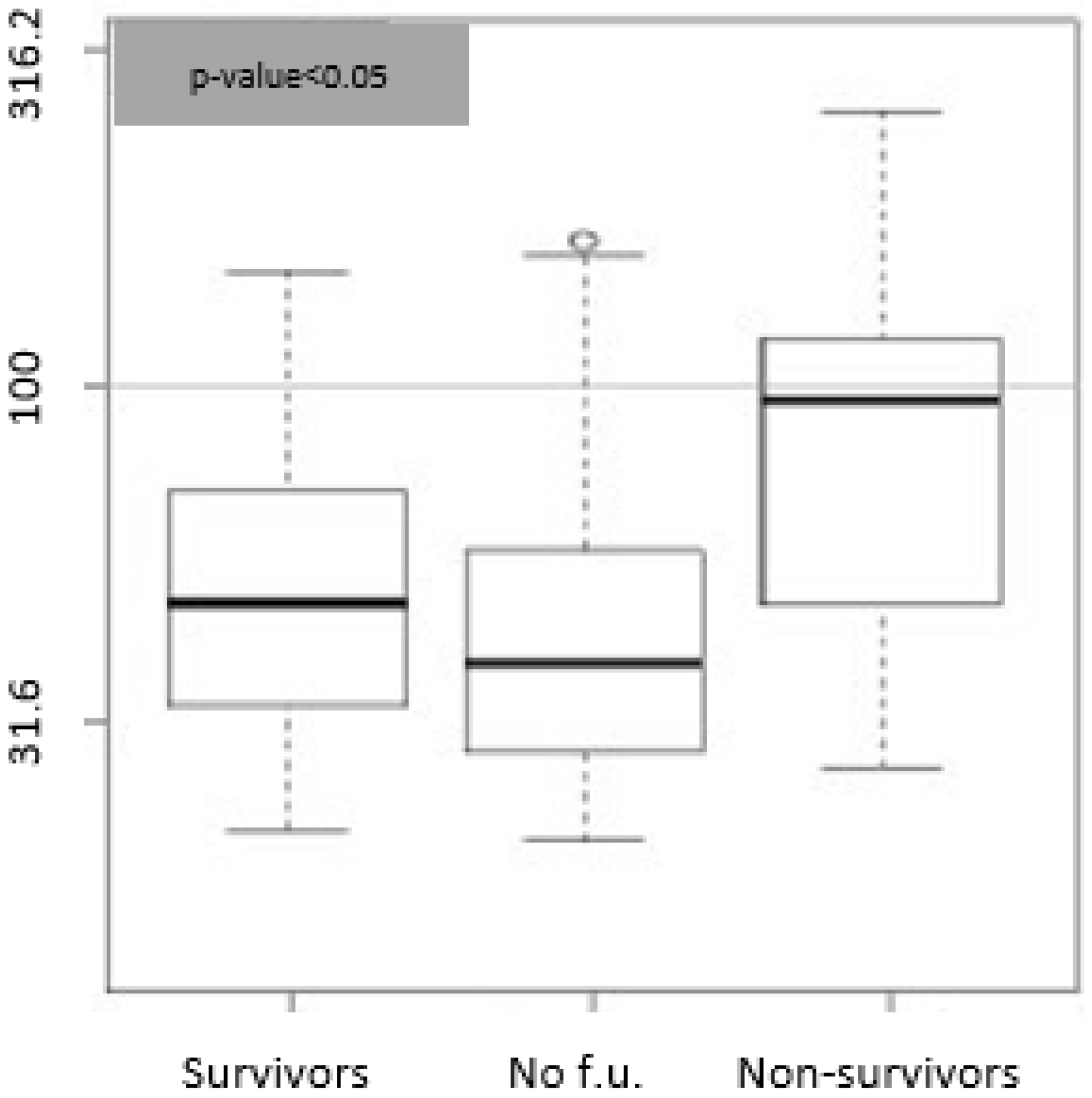
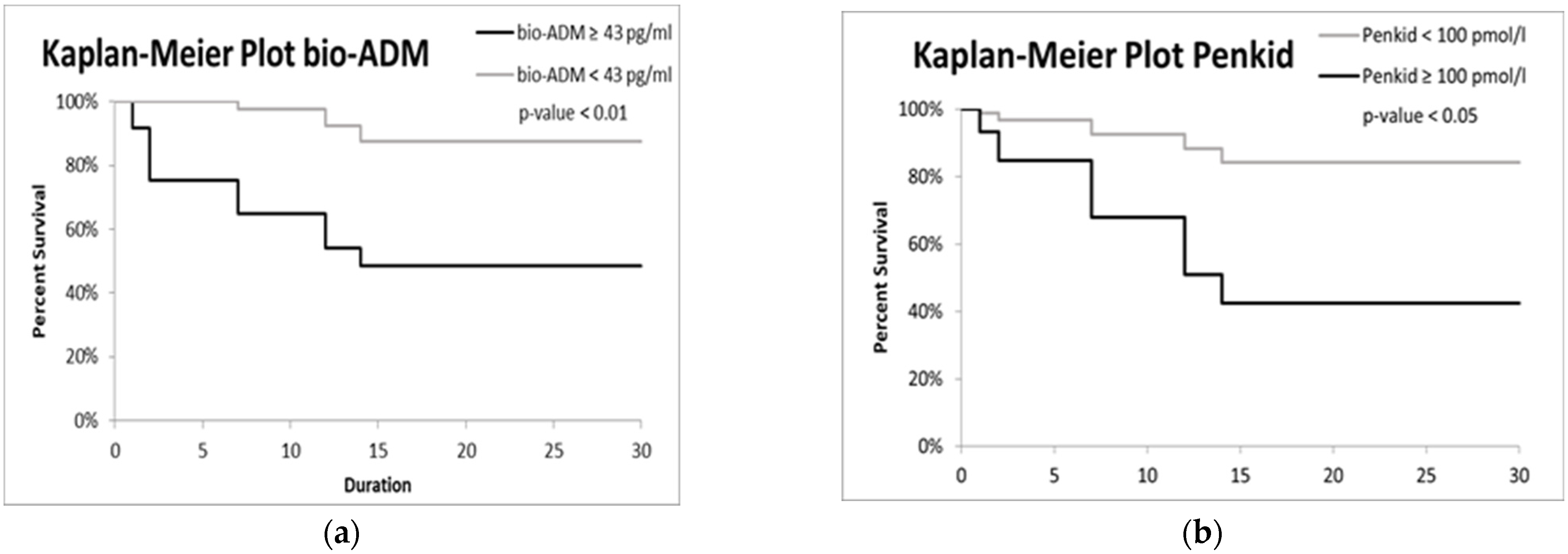
3.2. Biomarkers and Etiology Correlation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pneumonia of Unknown Cause—China n.d. Available online: https://www.who.int/emergencies/disease-outbreak-news/item/2020-DON229 (accessed on 19 October 2022).
- COVID-19 Response|United Nations n.d. Available online: https://www.un.org/en/coronavirus?gclid=Cj0KCQiA-JacBhC0ARIsAIxybyMv2M4dx8u_RFXLd6qOdZO53NZY1xZdT1pWXXoe0B-XyoT2v-NmqrAaAq4fEALw_wcB (accessed on 29 November 2022).
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.A.; Hall, L.E.; John, J.N.; Rapoport, A.B. The Early Natural History of SARS-CoV-2 Infection: Clinical Observations From an Urban, Ambulatory COVID-19 Clinic. Mayo Clin. Proc. 2020, 95, 1124–1126. [Google Scholar] [CrossRef] [PubMed]
- Wolff, D.; Nee, S.; Hickey, N.S.; Marschollek, M. Risk factors for COVID-19 severity and fatality: A structured literature review. Infection 2020, 49, 15–28. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Clinical Management of Severe Acute Respiratory Infection (SARI) When COVID-19 Disease is Suspected: Interim Guidance, 13 March 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Valli, G.; Galati, E.; De Marco, F.; Bucci, C.; Fratini, P.; Cennamo, E.; Ancona, C.; Volpe, N.; Ruggieri, M.P. In-hospital mortality in the emergency department: Clinical and etiological differences between early and late deaths among patients awaiting admission. Clin. Exp. Emerg. Med. 2021, 8, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Valli, G.; Fratini, P.; Volpe, N.; De Marco, F.; Pandolfi, C.; Ancona, C.; Ruggieri, M.P. Analysis of the costs of emergency room management of critically ill patients. Ital. J. Emerg. Med. 2020, 9, 20–28. [Google Scholar] [CrossRef]
- Coronavirus Disease (COVID-19)—World Health Organization n.d. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 19 October 2022).
- Marino, R.; Struck, J.; Maisel, A.S.; Magrini, L.; Bergmann, A.; Di Somma, S. Plasma adrenomedullin is associated with short-term mortality and vasopressor requirement in patients admitted with sepsis. Crit. Care 2014, 18, R34. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Hur, M.; Struck, J.; Bergmann, A.; Di Somma, S. Circulating Biologically Active Adrenomedullin Predicts Organ Failure and Mortality in Sepsis. Ann. Lab. Med. 2019, 39, 454–463. [Google Scholar] [CrossRef]
- Molvin, J.; Jujic, A.; Navarin, S.; Melander, O.; Zoccoli, G.; Hartmann, O.; Bergmann, A.; Struck, J.; Bachus, E.; Di Somma, S.; et al. Bioactive adrenomedullin, proenkephalin A and clinical outcomes in an acute heart failure setting. Open Heart 2019, 6, e001048. [Google Scholar] [CrossRef]
- Simon, T.-P.; Stoppe, C.; Breuer, T.; Stiehler, L.; Dreher, M.; Kersten, A.; Kluge, S.; Karakas, M.; Zechendorf, E.; Marx, G.; et al. Prognostic Value of Bioactive Adrenomedullin in Critically Ill Patients with COVID-19 in Germany: An Observational Cohort Study. J. Clin. Med. 2021, 10, 1667. [Google Scholar] [CrossRef]
- Hollinger, A.; Wittebole, X.; François, B.; Pickkers, P.; Antonelli, M.; Gayat, E.; Chousterman, B.G.; Lascarrou, J.-B.; Dugernier, T.; Di Somma, S.; et al. Proenkephalin A 119–159 (Penkid) Is an Early Biomarker of Septic Acute Kidney Injury: The Kidney in Sepsis and Septic Shock (Kid-SSS) Study. Kidney Int. Rep. 2018, 3, 1424–1433. [Google Scholar] [CrossRef] [PubMed]
- Marino, R.; Struck, J.; Hartmann, O.; Maisel, A.S.; Rehfeldt, M.; Magrini, L.; Melander, O.; Bergmann, A.; Di Somma, S. Diagnostic and short-term prognostic utility of plasma pro-enkephalin (pro-ENK) for acute kidney injury in patients admitted with sepsis in the emergency department. J. Nephrol. 2014, 28, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 180, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Rosenqvist, M.; Bronton, K.; Hartmann, O.; Bergmann, A.; Struck, J.; Melander, O. Proenkephalin a 119–159 (penKid)—A novel biomarker for acute kidney injury in sepsis: An observational study. BMC Emerg. Med. 2019, 19, 75. [Google Scholar] [CrossRef] [PubMed]
- Elicker, B.M.; Webb, W.R. Fundamentals of High-Resolution Lung CT: Common Findings, Common Patterns, Common Diseases, and Differential Diagnosis; Wolters Kluwer Health: Philadelphia, PA, USA; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2013. [Google Scholar]
- Yang, R.; Li, X.; Liu, H.; Zhen, Y.; Zhang, X.; Xiong, Q.; Luo, Y.; Gao, C.; Zeng, W. Chest CT Severity Score: An Imaging Tool for Assessing Severe COVID-19. Radiol. Cardiothorac. Imaging 2020, 2, e200047. [Google Scholar] [CrossRef] [PubMed]
- Guarnera, A.; Santini, E.; Podda, P. Idiopathic Interstitial Pneumonias and COVID-19 Pneumonia: Review of the Main Radiological Features and Differential Diagnosis. Tomography 2021, 7, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.; Sachse, J.; Bergmann, S.; Sparwaßer, A.; Struck, J.; Bergmann, A. Sandwich Immunoassay for Bioactive Plasma Adrenomedullin. J. Appl. Lab. Med. 2017, 2, 222–233. [Google Scholar] [CrossRef]
- Ter Maaten, J.M.; Kremer, D.; Demissei, B.G.; Struck, J.; Bergmann, A.; Anker, S.D.; Ng, L.L.; Dickstein, K.; Metra, M.; Samani, N.J.; et al. Bio-adrenomedullin as a marker of congestion in patients with new-onset and worsening heart failure. Eur. J. Heart Fail. 2019, 21, 732–743. [Google Scholar] [CrossRef]
- Ng, L.L.; Sandhu, J.K.; Narayan, H.; Quinn, P.A.; Squire, I.B.; Davies, J.E.; Bergmann, A.; Maisel, A.; Jones, D. Proenkephalin and Prognosis After Acute Myocardial Infarction. J. Am. Coll. Cardiol. 2013, 63, 280–289. [Google Scholar] [CrossRef]
- Lundberg, O.H.M.; Rosenqvist, M.; Bronton, K.; Schulte, J.; Friberg, H.; Melander, O. Bioactive adrenomedullin in sepsis patients in the emergency department is associated with mortality, organ failure and admission to intensive care. PLoS ONE 2022, 17, e0267497. [Google Scholar] [CrossRef]
- Murthy, S.; Gomersall, C.D.; Fowler, R.A. Care for Critically Ill Patients With COVID-19. JAMA 2020, 323, 1499–1500. [Google Scholar] [CrossRef] [PubMed]
- Caironi, P.; Latini, R.; Struck, J.; Hartmann, O.; Bergmann, A.; Maggio, G.; Cavana, M.; Tognoni, G.; Pesenti, A.; Gattinoni, L.; et al. Circulating Biologically Active Adrenomedullin (bio-ADM) Predicts Hemodynamic Support Requirement and Mortality During Sepsis. Chest 2017, 152, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Mebazaa, A.; Geven, C.; Hollinger, A.; Wittebole, X.; Chousterman, B.G.; Blet, A.; Gayat, E.; Hartmann, O.; Scigalla, P.; Struck, J.; et al. Circulating adrenomedullin estimates survival and reversibility of organ failure in sepsis: The prospective observational multinational Adrenomedullin and Outcome in Sepsis and Septic Shock-1 (AdrenOSS-1) study. Crit. Care 2018, 22, 354. [Google Scholar] [CrossRef] [PubMed]
- Blet, A.; de Roquetaillade, C.; Hartmann, O.; Struck, J.; Mebazaa, A.; Chousterman, B.G. Added value of serial bio-adrenomedullin measurement in addition to lactate for the prognosis of septic patients admitted to ICU. Crit. Care 2020, 24, 69. [Google Scholar] [CrossRef] [PubMed]
- Geven, C.; Peters, E.; Schroedter, M.; Struck, J.; Bergmann, A.; McCook, O.; Radermacher, P.; Kox, M.; Pickkers, P. Effects of the Humanized Anti-Adrenomedullin Antibody Adrecizumab (HAM8101) on Vascular Barrier Function and Survival in Rodent Models of Systemic Inflammation and Sepsis. Shock 2018, 50, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Geven, C.; van Lier, D.; Blet, A.; Peelen, R.; Elzen, B.T.; Mebazaa, A.; Kox, M.; Pickkers, P. Safety, tolerability and pharmacokinetics/pharmacodynamics of the adrenomedullin antibody adrecizumab in a first-in-human study and during experimental human endotoxaemia in healthy subjects. Br. J. Clin. Pharmacol. 2018, 84, 2129–2141. [Google Scholar] [CrossRef]
- Deniau, B.; Takagi, K.; Asakage, A.; Mebazaa, A. Adrecizumab: An investigational agent for the biomarker-guided treatment of sepsis. Expert Opin. Investig. Drugs 2020, 30, 95–102. [Google Scholar] [CrossRef]
- Laterre, P.-F.; Pickkers, P.; Marx, G.; Wittebole, X.; Meziani, F.; Dugernier, T.; Huberlant, V.; Schuerholz, T.; François, B.; Lascarrou, J.-B.; et al. Safety and tolerability of non-neutralizing adrenomedullin antibody adrecizumab (HAM8101) in septic shock patients: The AdrenOSS-2 phase 2a biomarker-guided trial. Intensive Care Med. 2021, 47, 1284–1294. [Google Scholar] [CrossRef]
- Sterner, G.; Frennby, B.; Mansson, S.; Nyman, U.; van Westen, D.; Almén, T. Determining ‘true’ glomerular filtration rate in healthy adults using infusion of inulin and comparing it with values obtained using other clearance techniques or prediction equations. Scand. J. Urol. Nephrol. 2008, 42, 278–285. [Google Scholar] [CrossRef]
- Beunders, R.; Van Groenendael, R.; Leijte, G.P.; Kox, M.; Pickkers, P. Proenkephalin Compared to Conventional Methods to Assess Kidney Function in Critically Ill Sepsis Patients. Shock 2020, 54, 308–314. [Google Scholar] [CrossRef]
- Kim, H.; Hur, M.; Lee, S.; Marino, R.; Magrini, L.; Cardelli, P.; Struck, J.; Bergmann, A.; Hartmann, O.; Di Somma, S.; et al. Proenkephalin, Neutrophil Gelatinase-Associated Lipocalin, and Estimated Glomerular Filtration Rates in Patients With Sepsis. Ann. Lab. Med. 2017, 37, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Kieneker, L.M.; Hartmann, O.; Struck, J.; Bergmann, A.; Gansevoort, R.T.; Joosten, M.M.; Berg, E.V.D.; de Boer, R.A.; Bakker, S.J. Plasma Proenkephalin and Poor Long-Term Outcome in Renal Transplant Recipients. Transplant. Direct 2017, 3, e190. [Google Scholar] [CrossRef] [PubMed]
- Fricker, M.; Wiesli, P.; Brändle, M.; Schwegler, B.; Schmid, C. Impact of thyroid dysfunction on serum cystatin C. Kidney Int. 2003, 63, 1944–1947. [Google Scholar] [CrossRef] [PubMed]
- Johnston, N.; Jernberg, T.; Lindahl, B.; Lindbäck, J.; Stridsberg, M.; Larsson, A.; Venge, P.; Wallentin, L. Biochemical indicators of cardiac and renal function in a healthy elderly population. Clin. Biochem. 2004, 37, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Ronco, C. Acute kidney injury: From clinical to molecular diagnosis. Crit. Care 2016, 20, 201. [Google Scholar] [CrossRef] [PubMed]
- Frigyesi, A.; Boström, L.; Lengquist, M.; Johnsson, P.; Lundberg, O.H.M.; Spångfors, M.; Annborn, M.; Cronberg, T.; Nielsen, N.; Levin, H.; et al. Plasma proenkephalin A 119–159 on intensive care unit admission is a predictor of organ failure and 30-day mortality. Intensive Care Med. Exp. 2021, 9, 36. [Google Scholar] [CrossRef] [PubMed]

| All (153) | COVID+ (101) | COVID− (52) | p-Value | |
|---|---|---|---|---|
| DEMOGRAPHICS | ||||
| Age (mean ± SD) | 70 ± 17 | 68 ± 17 | 72 ± 17 | 0.2 |
| Female n. (%) | 69 (45.1%) | 46 (45.5%) | 23 (44.2%) | 0.88 |
| COEXISTHING CONDITIONS n. (%) | ||||
| Hypertension | 69 (45.1%) | 46 (44.5%) | 23 (44.2%) | 0.88 |
| Diabetes | 24 (15.7%) | 14 (13.9) | 10 (19.2%) | 0.38 |
| IHD | 11 (7.2%) | 7 (6.9%) | 4 (7.7%) | 0.86 |
| CKD | 6 (3.9%) | 3 (3.0%) | 3 (5.7%) | 0.39 |
| COPD | 19 (12.4%) | 9 (8.9%) | 10 (19.2%) | 0.07 |
| SYMPTOMS OF PRESENTATION IN ED n. (%) | ||||
| Dyspnea | 90 (60.1%) | 61 (60.4%) | 31 (59.6%) | 0.92 |
| Cough | 36 (23.5%) | 26 (25.4%) | 10 (19.2%) | 0.36 |
| Fever | 72 (47.1%) | 58 (57.4%) | 14 (26.9%) | 0.001 |
| Other | 51 (33.3%) | 38 (37.6%) | 13 (25.5%) | 0.11 |
| More than one | 73 (47.7%) | 58 (57.4%) | 15 (28.8%) | 0.0008 |
| DEATH n. (%) | 15 (9.8%) | 12 (11.9%) | 3 (5.8%) | 0.22 |
| COMPLICATIONS n. (%) | ||||
| Major complications | 23 (14.6%) | 20 (19,6%) | 3 (5.8%) | 0.053 |
| AKI | 4 (2.6%) | 3 (2.97%) | 1 (1.92%) | 0.70 |
| Light complications | 19 (12.4%) | 16 (15.8%) | 3 (5.8%) | 0.15 |
| DESTINATION n. (%) | ||||
| Low intensive care unit | 77 (50.3%) | 63 (62.4%) | 14 (26.9%) | 0.00003 |
| Sub-intensive therapy | 73 (47.7%) | 36 (35.6%) | 37 (71.2%) | 0.00003 |
| Intensive care unit | 3 (2.0%) | 2 (2%) | 1 (0.9%) | 0.98 |
| HOME THERAPY n. (%) | ||||
| Oral anticoagulants | 17 (11.1%) | 12 (11.9%) | 5 (9.4%) | 0.70 |
| Antiplatelets | 30 (19.6%) | 17 (16.8%) | 13 (25%) | 0.22 |
| Antihypertensive | 59 (38.6%) | 42 (41.6%) | 17 (32.7%) | 0.28 |
| Cortisone | 9 (5.9%) | 5 (5%) | 4 (7.7%) | 0.49 |
| Oral hypoglycemic | 17 (11.1%) | 12 (11.9%) | 5 (9.6%) | 0.70 |
| Insulin | 8 (5.2%) | 6 (5.9%) | 2 (3.8%) | 0.58 |
| Anti-arrhythmic | 21 (13.7%) | 17 (16.8%) | 4 (7.7%) | 0.12 |
| Chemotherapy | 2 (1.3%) | 1 (1.0%) | 1 (1.9%) | 0.63 |
| Diuretics | 29 (19%) | 19 (18.8%) | 10 (19.2%) | <0.05 |
| SOFA | 4 ± 3 | 3 ± 3 | 6 ± 3 | <0.001 |
| RR (b.p.r) | 20 ± 5 | 20 ± 5 | 21 ± 6 | 0.31 |
| SpO2 (%) | 94 ± 6 | 94 ± 5 | 94 ± 7 | 0.18 |
| MBP (mmHg) | 95 ± 15 | 95 ± 13 | 95 ± 19 | 0.9 |
| HR (b.p.m) | 87 ± 18 | 87 ± 20 | 86 ± 14 | 0.74 |
| BT (°C) | 36.7 ± 0.9 | 36.9 ± 1.0 | 36.3 ± 0.6 | <0.001 |
| P/F (mmHg/%) | 360 ± 156 | 359 ± 168 | 363 ± 134 | 0.9 |
| Hb (g/dL) | 13 ± 2 | 13 ± 2 | 12 ± 3 | <0.01 |
| pH | 7.44 ± 0.06 | 7.44 ± 0.06 | 7.42 ± 0.05 | <0.01 |
| Lac (mmol/L) | 1.9 ± 1.2 | 1.8 ± 0.8 | 2.1 ± 1.8 | 0.08 |
| WBCs (×103/uL) | 8 ± 5 | 6 ± 3 | 12 ± 7 | <0.001 |
| Neutrophils (%) | 76 ± 14 | 78 ± 13 | 67 ± 22 | <0.05 |
| Plt (×103/uL) | 215 ± 104 | 193 ± 90 | 259 ± 117 | <0.001 |
| CRP (ng/mL) | 7 ± 7 | 7 ± 7 | 7 ± 8 | 0.9 |
| PCT (ug/mL) | 1 ± 5 | 1 ± 6 | 1 ± 5 | 0.9 |
| Creat (mg/dL) | 1.1 ± 0.6 | 1.0 ± 0.5 | 1.2 ± 0.7 | 0.05 |
| Bilirubin Tot. (mg/dL) | 0.9 ± 1.6 | 0.8 ± 0.6 | 1.2 ± 2.6 | 0.14 |
| INR | 1.3 ± 0.8 | 1.2 ± 0.5 | 1.4 ± 1.2 | 0.14 |
| LDH (U/L) | 356 ± 219 | 357 ± 177 | 354 ± 288 | 0.95 |
| K (mmol/L) | 3.7 ± 0.5 | 3.9 ± 0.5 | 4.1 ± 0.6 | 0.05 |
| Na (mmol/L) | 137 ± 4 | 137 ± 4 | 138 ± 6 | 0.22 |
| HSTn (ng/L) | 316 ± 1866 | 257 ± 661 | 426 ± 2213 | 0.21 |
| BNP (pg/mL) | 313 ± 657 | 233 ± 612 | 481 ± 724 | 0.05 |
| Covariate | Beta | Sdt. Err. | Wald | p-Value | HR | LCL | UCL |
|---|---|---|---|---|---|---|---|
| Bio-ADM > 43 pg/mL | 1.08 | 0.47 | 5.21 | 0.02 | 2.95 | 1.16 | 7.46 |
| PenKid > 100 mmol/L | 0.02 | 0.47 | 0.00 | 0.96 | 1.02 | 0.41 | 2.55 |
| Creat_T0 | 0.003 | 0.17 | 0.00 | 0.98 | 1.00 | 0.72 | 1.39 |
| COVID+ | 0.37 | 0.40 | 0,86 | 0.35 | 1.45 | 0.66 | 3.18 |
| Age, years | 0.09 | 0.02 | 15.96 | 0.0001 | 1.09 | 1.05 | 1.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papasidero, I.D.; Valli, G.; Marin, D.; Del Sasso, A.; De Magistris, A.; Cennamo, E.; Casalboni, S.; De Marco, F.; Rocchi, R.; Ndogmo Beumo, B.; et al. Utility of Measuring Circulating Bio-Adrenomedullin and Proenkephalin for 30-Day Mortality Risk Prediction in Patients with COVID-19 and Non-COVID-19 Interstitial Pneumonia in the Emergency Department. Medicina 2022, 58, 1852. https://doi.org/10.3390/medicina58121852
Papasidero ID, Valli G, Marin D, Del Sasso A, De Magistris A, Cennamo E, Casalboni S, De Marco F, Rocchi R, Ndogmo Beumo B, et al. Utility of Measuring Circulating Bio-Adrenomedullin and Proenkephalin for 30-Day Mortality Risk Prediction in Patients with COVID-19 and Non-COVID-19 Interstitial Pneumonia in the Emergency Department. Medicina. 2022; 58(12):1852. https://doi.org/10.3390/medicina58121852
Chicago/Turabian StylePapasidero, Ilaria Dafne, Gabriele Valli, Dario Marin, Alberto Del Sasso, Antonio De Magistris, Elisa Cennamo, Silvia Casalboni, Francesca De Marco, Roberta Rocchi, Brice Ndogmo Beumo, and et al. 2022. "Utility of Measuring Circulating Bio-Adrenomedullin and Proenkephalin for 30-Day Mortality Risk Prediction in Patients with COVID-19 and Non-COVID-19 Interstitial Pneumonia in the Emergency Department" Medicina 58, no. 12: 1852. https://doi.org/10.3390/medicina58121852
APA StylePapasidero, I. D., Valli, G., Marin, D., Del Sasso, A., De Magistris, A., Cennamo, E., Casalboni, S., De Marco, F., Rocchi, R., Ndogmo Beumo, B., Cusani, V., Gaudio, M., Hartmann, O., Bergman, A., Ruggieri, M. P., & Di Somma, S. (2022). Utility of Measuring Circulating Bio-Adrenomedullin and Proenkephalin for 30-Day Mortality Risk Prediction in Patients with COVID-19 and Non-COVID-19 Interstitial Pneumonia in the Emergency Department. Medicina, 58(12), 1852. https://doi.org/10.3390/medicina58121852








