Myofascial Injection Using Fascial Layer-Specific Hydromanipulation Technique (FLuSH) and the Delineation of Multifactorial Myofascial Pain
Abstract
1. Introduction
2. Materials and Methods
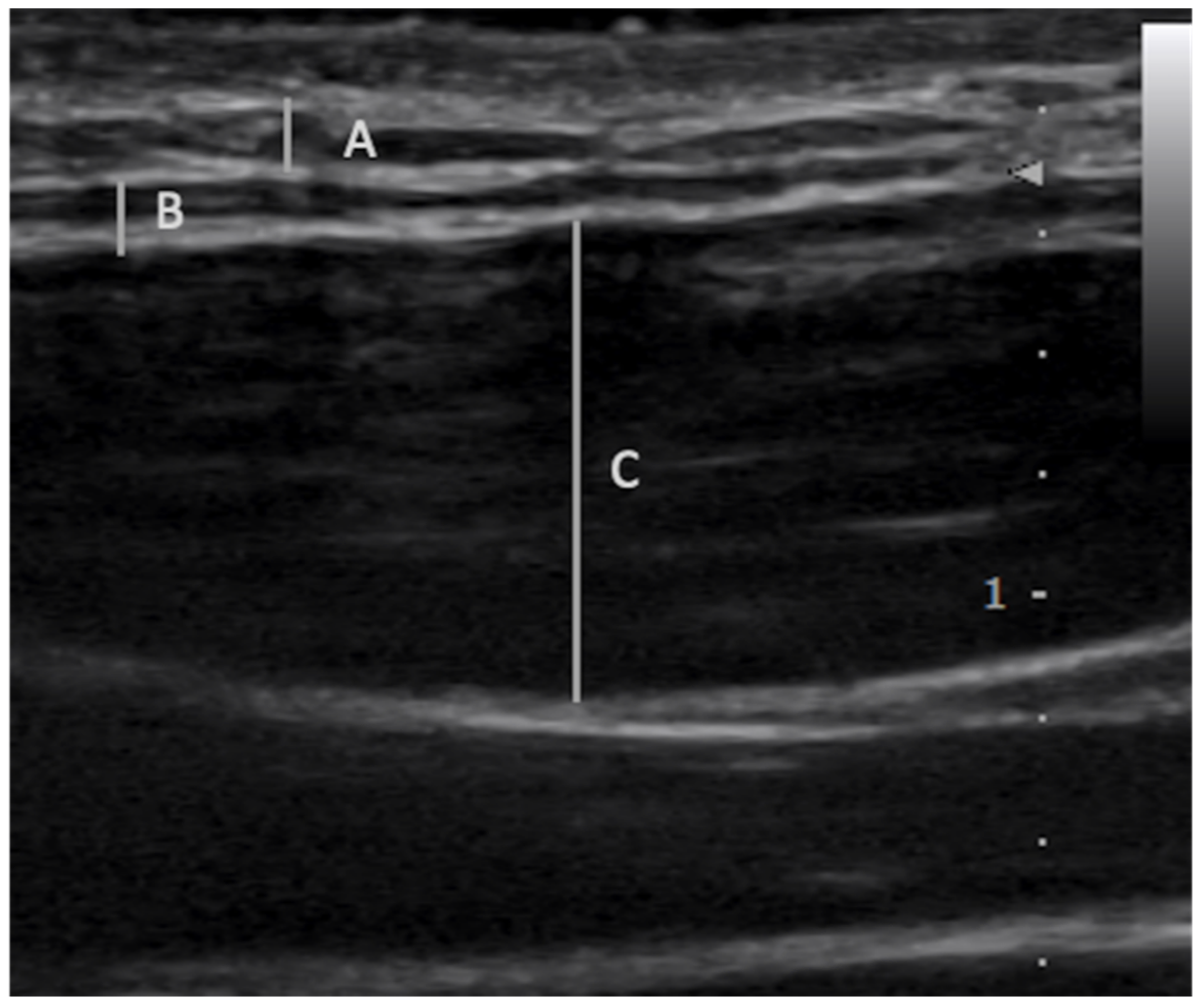
2.1. Fascial Layer Specific Hydromanipulation (FLuSH)
2.2. Data Collection
2.3. Outcomes
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Skootsky, S.A.; Jaeger, B.; Oye, R.K. Prevalence of myofascial pain in general internal medicine practice. West. J. Med. 1989, 151, 157–160. [Google Scholar] [PubMed]
- Thomae, M.K.; Porteous, J.E.; Brock, J.R.; Allen, G.D.; Heller, R.F. Back pain in Australian military helicopter pilots: A preliminary study. Aviat. Space Environ. Med. 1998, 69, 468–473. [Google Scholar] [PubMed]
- Donnelly, J.M.; Simons, D.G. (Eds.) Travell, Simons & Simons’ Myofascial Pain and Dysfunction: The Trigger Point Manual, 3rd ed.; Wolters Kluwer Health: Philadelphia, PA, USA, 2019; 935p. [Google Scholar]
- Simons, G.D.; Mense, S. Understanding and measurement of muscle tone as related to clinical muscle pain. Pain 1998, 75, 1–17. [Google Scholar] [CrossRef]
- Hua, N.K.; Van Der Does, E. The occurrence and inter-rater reliability of myofascial trigger points in the quadratus lumborum and gluteus medius: A prospective study in non-specific low back pain patients and controls in general practice. Pain 1994, 58, 317–323. [Google Scholar] [CrossRef]
- Bogduk, N. The anatomy and pathophysiology of neck pain. Phys. Med. Rehabil. Clin. N. Am. 2003, 14, 455–472. [Google Scholar] [CrossRef]
- Schroeder, B.; Sanfilippo, J.S.; Hertweck, S. Musculoskeletal pelvic pain in a pediatric and adolescent gynecology practice. J. Pediatr. Adolesc. Gynecol. 2000, 13, 90. [Google Scholar] [CrossRef]
- Stecco, C.; Day, J.A. The fascial manipulation technique and its biomechanical model: A guide to the human fascial system. Int. J. Ther. Massage. Bodyw. 2010, 3, 38–40. [Google Scholar]
- Gebhart, G.F.; Hansmond, D.L.; Jensen, T.S. Progress in Pain Research and Management, Vol. 2. In Proceedings of the 7th World Congress on Pain Progress in Pain Research and Management, Seattle, WA, USA, 1 August 1994. [Google Scholar]
- Affaitati, G.; Costantini, R.; Fabrizio, A.; Lapenna, D.; Tafuri, E.; Giamberardino, M.A. Effects of treatment of peripheral pain generators in fibromyalgia patients. Eur. J. Pain 2011, 15, 61–69. [Google Scholar] [CrossRef]
- Giamberardino, M.A.; Tafuri, E.; Savini, A.; Fabrizio, A.; Affaitati, G.; Lerza, R.; Di Ianni, L.; Lapenna, D.; Mezzetti, A. Contribution of Myofascial Trigger Points to Migraine Symptoms. J. Pain 2007, 8, 869–878. [Google Scholar] [CrossRef]
- Schilder, A.; Hoheisel, U.; Magerl, W.; Benrath, J.; Klein, T.; Treede, R.-D. Sensory findings after stimulation of the thoracolumbar fascia with hypertonic saline suggest its contribution to low back pain. Pain 2014, 155, 222–231. [Google Scholar] [CrossRef]
- Sikdar, S.; Shah, J.P.; Gebreab, T.; Yen, R.-H.; Gilliams, E.; Danoff, J.; Gerber, L.H. Novel Applications of Ultrasound Technology to Visualize and Characterize Myofascial Trigger Points and Surrounding Soft Tissue. Arch. Phys. Med. Rehabil. 2009, 90, 1829–1838. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.E.E.; Aranha, M.F.M.; Gavião, M.B.D. Two-Dimensional Ultrasound and Ultrasound Elastography Imaging of Trigger Points in Women with Myofascial Pain Syndrome Treated by Acupuncture and Electroacupuncture: A Double-Blinded Randomized Controlled Pilot Study. Ultrason. Imaging 2015, 37, 152–167. [Google Scholar] [CrossRef] [PubMed]
- Turo, D.; Otto, P.; Hossain, M.; Gebreab, T.; Armstrong, K.; Rosenberger, W.F.; Shao, H.; Shah, J.P.; Gerber, L.H.; Sikdar, S. Novel Use of Ultrasound Elastography to Quantify Muscle Tissue Changes After Dry Needling of Myofascial Trigger Points in Patients With Chronic Myofascial Pain. J. Ultrasound Med. 2015, 34, 2149–2161. [Google Scholar] [CrossRef] [PubMed]
- Margalef, R.; Sisquella, M.; Bosque, M.; Romeu, C.; Mayoral, O.; Monterde, S.; Priego, M.; Guerra-Perez, R.; Ortiz, N.; Tomàs, J.; et al. Experimental myofascial trigger point creation in rodents. J. Appl. Physiol. 2019, 126, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Stecco, A.; Meneghini, A.; Stern, R.; Stecco, C.; Imamura, M. Ultrasonography in myofascial neck pain: Randomized clinical trial for diagnosis and follow-up. Surg. Radiol. Anat. 2013, 36, 243–253. [Google Scholar] [CrossRef]
- Menon, R.G.; Oswald, S.F.; Raghavan, P.; Regatte, R.R.; Stecco, A. T1ρ-Mapping for Musculoskeletal Pain Diagnosis: Case Series of Variation of Water Bound Glycosaminoglycans Quantification before and after Fascial Manipulation® in Subjects with Elbow Pain. Int. J. Environ. Res. Public Health 2020, 17, 708. [Google Scholar] [CrossRef]
- Menon, R.G.; Raghavan, P.; Regatte, R.R. Quantifying muscle glycosaminoglycan levels in patients with post-stroke muscle stiffness using T1ρ MRI. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef]
- Pavan, P.G.; Stecco, A.; Stern, R.; Stecco, C. Painful Connections: Densification Versus Fibrosis of Fascia. Curr. Pain Headache Rep. 2014, 18, 1–8. [Google Scholar] [CrossRef]
- Fede, C.; Porzionato, A.; Petrelli, L.; Fan, C.; Pirri, C.; Biz, C.; De Caro, R.; Stecco, C. Fascia and soft tissues innervation in the human hip and their possible role in post-surgical pain. J. Orthop. Res. 2020, 38, 1646–1654. [Google Scholar] [CrossRef]
- Fernández-de-las-Peñas, C.; Dommerholt, J. Myofascial Trigger Points: Peripheral or Central Phenomenon? Curr. Rheumatol. Rep. 2014, 16, 395. [Google Scholar] [CrossRef]
- Cotti, A.; Del Corso, M.; Diana, R.; Cornale, L.; Sudanese, A.; Stecco, A.; Branchini, M. Inter and Intra Operator Reliability of Motor and Palpation Evaluation in Fascial Manipulation in individuals with coxarthrosis. J. Man. Manip. Ther. 2019, 28, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.J.; Kim, J.; Park, S.; Paek, S.; Kim, T.; Kim, D.K. Feasibility of Ultrasound-Guided Trigger Point Injection in Patients with Myofascial Pain Syndrome. Healthcare 2019, 7, 118. [Google Scholar] [CrossRef] [PubMed]
- Gerwin, R.D. Myofascial Trigger Point Pain Syndromes. Semin. Neurol. 2016, 36, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Perreault, T.; Dunning, J.; Butts, R. The local twitch response during trigger point dry needling: Is it necessary for successful outcomes? J. Bodyw. Mov. Ther. 2017, 21, 940–947. [Google Scholar] [CrossRef]
- Park, G.; Kim, C.W.; Park, S.B.; Kim, M.J.; Jang, S.H. Reliability and Usefulness of the Pressure Pain Threshold Measurement in Patients with Myofascial Pain. Ann. Rehabil. Med. 2011, 35, 412–417. [Google Scholar] [CrossRef]
- Hong, C.-Z. Algometry in Evaluation of Trigger Points and Referred Pain. J. Musculoskelet. Pain 1998, 6, 47–59. [Google Scholar] [CrossRef]
- Wilke, J.; Schleip, R.; Yucesoy, C.A.; Banzer, W. Not merely a protective packing organ? A review of fascia and its force transmission capacity. J. Appl. Physiol. 2018, 124, 234–244. [Google Scholar] [CrossRef]
- Roldan, C.J.; Osuagwu, U.; Cardenas-Turanzas, M.; Huh, B.K. Normal Saline Trigger Point Injections vs Conventional Active Drug Mix for Myofascial Pain Syndromes. Am. J. Emerg. Med. 2020, 38, 311–316. [Google Scholar] [CrossRef]
- Tantisiriwat, N.; Kongsagul, S.; Vitoonpong, T.; Kitisomprayoonkul, W. Ultrasound-guided physiological saline injection for patients with myofascial pain. J. Med. Ultrasound 2020, 28, 99–103. [Google Scholar] [CrossRef]
- Lam, S.K.H.; Hung, C.-Y.; Chiang, Y.-P.; Onishi, K.; Su, D.C.J.; Clark, T.B.; Reeves, K.D. Ultrasound-Guided Nerve Hydrodissection for Pain Management: Rationale, Methods, Current Literature, and Theoretical Mechanisms. J. Pain Res. 2020, 13, 1957–1968. [Google Scholar] [CrossRef]
- Misra, S.; Reed, K.B.; Schafer, B.W.; Ramesh, K.T.; Okamura, A.M. Mechanics of Flexible Needles Robotically Steered through Soft Tissue. Int. J. Robot. Res. 2010, 29, 1640–1660. [Google Scholar] [CrossRef] [PubMed]
- De Meulemeester, K.; Castelein, B.; Coppieters, I.; Barbe, T.; Cools, A.; Cagnie, B. Comparing Trigger Point Dry Needling and Manual Pressure Technique for the Management of Myofascial Neck/Shoulder Pain: A Randomized Clinical Trial. J. Manip. Physiol. Ther. 2017, 40, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Hoheisel, U.; Rosner, J.; Mense, S. Innervation changes induced by inflammation of the rat thoracolumbar fascia. Neuroscience 2015, 300, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Vergara, D.M.; Berrueta, L.; Carmody, C.; An, X.; Wayne, P.M.; Zavacki, A.M.; Langevin, H.M. Establishment of a novel porcine model to study the impact of active stretching on a local Carrageenan-induced inflammation. Am. J. Phys. Med. Rehabil. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mense, S. Innervation of the thoracolumbar fascia. Eur. J. Transl. Myol. 2019, 29. Available online: https://pagepressjournals.org/index.php/bam/article/view/8297 (accessed on 7 January 2020). [CrossRef]
- Matteini, P.; Dei, L.; Carretti, E.; Volpi, N.; Goti, A.; Pini, R. Structural Behavior of Highly Concentrated Hyaluronan. Biomacromolecules 2009, 10, 1516–1522. [Google Scholar] [CrossRef]
- Langevin, H.M.; Fox, J.R.; Koptiuch, C.; Badger, G.J.; Naumann, A.C.G.; Bouffard, N.A.; Konofagou, E.E.; Lee, W.-N.; Triano, J.J.; Henry, S.M. Reduced thoracolumbar fascia shear strain in human chronic low back pain. BMC Musculoskelet Disord. 2011, 12, 203. [Google Scholar] [CrossRef]
- Shah, J.P.; Gilliams, E.A. Uncovering the biochemical milieu of myofascial trigger points using in vivo microdialysis: An application of muscle pain concepts to myofascial pain syndrome. J. Bodyw. Mov. Ther. 2008, 12, 371–384. [Google Scholar] [CrossRef]
- Fede, C.; Angelini, A.; Stern, R.; Macchi, V.; Porzionato, A.; Ruggieri, P.; De Caro, R.; Stecco, C. Quantification of hyaluronan in human fasciae: Variations with function and anatomical site. J. Anat. 2018, 233, 552–556. [Google Scholar] [CrossRef]
- Stecco, C.; Giordani, F.; Fan, C.; Biz, C.; Pirri, C.; Frigo, A.C.; Fede, C.; Macchi, V.; Masiero, S.; De Caro, R. Role of fasciae around the median nerve in pathogenesis of carpal tunnel syndrome: Microscopic and ultrasound study. J. Anat. 2020, 236, 660–667. [Google Scholar] [CrossRef]
- Stecco, C.; Stern, R.; Porzionato, A.; Macchi, V.; Masiero, S.; Stecco, A.; De Caro, R. Hyaluronan within fascia in the etiology of myofascial pain. Surg. Radiol. Anat. 2011, 33, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Casato, G.; Stecco, C.; Busin, R. Role of fasciae in nonspecific low back pain. Eur. J. Transl. Myol. 2019, 29, 8330. [Google Scholar] [CrossRef] [PubMed]
- Kääriäinen, M.; Kääriäinen, J.; Järvinen, T.L.N.; Sievänen, H.; Kalimo, H.; Järvinen, M. Correlation between biomechanical and structural changes during the regeneration of skeletal muscle after laceration injury: Laceration injury of skeletal muscle. J. Orthop. Res. 1998, 16, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Äärimaa, V.; Kääriäinen, M.; Vaittinen, S.; Tanner, J.; Järvinen, T.; Best, T.; Kalimo, H. Restoration of myofiber continuity after transection injury in the rat soleus. Neuromuscul. Disord. 2004, 14, 421–428. [Google Scholar] [CrossRef]
- Huijing, P.; Baan, G.C. Myofascial force transmission: Muscle relative position and length determine agonist and synergist muscle force. J. Appl. Physiol. 2003, 94, 1092–1107. [Google Scholar] [CrossRef] [PubMed]
- Srbely, J.Z.; Dickey, J.P.; Bent, L.R.; Lee, D.; Lowerison, M. Capsaicin-Induced Central Sensitization Evokes Segmental Increases in Trigger Point Sensitivity in Humans. J. Pain 2010, 11, 636–643. [Google Scholar] [CrossRef]
- Hoheisel, U.; Vogt, M.; Palme, R.; Gass, P.; Mense, S. Immobilization stress sensitizes rat dorsal horn neurons having input from the low back. Eur. J. Pain 2015, 19, 861–870. [Google Scholar] [CrossRef]
- Cerritelli, F.; Chiacchiaretta, P.; Gambi, F.; Ferretti, A. Effect of Continuous Touch on Brain Functional Connectivity Is Modified by the Operator’s Tactile Attention. Front. Hum. Neurosci. 2017, 11, 368. [Google Scholar] [CrossRef]
- Stecco, C.; Macchi, V.; Porzionato, A.; Duparc, F.; De Caro, R. The fascia: The forgotten structure. Ital. J. Anat. Embryol. 2011, 116, 127–138. [Google Scholar]
- Kurosawa, A.; Kobayashi, T.; Namiki, H. Ultrasound-Guided Dry Needling for Abnormal Fascia Between the Deltoid Muscle and the Supraspinatus Tendon. Pain Med. 2019, 21, 863–864. [Google Scholar] [CrossRef]

| Number of Treated Myofascial Points (% of All Treated Myofascial Points) | |
|---|---|
| Muscle only | 5 (6%) |
| Deep fascia only | 18 (23%) |
| Superficial fascia only | 11 (14%) |
| Muscle + Superficial Fascia | 7 (9%) |
| Muscle + Deep Fascia | 14 (18%) |
| Deep Fascia + Superficial Fascia | 17 (22%) |
| Muscle + Deep Fascia + Superficial Fascia | 7 (9%) |
| None | 8 (10%) |
| Muscle Only | Deep Fascia Only | Superficial Fascia Only | Muscle + Superficial Fascia | Muscle + Deep Fascia | Deep Fascia + Superficial Fascia | Muscle + Deep Fascia + Superficial Fascia | None | |
|---|---|---|---|---|---|---|---|---|
| VAS (mean ± standard deviation) | 5.8 ± 1.8 | 6.1 ± 2.7 | 4.5 ± 1.7 | 6.0 ± 1.5 | 5.9 ± 1.8 | 5.8 ± 1.6 | 7.0 ± 1.7 | 5.9 ± 2.3 |
| Radiating (% of layer specific radiation) | 60% | 89% | 82% | 100% | 93% | 73% | 100% | 0% |
| Number of Treated Myofascial Points (% of All Treated Myofascial Points) | |
|---|---|
| Trapezius | 16 (18%) |
| Torso | 14 (16%) |
| Arm | 22 (25%) |
| Leg | 35 (40%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Vahdatinia, R.; Humbert, S.; Stecco, A. Myofascial Injection Using Fascial Layer-Specific Hydromanipulation Technique (FLuSH) and the Delineation of Multifactorial Myofascial Pain. Medicina 2020, 56, 717. https://doi.org/10.3390/medicina56120717
Wang T, Vahdatinia R, Humbert S, Stecco A. Myofascial Injection Using Fascial Layer-Specific Hydromanipulation Technique (FLuSH) and the Delineation of Multifactorial Myofascial Pain. Medicina. 2020; 56(12):717. https://doi.org/10.3390/medicina56120717
Chicago/Turabian StyleWang, Tina, Roya Vahdatinia, Sarah Humbert, and Antonio Stecco. 2020. "Myofascial Injection Using Fascial Layer-Specific Hydromanipulation Technique (FLuSH) and the Delineation of Multifactorial Myofascial Pain" Medicina 56, no. 12: 717. https://doi.org/10.3390/medicina56120717
APA StyleWang, T., Vahdatinia, R., Humbert, S., & Stecco, A. (2020). Myofascial Injection Using Fascial Layer-Specific Hydromanipulation Technique (FLuSH) and the Delineation of Multifactorial Myofascial Pain. Medicina, 56(12), 717. https://doi.org/10.3390/medicina56120717






