Performance of the IOTA ADNEX Model on Selected Group of Patients with Borderline Ovarian Tumours
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Design
2.3. Patients and Ultrasound Collection Parameters
2.4. IOTA ADNEX Model Specifications and Interpretation
2.5. Statistical Analysis
3. Results
3.1. Main Characteristics of Study Population
3.2. Performance of ADNEX Model According to Absolute and Relative Risk
3.3. The Value of Ca125 Marker in ADNEX Model for BOT Classification
3.4. The Performance of the ADNEX Model Between Different Histological Subtypes of BOTs
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tinelli, R.; Tinelli, A.; Tinelli, F.G.; Cicinelli, E.; Malvasi, A. Conservative surgery for borderline ovarian tumors: A review. Gynecol. Oncol. 2006, 100, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Trimble, C.L.; Trimble, E.L. Management of epithelial ovarian tumors of low malignant potential. Gynecol. Oncol. 1994, 55, S52–S61. [Google Scholar] [CrossRef] [PubMed]
- Morotti, M.; Menada, M.V.; Gillott, D.J.; Venturini, P.L.; Ferrero, S. The preoperative diagnosis of borderline ovarian tumors: A review of current literature. Arch. Gynecol. Obstet. 2012, 285, 1103–1112. [Google Scholar] [CrossRef] [PubMed]
- Skirnisdottir, I.; Garmo, H.; Wilander, E.; Holmberg, L. Borderline ovarian tumors in Sweden 1960-2005: Trends in incidence and age at diagnosis compared to ovarian cancer. Int. J. Cancer 2008, 123, 1897–1901. [Google Scholar] [CrossRef] [PubMed]
- Colombo, N.; Sessa, C.; du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent diseaset. Ann. Oncol 2019, 30, 672–705. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, I.; de Sousa Mendes, M. Conservative surgery in ovarian borderline tumours: A meta-analysis with emphasis on recurrence risk. Eur. J. Cancer 2015, 51, 620–631. [Google Scholar] [CrossRef] [PubMed]
- Kaijser, J. Towards an evidence-based approach for diagnosis and management of adnexal masses: Findings of the International Ovarian Tumour Analysis (IOTA) studies. Facts Views Vis. Obgyn 2015, 7, 42–59. [Google Scholar] [PubMed]
- Timmerman, D.; Schwärzler, P.; Collins, W.P.; Claerhout, F.; Coenen, M.; Amant, F.; Vergote, I.; Bourne, T.H. Subjective assessment of adnexal masses with the use of ultrasonography: An analysis of interobserver variability and experience. Ultrasound Obstet. Gynecol. 1999, 13, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Fruscella, E.; Testa, A.C.; Ferrandina, G.; De Smet, F.; Van Holsbeke, C.; Scambia, G.; Zannoni, G.F.; Ludovisi, M.; Achten, R.; Amant, F.; et al. Ultrasound features of different histopathological subtypes of borderline ovarian tumors. Ultrasound Obstet. Gynecol. 2005, 26, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Van Calster, B.; Van Hoorde, K.; Valentin, L.; Testa, A.C.; Fischerova, D.; Van Holsbeke, C.; Savelli, L.; Franchi, D.; Epstein, E.; Kaijser, J.; et al. Evaluating the risk of ovarian cancer before surgery using the ADNEX model to differentiate between benign, borderline, early and advanced stage invasive, and secondary metastatic tumours: Prospective multicentre diagnostic study. BMJ 2014, 349, 5920. [Google Scholar] [CrossRef] [PubMed]
- Van Calster, B.; Van Hoorde, K.; Froyman, W.; Kaijser, J.; Wynants, L.; Landolfo, C.; Anthoulakis, C.; Vergote, I.; Bourne, T.; Timmerman, D.; et al. Practical guidance for applying the ADNEX model from the IOTA group to discriminate between different subtypes of adnexal tumors. Facts Views Vis. Obgyn 2015, 7, 32–41. [Google Scholar] [PubMed]
- Eltabbakh, G.H.; Yadav, P.R.; Morgan, A. Clinical picture of women with early stage ovarian cancer. Gynecol. Oncol. 1999, 75, 476–479. [Google Scholar] [CrossRef]
- Fotopoulou, C.; Sehouli, J.; Ewald-Riegler, N.; de Gregorio, N.; Reuss, A.; Richter, R.; Mahner, S.; Kommoss, F.; Schmalfeldt, B.; Fehm, T.; et al. The Value of Serum CA125 in the Diagnosis of Borderline Tumors of the Ovary: A Subanalysis of the Prospective Multicenter ROBOT Study. Int. J. Gynecol. Cancer 2015, 25, 1248–1252. [Google Scholar] [CrossRef]
- May, J.; Skorupskaite, K.; Congiu, M.; Ghaoui, N.; Walker, G.A.; Fegan, S.; Martin, C.W.; O’Donnell, R.L. Borderline Ovarian Tumors: Fifteen Years’ Experience at a Scottish Tertiary Cancer Center. Int. J. Gynecol. Cancer 2018, 28, 1683–1691. [Google Scholar] [CrossRef] [PubMed]
- Moro, F.; Baima Poma, C.; Zannoni, G.F.; Vidal Urbinati, A.; Pasciuto, T.; Ludovisi, M.; Moruzzi, M.C.; Carinelli, S.; Franchi, D.; Scambia, G.; et al. Imaging in gynecological disease (12): Clinical and ultrasound features of invasive and non-invasive malignant serous ovarian tumors. Ultrasound Obstet. Gynecol. 2017, 50, 788–799. [Google Scholar] [CrossRef]
- Pascual, A.; Guerriero, S.; Rams, N.; Juez, L.; Ajossa, S.; Graupera, B.; Hereter, L.; Cappai, A.; Pero, M.; Perniciano, M.; et al. Clinical and ultrasound features of benign, borderline, and malignant invasive mucinous ovarian tumors. Eur. J. Gynaecol. Oncol. 2017, 38, 382–386. [Google Scholar]
| Median Age (Range) | 46.1 (17–83) |
|---|---|
| Tumours histology: | |
| Serous | 57 (67.1%) |
| Mucinous | 22 (25.9%) |
| Other: | |
| Endometrioid | 4 (4.7 %) |
| Mixed | 2 (2.3%) |
| FIGO Stage: | |
| I | 74 (87.1 %) |
| II–III | 11 (12.9 %) |
| Median value of Ca125 tumour marker (range), U/mL | 72.8 (5.9–918.0) * |
| Ultrasound Feature | Median (Interval) or n (%) |
|---|---|
| Maximal diameter of the tumours (mm) | 80.0 (20–639) |
| Maximal diameter of the solid part (mm) | 15.0 (3–73) |
| Type of tumour: | |
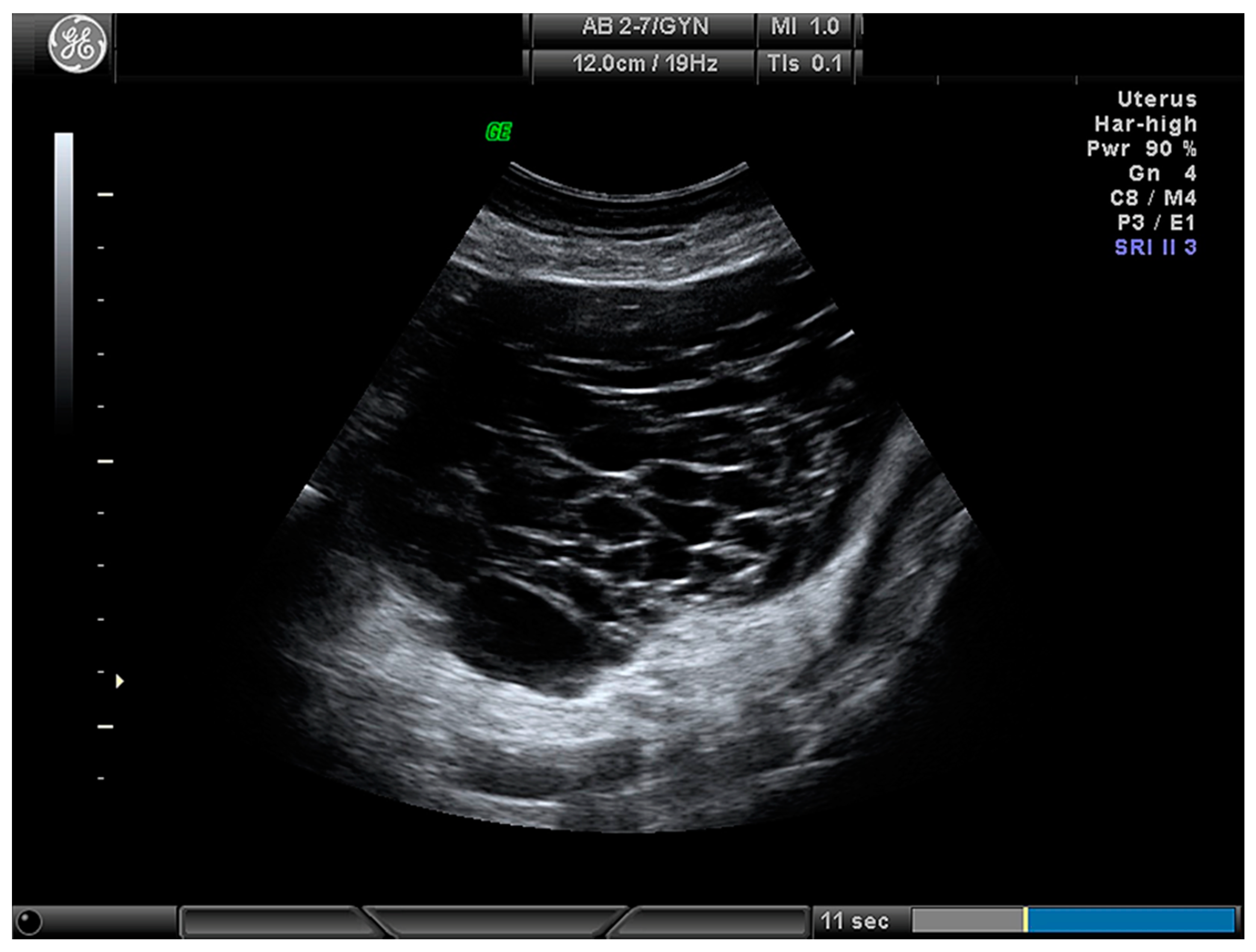
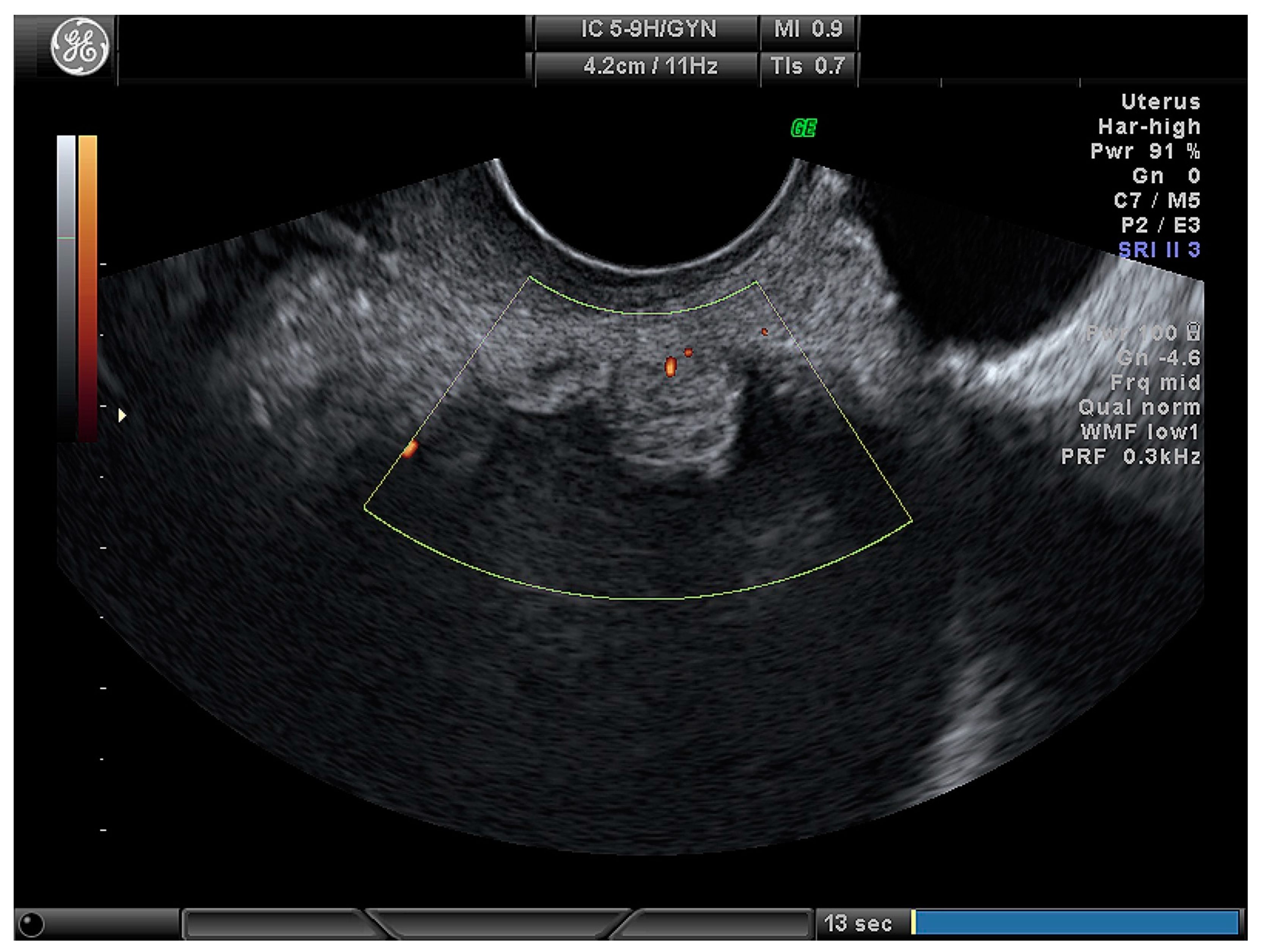
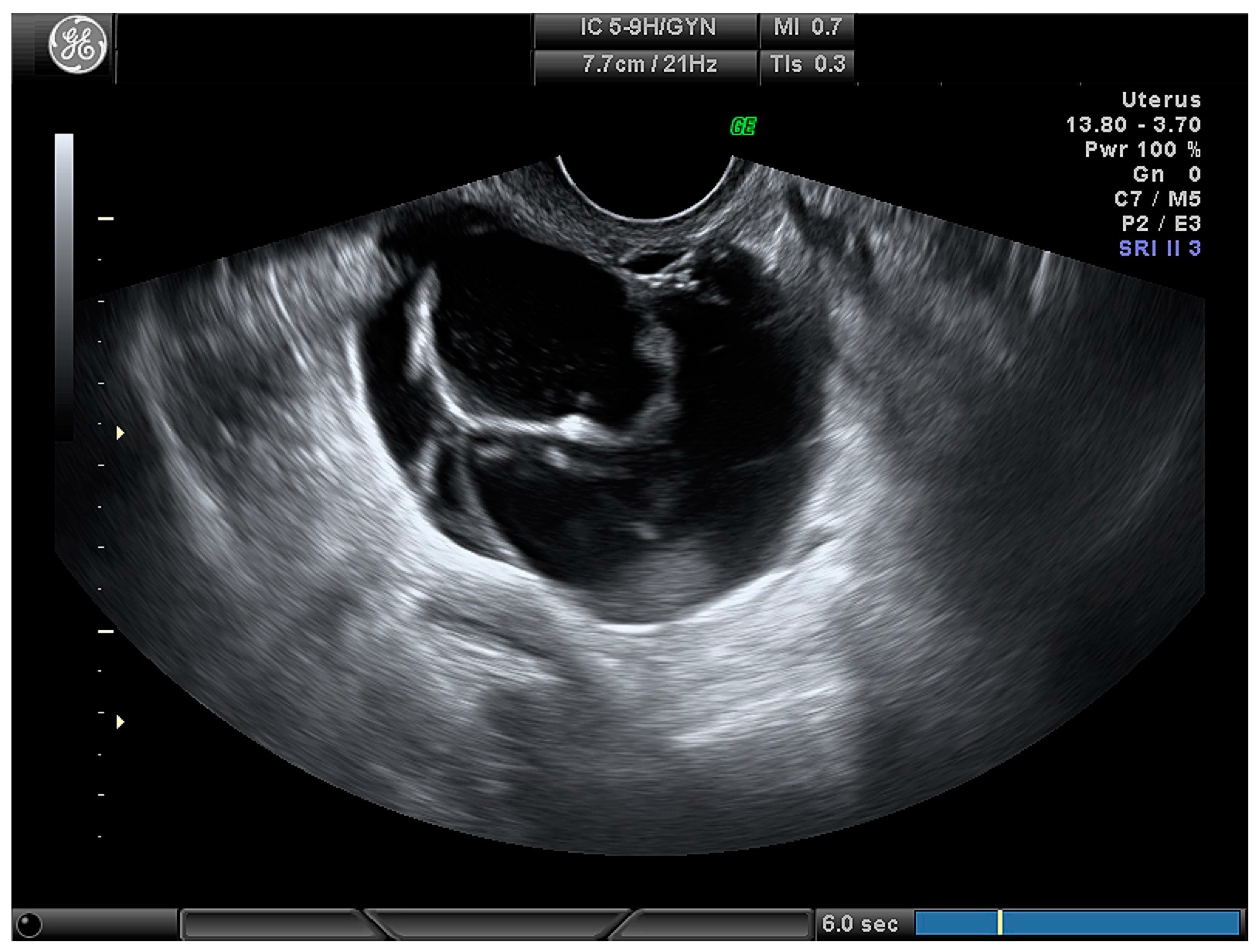
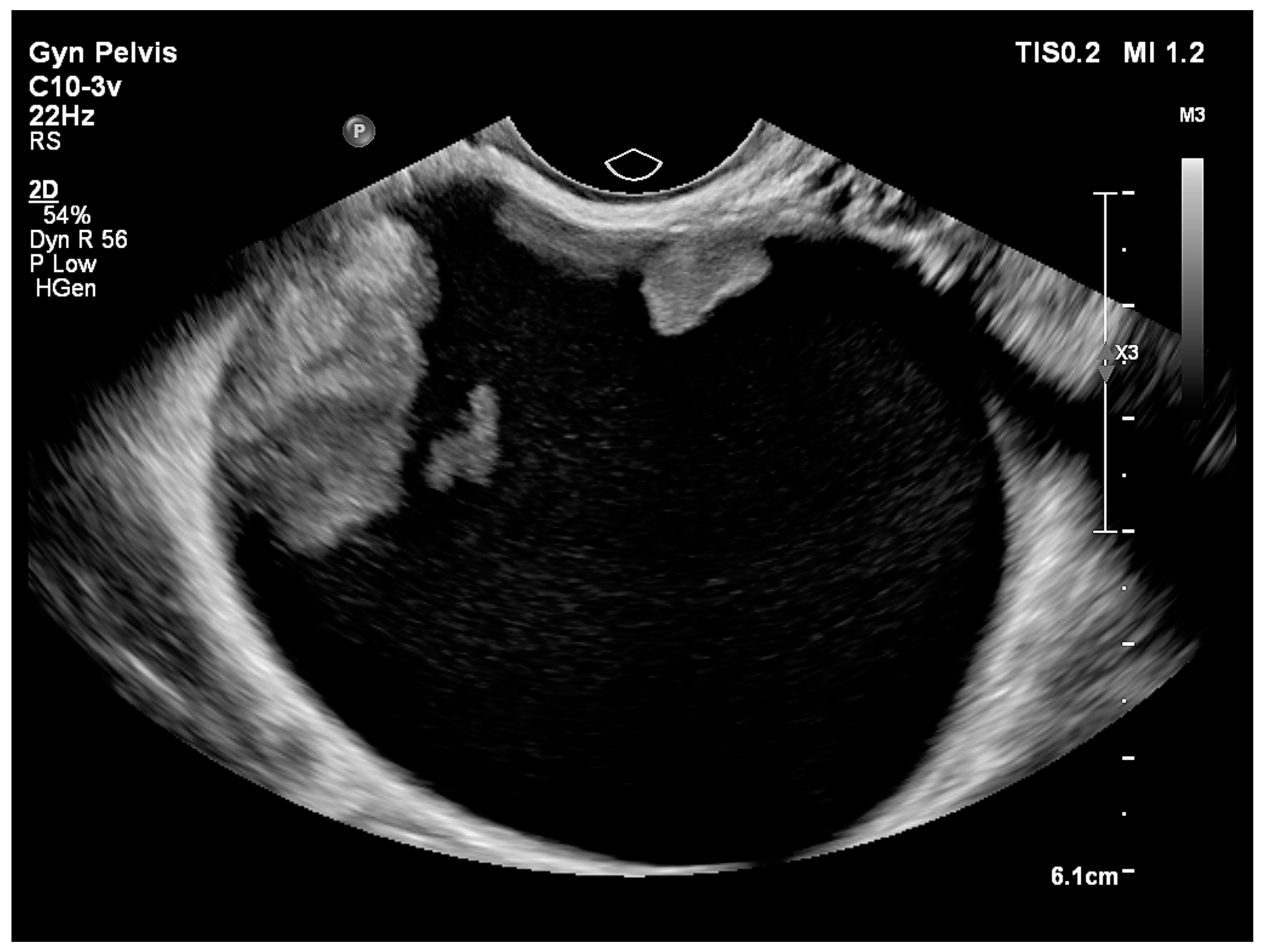
| Cystic (Figure 3) | 24 (28.2) |
| Cystic–solid (Figure 4) | 61 (71.8) |
| Number of locules: | |
| Unilocular | 41 (48.2) |
| Multiloculcar (total) | 44 (51.8) |
| More than 10 locules | 30 (35.3) |
| Number of papillary projections: | |
| None | 30 (35.3) |
| 1 | 15 (17.6) |
| 2 | 5 (5.9) |
| 3 | 2 (2.4) |
| More than 3 | 33 (38.8) |
| Ascites present | 6 (7.1) |
| Ultrasound Feature | Serous BOT | Mucinous BOT | Serous vs. Mucinous |
|---|---|---|---|
| (n = 57) | (n = 22) | ||
| Median (Range) or n (%) | |||
| Type of tumour: | p < 0.001 | ||
| Cystic | 7 (12.3) | 12 (54.5) | |
| Cystic–solid | 50 (87.7) | 10 (45.5) | |
| Number of locules: | p = 0.008 | ||
| Uniloculcar | 32 (56.1) | 5 (22.7) | |
| Multilocular | 25 (43.9) | 17 (77.3) | |
| More than 10 locules present | 14 (24.6) | 14 (63.6) | p = 0.001 |
| Presence of papillary projections | 46 (80.7) | 8 (36.4) | p < 0.001 |
| Maximal diameter of the tumour (mm) | 72.0 (20.5–639.0) | 135 (25.7–300) | p = 0.003 |
| Maximal diameter of solid part (mm) | 15.0 (3.0–73.0) | 21.5 (4.0–66.0) | p = 0.003 |
| ADNEX Result | Number (%) of Cases According to AR with Different Cut-Off Values | Number (%) of Cases According to RR | |||
|---|---|---|---|---|---|
| 3% | 5% | 10% | 20% | ||
| Benign | 1 (1.3%) | 5 (6.4%) | 12 (15.4%) | 21 (26.9%) | 13 (16.7%) |
| BOT | 67 (85.9%) | 63 (80.8%) | 56 (71.2%) | 47 (60.3%) | 59 (75.6%) |
| Stage I invasive Ca | 3 (3.8%) | 3 (3.8%) | 3 (3.8%) | 3 (3.8%) | 5 (6.4%) |
| Stage II–IV invasive Ca | 7 (9.0%) | 7 (9.0%) | 7 (9.0%) | 7 (9.0%) | 1 (1.3%) |
| Cut-Off | Correctly Classified BOT Cases N (%) | Absolute Change in Case Number (%) | p-Value |
|---|---|---|---|
| 20% | 47 (60.3%) | − | − |
| 10% | 56 (71.2%) | +9 (11.5 %) | 0.004 |
| 5% | 63 (80.8%) | +7 (8.97%) | 0.016 |
| 3% | 67 (85.9%) | +4 (5.1%) | 0.375 |
| Cut-Off Value | Difference of the Case Number According to AR | p-Value |
|---|---|---|
| 20% | −12 (15.4%) | <0.001 |
| 10% | −3 (3.9%) | 0.375 |
| 5% | +4 (5.1%) | 0.388 |
| 3% | +7 (9.0%) | 0.118 |
| Without Ca125 | Serous BOTs (n = 57) | Mucinous BOTs (n = 22) | p-Value (Serous vs. Mucinous) |
|---|---|---|---|
| AR (cut-off 10%) | 40 (70.2%) | 16 (72.7%) | 0.823 |
| RR | 44 (77.2%) | 15 (68.2%) | 0.409 |
| p-value (AR vs. RR) | 0.289 | 1.000 | − |
| With Ca125 | Serous BOTs (n = 53) | Mucinous BOTs (n = 20) | p-Value (Serous vs. Mucinous) |
| AR (cut-off 10%) | 40 (75.5%) | 15 (75.0%) | 0.967 |
| RR | 42 (79.2%) | 15 (75.0%) | 0.696 |
| p-value (AR vs. RR) | 0.625 | 1.000 | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaurilcikas, A.; Gedgaudaite, M.; Cizauskas, A.; Atstupenaite, V.; Paskauskas, S.; Gaurilcikiene, D.; Birzietis, T.; Vaitkiene, D.; Nadisauskiene, R.J. Performance of the IOTA ADNEX Model on Selected Group of Patients with Borderline Ovarian Tumours. Medicina 2020, 56, 690. https://doi.org/10.3390/medicina56120690
Gaurilcikas A, Gedgaudaite M, Cizauskas A, Atstupenaite V, Paskauskas S, Gaurilcikiene D, Birzietis T, Vaitkiene D, Nadisauskiene RJ. Performance of the IOTA ADNEX Model on Selected Group of Patients with Borderline Ovarian Tumours. Medicina. 2020; 56(12):690. https://doi.org/10.3390/medicina56120690
Chicago/Turabian StyleGaurilcikas, Adrius, Migle Gedgaudaite, Arvydas Cizauskas, Vaida Atstupenaite, Saulius Paskauskas, Dovile Gaurilcikiene, Tomas Birzietis, Daiva Vaitkiene, and Ruta Jolanta Nadisauskiene. 2020. "Performance of the IOTA ADNEX Model on Selected Group of Patients with Borderline Ovarian Tumours" Medicina 56, no. 12: 690. https://doi.org/10.3390/medicina56120690
APA StyleGaurilcikas, A., Gedgaudaite, M., Cizauskas, A., Atstupenaite, V., Paskauskas, S., Gaurilcikiene, D., Birzietis, T., Vaitkiene, D., & Nadisauskiene, R. J. (2020). Performance of the IOTA ADNEX Model on Selected Group of Patients with Borderline Ovarian Tumours. Medicina, 56(12), 690. https://doi.org/10.3390/medicina56120690





