Vitamin D Deficiency: Consequence or Cause of Obesity?
Abstract
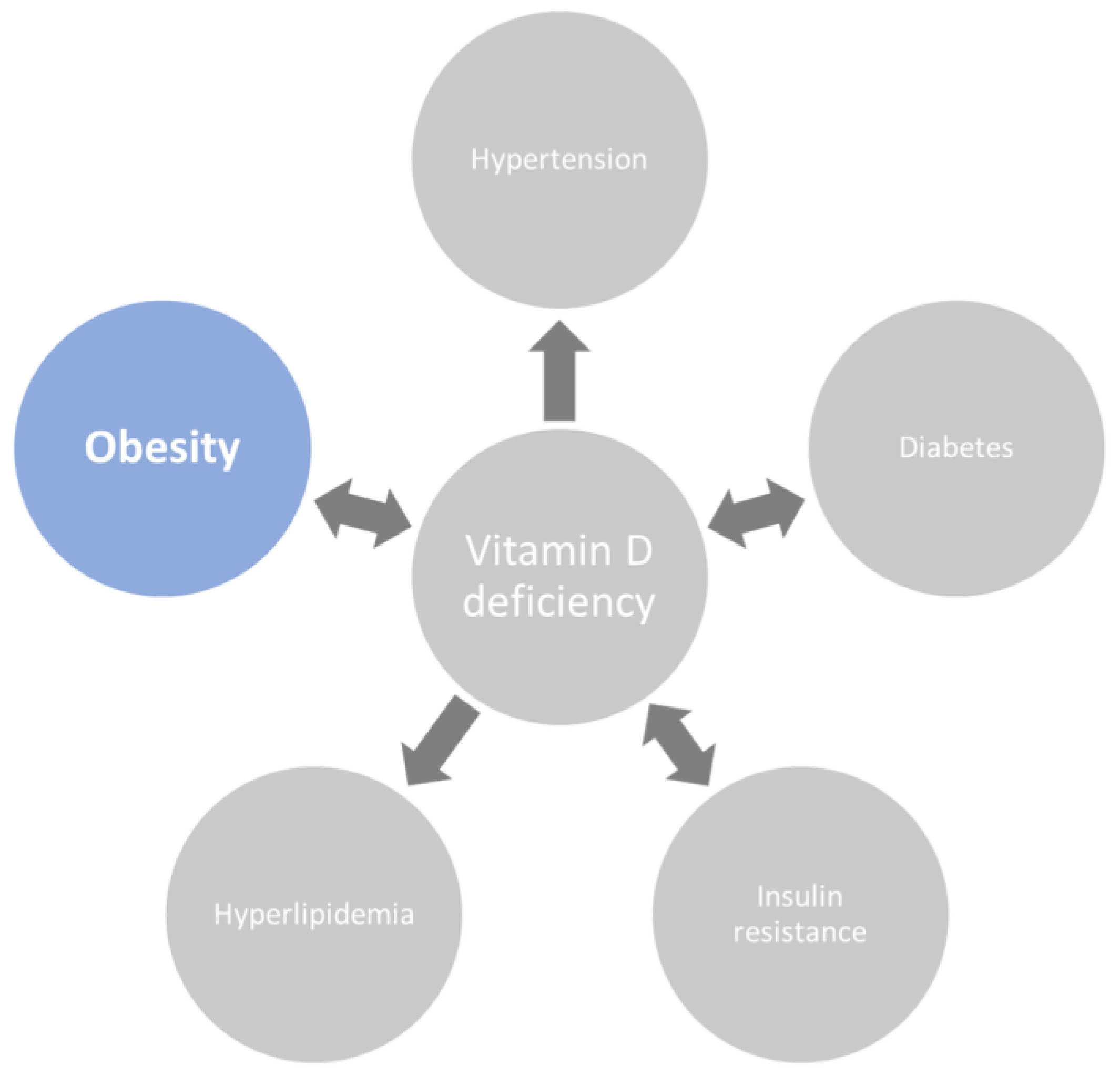
1. Introduction
2. Clinical Consequences of Lower 25(OH)D
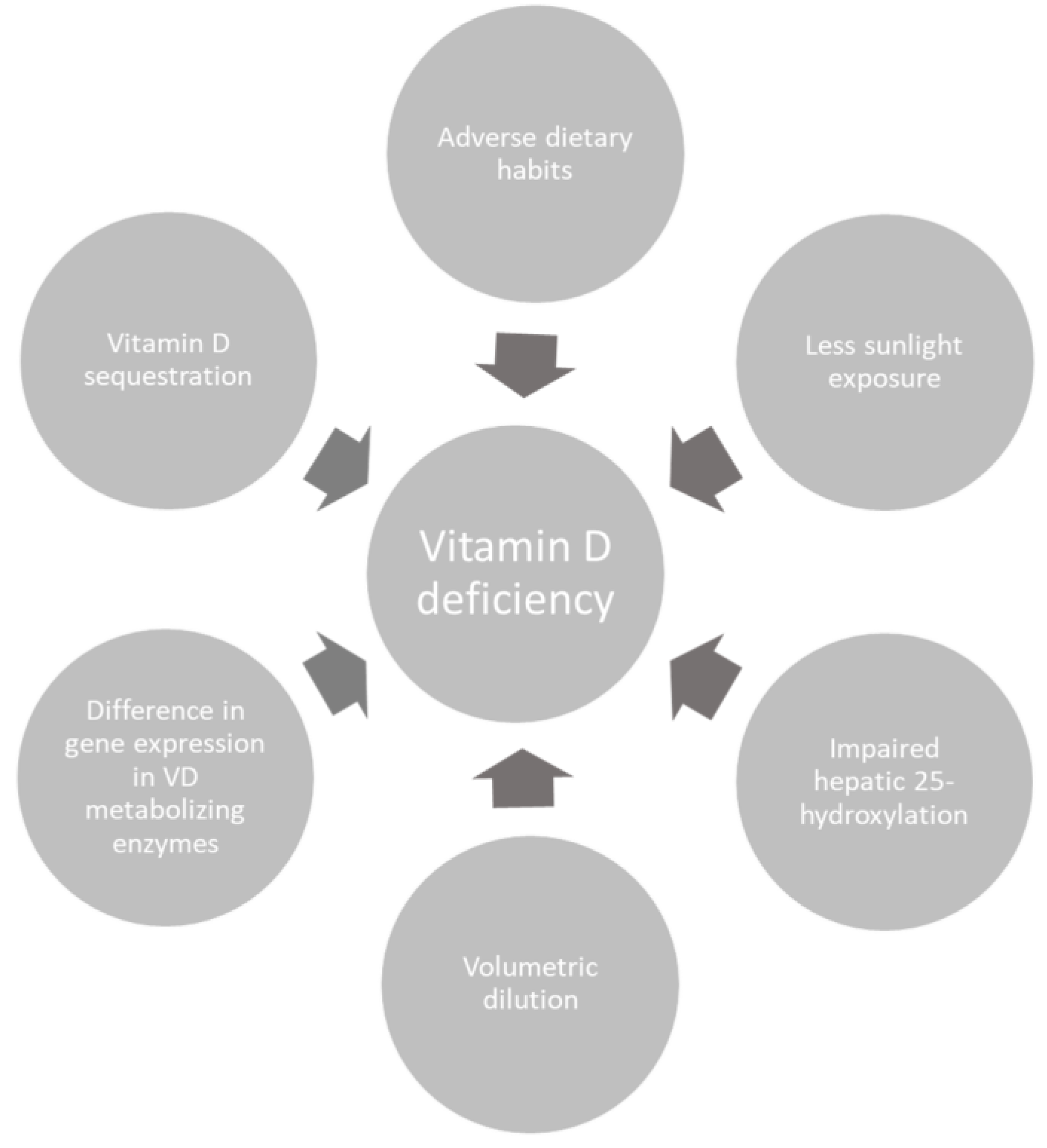
3. Causes of Low 25-Hydroxyvitamin D in Obese Patients
4. Effects of Vitamin D Supplementation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lips, P. Vitamin D physiology. Prog. Biophys. Mol. Biol. 2006, 92, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 Report on Dietary Reference Intakes for Calcium and Vitamin D from the Institute of Medicine: What Clinicians Need to Know. Obstet. Gynecol. Surv. 2011, 66, 356–357. [Google Scholar] [CrossRef]
- Costa, P.R.F.; Assis, A.M.O.; Santos, C.A.S.T.; Santos, D.B.; Pereira-Santos, M.; Pereira-Santos, M.; Pereira-Santos, M. Obesity and vitamin D deficiency: A systematic review and meta-analysis. Obes. Rev. 2015, 16, 341–349. [Google Scholar]
- Milic, S.; Mikolasevic, I.; Krznaric-Zrnic, I.; Stanic, M.; Poropat, G.; Stimac, D.; Vlahović-Palčevski, V.; Orlic, L. Nonalcoholic steatohepatitis: Emerging targeted therapies to optimize treatment options. Drug Des. Dev. Ther. 2015, 9, 4835–4845. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.L.; Paggiosi, M.A.; Eastell, R.; Walsh, J.S. Bone Density, Microstructure and Strength in Obese and Normal Weight Men and Women in Younger and Older Adulthood. J. Bone Miner. Res. 2015, 30, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.S.; Bowles, S.; Evans, A.L. Vitamin D in obesity. Curr. Opin. Endocrinol. Diabetes Obes. 2017, 24, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.B.; Ames, R.W.; Mason, B.H.; Horne, A.M.; Gamble, G.D.; Reid, I.R. The effects of seasonal variation of 25-hydroxyvitamin D and fat mass on a diagnosis of vitamin D sufficiency. Am. J. Clin. Nutr. 2007, 86, 959–964. [Google Scholar] [CrossRef]
- Gallagher, J.C.; Yalamanchili, V.; Smith, L.M. The effect of vitamin D supplementation on serum 25(OH)D in thin and obese women. J. Steroid Biochem. Mol. Biol. 2013, 136, 195–200. [Google Scholar] [CrossRef]
- Drincic, A.; Fuller, E.; Heaney, R.P.; Armas, L.A.G. 25-Hydroxyvitamin D Response to Graded Vitamin D3Supplementation Among Obese Adults. J. Clin. Endocrinol. Metab. 2013, 98, 4845–4851. [Google Scholar] [CrossRef]
- Carrelli, A.; Bucovsky, M.; Horst, R. Vitamin D storage in adipose tissue of obese and normal weight women. J. Bone Miner. Res. 2017, 32, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Wamberg, L.; Christiansen, T.; Paulsen, S.K. Expression of vitamin D-metabolizing enzymes in human adipose tissue—The effect of obesity and diet-induced weight loss. Int. J. Obes. 2013, 37, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Emond, J.A.; Flatt, S.W.; Heath, D.D.; Karanja, N.; Pakiz, B.; Sherwood, N.E.; Thomson, C.A. Weight Loss Is Associated with Increased Serum 25-Hydroxyvitamin D in Overweight or Obese Women. Obesity 2012, 20, 2296–2301. [Google Scholar] [CrossRef] [PubMed]
- Mason, C.; Xiao, L.; Imayama, I.; Duggan, C.R.; Bain, C.; Foster-Schubert, K.E.; Kong, A.; Campbell, K.L.; Wang, C.-Y.; Neuhouser, M.L.; et al. Effects of weight loss on serum vitamin D in postmenopausal women12345. Am. J. Clin. Nutr. 2011, 94, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Wortsman, J.; Matsuoka, L.Y.; Chen, T.C.; Lu, Z.; Holick, M.F. Decreased bioavailability of vitamin D in obesity. Am. J. Clin. Nutr. 2000, 72, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Gangloff, A.; Bergeron, J.; Lemieux, I.; Després, J.-P. Changes in circulating vitamin D levels with loss of adipose tissue. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 464–470. [Google Scholar] [CrossRef]
- Targher, G.; Bertolini, L.; Scala, L.; Cigolini, M.; Zenari, L.; Falezza, G.; Arcaro, G. Associations between serum 25-hydroxyvitamin D3 concentrations and liver histology in patients with non-alcoholic fatty liver disease. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 517–524. [Google Scholar] [CrossRef]
- Mikolasević, I.; Milic, S.; Wensveen, T.T.; Grgic, I.; Jakopcic, I.; Štimac, D.; Wensveen, F.; Orlic, L. Nonalcoholic fatty liver disease—A multisystem disease? World J. Gastroenterol. 2016, 22, 9488–9505. [Google Scholar] [CrossRef]
- Walsh, J.S.; Evans, A.L.; Bowles, S.; Naylor, K.E.; Jones, K.S.; Schoenmakers, I.; Jacques, R.M.; Eastell, R. Free 25-hydroxyvitamin D is low in obesity, but there are no adverse associations with bone health. Am. J. Clin. Nutr. 2016, 103, 1465–1471. [Google Scholar] [CrossRef]
- Macdonald, H.M.; Mavroeidi, A.; Aucott, L.A.; Diffey, B.L.; Fraser, W.D.; Ormerod, A.D.; Reid, D.M. Skin Color Change in Caucasian Postmenopausal Women Predicts Summer-Winter Change in 25-Hydroxyvitamin D: Findings from the ANSAViD Cohort Study. J. Clin. Endocrinol. Metab. 2011, 96, 1677–1686. [Google Scholar] [CrossRef][Green Version]
- Wamberg, L.; Pedersen, S.B.; Rejnmark, L.; Richelsen, B. Causes of Vitamin D Deficiency and Effect of Vitamin D Supplementation on Metabolic Complications in Obesity: A Review. Curr. Obes. Rep. 2015, 4, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Wood, R.J. Vitamin D and adipogenesis: New molecular insights. Nutr. Rev. 2008, 66, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Martini, L.A.; Wood, R.J. Vitamin D Status and the Metabolic Syndrome. Nutr. Rev. 2006, 64, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, J.M.; Tzameli, I.; Astapova, I.; Lam, F.S.; Flier, J.S.; Hollenberg, A.N. Complex Role of the Vitamin D Receptor and Its Ligand in Adipogenesis in 3T3-L1 Cells. J. Boil. Chem. 2006, 281, 11205–11213. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Bae, S.; Yoon, Y. Anti-adipogenic effects of 1,25-dihydroxyvitamin D3 are mediated by the maintenance of the wingless-type MMTV integration site/beta-catenin pathway. Int. J. Mol. Med. 2012, 30, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Leblanc, E.S.; Rizzo, J.H.; Pedula, K.L.; Ensrud, K.E.; Cauley, J.; Hochberg, M.; Hillier, T.A. Associations Between 25-Hydroxyvitamin D and Weight Gain in Elderly Women. J. Women’s Health 2012, 21, 1066–1073. [Google Scholar] [CrossRef]
- Mai, X.M.; Chen, Y.; Camargo, C.A., Jr. Cross-sectional and prospective cohort study of serum 25-hydroxyvitamin D level and obesity in adults: The HUNT study. Am. J. Epidemiol. 2012, 175, 1029–1036. [Google Scholar] [CrossRef]
- Zittermann, A.; Frisch, S.; Berthold, H.K.; Götting, C.; Kuhn, J.; Kleesiek, K.; Stehle, P.; Koertke, H.; Koerfer, R. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers. Am. J. Clin. Nutr. 2009, 89, 1321–1327. [Google Scholar] [CrossRef]
- Wamberg, L.; Kampmann, U.; Stødkilde-Jørgensen, H.; Rejnmark, L.; Pedersen, S.B.; Richelsen, B. Effects of vitamin D supplementation on body fat accumulation, inflammation, and metabolic risk factors in obese adults with low vitamin D levels—Results from a randomized trial. Eur. J. Intern. Med. 2013, 24, 644–649. [Google Scholar] [CrossRef]
- Kampmann, U.; Mosekilde, L.; Juhl, C.; Moller, N.; Christensen, B.; Rejnmark, L.; Wamberg, L.; Orskov, L. Effects of 12weeks high dose vitamin D3 treatment on insulin sensitivity, beta cell function, and metabolic markers in patients with type 2 diabetes and vitamin D insufficiency—A double-blind, randomized, placebo-controlled trial. Metabolism 2014, 63, 1115–1124. [Google Scholar] [CrossRef]
- Mason, C.; Xiao, L.; Imayama, I.; Duggan, C.; Wang, C.-Y.; Korde, L.; McTiernan, A. Vitamin D3 supplementation during weight loss: A double-blind randomized controlled trial123. Am. J. Clin. Nutr. 2014, 99, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, L.A.; Struthers, A.D.; Khan, F. D-PRESSURE Collaboration. Effect of Vitamin D Supplementation on Blood Pressure: A Systematic Review and Meta-analysis Incorporating Individual Patient Data. JAMA Intern Med. 2015, 175, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, L.A.; Witham, M.D. Controversy in the link between vitamin D supplementation and hypertension. Expert Rev. Cardiovasc. Ther. 2015, 13, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Barnes, G.T.; Yang, Q.; Tan, G.; Yang, D.; Chou, C.J.; Sole, J.; Nichols, A.; Ross, J.S.; Tartaglia, L.A.; et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Investig. 2003, 112, 1821–1830. [Google Scholar] [CrossRef] [PubMed]
- Berg, A.H.; Scherer, P.E. Adipose Tissue, Inflammation, and Cardiovascular Disease. Circ. Res. 2005, 96, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Wamberg, L.; Cullberg, K.; Rejnmark, L.; Richelsen, B.; Pedersen, S.B. Investigations of the Anti-inflammatory Effects of Vitamin D in Adipose Tissue: Results from an In Vitro Study and a Randomized Controlled Trial. Horm. Metab. Res. 2013, 45, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Lorente-Cebrian, S.; Eriksson, A.; Dunlop, T. Differential effects of 1alpha,25-dihydroxycholecalciferol on MCP-1 and adiponectin production in human white adipocytes. Eur. J. Nutr. 2012, 51, 335–342. [Google Scholar] [CrossRef]
- Schleithoff, S.S.; Zittermann, A.; Tenderich, G.; Berthold, H.K.; Stehle, P.; Koerfer, R. Vitamin D supplementation improves cytokine profiles in patients with congestive heart failure: A double-blind, randomized, placebo-controlled trial. Am. J. Clin. Nutr. 2006, 83, 754–759. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Harris, S.S.; Stark, P.C.; Dawson-Hughes, B.; Msc, D. The Effects of Calcium and Vitamin D Supplementation on Blood Glucose and Markers of Inflammation in Nondiabetic Adults. Diabetes Care 2007, 30, 980–986. [Google Scholar] [CrossRef]
- Tai, K.; Need, A.G.; Horowitz, M.; Chapman, I.M. Vitamin D, glucose, insulin, and insulin sensitivity. Nutrition 2008, 24, 279–285. [Google Scholar] [CrossRef]
- Need, A.G.; O’Loughlin, P.D.; Horowitz, M.; Nordin, B.E.C. Relationship between fasting serum glucose, age, body mass index and serum 25 hydroxyvitamin D in postmenopausal women. Clin. Endocrinol. 2005, 62, 738–741. [Google Scholar] [CrossRef] [PubMed]
- Baynes, K.C.R.; Boucher, B.J.; Feskens, E.J.M.; Kromhout, D. Vitamin D, glucose tolerance and insulinaemia in elderly men. Diabetologia 1997, 40, 344–347. [Google Scholar] [CrossRef] [PubMed]
- Chiu, K.C.; Chu, A.; Go, V.L.W. Hypovitaminosis D is associated with insulin resistance and beta cell dysfunction. Am. J. Clin. Nutr. 2004, 79, 820–825. [Google Scholar] [CrossRef]
- von Hurst, P.R.; Stonehouse, W.; Coad, J. Vitamin D supplementation reduces insulin resistance in South Asian women living in New Zealand who are insulin resistant and vitamin D deficient—A randomised, placebo-controlled trial. Br. J. Nutr. 2010, 103, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Grimnes, G.; Emaus, N.; Joakimsen, R.M.; Figenschau, Y.; Jenssen, T.; Njølstad, I.; Schirmer, H.; Jorde, R. Baseline serum 25-hydroxyvitamin D concentrations in the Tromsø Study 1994-95 and risk of developing type 2 diabetes mellitus during 11 years of follow-up. Diabet. Med. 2010, 27, 1107–1115. [Google Scholar] [CrossRef]
- Tai, K.; Need, A.G.; Horowitz, M.; Chapman, I.M. Glucose tolerance and vitamin D: Effects of treating vitamin D deficiency. Nutrition 2008, 24, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Drincic, A.T.; Armas, L.A.; Van Diest, E.E.; Heaney, R.P. Volumetric Dilution, Rather Than Sequestration Best Explains the Low Vitamin D Status of Obesity. Obesity 2012, 20, 1444–1448. [Google Scholar] [CrossRef]
| Ref | Study Design | Study Size | Major Findings |
|---|---|---|---|
| Evans et al. [5] | Cross-sectional case-control study | 200 | Obesity has a positive impact on peak bone mass acquisition and also obese subjects have greater cortical thickness and cortical tissue mineral density. |
| Bolland et al. [8] | Cross-sectional study | 1984 | Vitamin D serum levels show seasonal variations. |
| Gallagher et al. [9] | Randomized, double-blind placebo controlled study | 163 | Obese subjects respond with smaller 25(OH)D rise in serum after VD supplementation in comparison to normal weight group. |
| Drincic et al. [10] | Randomized, single-blind study | 67 | 25(OH)D response after VD supplementation is 30% lower in the obese group. VD supplementation should be adjusted according for body size. |
| Wamberg et al. [12] | Cross-sectional study | 40 | Due to different expression of liver enzymes between obese and normal weight groups, adipose tissue can metabolize VD locally which can be altered after weight loss. |
| Rock et al. [13] | Prospective randomized clinical trial | 383 | Weight loss increases 25(OH)D serum concentration |
| Mason et al. [14] | Prospective randomized controlled trial | 439 | Weight loss of 15% of body weight and above increases 25(OH)D significantly, otherwise weight loss does not impact on serum 25(OH)D |
| Walsh et al. [19] | Cross-sectional observational study | 223 | Total and free 25(OH)D and 1,25(OH)2D are lower at higher BMI does not impact bone structure and health. |
| Macdonald et al. [20] | Prospective observational cohort study | 314 | Vitamin D deficiency is not related to reduced sun exposure in obese woman. Vitamin D serum concentrations seasonally changes. |
| LeBlanc et al. [26] | Prospective longitudinal study | 9704 | Vitamin D deficiency predisposes for obesity, higher doses of VD are related to lower weight gain |
| Mai et al. [27] | Cross-sectional and prospective cohort study | 25,616 | Low plasma 25(OH)D level (less than 50 nmol/L) was related to higher BMI and waist circumference. |
| Zittermann et al. [28] | Randomized, double-blind placebo-controlled study | 200 | VD supplementation has positive impact on several cardiovascular disease risk markers in obese, but does not adversely affect weight loss |
| Wamberg et al. [29] | Randomized, double-blind placebo-controlled study | 52 | VD supplementation has no effect on obesity-related complication nor on body weight reduction. |
| Kampmann et al. [30] | Randomized, double-blind placebo-controlled study | 16 | VD supplementation does not improve insulin resistance, blood pressure, inflammation or HbA1c, but might increase insulin secretion. |
| Mason et al. [31] | Randomized, double-blind placebo-controlled study | 218 | VD supplementation does not reduce body weight. |
| Wamberg et al. [36] | Randomized, double-blind placebo-controlled study | 40 | Inflammatory cytokines and inflammatory markers expression in adipose tissue were not reduced after VD supplementation, nevertheless it had significant inflammatory effects in AT in vitro. |
| Schleithoff et al. [38] | Randomized, double-blind placebo-controlled study | 123 | Improvement of VD status decreased plasma proinflammatory cytokines in patients with congestive heart failure. |
| Pittas et al. [39] | Randomized, double-blind placebo-controlled study | 314 | VD and calcium supplementation did not affect circulating levels of cytokines but attenuate increases in glycemia and insulin resistance in nondiabetic subjects. |
| Baynes et al. [42] | Prospective population-based cross-sectional study | 142 | VD hypovitaminosis is associated to hyperglycemia. |
| van Hurst et al. [44] | Randomized, double-blind placebo-controlled study | 81 | VD supplementation improves insulin sensitivity, but has no effect on insulin secretion. |
| Drincic et al. [47] | Cross-sectional study | 686 | Body weight and VD plasma levels are conversely associated due to volumetric dilution. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vranić, L.; Mikolašević, I.; Milić, S. Vitamin D Deficiency: Consequence or Cause of Obesity? Medicina 2019, 55, 541. https://doi.org/10.3390/medicina55090541
Vranić L, Mikolašević I, Milić S. Vitamin D Deficiency: Consequence or Cause of Obesity? Medicina. 2019; 55(9):541. https://doi.org/10.3390/medicina55090541
Chicago/Turabian StyleVranić, Luka, Ivana Mikolašević, and Sandra Milić. 2019. "Vitamin D Deficiency: Consequence or Cause of Obesity?" Medicina 55, no. 9: 541. https://doi.org/10.3390/medicina55090541
APA StyleVranić, L., Mikolašević, I., & Milić, S. (2019). Vitamin D Deficiency: Consequence or Cause of Obesity? Medicina, 55(9), 541. https://doi.org/10.3390/medicina55090541




