Effects of Different Light Sources on Neural Activity of the Paraventricular Nucleus in the Hypothalamus
Abstract
1. Introduction
2. Methods
2.1. Experimental Animals
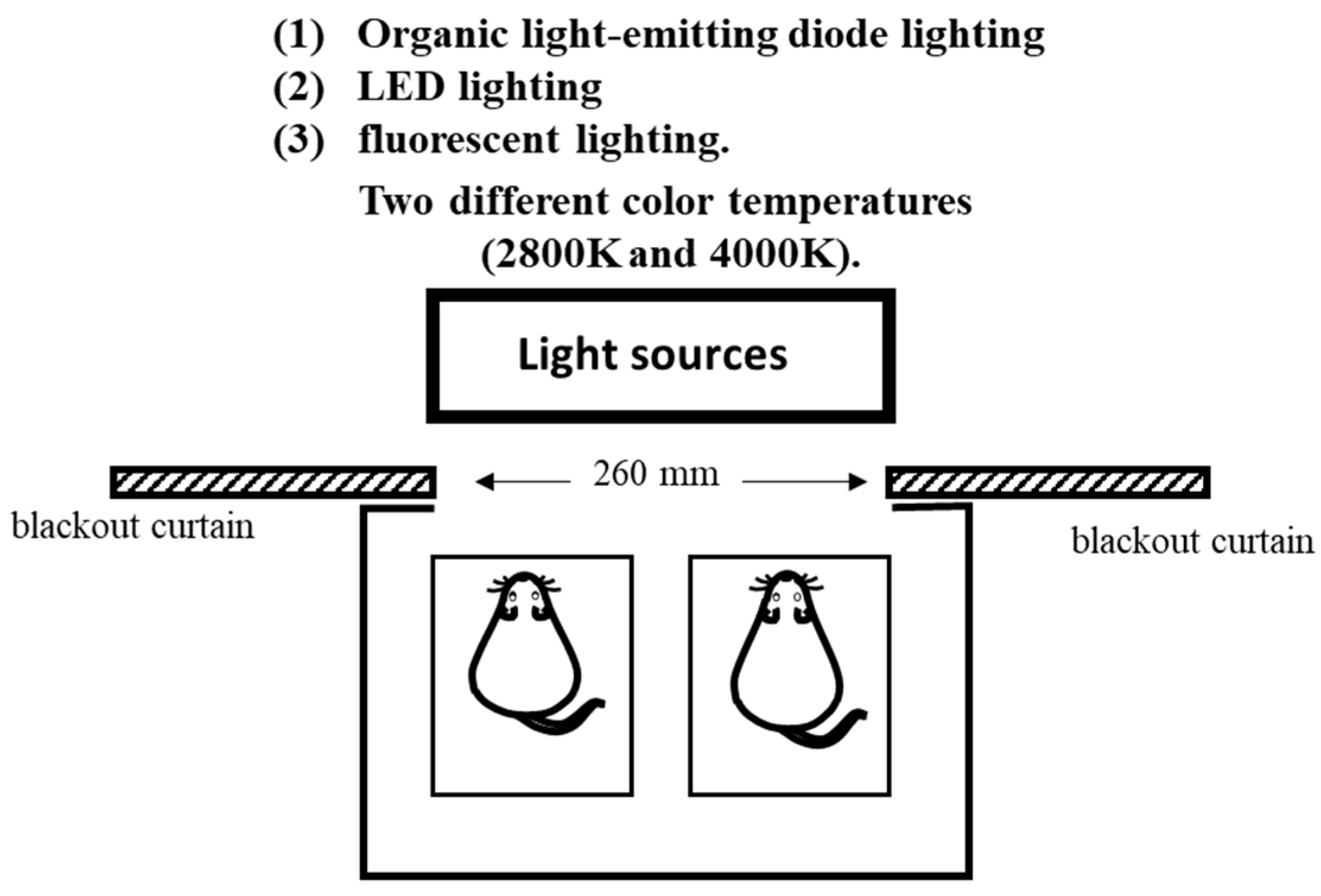
2.2. Experimental Design
2.3. Immunohistochemistry
2.4. Data Analysis
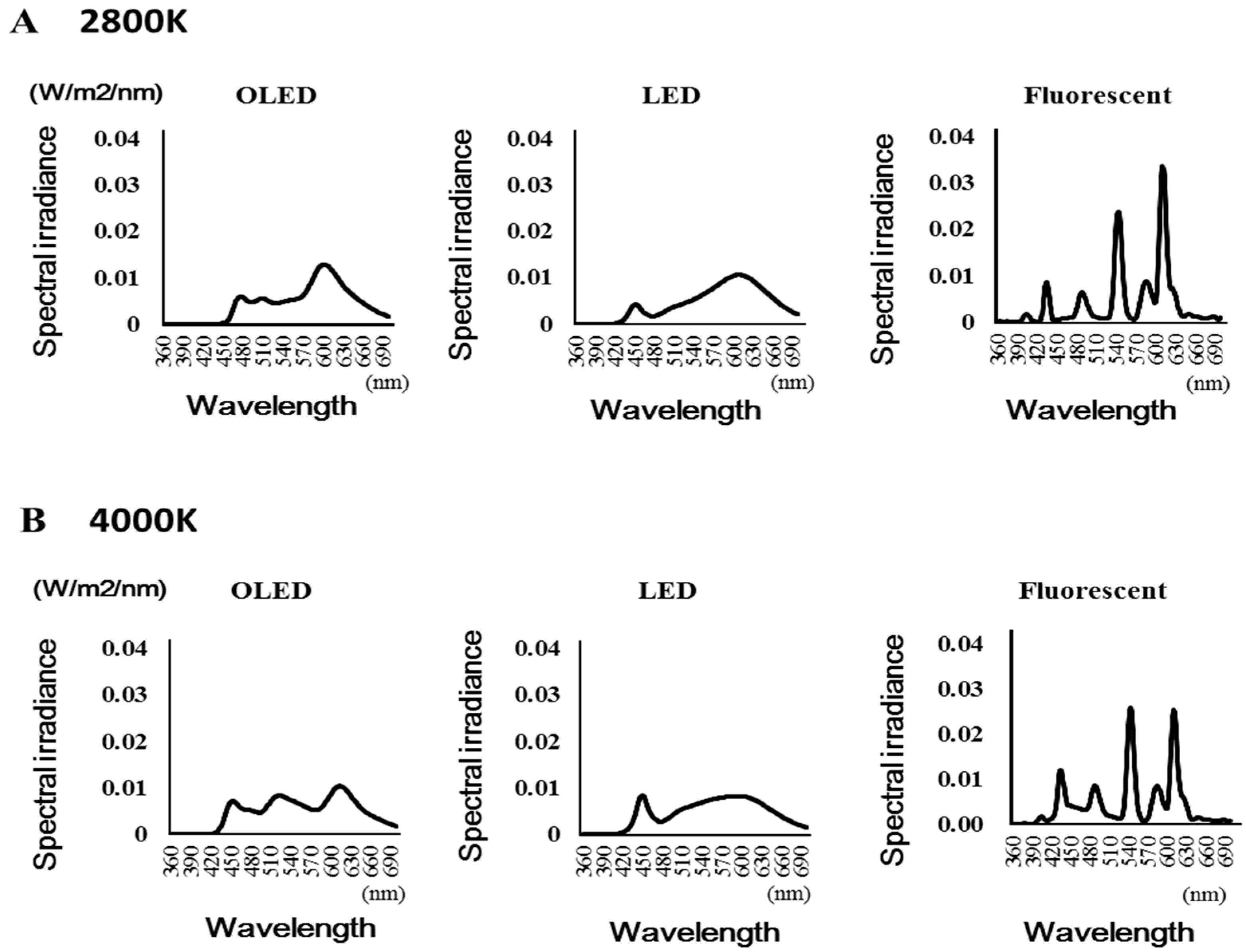
2.4.1. Light Source Analysis
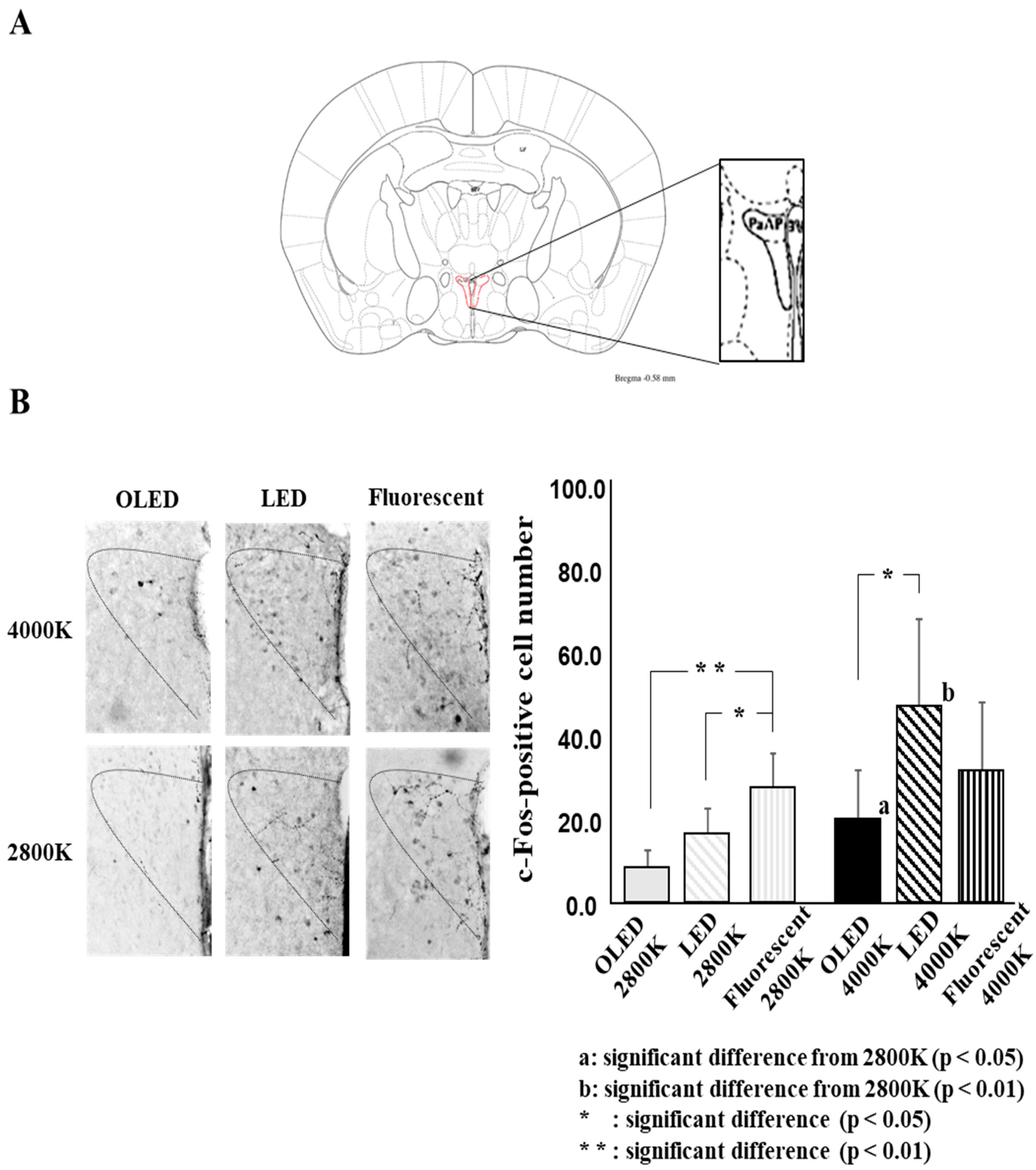
2.4.2. Analysis of the c-Fos-Positive Cell Number
2.5. Statistics
3. Results
3.1. Light Irradiation
3.2. c-Fos-Positive Cell Number
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chellappa, S.L.; Lasauskaite, R.; Cajochen, C. In a Heartbeat: Light and Cardiovascular Physiology. Front. Neurol. 2017, 8, 541. [Google Scholar] [CrossRef] [PubMed]
- Fisk, A.S.; Tam, S.K.E.; Brown, L.A.; Vyazovskiy, V.V.; Bannerman, D.M.; Peirson, S.N. Light and Cognition: Roles for Circadian Rhythms, Sleep, and Arousal. Front. Neurol. 2018, 9, 56. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.M.; Khalsa, S.B.; Scheer, F.A.; Cajochen, C.; Lockley, S.W.; Czeisler, C.A.; Wright, K.P., Jr. Acute effects of bright light exposure on cortisol levels. J. Biol. Rhythms 2010, 25, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Lockley, S.W.; Brainard, G.C.; Czeisler, C.A. High sensitivity of the human circadian melatonin rhythm to resetting by short wavelength light. J. Clin. Endocrinol. Metab. 2003, 88, 4502–4505. [Google Scholar] [CrossRef]
- Berson, D.M.; Dunn, F.A.; Takao, M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 2002, 295, 1070–1073. [Google Scholar] [CrossRef]
- Hattar, S.; Liao, W.; Takao, M.; Berson, D.M.; Yau, K.W. Melanopsin-containing retinal ganglion cells: Architecture, projections, and intrinsic photosensitivity. Science 2002, 295, 1065–1070. [Google Scholar] [CrossRef]
- Fan, S.M.Y.; Chang, Y.T.; Chen, C.L.; Wang, W.H.; Pan, M.K.; Chen, W.P.; Lin, S.J. External light activates hair follicle stem cells through eyes via an ipRGC SCN sympathetic neural pathway. Proc. Natl. Acad. Sci. USA 2018, 115, E6880–E6889. [Google Scholar] [CrossRef]
- Hattar, S.; Kumar, M.; Park, A.; Tong, P.; Tung, J.; Yau, K.W.; Berson, D.M. Central projections of melanopsin-expressing retinal ganglion cells in the mouse. J. Comp. Neurol. 2006, 497, 326–349. [Google Scholar] [CrossRef]
- Hull, J.T.; Czeisler, C.A.; Lockley, S.W. Suppression of Melatonin Secretion in Totally Visually Blind People by Ocular Exposure to White Light: Clinical Characteristics. Ophthalmology 2018, 125, 1160–1171. [Google Scholar] [CrossRef]
- Moldavan, M.G.; Allen, C.N. Retinohypothalamic tract synapses in the rat suprachiasmatic nucleus demonstrate short-term synaptic plasticity. J. Neurophysiol. 2010, 103, 2390–2399. [Google Scholar] [CrossRef]
- Ohi, K.; Takashima, M.; Nishikawa, T.; Takahashi, K. N-methyl-D-aspartate receptor participates in neuronal transmission of photic information through the retinohypothalamic tract. Neuroendocrinology 1991, 53, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Gabel, V.; Reichert, C.F.; Maire, M.; Schmidt, C.; Schlangen, L.J.M.; Kolodyazhniy, V.; Garbazza, C.; Cajochen, C.; Viola, A.U. Differential impact in young and older individuals of blue-enriched white light on circadian physiology and alertness during sustained wakefulness. Sci. Rep. 2017, 7, 7620. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, J.P.; Lockley, S.W.; Cecil, K.; West, K.; Jablonski, M.; Warfield, B.; James, M.; Ayers, M.; Byrne, B.; Gerner, E.; et al. Randomized trial of polychromatic blue-enriched light for circadian phase shifting, melatonin suppression, and alerting responses. Physiol. Behav. 2019, 198, 57–66. [Google Scholar] [CrossRef] [PubMed]
- West, K.E.; Jablonski, M.R.; Warfield, B.; Cecil, K.S.; James, M.; Ayers, M.A.; Maida, J.; Bowen, C.; Sliney, D.H.; Rollag, M.D.; et al. Blue light from light-emitting diodes elicits a dose-dependent suppression of melatonin in humans. J. Appl. Physiol. 2011, 110, 619–626. [Google Scholar] [CrossRef]
- Alkozei, A.; Smith, R.; Pisner, D.A.; Vanuk, J.R.; Berryhill, S.M.; Fridman, A.; Shane, B.R.; Knight, S.A.; Killgore, W.D. Exposure to Blue Light Increases Subsequent Functional Activation of the Prefrontal Cortex during Performance of a Working Memory Task. Sleep 2016, 39, 1671–1680. [Google Scholar] [CrossRef] [PubMed]
- Alkozei, A.; Smith, R.; Dailey, N.S.; Bajaj, S.; Killgore, W.D.S. Acute exposure to blue wavelength light during memory consolidation improves verbal memory performance. PLoS ONE 2007, 12, e0184884. [Google Scholar] [CrossRef]
- Knaier, R.; Schäfer, J.; Rossmeissl, A.; Klenk, C.; Hanssen, H.; Höchsmann, C.; Cajochen, C.; Schmidt-Trucksäss, A. Effects of bright and blue light on acoustic reaction time and maximum handgrip strength in male athletes: A randomized controlled trial. Eur. J. Appl. Physiol. 2017, 117, 1689–1696. [Google Scholar] [CrossRef]
- Bajaj, S.; Vanuk, J.R.; Smith, R.; Dailey, N.S.; Killgore, W.D.S. Blue-Light Therapy Following Mild Traumatic Brain Injury: Effects on White Matter Water Diffusion in the Brain. Front. Neurol. 2017, 8, 616. [Google Scholar] [CrossRef]
- Strong, R.E.; Marchant, B.K.; Reimherr, F.W.; Williams, E.; Soni, P.; Mestas, R. Narrow-band blue-light treatment of seasonal affective disorder in adults and the influence of additional nonseasonal symptoms. Depress Anxiety 2009, 26, 273–278. [Google Scholar] [CrossRef]
- Saeb-Parsy, K.; Lombardelli, S.; Khan, F.Z.; McDowall, K.; Au-Yong, I.T.; Dyball, R.E. Neural connections of hypothalamic neuroendocrine nuclei in the rat. J. Neuroendocrinol. 2000, 12, 635–648. [Google Scholar] [CrossRef]
- Vrang, N.; Larsen, P.J.; Mikkelsen, J.D. Direct projection from the suprachiasmatic nucleus to hypophysiotrophic corticotropin-releasing factor immunoreactive cells in the paraventricular nucleus of the hypothalamus demonstrated by means of Phaseolus vulgaris-leucoagglutinin tract tracing. Brain Res. 1995, 684, 61–69. [Google Scholar] [CrossRef]
- Askaripoor, T.; Motamedzadeh, M.; Golmohammadi, R.; Farhadian, M.; Babamiri, M.; Samavati, M. Non-Image Forming Effects of Light on Brainwaves, Autonomic Nervous Activity, Fatigue, and Performance. J. Circadian Rhythms 2018, 16, 9. [Google Scholar] [CrossRef] [PubMed]
- Deguchi, T.; Sato, M. The effect of color temperature of lighting sources on mental activity level. Ann. Physiol. Anthropol. 1992, 11, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Kapogiannatou, A.; Paronis, E.; Paschidis, K.; Polissidis, A.; Kostomitsopoulos, N.G. Effect of light colour temperature and intensity on the behaviour of male C57CL/6J mice. Appl. Anim. Behav. Sci. 2016, 184, 135–140. [Google Scholar] [CrossRef]
- Rahman, S.A.; St Hilaire, M.A.; Lockley, S.W. The effects of spectral tuning of evening ambient light on melatonin suppression, alertness and sleep. Physiol. Behav. 2017, 177, 221–229. [Google Scholar] [CrossRef]
- Yuda, E.; Yoshida, Y.; Ogasawara, H.; Hayano, J. Relaxation effects of organic light emitting diode lighting. Jpn. J. Physiol. Anthropol. 2018, 23, 23–28. (In Japanese) [Google Scholar]
- Lucas, R.J.; Peirson, S.N.; Berson, D.M.; Brown, T.M.; Cooper, H.M.; Czeisler, C.A.; Figueiro, M.G.; Gamlin, P.D.; Lockley, S.W.; O’Hagan, J.B.; et al. Measuring and using light in the melanopsin age. Trends Neurosci. 2014, 37, 19. [Google Scholar] [CrossRef]
- Measuring Melanopic Illuminance. Available online: http://lucasgroup.lab.manchester.ac.uk/research/measuringmelanopicilluminance/ (accessed on 15 October 2019).
- Kalsbeek, A.; Scheer, F.A.; Perreau-Lenz, S.; La Fleur, S.E.; Yi, C.X.; Fliers, E.; Buijs, R.M. Circadian disruption and SCN control of energy metabolism. FEBS Lett. 2011, 585, 1412–1426. [Google Scholar] [CrossRef]
- Ni, J.; Shen, T.; Ebara, Y.; Koyamada, K.; Kawakami, Y.; Nakamura, Y. Reduction of the blue light hazard by adding a cyan light LED. J. Adv. Simul. Sci. Eng. 2018, 4, 44–63. [Google Scholar] [CrossRef]
- Peng, M.-L.; Tsai, C.-Y.; Chien, C.-L.; Hsiao, J.C.-J.; Huang, S.-Y.; Lee, C.-J.; Lin, H.-Y.; Wen, Y.-C.; Tseng, K.-W. The influence of low-powered family LED lighting on eyes in mice experimental model. Life Sci. J. 2012, 9, 477–482. [Google Scholar]
- Kraneburg, A.; Franke, S.; Methling, R.; Griefahn, B. Effect of color temperature on melatonin production for illumination of working environments. Appl. Ergon. 2017, 58, 446–453. [Google Scholar] [CrossRef]
- Jacobs, G.H.; Neitz, J.; Deegan, J.F. Retinal receptors in rodents maximally sensitive to ultraviolet light. Nature 1991, 353, 655–656. [Google Scholar] [CrossRef]
- Badoer, E. Hypothalamic paraventricular nucleus and cardiovascular regulation. Clin. Exp. Pharmacol. Physiol. 2001, 28, 95–99. [Google Scholar] [CrossRef]
- Ebner, K.; Singewald, N. Individual differences in stress susceptibility and stress inhibitory mechanisms. Curr. Opin. Behav. Sci. 2017, 14, 54–64. [Google Scholar] [CrossRef]
- Smith, S.M.; Vale, W.W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 2006, 8, 383–395. [Google Scholar]
- Alves-Simoes, M.; Coleman, G.; Canal, M.M. Effects of type of light on mouse circadian behaviour and stress levels. Lab. Anim. 2016, 50, 21–29. [Google Scholar] [CrossRef]
- Chang, S.W.; Kim, H.I.; Kim, G.H.; Park, S.J.; Kim, I.B. Increased expression of osteopontin in retinal degeneration induced by blue light-emitting diode exposure in mice. Front. Mol. Neurosci. 2016, 9, 58. [Google Scholar] [CrossRef]
- Jaadane, I.; Villalpando Rodriguez, G.E.; Boulenguez, P.; Chahory, S.; Carré, S.; Savoldelli, M.; Jonet, L.; Behar-Cohen, F.; Martinsons, C.; Torriglia, A. Effects of white light-emitting diode (LED) exposure on retinal pigment epithelium in vivo. J. Cell Mol. Med. 2017, 21, 3453–3466. [Google Scholar] [CrossRef]
- Nakamura, M.; Kuse, Y.; Tsuruma, K.; Shimazawa, M.; Hara, H. The Involvement of the Oxidative Stress in Murine Blue LED Light-Induced Retinal Damage Model. Biol. Pharm. Bull. 2017, 40, 1219–1225. [Google Scholar] [CrossRef]
- Vicente-Tejedor, J.; Marchena, M.; Ramírez, L.; García-Ayuso, D.; Gómez-Vicente, V.; Sánchez-Ramos, C.; de la Villa, P.; Germain, F. Removal of the blue component of light significantly decreases retinal damage after high intensity exposure. PLoS ONE 2018, 13, e0194218. [Google Scholar] [CrossRef]
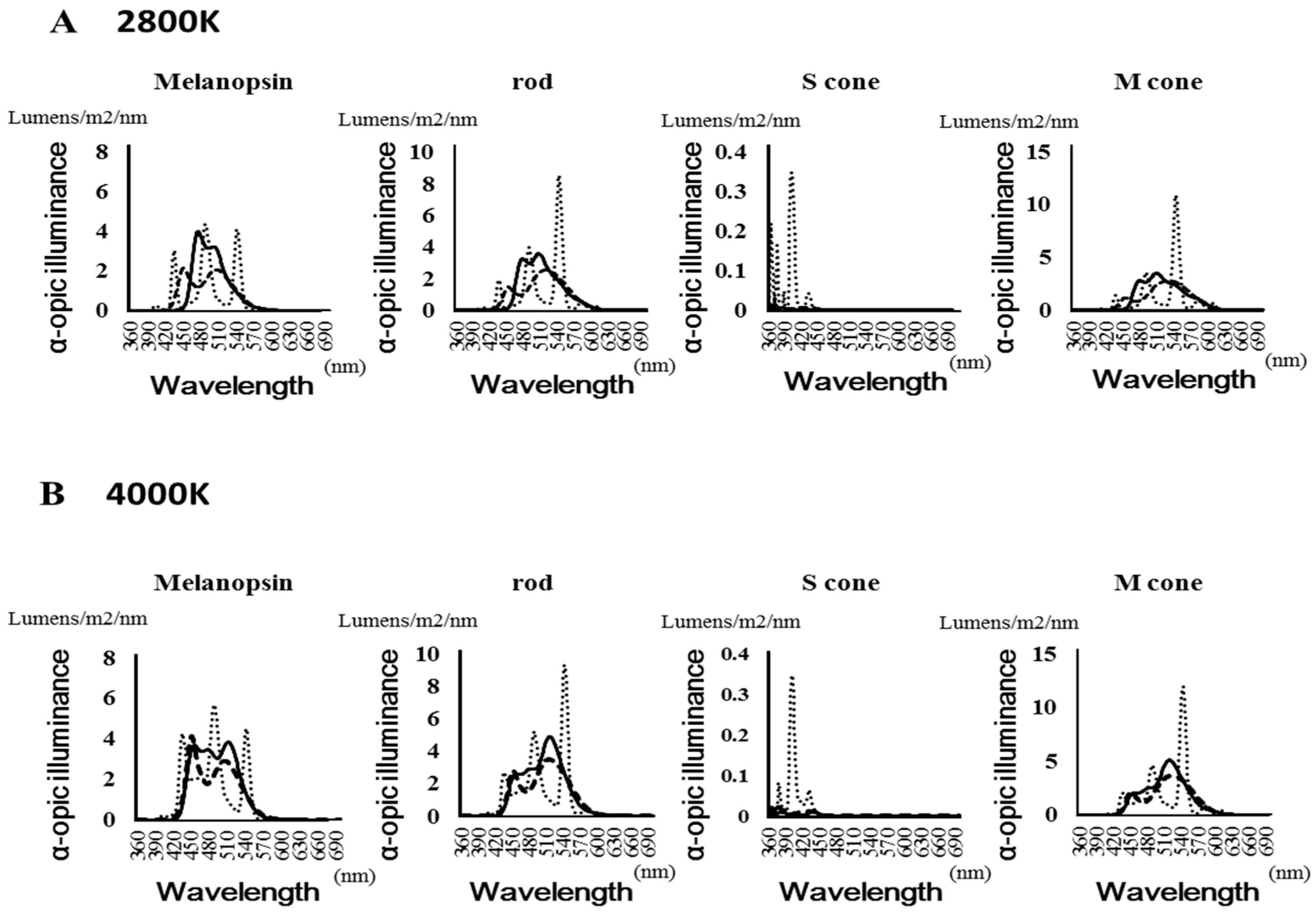
| Melanopsin | Rods | S cone | M cone | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Color Temperature | Light Source | α-Opic Lux Value | Peak Wavelength | α-Opic Lux Value | Peak Wavelength | α-Opic Lux Value | Peak Wavelength | α-Opic Lux Value | Peak Wavelength |
| (Lumens/m2/nm) | (nm) | (Lumens/m2/nm) | (nm) | (Lumens/m2/nm) | (nm) | (Lumens/m2/nm) | (nm) | ||
| 2800 K | OLED | 3.973 | 477 | 3.552 | 507 | 0.023 | 360 | 3.47 | 509 |
| LED | 2.143 | 452 | 2.513 | 519 | 0.029 | 372 | 2.75 | 531 | |
| Fluorescent | 4.362 | 489 | 8.506 | 543 | 0.347 | 402 | 10.888 | 544 | |
| 4000 K | OLED | 3.854 | 512 | 4.887 | 517 | 0.015 | 387 | 5.11 | 520 |
| LED | 4.11 | 452 | 3.477 | 516 | 0.03 | 368 | 3.672 | 525 | |
| Fluorescent | 5.719 | 489 | 9.345 | 543 | 0.348 | 402 | 11.913 | 543 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yokoyama, M.; Chang, H.; Anzai, H.; Kato, M. Effects of Different Light Sources on Neural Activity of the Paraventricular Nucleus in the Hypothalamus. Medicina 2019, 55, 732. https://doi.org/10.3390/medicina55110732
Yokoyama M, Chang H, Anzai H, Kato M. Effects of Different Light Sources on Neural Activity of the Paraventricular Nucleus in the Hypothalamus. Medicina. 2019; 55(11):732. https://doi.org/10.3390/medicina55110732
Chicago/Turabian StyleYokoyama, Michio, Hyukki Chang, Hiroshi Anzai, and Morimasa Kato. 2019. "Effects of Different Light Sources on Neural Activity of the Paraventricular Nucleus in the Hypothalamus" Medicina 55, no. 11: 732. https://doi.org/10.3390/medicina55110732
APA StyleYokoyama, M., Chang, H., Anzai, H., & Kato, M. (2019). Effects of Different Light Sources on Neural Activity of the Paraventricular Nucleus in the Hypothalamus. Medicina, 55(11), 732. https://doi.org/10.3390/medicina55110732




