Pleurotus highking Mushroom Induces Apoptosis by Altering the Balance of Proapoptotic and Antiapoptotic Genes in Breast Cancer Cells and Inhibits Tumor Sphere Formation
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Extraction of Crude Mushroom Extract
2.3. Fractionation of Extract
2.4. Cell Line and Cell Culture
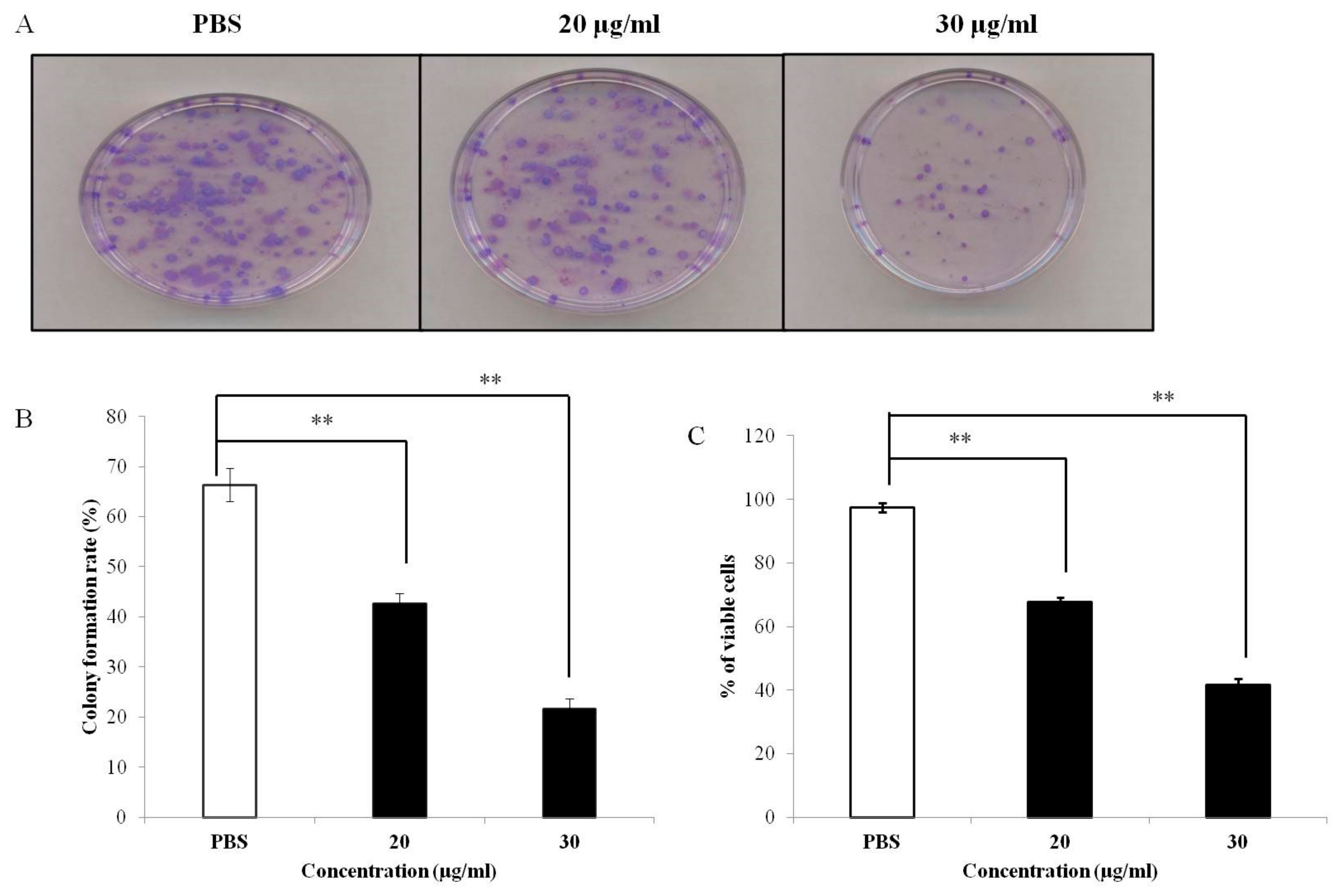
2.5. Colony Formation Assay
2.6. Cell Viability/Proliferation Assay
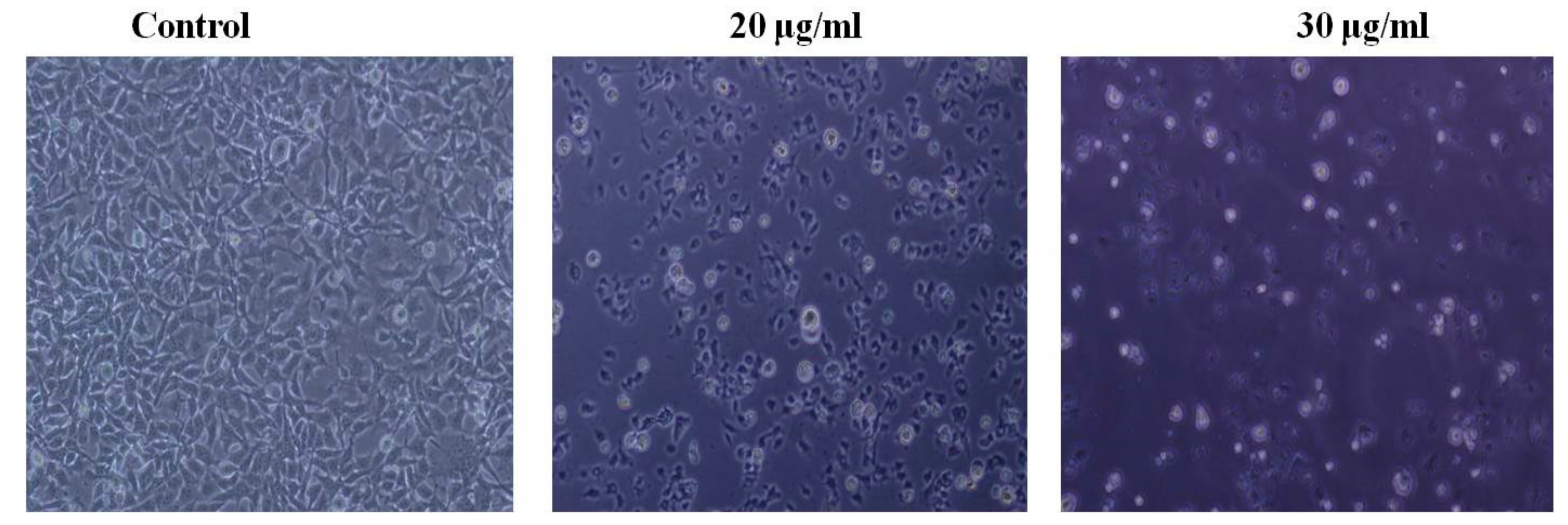
2.7. Morphological Study Using an Inverted Light Microscope
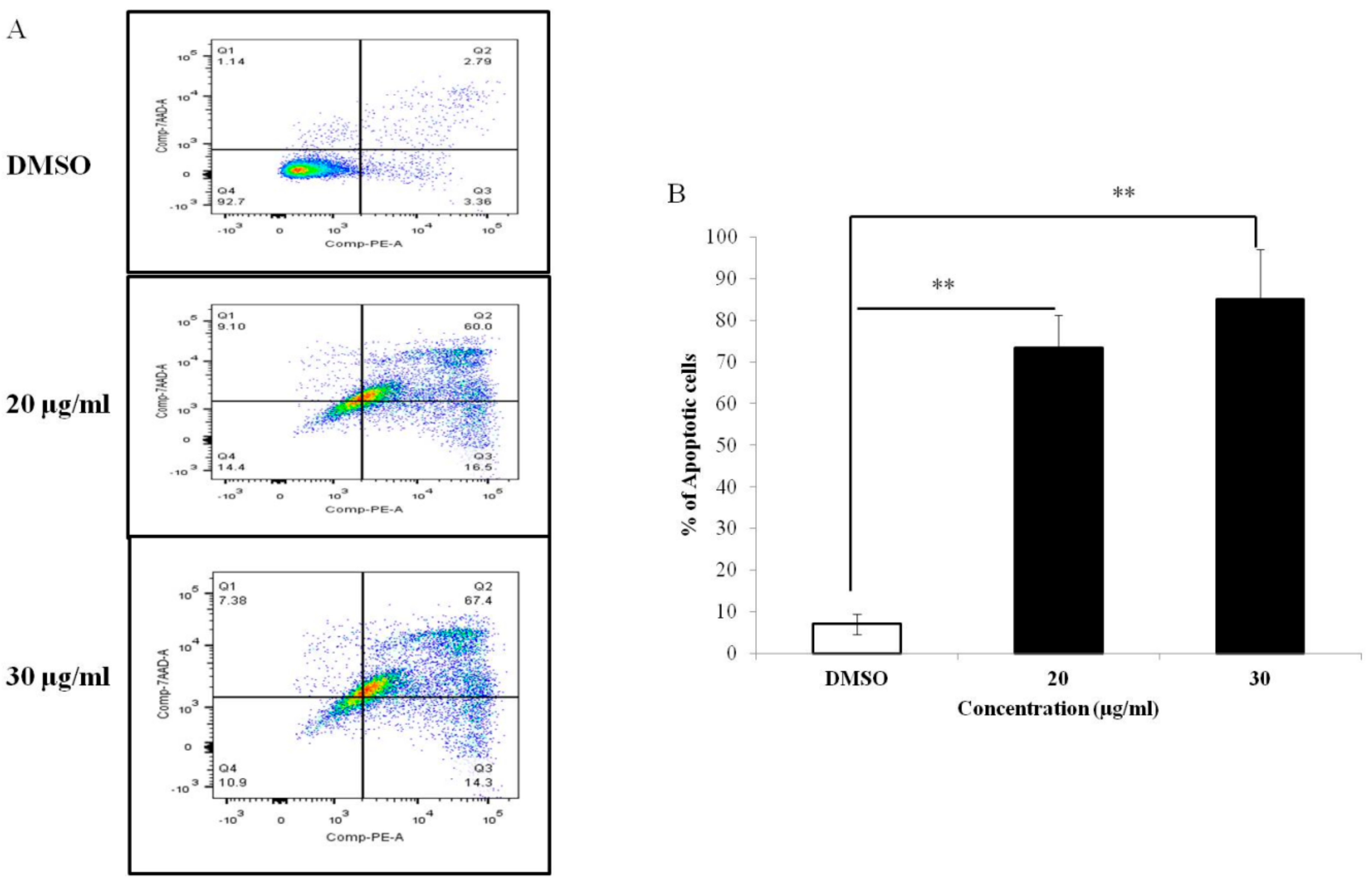
2.8. Annexin V-Phycoerythrin and PI Staining for Apoptosis
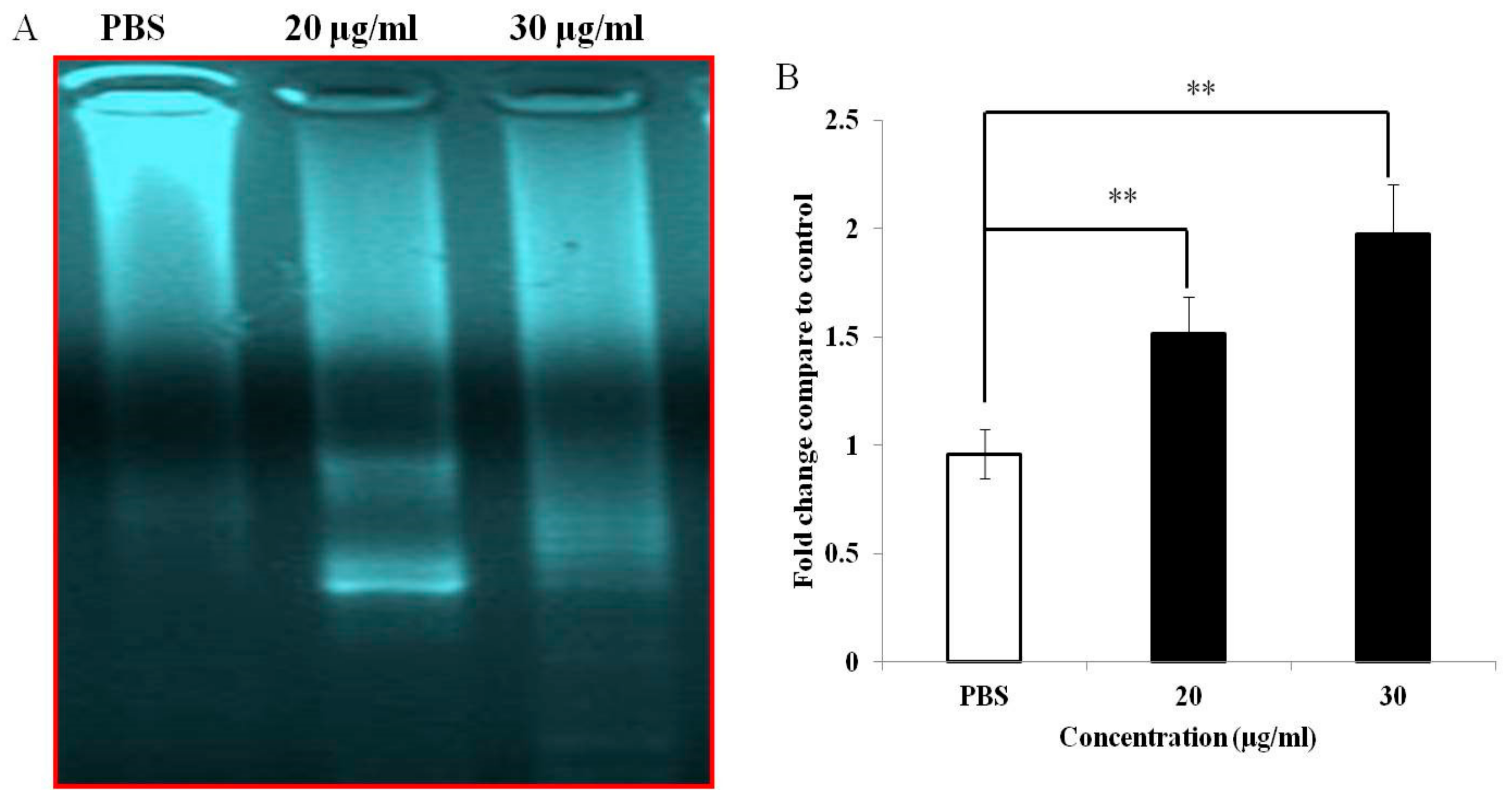
2.9. Investigation of DNA Fragmentation
2.10. Caspase 3/7 Activity Assay
2.11. SDS-PAGE and Western Blot Analysis
2.12. In Vitro Tumor Sphere Formation Assay
2.13. Statistical Analysis
3. Results
3.1. PEF-III Decreases Proliferation of Breast Cancer Cells
3.2. PEF-III Induced Apoptosis-Related Morphological Characteristics
3.3. PEF-III Induces Cell Death through Apoptosis
3.4. PEF-III Induces DNA Fragmentation and Increases Caspase 3/7 Activity
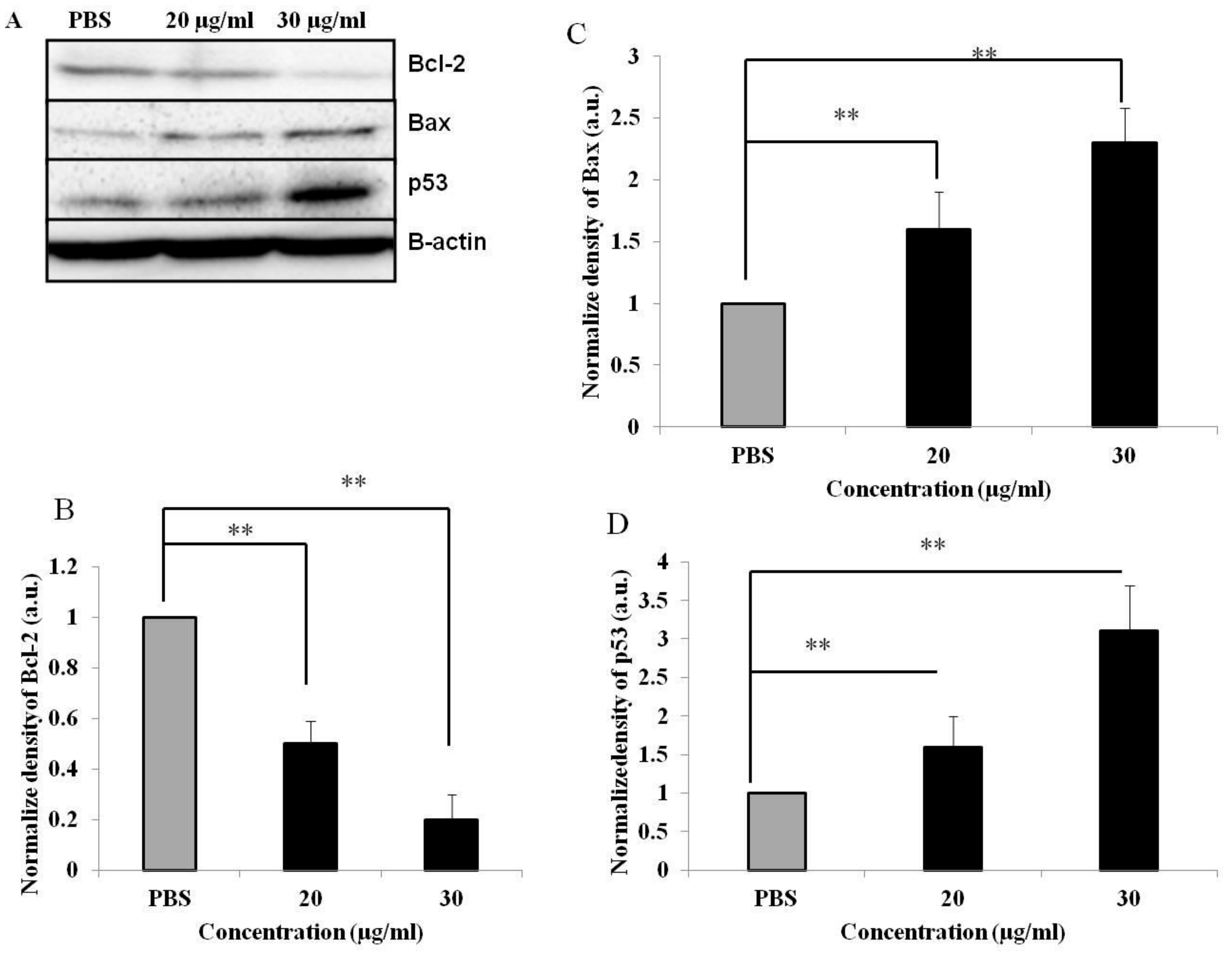
3.5. PEF-III Alters the Expression of Proapoptotic and Antiapoptotic Genes
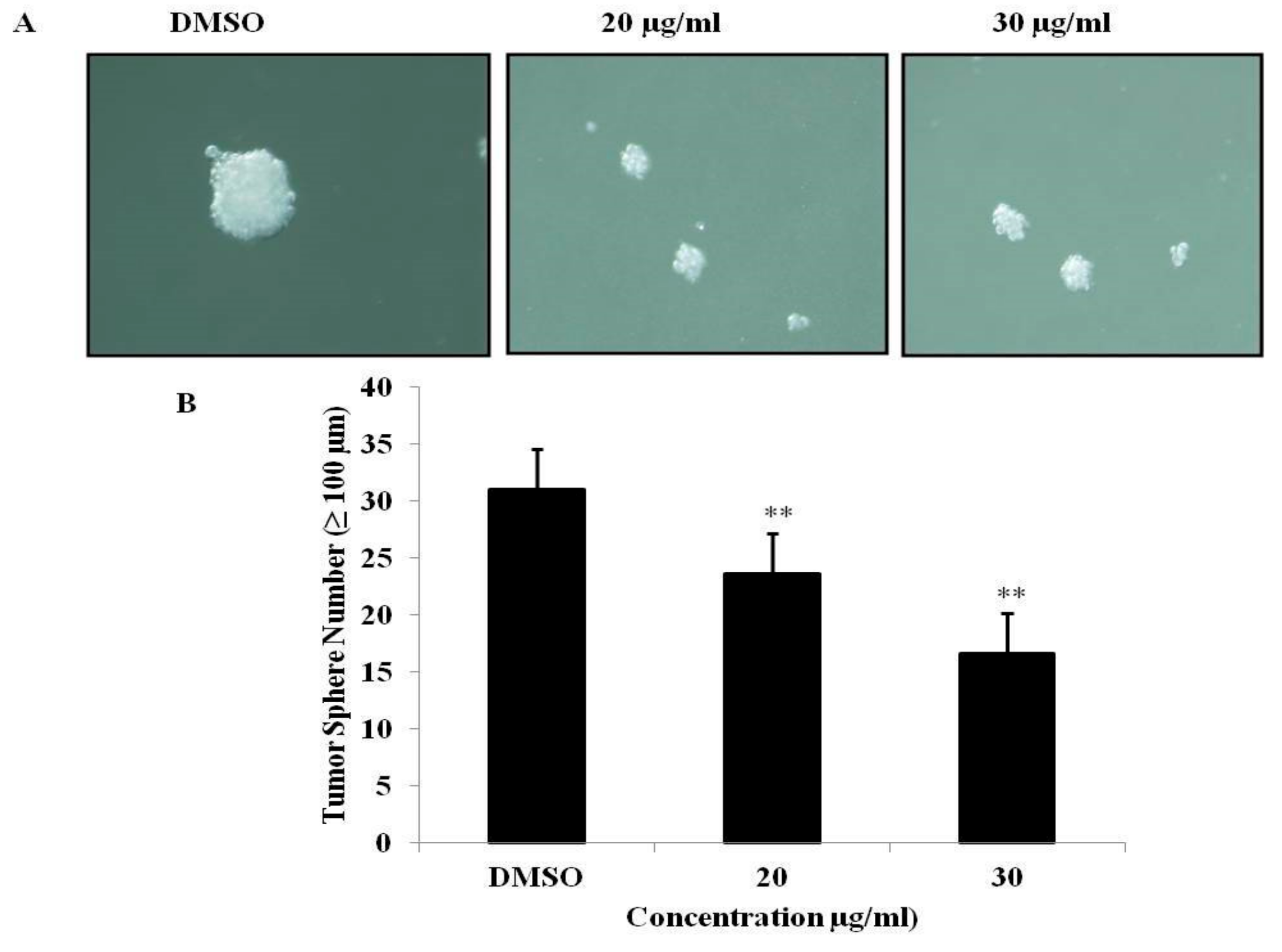
3.6. PEF-III Inhibits Tumor Sphere Formation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dolai, N.; Islab, A.; Haldar, P.K. Antiproliferative activity and apoptosis inducing mechanism of Anthocephalus cadamba on Dalton’s lymphoma ascites cells. Iran. J. Pharm. Res. 2016, 15, 505–514. [Google Scholar] [PubMed]
- Salem, M.L.; Shoukry, N.M.; Teleb, W.K.; Abdel-Daim, M.M.; Rahman, M.A.A. In vitro and in vivo antitumor effects of the Egyptian scorpion Androctonus amoreuxi venom in an Ehrlich ascites tumor model. Springerplus 2016, 5, 570. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Biswas, G.; Chandra, S.; Saha, G.K.; Acharya, K. Apoptogenic effects of Tricholoma giganteum on Ehrlich’s ascites carcinoma cell. Bioprocess Biosyst. Eng. 2013, 36, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.A. Mushrooms: The ultimate health food but little research in U.S to prove it. Mushroom News. 1993, 41, 29–46. [Google Scholar]
- Bala, N.; Aitken, E.A.B.; Fechner, N.; Cusack, A.; Steadman, K.J. Evaluation of antibacterial activity of Australian basidiomycetous macrofungi using a high-throughput 96-well plate assay. Pharm. Biol. 2011, 49, 492–500. [Google Scholar] [CrossRef][Green Version]
- Haque, M.A.; Sarker, A.K.; Khatun, A.; Islam, M.S.; Islam, M.A.U.; Anisuzzaman, A.S.M. Evaluation of susceptibility of methanol extract of Pleurotus highking, an edible mushroom cultivated in Bangladesh. Int. J. Sci. Technol. Res. 2014, 3, 275–278. [Google Scholar]
- Piraino, F.; Brandt, C.R. Isolation and partial characterization of an antiviral, RC-183, from the edible mushroom Rozites caperata. Antivir. Res. 1999, 3, 67–78. [Google Scholar] [CrossRef]
- Maiti, S.; Mallick, S.K.; Bhutia, S.K.; Behera, B.; Mandal, M.; Maiti, T.K. Antitumor effect of culinary-medicinal oyster mushroom, Pleurotus ostreatus (Jacq.: Fr.) P. Kumm., derived protein 84 fraction on tumor-bearing mice models. Int. J. Med. Mushrooms 2011, 13, 427–440. [Google Scholar] [CrossRef]
- Haque, M.A.; Rana, M.M.; Aktar, K.; Haque, M.U.; Chouduri, M.A.U.; Islam, M.A.U. In-vitro antioxidant and cytotoxic activity of crude extracts of Pleurotus highking, a potential oyster mushroom. Br. J. Pharm. Res. 2017, 14, BJPR.30594. [Google Scholar] [CrossRef]
- Alam, N.; Yoon, K.N.; Lee, T.S. Antihyperlipidemic activities of Pleurotus ferulae on biochemical and histological function in hypercholesterolemic rats. J. Res. Med. Sci. 2011, 16, 776–786. [Google Scholar]
- Miyazawa, N.; Okazaki, M.; Ohga, S. Antihypertensive effect of Pleurotus nebrodensis in spontaneously hypertensive rats. J. Oleo Sci. 2008, 57, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Nosalova, V.; Bobek, P.; Cerna, S.; Galbavy, S.; Stvrtina, S. Effects of pleuran (beta glucan isolated from Pleurotus ostreatus) on experimental colitis in rats. Physiol. Res. 2001, 50, 5755–5781. [Google Scholar]
- Wasser, S.P. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [PubMed]
- Tor, Y.S.; Yazan, L.S.; Foo, J.B.; Armania, N.; Cheah, Y.K.; Abdullah, R.; Imam, M.U.; Ismail, N.; Ismail, M. Induction of apoptosis through oxidative stress-related pathways in MCF-7, human breast cancer cells, by ethyl acetate extract of Dillenia suffruticosa. BMC Compl. Alter. Med. 2014, 14, 55. [Google Scholar] [CrossRef] [PubMed]
- Tayeb, K.; William, T.B. Merbarone, a catalytic inhibitor of DNA topoisomerase II, induces apoptosis in CEM cells through activation of ICE/CED-3-like protease. Mol. Pharmacol. 1999, 55, 548–556. [Google Scholar]
- Wu, C.H.; Liu, F.C.; Pan, C.H.; Lai, M.T.; Lan, S.J.; Wu, C.H.; Sheu, M.J. Suppression of cell growth, migration and drug resistance by ethanolic extract of Antrodia cinnamomea in human lung cancer A549 cells and C57BL/6J allograft tumor model. Int. J. Mol. Sci. 2018, 19, 791. [Google Scholar] [CrossRef]
- Zhu, Z.W.; Chen, L.; Liu, J.X.; Huang, J.W.; Wu, G.; Zheng, Y.F.; Yao, K.T. A novel three-dimensional tumorsphere culture system for the efficient and low-cost enrichment of cancer stem cells with natural polymers. Exp. Ther. Med. 2018, 15, 85–92. [Google Scholar] [CrossRef]
- Lee, C.H.; Yu, C.C.; Wang, B.Y.; Chang, W.W. Tumorsphere as an effective in vitro platform for screening anti-cancer stem cell drugs. Oncotarget 2016, 7, 1215–1226. [Google Scholar] [CrossRef]
- Somasagara, R.R.; Hegde, M.; Chiruvella, K.K.; Musini, A.; Choudhary, B.; Raghavan, S.C. Extracts of strawberry fruits induce intrinsic pathway of apoptosis in breast cancer cells and inhibits tumor progression in mice. PLoS ONE 2012, 7, e47021. [Google Scholar] [CrossRef]
- Andrej, J.; Daniel, S. Pleurotus ostreatus inhibits proliferation of human breast and colon cancer cells through p53-dependent as well as p53-independent pathway. Int. J. Oncol. 2008, 33, 1307–1313. [Google Scholar]
- Yang, S.; Zhao, Q.; Xiang, H.; Liu, M.; Zhang, Q.; Xue, W.; Song, B.; Yang, S. Antiproliferative activity and apoptosis-inducing mechanism of constituents from Toona sinensis on human cancer cells. Cancer Cell Int. 2013, 13, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Choudhuri, T.; Pal, S.; Chattopadhyay, S.; Datta, G.K.; Sa, G.; Das, T. Apoptogenic effects of black tea on Ehrlich’s ascites carcinoma cell. Carcinogenesis 2003, 24, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Giannakakou, P.; Robey, R.; Fojo, T.; Blagosklonny, M.V. Low concentrations of paclitaxel induce cell type dependent p53, p21 and G1/G2 arrest instead of mitotic arrest: Molecular determinants of paclitaxel induced cytotoxicity. Oncogene 2001, 20, 3806–3813. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, L. PUMA, a potent killer with or without p53. Oncogene 2008, 27, S71–S83. [Google Scholar] [CrossRef]
- Borner, C. The Bcl-2 protein family: Sensors and checkpoints for life-or-death decisions. Mol. Immunol. 2003, 39, 615–647. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haque, M.A.; Islam, M.A.U. Pleurotus highking Mushroom Induces Apoptosis by Altering the Balance of Proapoptotic and Antiapoptotic Genes in Breast Cancer Cells and Inhibits Tumor Sphere Formation. Medicina 2019, 55, 716. https://doi.org/10.3390/medicina55110716
Haque MA, Islam MAU. Pleurotus highking Mushroom Induces Apoptosis by Altering the Balance of Proapoptotic and Antiapoptotic Genes in Breast Cancer Cells and Inhibits Tumor Sphere Formation. Medicina. 2019; 55(11):716. https://doi.org/10.3390/medicina55110716
Chicago/Turabian StyleHaque, Md. Anwarul, and Md. Anwar Ul Islam. 2019. "Pleurotus highking Mushroom Induces Apoptosis by Altering the Balance of Proapoptotic and Antiapoptotic Genes in Breast Cancer Cells and Inhibits Tumor Sphere Formation" Medicina 55, no. 11: 716. https://doi.org/10.3390/medicina55110716
APA StyleHaque, M. A., & Islam, M. A. U. (2019). Pleurotus highking Mushroom Induces Apoptosis by Altering the Balance of Proapoptotic and Antiapoptotic Genes in Breast Cancer Cells and Inhibits Tumor Sphere Formation. Medicina, 55(11), 716. https://doi.org/10.3390/medicina55110716




