Observational Study of Antibiotic Usage at the Children’s Clinical University Hospital in Riga, Latvia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Setting
2.3. Participants
2.4. Data Collection
2.5. Intervention
2.6. Main Outcome Measure
2.7. Data Analysis
2.8. Quality Indicators
2.9. Ethics
3. Results
3.1. Point Prevalence Surveys’ Results
3.2. Defined Daily Dosage Method’s Results
Antibiotic Groups Used during the Study Period
3.3. Antibiotic Consumption 90%DU Analysis
4. Discussion
Strength and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Global Antimicrobial Resistance Surveillance System (GLASS) Report: Early Implementation 2016–2017. 2017. Available online: http://apps.who.int/iris/bitstream/handle/10665/259744/9789241513449-eng.pdf;jsessionid=83D25F98539A6F4BF92B66CA99FD0D13?sequence=1 (accessed on 20 June 2018).
- Raveh, D.; Yinnon, A.M.; Broide, E.; Rudensky, B. Susceptibilities of ESCL-producing Enterobacteriaceae to ertapenem, meropenem and piperacillin-tazobactam with and without clavulanic acid. Chemotherapy 2007, 53, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Gerber, J.S.; Kronman, M.P.; Ross, R.K.; Hersh, A.L.; Newland, J.G.; Metjian, T.A.; Zaoutis, T.E. Identifying targets for antimicrobial stewardship in children’s hospitals. Infect. Control Hosp. Epidemiol. 2013, 34, 1252–1258. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.J.; Gerber, J.S.; Hersh, A.L. Inpatient Antimicrobial Stewardship in Pediatrics: A Systematic Review. J. Pediatr. Infect. Dis. Soc. 2015, 4, e127–e135. [Google Scholar] [CrossRef] [PubMed]
- Dellit, T.H.; Owens, R.C.; McGowan, J.E.; Gerding, D.N.; Weinstein, R.A.; Burke, J.P.; Huskins, W.C.; Paterson, D.L.; Fishman, N.O.; Carpenter, C.F.; et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin. Infect. Dis. 2007, 44, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Levy, E.R.; Swami, S.; Dubois, S.G.; Wendt, R.; Banerjee, R. Rates and appropriateness of antimicrobial prescribing at an academic children’s hospital, 2007–2010. Infect. Control Hosp. Epidemiol. 2012, 33, 346–353. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antimicrobial Resistance: Global Report on Surveillance. 2014. Available online: http://apps.who.int/iris/bitstream/10665/112642/1/9789241564748_eng.pdf?ua=1 (accessed on 10 November 2017).
- Davey, P.; Marwick, C.A.; Scott, C.L.; Charani, E.; McNeil, K.; Brown, E.; Gould, I.M.; Ramsay, C.R.; Michie, S. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst. Rev. 2017, 2, CD003543. [Google Scholar] [CrossRef] [PubMed]
- De Jong, J.; van den Berg, P.B.; Visser, S.T.; de Vries, T.W.; de Jong-van den Berg, L.T. Antibiotic usage, dosage and course length in children between 0 and 4 years. Acta Paediatr. 2009, 98, 1142–1148. [Google Scholar] [CrossRef] [PubMed]
- Schindler, C.; Krappweis, J.; Morgenstern, I.; Kirch, W. Prescriptions of systemic antibiotics for children in Germany aged between 0 and 6 years. Pharmacoepidemiol. Drug Saf. 2003, 12, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Gerber, J.S.; Newland, J.G.; Coffin, S.E.; Hall, M.; Thurm, C.; Prasad, P.A.; Feudtner, C.; Zaoutis, T.E. Variability in Antibiotic Use at Children’s Hospitals. Pediatric 2010, 126, 1067–1073. [Google Scholar] [CrossRef] [PubMed]
- Godbout, E.J.; Pakyz, A.L.; Markley, J.D.; Noda, A.J.; Stevens, M.P. Pediatric Antimicrobial Stewardship: State of the Art. Curr. Infect. Dis. Rep. 2018, 20, 39. [Google Scholar] [CrossRef] [PubMed]
- Brett, A.; Bielicki, J.; Newland, J.G.; Rodrigues, F.; Schaad, U.B.; Sharland, M. Neonatal and Pediatric Antimicrobial Stewardship Programs in Europe—Defining the Research Agenda. Pediatr. Infect. Dis. J. 2013, 32, e456–e465. [Google Scholar] [CrossRef] [PubMed]
- Versporten, A.; Sharland, M.; Bielicki, J.; Drapier, N.; Vankerckhoven, V.; Goossens, H.; For the ARPEC Project Group Members. The antibiotic resistance and prescribing in European Children project: A neonatal and pediatric antimicrobial web-based point prevalence survey in 73 hospitals worldwide. Pediatr. Infect. Dis. J. 2013, 2, e242–e253. [Google Scholar] [CrossRef] [PubMed]
- Spyridis, N.; Syridou, G.; Goossens, H.; Versporten, A.; Kopsidas, J.; Kourlaba, G.; Bielicki, J.; Drapier, N.; Zaoutis, T.; Tsolia, M.; et al. Variation in paediatric hospital antibiotic guidelines in Europe. Arch. Dis. Child. 2016, 101, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Zingg, W.; Hopkins, S.; Gayet-Ageron, A.; Holmes, A.; Sharland, M.; Suetens, C. Health-care-associated infections in neonates, children, and adolescents: An analysis of paediatric data from the European Centre for Disease Prevention and Control point-prevalence survey. Lancet Infect. Dis. 2017, 17, 381–389. [Google Scholar] [CrossRef]
- Zhang, W.; Shen, X.; Wang, Y.; Chen, Y.; Huang, M.; Zeng, Q.; Wei, J.; Lu, Q.; Wang, G.; Deng, L.; et al. Antibiotic use in five children’s hospitals during 2002–2006: The impact of antibiotic guidelines issued by the Chinese Ministry of Health. Pharmacoepidemiol. Drug Saf. 2008, 17, 306–311. [Google Scholar] [CrossRef] [PubMed]
- WHO. Collaborating Centre for Drug Statistics Methodology (WHOCC). Available online: http://www.whocc.no/atc_ddd_index/ (accessed on 12 February 2018).
- Zhang, W.; Shen, X.; Bergman, U.; Wang, Y.; Chen, Y.; Huang, M.; Zeng, Q.; Wei, J.; Lu, Q.; Wang, G.; et al. Drug utilisation 90% (DU90%) profiles of antibiotics in five Chinese children’s hospitals (2002–2006). Int. J. Antimicrob. Agents 2008, 32, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Dimiņa, E.; Kūla, M.; Caune, U.; Vīgante, D.; Liepiņs, M.; Zeidaka, L.; Nikitina, O.; Kūriņa, D.; Mironovska, A.; Dumpis, U. Repeated prevalence studies on antibiotic use in Latvia, 2003–2007. Euro Surveill 2009, 14, 127–136. [Google Scholar] [CrossRef]
- Ansari, F.; Molana, H.; Goossens, H.; Davey, P. ESAC II Hospital Care Study Group. Development of standardized methods for analysis of changes in antibacterial use in hospitals from 18 European countries: The European Surveillance of Antimicrobial Consumption (ESAC) longitudinal survey, 2000–06. J. Antimicrob. Chemother. 2010, 65, 2685–2691. [Google Scholar] [CrossRef] [PubMed]
- Ansari, F.; Erntell, M.; Goossens, H.; Davey, P. The European Surveillance of Antimicrobial Consumption (ESAC) Point-Prevalence Survey of Antibacterial Use in 20 European Hospitals in 2006. Clin. Infect. Dis. 2009, 49, 1496–1504. [Google Scholar] [CrossRef] [PubMed]
- Thu, T.A.; Rahman, M.; Coffin, S.; Harun-Or-Rashid, M.; Sakamoto, J.; Hung, N.V. Antibiotic use in Vietnamese hospitals: A multicenter point-prevalence study. Am. J. Infect. Control 2012, 40, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.S.; Xiang, L.L.; Li, R.; Hu, Q.; Luo, Q.Q.; Xiong, W. A multicenter point-prevalence survey of antibiotic use in 13 Chinese hospitals. J. Infect. Public Health 2015, 8, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Laine, N.; Hoppu, K.; Airaksinen, M.; Saxen, H. Antimicrobial consumption in a tertiary children’s hospital in Finland (2003–2013). Eur. J. Hosp. Pharm. 2016, 23, 266–271. [Google Scholar] [CrossRef]
- Public Health England. Start Smart Then Focus. 2015. Available online: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/417032/Start_Smart_Then_Focus_FINAL.PDF (accessed on 15 May 2018).
- Haug, J.B.; Berild, D.; Walberg, M.; Reikvam, A. Hospital- and patient-related factors associated with differences in hospital antibiotic use: Analysis of national surveillance results. Antimicrob. Resist. Infect. Control 2014, 3, 40. [Google Scholar] [CrossRef] [PubMed]
- Müller, A.; Monnet, D.L.; Talon, D.; Hénon, T.; Bertrand, X. Discrepancies between prescribed daily doses and WHO defined daily doses of antibacterials at a university hospital. Br. J. Clin. Pharmacol. 2006, 61, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Gharbi, M.; Doerholt, K.; Vergnano, S.; Bielicki, J.A.; Paulus, S.; Menson, E.; Riordan, A.; Lyall, H.; Patel, S.V.; Bernatoniene, J.; et al. Using a simple point-prevalence survey to define appropriate antibiotic prescribing in hospitalised children across the UK. BMJ Open 2016, 6, e012675. [Google Scholar] [CrossRef] [PubMed]
- Porta, A.; Hsia, Y.; Doerholt, K.; Spyridis, N.; Bielicki, J.; Menson, E.; Tsolia, M.; Esposito, S.; Wong, I.C.; Sharland, M. Comparing neonatal and paediatric antibiotic prescribing between hospitals: A new algorithm to help international benchmarking. J. Antimicrob. Chemother. 2012, 67, 1278–1286. [Google Scholar] [CrossRef] [PubMed]
- Norberg, S.; Struwe, J.; Grunewald, M.; Ternhag, A. A pilot study of risk adjustment for benchmarking antibiotic use between hospitals in Sweden. J. Glob. Antimicrob. Resist. 2014, 2, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Thaulow, C.M.; Berild, D.; Eriksen, B.H.; Myklebust, T.Å.; Blix, H.S. Potential for More Rational Use of Antibiotics in Hospitalized Children in a Country with Low Resistance–Data From Eight Point Prevalence Surveys. Pediatr. Infect. Dis. J. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gundlapalli, A.V.; Beekmann, S.E.; Graham, D.R.; Polgreen, P.M.; Members of the Emerging Infections Network. Antimicrobial Agent Shortages: The New Norm for Infectious Diseases Physicians. Open Forum Infect. Dis. 2018, 5. [Google Scholar] [CrossRef] [PubMed]
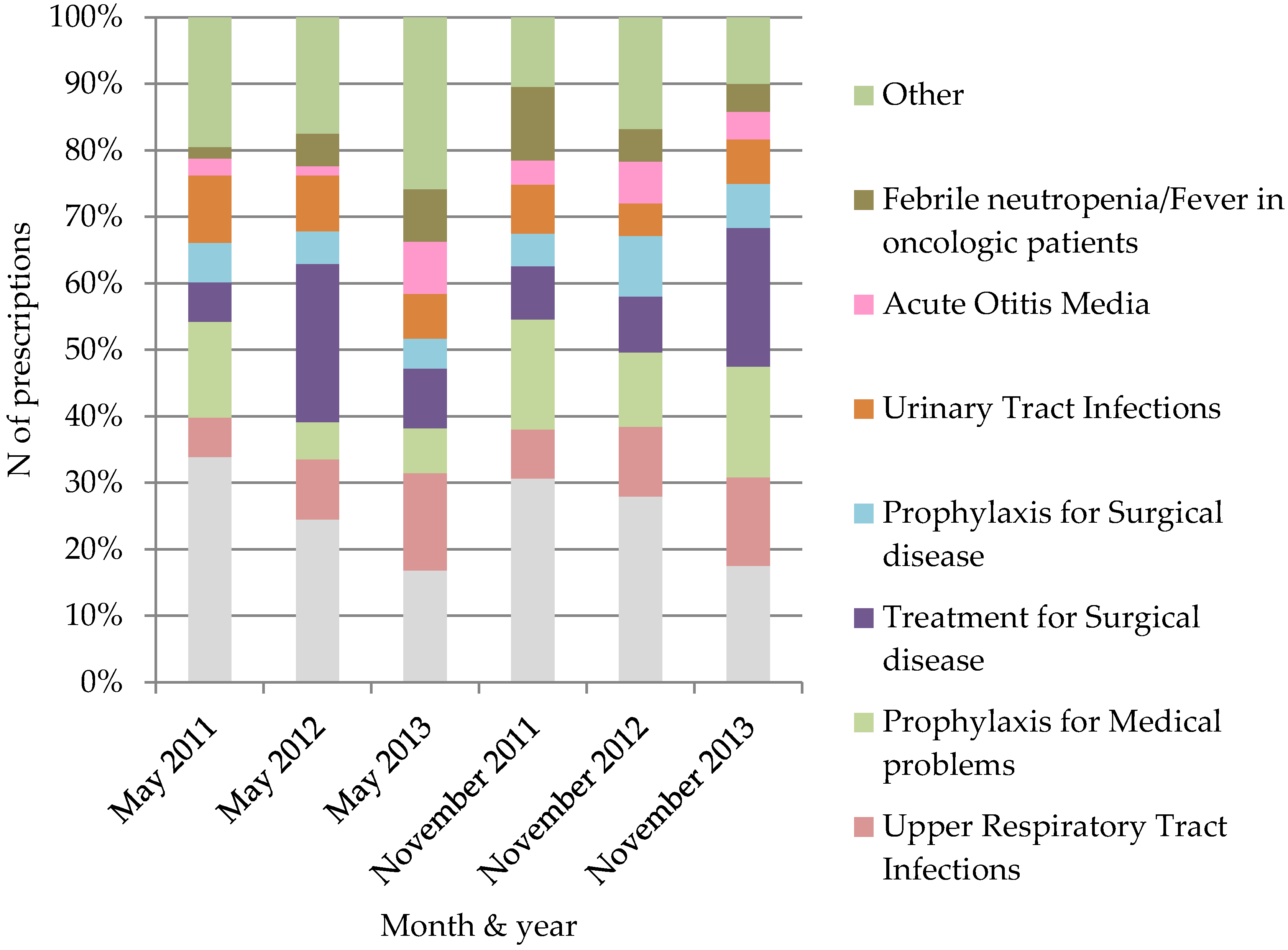
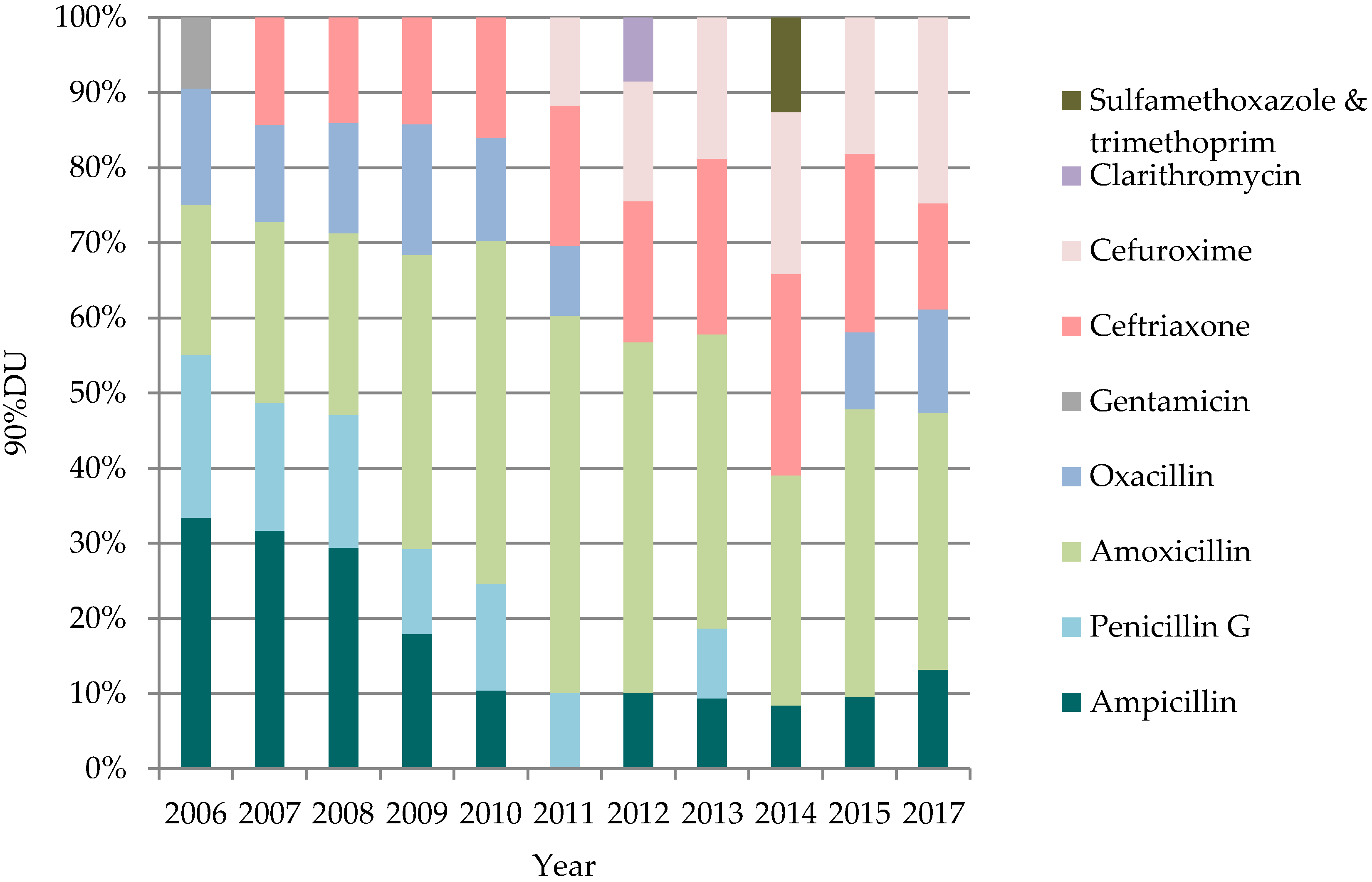
| Year | 2011 | 2012 | 2013 | 2011 | 2012 | 2013 |
|---|---|---|---|---|---|---|
| Month | May | May | May | November | November | November |
| Patients | N (%) (95% CI) | N (%) (95% CI) | N (%) (95% CI) | N (%) (95% CI) | N (%) (95% CI) | N (%) (95% CI) |
| Total number | 418 | 395 | 335 | 424 | 358 | 320 |
| Male | 230 (55) (50–60) | 215 (54) (50–59) | 178 (53) (48–58) | 225 (53) (48–58) | 196 (55) (50–60) | 159 (50) (44–55) |
| Female | 188 (45) (40–50) | 180 (46) (41–51) | 157 (47) (42–52) | 199 (47) (42–52) | 162 (45) (40–51) | 161 (50) (45–56) |
| Patients on antibiotics | 125 (30) (26–34) | 128 (32) (28–37) | 88 (26) (22–31) | 159 (38) (33–42) | 130 (36) (31–41) | 111 (35) (30–40) |
| Gender: | ||||||
| Male | 63 (50) (42–59) | 59 (46) (38–55) | 47 (53) (43–64) | 90 (57) (49–63) | 74 (57) (48–65) | 57 (51) (42–61) |
| Female | 62 (50) (41–58) | 69 (54) (45–63) | 41 (47) (36–57) | 69 (43) (36–51) | 56 (43) (35–52) | 54 (49) (39–58) |
| Age groups: | ||||||
| 0–<1 month | 20 (16) | 10 (8) | 10 (11) | 19 (12) | 12 (9) | 19 (17) |
| ≥1 month–<1 year | 14 (11) | 27 (21) | 13 (15) | 24 (15) | 21 (16) | 20 (18) |
| ≥1–<5 years | 52 (42) | 33 (26) | 32 (36) | 54 (34) | 36 (28) | 24 (22) |
| ≥5–<12 years | 27 (22) | 29 (3) | 14 (16) | 33 (21) | 35 (27) | 27 (24) |
| ≥12–<18 years | 12 (10) | 29 (23) | 19 (20) | 29 (19) | 26 (20) | 21 (19) |
| How many antibiotics received one patient: | ||||||
| 1 antibiotic | 108 (86) | 102 (79) | 69 (79) | 141 (88) | 102 (79) | 95 (85) |
| 2 antibiotics | 17 (14) | 23 (18) | 15 (17) | 8 (5) | 23 (18) | 12 (11) |
| 3 antibiotics | 0 | 2 (2) | 3 (3) | 6 (4) | 2 (1) | 2 (2) |
| 4 antibiotics | 0 | 1 (1) | 1 (1) | 4 (3) | 3 (2) | 1 (1) |
| 5 antibiotics | 0 | 0 | 0 | 0 | 0 | 1 (1) |
| Year | 2011 | 2012 | 2013 | 2011 | 2012 | 2013 |
|---|---|---|---|---|---|---|
| Month | May | May | May | November | November | November |
| Antibiotics (prescriptions): | N (%) | N (%) | N (%) | N (%) | N (%) | N (%) |
| Total N of prescriptions | 142 | 157 | 106 | 192 | 162 | 132 |
| Aminoglycosides (J01G) | 11 (8) | 15 (10) | 9 (9) | 11 (6) | 13 (8) | 12 (9) |
| β-lactam AB, penicillins (J01C) | 51 (35.9) | 46 (29.3) | 32 (30.2) | 65 (34) | 49 (30) | 23 (17) |
| Macrolides and lincosamides (J01F) | 8 (6) | 6 (4) | 6 (6) | 12 (6.3) | 12 (7.4) | 9 (6.8) |
| Other β-lactam AB (J01D) | 46 (32) | 66 (42) | 42 (40) | 63 (33) | 67 (41) | 54 41) |
| Quinolones (J01M) | 1 (1) | 3 (2) | 2 (2) | 0 | 0 | 5 (4) |
| Other AB (J01X) | 7 (5) | 10 (6) | 7 (7) | 17 (9) | 9 (6) | 10 (8) |
| Sulfonamides and trimethoprim (J01E) | 18 (13) | 11 (7) | 8 (8) | 24 (13) | 12 (7) | 19 (14) |
| Six most often used antibiotics: | ||||||
| Ampicillin | 25 (18) | 10 (6) | 9 (8) | 17 (9) | 5 (3) | 6 (5) |
| Ceftriaxone | 12 (8) | 13 (8) | 16 (15) | 19 (10) | 25 (15) | 23 (17) |
| Amoxicillin | 11 (8) | 19 (12) | 8 (8) | 22 (11) | 21 913) | 7 (5) |
| Cefuroxime | 9 (6) | 22 (14) | 9 (8) | 12 (6) | 18 (11) | 15 (14) |
| Sulfamethoxazole & trimethoprim | 18 (13) | 11 (7) | 8 (8) | 24 (13) | 12 (7) | 15 (14) |
| Penicillin G | 14 (10) | 11 (7) | 11 (10) | 19 (10) | 17 (10) | 8 (6) |
| Route of administration: | ||||||
| Intravenous | 108 (76) | 135 (86) | 91 (86) | 146 (76) | 131 (81) | 100 (76) |
| Oral | 34 (24) | 22 (14) | 15 (14) | 46 (24) | 31 (19) | 32 (24) |
| Year | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2017 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| DDD/100 BD: | |||||||||||
| Aminoglycosides (J01G) | 3.9 | 4.0 | 3.3 | 3.3 | 2.5 | 3.3 | 3.0 | 3.6 | 3.5 | 2.3 | 2.3 |
| β-lactam AB, penicillins (J01C) | 23.0 | 24.2 | 19.7 | 20.4 | 22.0 | 22.0 | 19.8 | 19.7 | 14.2 | 18.7 | 21.2 |
| Macrolides and lincosamides (J01F) | 1.3 | 2.1 | 1.2 | 1.0 | 1.4 | 1.9 | 3.4 | 3.5 | 2.6 | 8.1 | 4.9 |
| Other β-lactam AB (J01D) | 6.8 | 10.3 | 9.2 | 8.3 | 9.4 | 11.2 | 12.2 | 16.4 | 16.7 | 15.7 | 16.8 |
| Quinolones (J01M) | 0.7 | 1.3 | 0.5 | 0.3 | 0.5 | 1.8 | 0.6 | 0.9 | 1.9 | 1.4 | 1.0 |
| Tetracyclines (J01A) | 0.2 | 0.5 | 0.6 | 0.4 | 0.5 | 0.6 | 0.4 | 0.2 | 0.4 | 0.3 | 0.2 |
| Other AB (J01X) | 1.9 | 2.7 | 1.1 | 2.2 | 2.3 | 2.4 | 3.3 | 4.0 | 3.4 | 4.3 | 5.2 |
| Sulfonamides and trimethoprim (J01E) | 1.6 | 1.6 | 2.0 | 2.1 | 1.8 | 0.5 | 0.4 | 0.8 | 3.2 | 2.4 | 2.7 |
| DDD/100 inpatients: | |||||||||||
| Aminoglycosides (J01G) | 22 | 22 | 19 | 18 | 18 | 16 | 13 | 15 | 18 | 12 | 11 |
| β-lactam AB, penicillins (J01C) | 131 | 136 | 112 | 107 | 111 | 90 | 88 | 82 | 74 | 57 | 104 |
| Macrolides and lincosamides (J01F) | 7 | 12 | 7 | 5 | 7 | 9 | 15 | 15 | 13 | 16 | 24 |
| Other β-lactam AB (J01D) | 39 | 58 | 52 | 43 | 47 | 53 | 54 | 69 | 85 | 83 | 82 |
| Quinolones (J01M) | 4 | 7 | 3 | 2 | 3 | 9 | 3 | 4 | 10 | 7 | 5 |
| Tetracyclines (J01A) | 1 | 3 | 3 | 2 | 2 | 3 | 2 | 1 | 2 | 0.3 | 1 |
| Other AB (J01X) | 11 | 15 | 6 | 12 | 12 | 11 | 15 | 17 | 18 | 22 | 25 |
| Sulfonamides and trimethoprim (J01E) | 9 | 9 | 11 | 11 | 9 | 2 | 2 | 3 | 17 | 12 | 13 |
| Year | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2017 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| DDD/100 BD: | |||||||||||
| Amoxicillin | 4.9 | 6.5 | 5.3 | 8.8 | 11.5 | 13.5 | 13.0 | 11.3 | 7.7 | 10.8 | 9.8 |
| Ampicillin | 8.1 | 8.6 | 6.4 | 4.0 | 2.6 | 2.4 | 2.7 | 2.7 | 2.1 | 2.7 | 3.7 |
| Penicillin G | 5.2 | 4.3 | 3.8 | 2.5 | 3.6 | 2.7 | 1.9 | 2.7 | 1.4 | 1.2 | 0.7 |
| Ceftriaxone | 1.3 | 3.9 | 3.0 | 3.2 | 3.9 | 5.0 | 5.1 | 6.7 | 7.4 | 6.7 | 4.0 |
| DDD/100 inpatients: | |||||||||||
| Amoxicillin | 27.8 | 36.5 | 29.8 | 46.2 | 57.9 | 64.7 | 56.2 | 47.1 | 40.2 | 56.8 | 47.9 |
| Ampicillin | 46.0 | 48.0 | 36.3 | 21.2 | 13.1 | 11.7 | 12.1 | 11.3 | 11.1 | 14.0 | 17.9 |
| Penicillin G | 30.0 | 25.8 | 21.7 | 13.3 | 18.0 | 13.0 | 8.3 | 11.2 | 7.3 | 6.5 | 3.5 |
| Ceftriaxone | 7.4 | 21.8 | 17.3 | 16.7 | 19.5 | 24.1 | 22.7 | 28.1 | 38.2 | 35.1 | 19.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sviestina, I.; Mozgis, D. Observational Study of Antibiotic Usage at the Children’s Clinical University Hospital in Riga, Latvia. Medicina 2018, 54, 74. https://doi.org/10.3390/medicina54050074
Sviestina I, Mozgis D. Observational Study of Antibiotic Usage at the Children’s Clinical University Hospital in Riga, Latvia. Medicina. 2018; 54(5):74. https://doi.org/10.3390/medicina54050074
Chicago/Turabian StyleSviestina, Inese, and Dzintars Mozgis. 2018. "Observational Study of Antibiotic Usage at the Children’s Clinical University Hospital in Riga, Latvia" Medicina 54, no. 5: 74. https://doi.org/10.3390/medicina54050074
APA StyleSviestina, I., & Mozgis, D. (2018). Observational Study of Antibiotic Usage at the Children’s Clinical University Hospital in Riga, Latvia. Medicina, 54(5), 74. https://doi.org/10.3390/medicina54050074





