The Pre-Transplant Drop in Panel-Reactive Antibodies Titer Evaluated Using Complement-Dependent Cytotoxicity (PRA-CDC) and the Risk of Early Acute Rejection in Sensitized Kidney Transplant Recipients
Abstract
1. Background
2. Materials and Methods
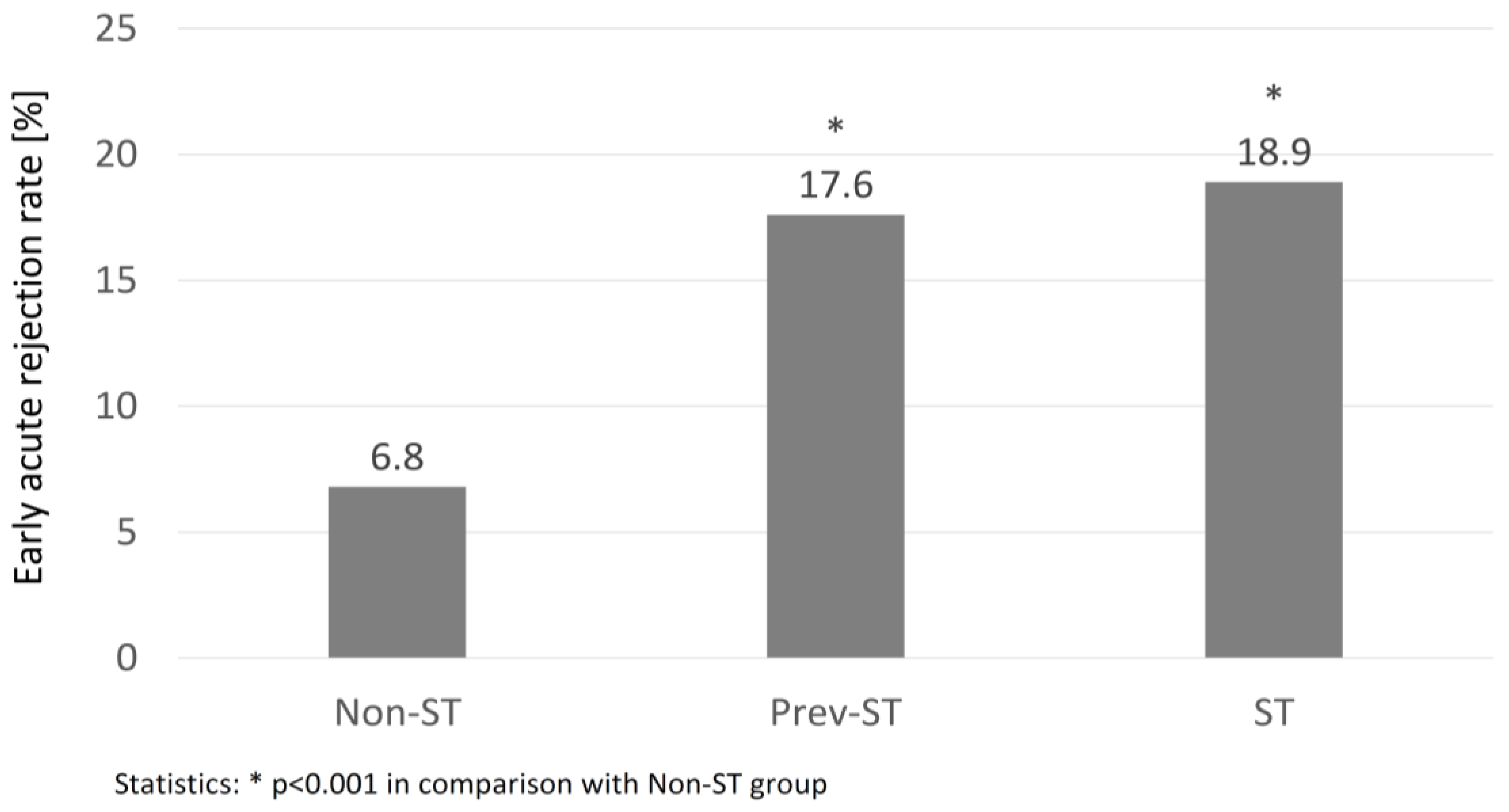
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Soosay, A.; O’Neill, D.; Counihan, A.; Hickey, D.; Keogan, M. Causes of sensitization in patients awaiting renal transplantation in Ireland. Ir. Med. J. 2003, 96, 109–112. [Google Scholar] [PubMed]
- Lieber, S.R.; Perez, F.V.T.; Tabossi, M.R.; Persoli, L.B.; Marques, S.B.; Mazzali, M.; Alves-Filho, G.; de Souza, C.A. Effects of panel-reactive antibodies in predicting crossmatch selection of cadaveric kidney recipients. Transplant. Proc. 2007, 39, 429–431. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Kim, S.J.; Lee, D.S.; Lee, H.H.; Joh, J.W.; Lee, S.K.; Oh, H.Y.; Kim, D.J.; Kim, Y.G.; Huh, W.S.; et al. Effect of panel-reactive antibody positivity on graft rejection before or after kidney transplantation. Transplant. Proc. 2004, 36, 2009–2010. [Google Scholar] [CrossRef] [PubMed]
- Jun, K.W.; Kim, M.H.; Hwang, J.K.; Kim, S.D.; Park, S.C.; Won, Y.S.; Moon, I.S.; Chung, B.H.; Choi, B.S.; Yang, C.W.; et al. Impact of pretransplant panel-reactive antibody level on renal graft survival in patients with a negative crossmatch and no donor-specific antibody. Transplant. Proc. 2016, 48, 770–772. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, H.; Moszkowska, G.; Debska-Slizien, A.; Zieliński, M.; Jankowska, M.; Rutkowski, B.; Trzonkowski, P. Diagnostic options in pre- and post-transplant immune risk assessment in kidney recipients. Nephrological. Forum 2011, 4, 320–330. [Google Scholar]
- Bączkowska, T.; Debska-Slizien, A.; Durlik, M.; Klinger, M.; Moszkowska, G.; Wiecek, A. Guidelines on Immunosuppressive Therapy in Solid Organ Transplant Recipients; Durlik, M., Zieniewicz, K., Eds.; Polish Transplantation Society: Warszawa, Poland, December 2016. [Google Scholar]
- Meng, H.L.; Jin, X.B.; Li, X.T.; Wang, H.W.; Lu, J.J. Impact of human leukocyte antigen matching and recipients’ panel reactive antibodies on two-year outcomes in presensitized renal allograft recipients. Chin. Med. J. 2009, 122, 420–426. [Google Scholar] [PubMed]
- Tahir, M.T.; Mehra, N.K.; Taneja, V.; Singal, D.P.; Dash, S.C.; Singh, R.; Mehta, S.N. Effect of panel reactive antibody on live related donor kidney transplantation: Indian experience. Transplant. Immunol. 1994, 2, 238–242. [Google Scholar] [CrossRef]
- Speiser, D.E.; Jeannet, M. Renal transplantation to sensitized patients: Decrease graft survival probability associated with a positive historical crossmatch. Transplant. Immunol. 1995, 3, 330–334. [Google Scholar] [CrossRef]
- Sanfilippo, F.; Vaughn, W.K.; Spees, E.K.; Bollinger, R.R. Cadaver renal transplantation ignoring peak-reactive sera in patients with markedly decreasing pretransplant sensitization. Transplantation 1984, 38, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Sanfilippo, F.; Vaughn, W.K.; Spees, E.K.; Bollinger, R.R. Influence of changes in pretransplant sensitization on patient and graft survival in cadaver renal transplantation. Transplantation 1984, 38, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Scomik, J.C.; Brunson, M.E.; Howard, R.J.; Pfaff, W.W. Alloimmunization, memory, and the interpretation of crossmatch results for renal transplantation. Transplantation 1992, 5, 389–394. [Google Scholar]
- Bostock, I.C.; Alberu, J.; Arvizu, A.; Hernández-Mendez, E.A.; De-Santiago, A.; González-Tableros, N.; López, M.; Castelán, N.; Contreras, A.G.; Morales-Buenrostro, L.E.; et al. Probability of deceased donor kidney transplantation based on % PRA. Transplant. Immunol. 2013, 28, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Gu, J.; Qiu, J.; Wang, C.; Fei, J.; Deng, S.; Li, J.; Huang, G.; Fu, Q.; Chen, L. Efficacy and safety of thymoglobulin and basiliximab in kidney transplant patients at high risk for acute rejection and delayed graft function. Exp. Clin. Transplant. 2013, 4, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Susal, C.; Seidl, C.; Schonemann, C.; Heinemann, F.M.; Kauke, T.; Gombos, P.; Kelsch, R.; Arns, W.; Bauerfeind, U.; Hallensleben, M.; et al. Determination of unacceptable HLA antigen mismatches in kidney transplant recipients: Recommendations of the German Society for Immunogenetics. Tissue Antigens 2015, 86, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Dukat-Mazurek, A.; Zielinska, H.; Moszkowska, G.; Bieniaszewska, M.; Trzonkowski, P. The role of virtual crossmatch in solid organ transplantation. Transplantol. Forum 2016, 3, 5–10. [Google Scholar]
| Group 1 | Group 2 | Group 3 | ANOVA/Chi2 | |
|---|---|---|---|---|
| Non-ST | Prev-ST | ST | ||
| N = 437 | N = 53 | N = 38 | ||
| Age [yrs] | 50.6 (49.3–51.8) | 51.5 (48.0–55.0) | 47.9 (43.7–52.1) | 0.40 |
| Gender (M/F) | 283/154 | 24/29 | 15/23 | <0.001 * |
| Dialysis vintage [mo] | 42 (39–45) | 65 (54–76) | 69 (52–86) | <0.001 for 2. and 3. vs. 1. |
| Retransplant [%] | 11.5 | 37.7 | 55.3 | <0.001 * |
| HLA mismatch I | 2.2 (2.1–2.3) | 1.9 (1.6–2.2) | 2.0 (1.7–2.3) | 0.16 |
| HLA mismatch II | 0.5 (0.5–0.6) | 0.6 (0.4–0.8) | 0.8 (0.5–1.0) | 0.02 |
| PRA last [%] | 0.4 (0.2–0.5) | 3.9 (2.3–5.5) | 48.6 (41.2–55.9) | <0.001 (0.01 for 2. vs. 1.) |
| PRA max [%] | 1.7 (1.3–2.1) | 38.4 (33.2–43.6) | 61.5 (53.3–69.7) | <0.001 * |
| CyA/Tc [n] | 83/351 | 4/49 | 5/33 | 0.09 |
| MMF [%] | 97.7 | 100 | 100 | 0.34 |
| Induction total n (%) | 155 (35.8) | 45 (84.9) | 38 (100) | <0.001 * |
| SIMU n (%) | 118 (27.2) | 20 (37.7) | 5 (13.2) | Chi2 test 0.03 |
| ATG n (%) | 37 (8.5) | 25 (47.2) | 33 (86.8) | <0.001 * |
| XM+ [%] | 18.4 (16.7–20.1) | 53.7 (46.6–60.7) | 76.9 (70.6–83.2) | <0.001 * |
| Time on the waitlist [mo] | 9.6 (8.7–10.4) | 14.8 (12.0–17.6) | 12.5 (8.9–16.0) | 0.01 for 2. vs. 1. |
| DGF [%] | 29.4 | 39.2 | 32.4 | 0.39 |
| PGN [%] | 1.6 | 1.9 | 2.6 | 0.89 |
| Independent Variable | Beta | SE | OR | 95% CI | p Value |
|---|---|---|---|---|---|
| Recipient gender (M vs. F) | −0.97 | 0.65 | 0.38 | 0.11–1.37 | 0.14 |
| Retransplant | 0.26 | 0.60 | 1.30 | 0.40–4.26 | 0.67 |
| Lack of induction therapy | 1.23 | 1.03 | 3.43 | 0.46–25.8 | 0.23 |
| Any HLA class II mismatch | 1.49 | 0.75 | 4.43 | 1.02–19.3 | <0.05 |
| PRA titer drop | 0.09 | 0.64 | 1.09 | 0.31–3.85 | 0.89 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolonko, A.; Bzoma, B.; Giza, P.; Styrc, B.; Sobolewski, M.; Chudek, J.; Dębska-Ślizień, A.; Więcek, A. The Pre-Transplant Drop in Panel-Reactive Antibodies Titer Evaluated Using Complement-Dependent Cytotoxicity (PRA-CDC) and the Risk of Early Acute Rejection in Sensitized Kidney Transplant Recipients. Medicina 2018, 54, 66. https://doi.org/10.3390/medicina54050066
Kolonko A, Bzoma B, Giza P, Styrc B, Sobolewski M, Chudek J, Dębska-Ślizień A, Więcek A. The Pre-Transplant Drop in Panel-Reactive Antibodies Titer Evaluated Using Complement-Dependent Cytotoxicity (PRA-CDC) and the Risk of Early Acute Rejection in Sensitized Kidney Transplant Recipients. Medicina. 2018; 54(5):66. https://doi.org/10.3390/medicina54050066
Chicago/Turabian StyleKolonko, Aureliusz, Beata Bzoma, Piotr Giza, Beata Styrc, Michał Sobolewski, Jerzy Chudek, Alicja Dębska-Ślizień, and Andrzej Więcek. 2018. "The Pre-Transplant Drop in Panel-Reactive Antibodies Titer Evaluated Using Complement-Dependent Cytotoxicity (PRA-CDC) and the Risk of Early Acute Rejection in Sensitized Kidney Transplant Recipients" Medicina 54, no. 5: 66. https://doi.org/10.3390/medicina54050066
APA StyleKolonko, A., Bzoma, B., Giza, P., Styrc, B., Sobolewski, M., Chudek, J., Dębska-Ślizień, A., & Więcek, A. (2018). The Pre-Transplant Drop in Panel-Reactive Antibodies Titer Evaluated Using Complement-Dependent Cytotoxicity (PRA-CDC) and the Risk of Early Acute Rejection in Sensitized Kidney Transplant Recipients. Medicina, 54(5), 66. https://doi.org/10.3390/medicina54050066






