Evaluation of the Sleep-Prolonging Effect of Lagenaria vulgaris and Cucurbita pepo Extracts on Pentobarbital-Induced Sleep and Possible Mechanisms of Action
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Collection and Extraction
2.3. Animals
2.4. Sleep Induction Protocol
2.5. Median Lethal Dose (LD50) Determination
2.6. Cytotoxicity and Neurotoxicity Assessment
2.7. Statistics
2.8. Ethical Disclosures
3. Results
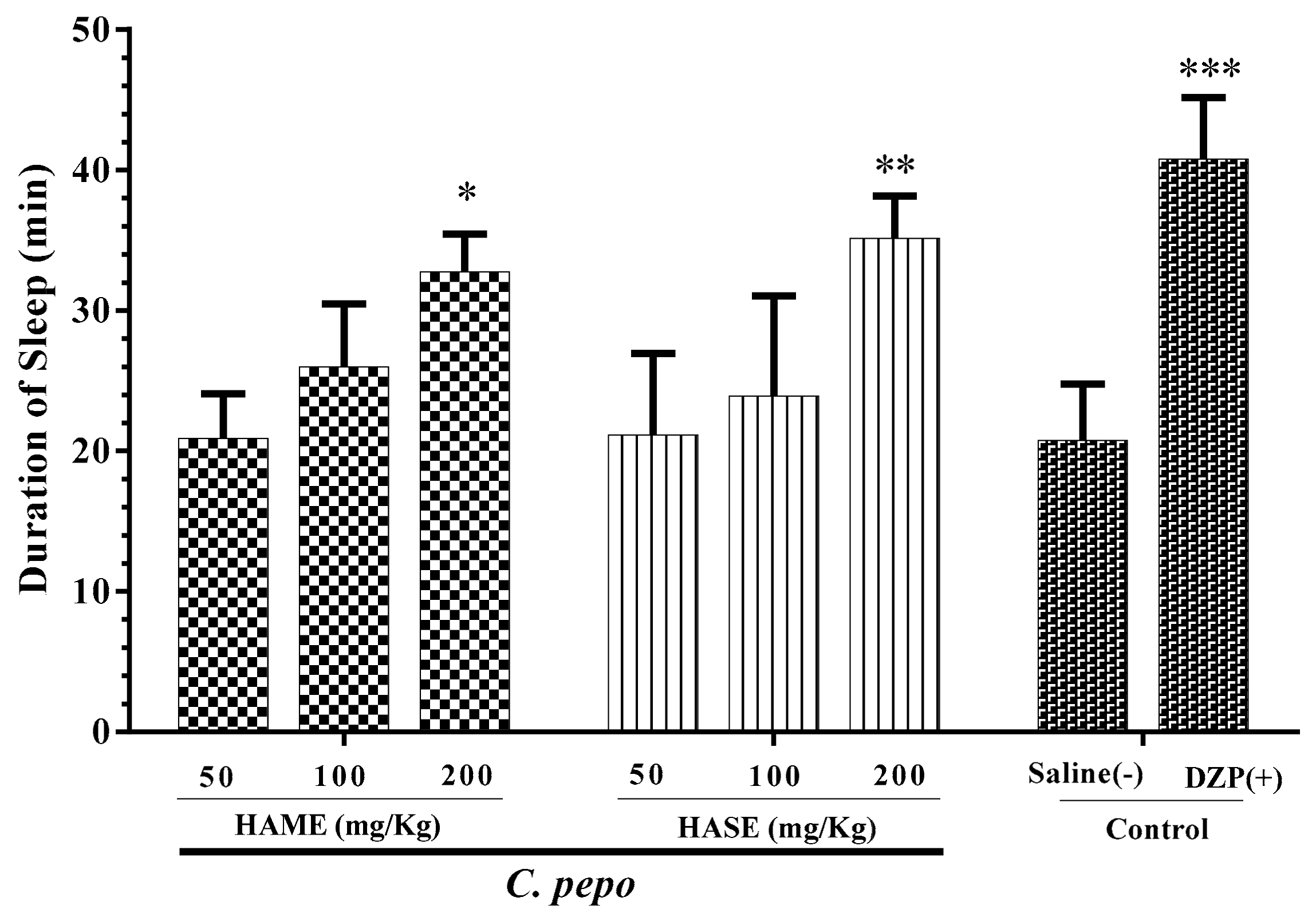
3.1. Effect of C. Pepo on Sleep Duration
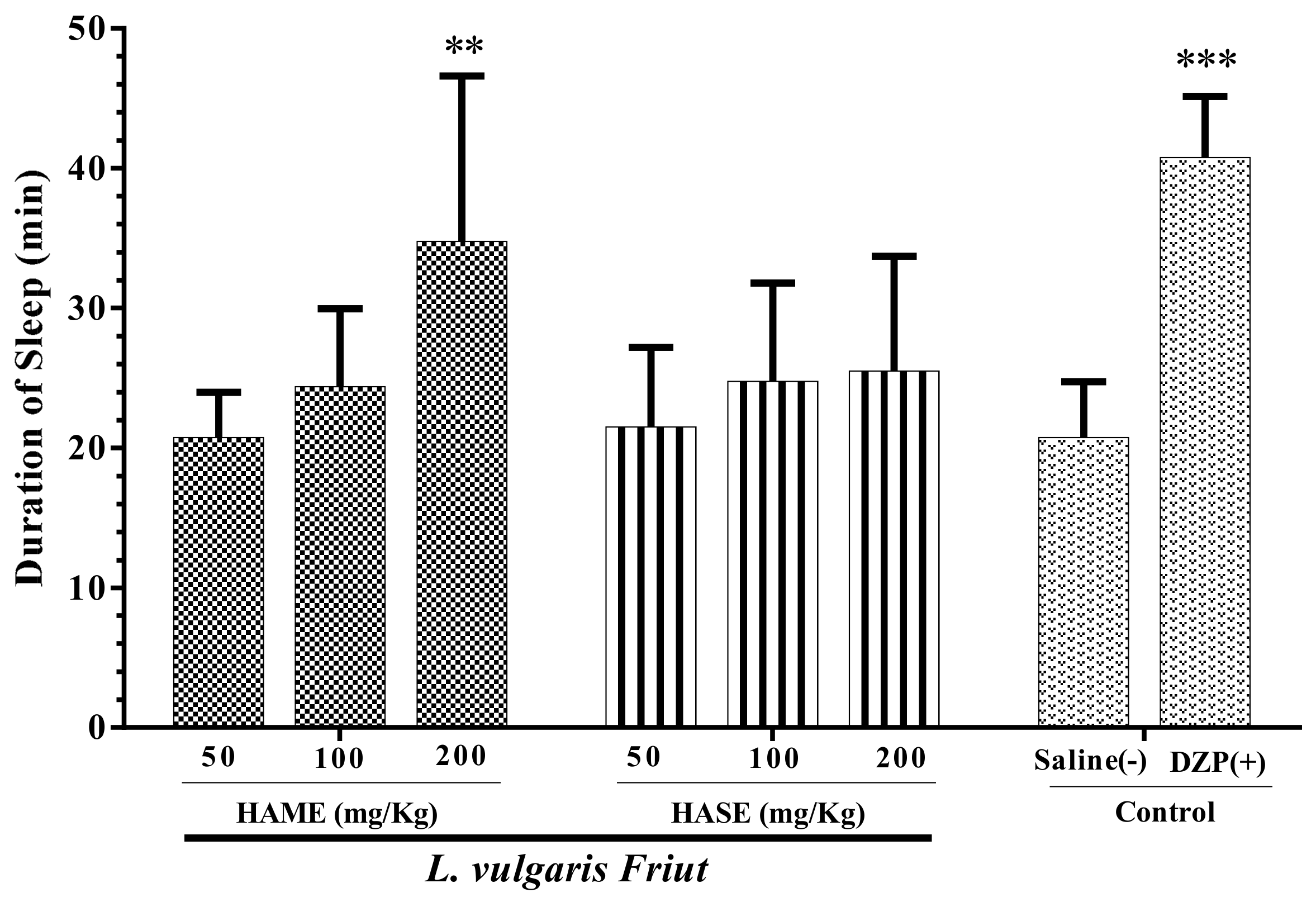
3.2. Effect of Fruit Extracts of L. vulgaris on Sleep Duration
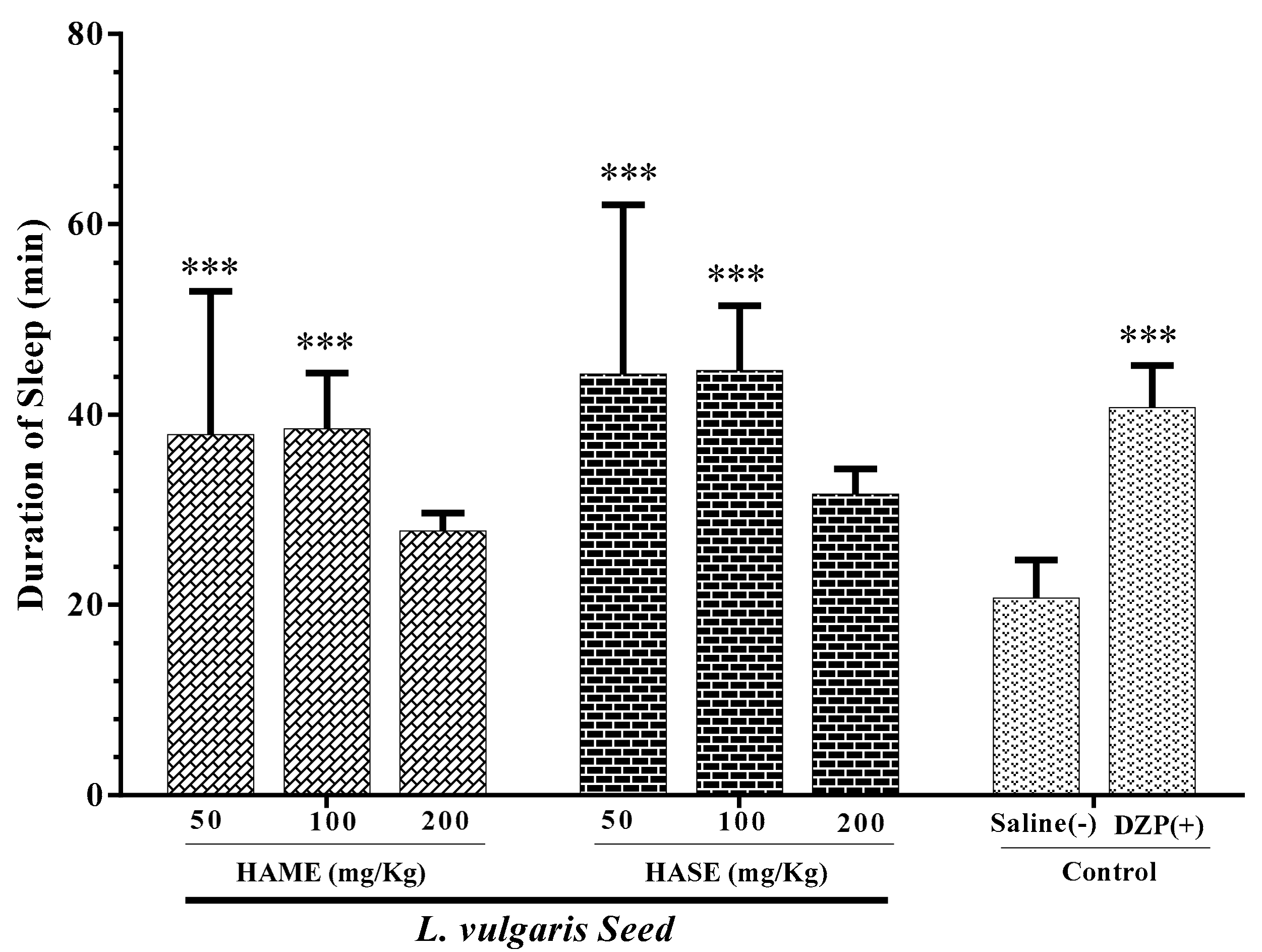
3.3. Effect of Seed Extracts of L. Vulgaris on Sleep Duration
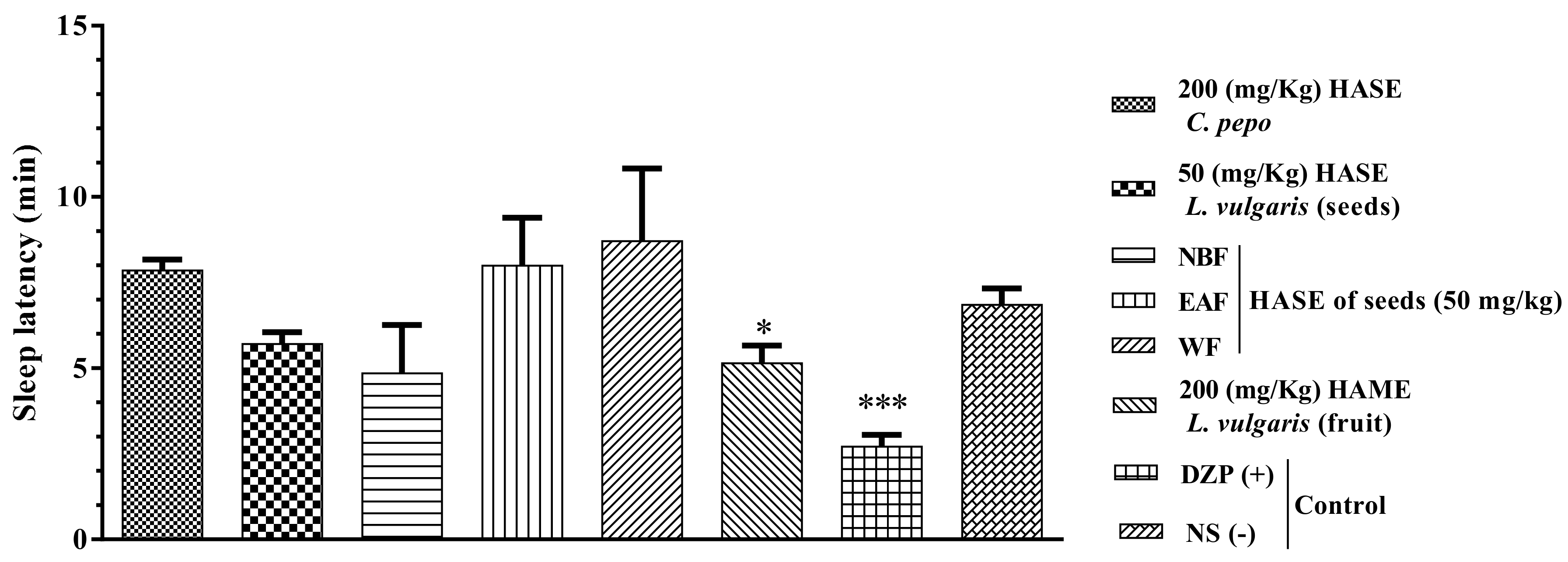
3.4. Sleep Latency
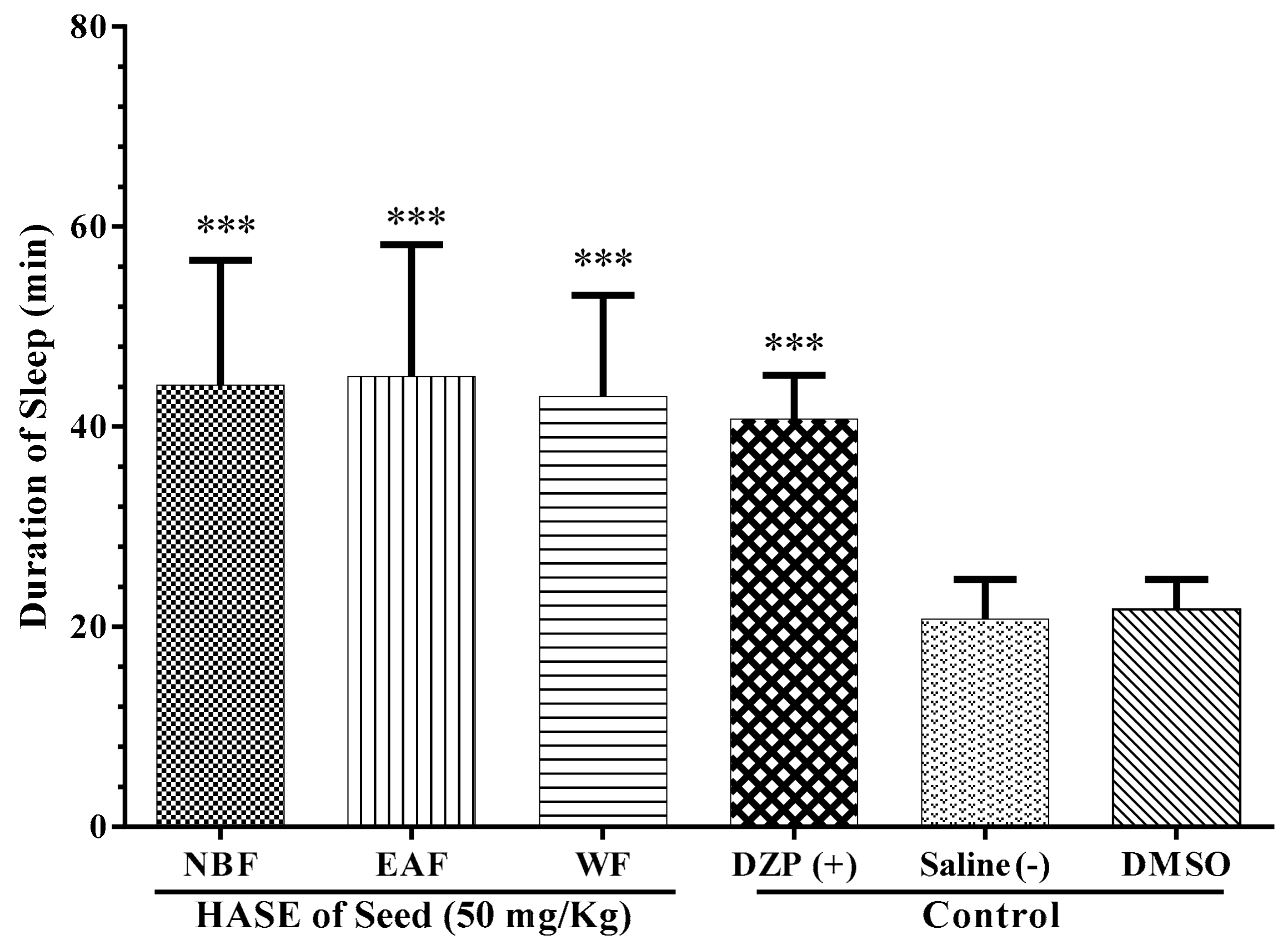
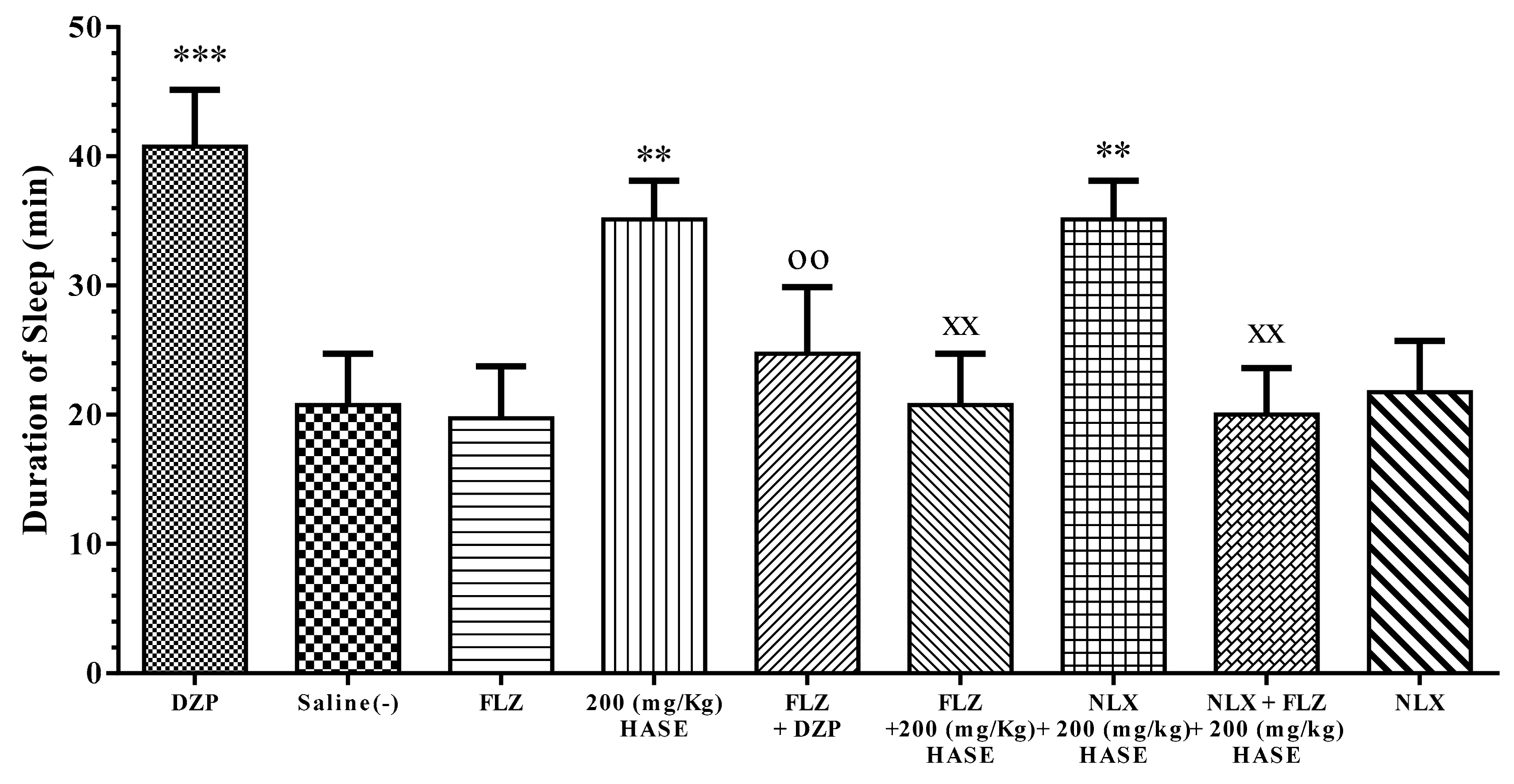
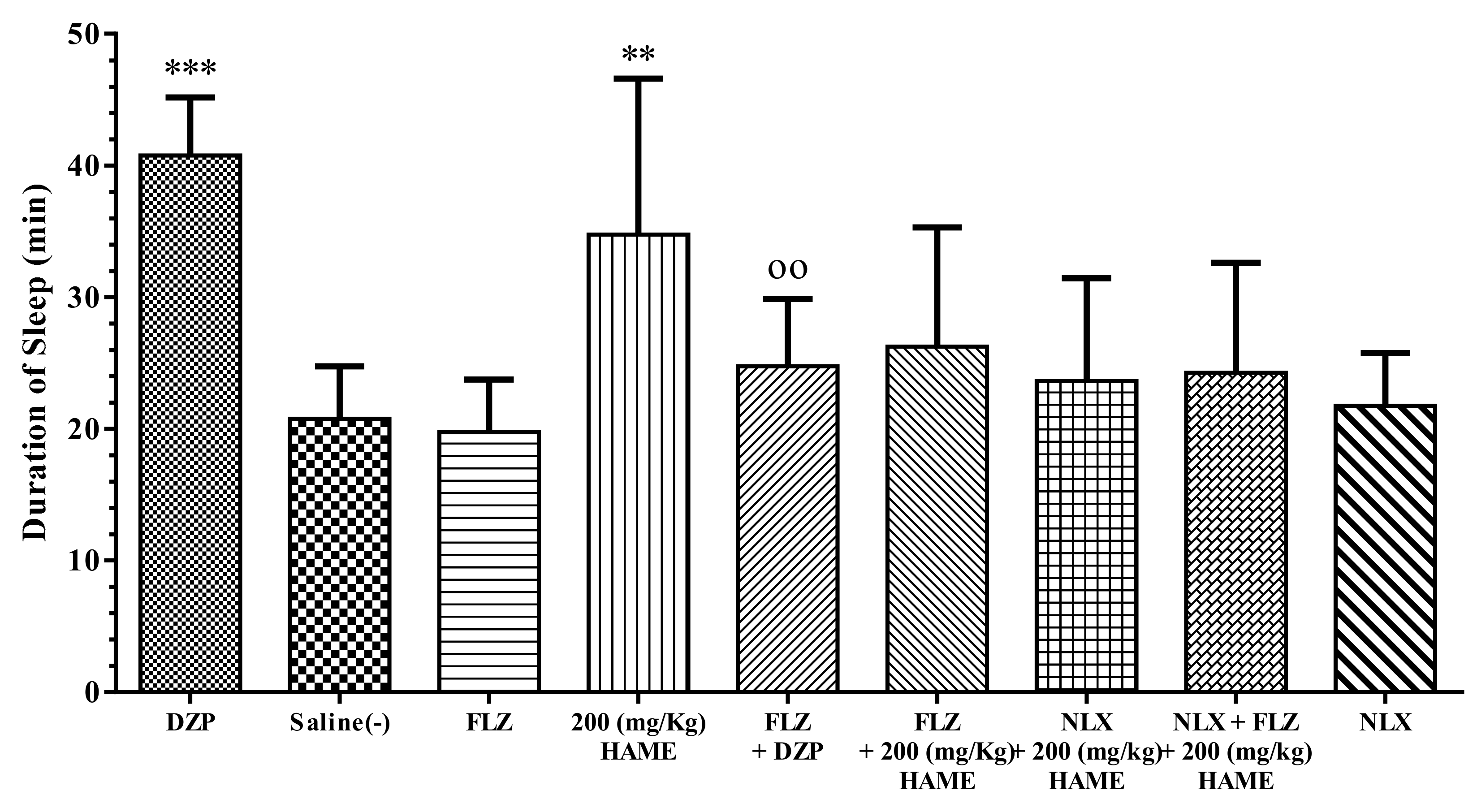
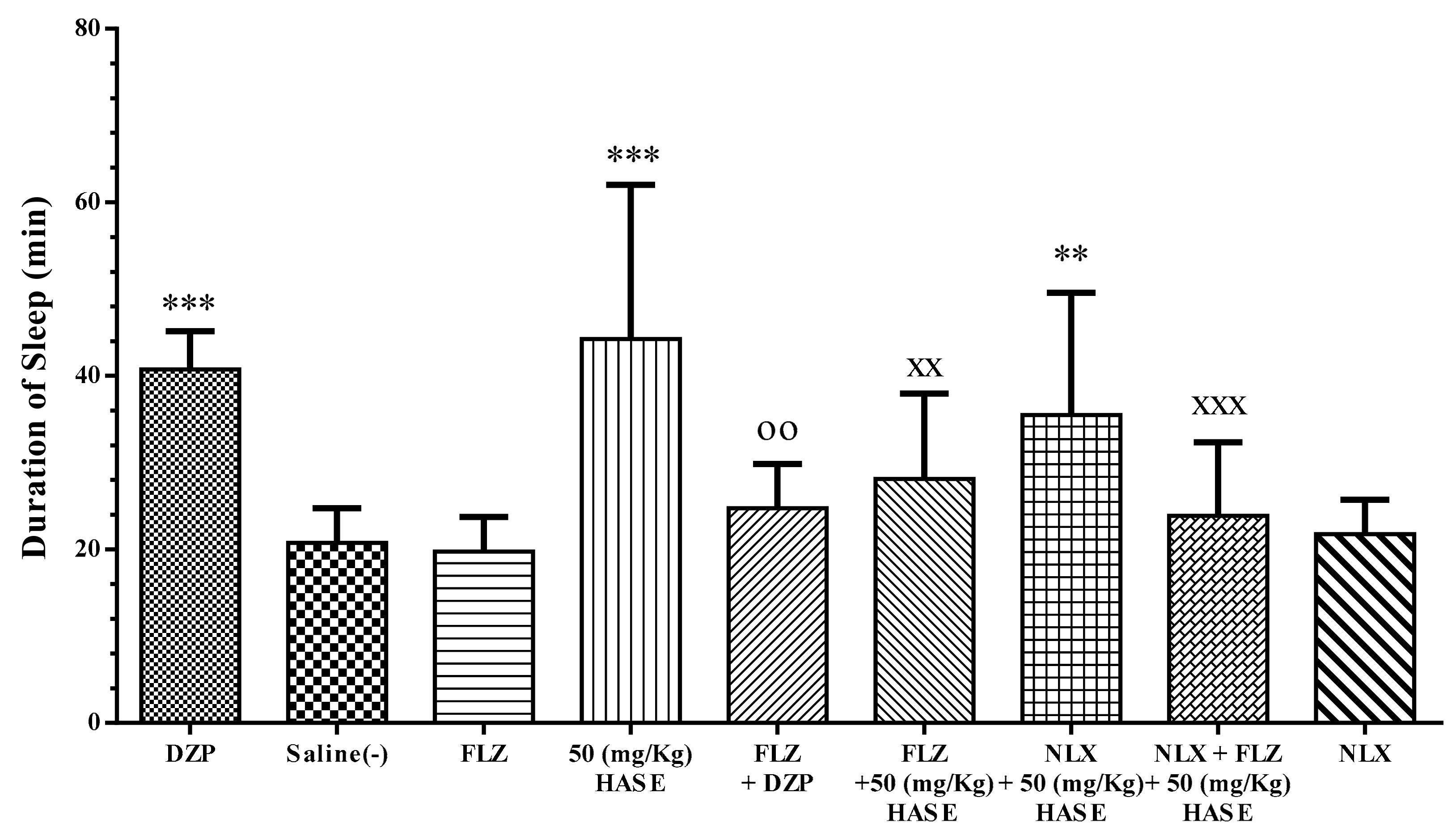
3.5. Sleep Mechanism
3.6. Toxicity Assessments
3.6.1. LD50 Determination
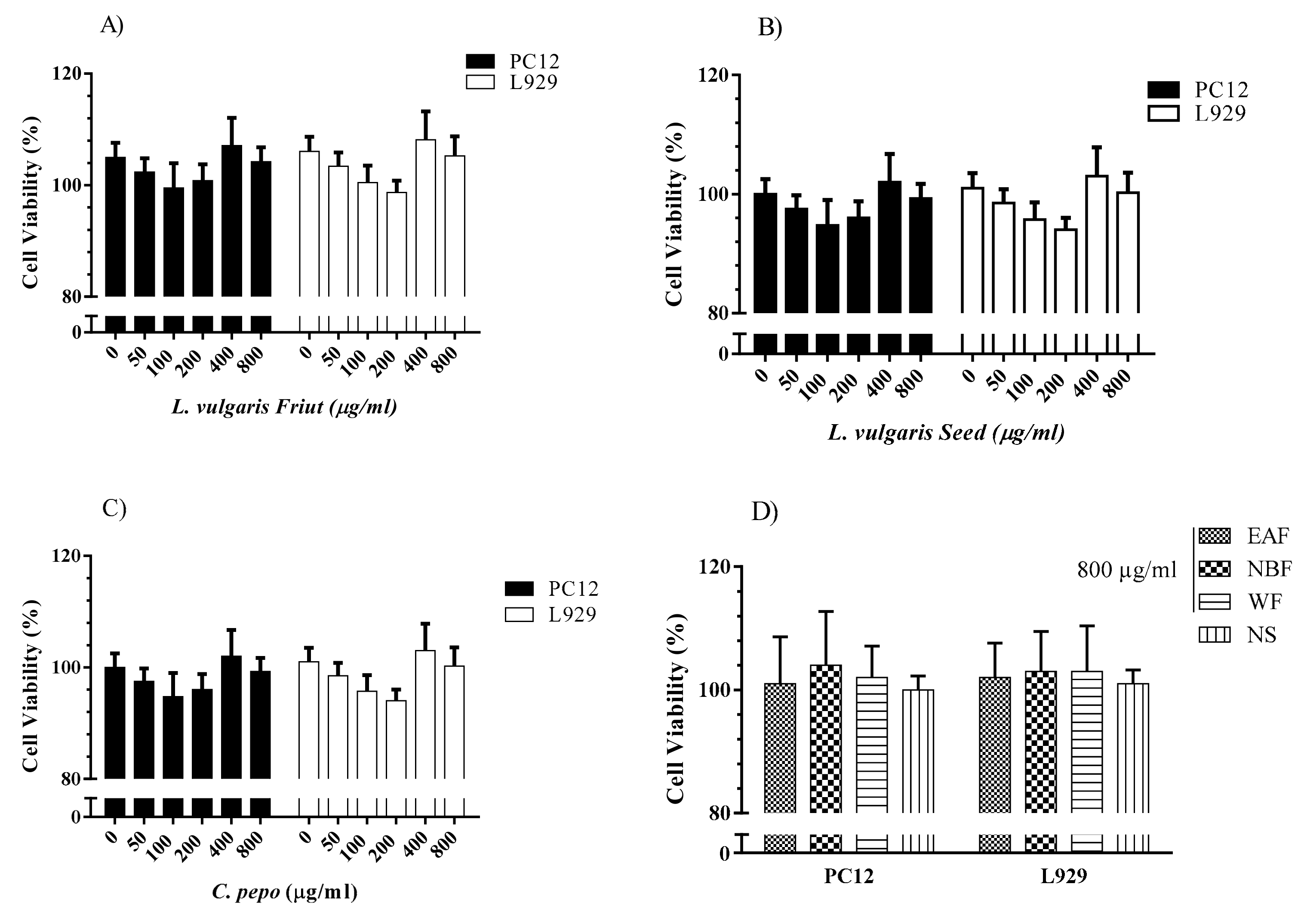
3.6.2. Evaluation of the Cytotoxicity Effects of C. pepo and L. vulgaris
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Stone, K.L.; Ensrud, K.E.; Ancoli-Israel, S. Sleep, insomnia and falls in elderly patients. Sleep Med. 2008, 9 (Suppl. 1), S18–S22. [Google Scholar] [CrossRef]
- Askari, V.R.; Rahimi, V.B.; Ghorbani, A.; Rakhshandeh, H. Hypnotic Effect of Ocimum basilicum on Pentobarbital-Induced Sleep in Mice. Iran. Red Crescent Med. J. 2016, 18, e24261. [Google Scholar] [CrossRef] [PubMed]
- Orzel-Gryglewska, J. Consequences of sleep deprivation. Int. J. Occup. Med. Environ. Health 2010, 23, 95–114. [Google Scholar] [CrossRef] [PubMed]
- Mason, R.H.; West, S.D.; Kiire, C.A.; Groves, D.C.; Lipinski, H.J.; Jaycock, A.; Chong, V.N.; Stradling, J.R. High prevalence of sleep disordered breathing in patients with diabetic macular edema. Retina 2012, 32, 1791–1798. [Google Scholar] [CrossRef] [PubMed]
- Postuma, R.B.; Gagnon, J.F.; Vendette, M.; Fantini, M.L.; Massicotte-Marquez, J.; Montplaisir, J. Quantifying the risk of neurodegenerative disease in idiopathic REM sleep behavior disorder. Neurology 2009, 72, 1296–1300. [Google Scholar] [CrossRef] [PubMed]
- Limandri, B.J. Insomnia: Will Medication Bring Rest? J. Psychosoc. Nurs. Ment. Health Serv. 2018, 56, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Roehrs, T.; Roth, T. Insomnia pharmacotherapy. Neurotherapeutics 2012, 9, 728–738. [Google Scholar] [CrossRef] [PubMed]
- Helft, P.R.; Williams, J.R.; Bandy, R.J. Opiate written behavioral agreements: A case for abandonment. Perspect. Biol. Med. 2014, 57, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Longo, L.P.; Johnson, B. Addiction: Part I. Benzodiazepines-side effects, abuse risk and alternatives. Am. Fam. Phys. 2000, 61, 2121–2128. [Google Scholar]
- Trauner, G.; Khom, S.; Baburin, I.; Benedek, B.; Hering, S.; Kopp, B. Modulation of GABAA receptors by valerian extracts is related to the content of valerenic acid. Planta Med. 2008, 74, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Butterweck, V.; Brattstroem, A.; Grundmann, O.; Koetter, U. Hypothermic effects of hops are antagonized with the competitive melatonin receptor antagonist luzindole in mice. J. Pharm. Pharmacol. 2007, 59, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Hajhashemi, V.; Safaei, A. Hypnotic effect of Coriandrum sativum, Ziziphus jujuba, Lavandula angustifolia and Melissa officinalis extracts in mice. Res. Pharm. Sci. 2015, 10, 477–484. [Google Scholar] [PubMed]
- Ziegler, G.; Ploch, M.; Miettinen-Baumann, A.; Collet, W. Efficacy and tolerability of valerian extract LI 156 compared with oxazepam in the treatment of non-organic insomnia—A randomized, double-blind, comparative clinical study. Eur. J. Med. Res. 2002, 7, 480–486. [Google Scholar] [PubMed]
- Grundmann, O.; Wang, J.; McGregor, G.P.; Butterweck, V. Anxiolytic activity of a phytochemically characterized Passiflora incarnata extract is mediated via the GABAergic system. Planta Med. 2008, 74, 1769–1773. [Google Scholar] [CrossRef] [PubMed]
- Rolland, A.; Fleurentin, J.; Lanhers, M.C.; Misslin, R.; Mortier, F. Neurophysiological effects of an extract of Eschscholzia californica Cham. (Papaveraceae). Phytother. Res. 2001, 15, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Boonen, G.; Haberlein, H. Influence of genuine kavapyrone enantiomers on the GABA-A binding site. Planta Med. 1998, 64, 504–506. [Google Scholar] [CrossRef] [PubMed]
- Yakoot, M.; Helmy, S.; Fawal, K. Pilot study of the efficacy and safety of lettuce seed oil in patients with sleep disorders. Int. J. Gen. Med. 2011, 4, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, M.K.; Kaul, S.C.; Wadhwa, R.; Yanagisawa, M.; Urade, Y. Triethylene glycol, an active component of Ashwagandha (Withania somnifera) leaves, is responsible for sleep induction. PLoS ONE 2017, 12, e0172508. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, M.K.; Aritake, K.; Takeuchi, A.; Yanagisawa, M.; Urade, Y. Octacosanol restores stress-affected sleep in mice by alleviating stress. Sci. Rep. 2017, 7, 8892. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, R.P.; Kalariya, M.; Parmar, S.K.; Sheth, N.R. Phytochemical and pharmacological review of Lagenaria sicereria. J. Ayurveda Integr. Med. 2010, 1, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Roopan, S.M.; Rajeswari, V.D.; Kalpana, V.N.; Elango, G. Biotechnology and pharmacological evaluation of Indian vegetable crop Lagenaria siceraria: An overview. Appl. Microbiol. Biotechnol. 2016, 100, 1153–1162. [Google Scholar] [CrossRef] [PubMed]
- Noor Mohammadi, M.R.; Haji, Y. Evaluating the Effect of Herbal Medicine in the Treatment of Depression, From the Viewpoint of Islam and Medical Sciences. Islam. Lifestyle Cent. Health 2013, 1, 37–42. [Google Scholar] [CrossRef]
- Askari, V.R.; Rahimi, V.B.; Zamani, P.; Fereydouni, N.; Rahmanian-Devin, P.; Sahebkar, A.H.; Rakhshandeh, H. Evaluation of the effects of Iranian propolis on the severity of post operational-induced peritoneal adhesion in rats. Biomed. Pharmacother. 2018, 99, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, V.B.; Askari, V.R.; Hosseini, M.; Yousefsani, B.S.; Sadeghnia, H.R. Anticonvulsant Activity of Viola tricolor against Seizures Induced by Pentylenetetrazol and Maximal Electroshock in Mice. Iran. J. Med. Sci. 2018, 2018. [Google Scholar]
- Rahimi, V.B.; Askari, V.R.; Emami, S.A.; Tayarani-Najaran, Z. Anti-melanogenic activity of Viola odorata different extracts on B16F10 murine melanoma cells. Iran. J. Basic Med. Sci. 2017, 20, 242–249. [Google Scholar] [PubMed]
- Akhila, J.S.; Shyamjith, D.; Alwar, M.C. Acute toxicity studies and determination of median lethal dose. Curr. Sci. 2007, 93, 917–920. [Google Scholar]
- Rahimi, V.B.; Askari, V.R.; Mehrdad, A.; Sadeghnia, H.R. Boswellia Serrata Has Promising Impact on Glutamate and Quinolinic Acid-Induced Toxicity on Oligodendroglia Cells: In Vitro Study. Acta Pol. Pharm. 2017, 74, 1803–1811. [Google Scholar]
- Huang, F.; Xiong, Y.; Xu, L.; Ma, S.; Dou, C. Sedative and hypnotic activities of the ethanol fraction from Fructus Schisandrae in mice and rats. J. Ethnopharmacol. 2007, 110, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Alnamer, R.; Alaoui, K.; Bouidida, E.H.; Benjouad, A.; Cherrah, Y. Sedative and Hypnotic Activities of the Methanolic and Aqueous Extracts of Lavandula officinalis from Morocco. Adv. Pharmacol. Sci. 2012, 2012, 270824. [Google Scholar] [PubMed]
- Sharma, M.; Rauniar, G.; Das, B. Experimental study of various central nervous system effects of eugenol in mice and rats. Health Renaiss. 2012, 10, 208–214. [Google Scholar] [CrossRef]
- Adnaik, R.S.; Mohite, S.K. Neuroprotective evaluation of Lagenaria vulgaris extract hypoxic neurotoxicity induced rats. Asian J. Pharm. Clin. Res. 2015, 8, 121–124. [Google Scholar]
- Bahramsoltani, R.; Farzaei, M.H.; Abdolghaffari, A.H.; Rahimi, R.; Samadi, N.; Heidari, M.; Esfandyari, M.; Baeeri, M.; Hassanzadeh, G.; Abdollahi, M.; et al. Evaluation of phytochemicals, antioxidant and burn wound healing activities of Cucurbita moschata Duchesne fruit peel. Iran. J. Basic Med. Sci. 2017, 20, 798–805. [Google Scholar] [PubMed]
- He, M.; Min, J.W.; Kong, W.L.; He, X.H.; Li, J.X.; Peng, B.W. A review on the pharmacological effects of vitexin and isovitexin. Fitoterapia 2016, 115, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, E.; Nassiri-Asl, M.; Shafeei, M.; Sheikhi, M. Neuroprotective effects of vitexin, a flavonoid, on pentylenetetrazole-induced seizure in rats. Chem. Biol. Drug Des. 2012, 80, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.J.; Park, H.J.; Gao, Q.; dela Pena, I.J.; Park, S.J.; Lee, H.E.; Woo, H.; Kim, H.J.; Cheong, J.H.; Hong, E.; et al. Ursolic acid enhances pentobarbital-induced sleeping behaviors via GABAergic neurotransmission in mice. Eur. J. Pharmacol. 2015, 762, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Fajemiroye, J.O.; Galdino, P.M.; Florentino, I.F.; Da Rocha, F.F.; Ghedini, P.C.; Polepally, P.R.; Zjawiony, J.K.; Costa, E.A. Plurality of anxiety and depression alteration mechanism by oleanolic acid. J. Psychopharmacol. 2014, 28, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.J.; Park, H.J.; Gao, Q.; Lee, H.E.; Park, S.J.; Hong, E.; Jang, D.S.; Shin, C.Y.; Cheong, J.H.; Ryu, J.H. Positive effects of β-amyrin on pentobarbital-induced sleep in mice via GABAergic neurotransmitter system. Behav. Brain Res. 2015, 291, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Askari, V.R.; Fereydouni, N.; Rahimi, V.B.; Askari, N.; Sahebkar, A.H.; Rahmanian-Devin, P.; Samzadeh-Kermani, A. β-Amyrin, the cannabinoid receptors agonist, abrogates mice brain microglial cells inflammation induced by lipopolysaccharide/interferon-γ and regulates Mφ1/Mφ2 balances. Biomed. Pharmacother. 2018, 101, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Librowski, T.; Czarnecki, R.; Mendyk, A.; Jastrzebska, M. Influence of new monoterpene homologues of GABA on the central nervous system activity in mice. Pol. J. Pharmacol. 2000, 52, 317–321. [Google Scholar] [PubMed]
- Nesterkina, M.; Kravchenko, I. Synthesis and Pharmacological Properties of Novel Esters Based on Monoterpenoids and Glycine. Pharmaceuticals 2017, 10, 47. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baradaran Rahimi, V.; Askari, V.R.; Tajani, A.S.; Hosseini, A.; Rakhshandeh, H. Evaluation of the Sleep-Prolonging Effect of Lagenaria vulgaris and Cucurbita pepo Extracts on Pentobarbital-Induced Sleep and Possible Mechanisms of Action. Medicina 2018, 54, 55. https://doi.org/10.3390/medicina54040055
Baradaran Rahimi V, Askari VR, Tajani AS, Hosseini A, Rakhshandeh H. Evaluation of the Sleep-Prolonging Effect of Lagenaria vulgaris and Cucurbita pepo Extracts on Pentobarbital-Induced Sleep and Possible Mechanisms of Action. Medicina. 2018; 54(4):55. https://doi.org/10.3390/medicina54040055
Chicago/Turabian StyleBaradaran Rahimi, Vafa, Vahid Reza Askari, Amineh Sadat Tajani, Azar Hosseini, and Hassan Rakhshandeh. 2018. "Evaluation of the Sleep-Prolonging Effect of Lagenaria vulgaris and Cucurbita pepo Extracts on Pentobarbital-Induced Sleep and Possible Mechanisms of Action" Medicina 54, no. 4: 55. https://doi.org/10.3390/medicina54040055
APA StyleBaradaran Rahimi, V., Askari, V. R., Tajani, A. S., Hosseini, A., & Rakhshandeh, H. (2018). Evaluation of the Sleep-Prolonging Effect of Lagenaria vulgaris and Cucurbita pepo Extracts on Pentobarbital-Induced Sleep and Possible Mechanisms of Action. Medicina, 54(4), 55. https://doi.org/10.3390/medicina54040055




