Advocacy of Precision Allergy Molecular Diagnosis in Decision Making for the Eligibility of Customized Allergen Immunotherapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Skin Prick Test, Blood Eosinophlis, and Serological Analysis
2.3. Statistical Analysis
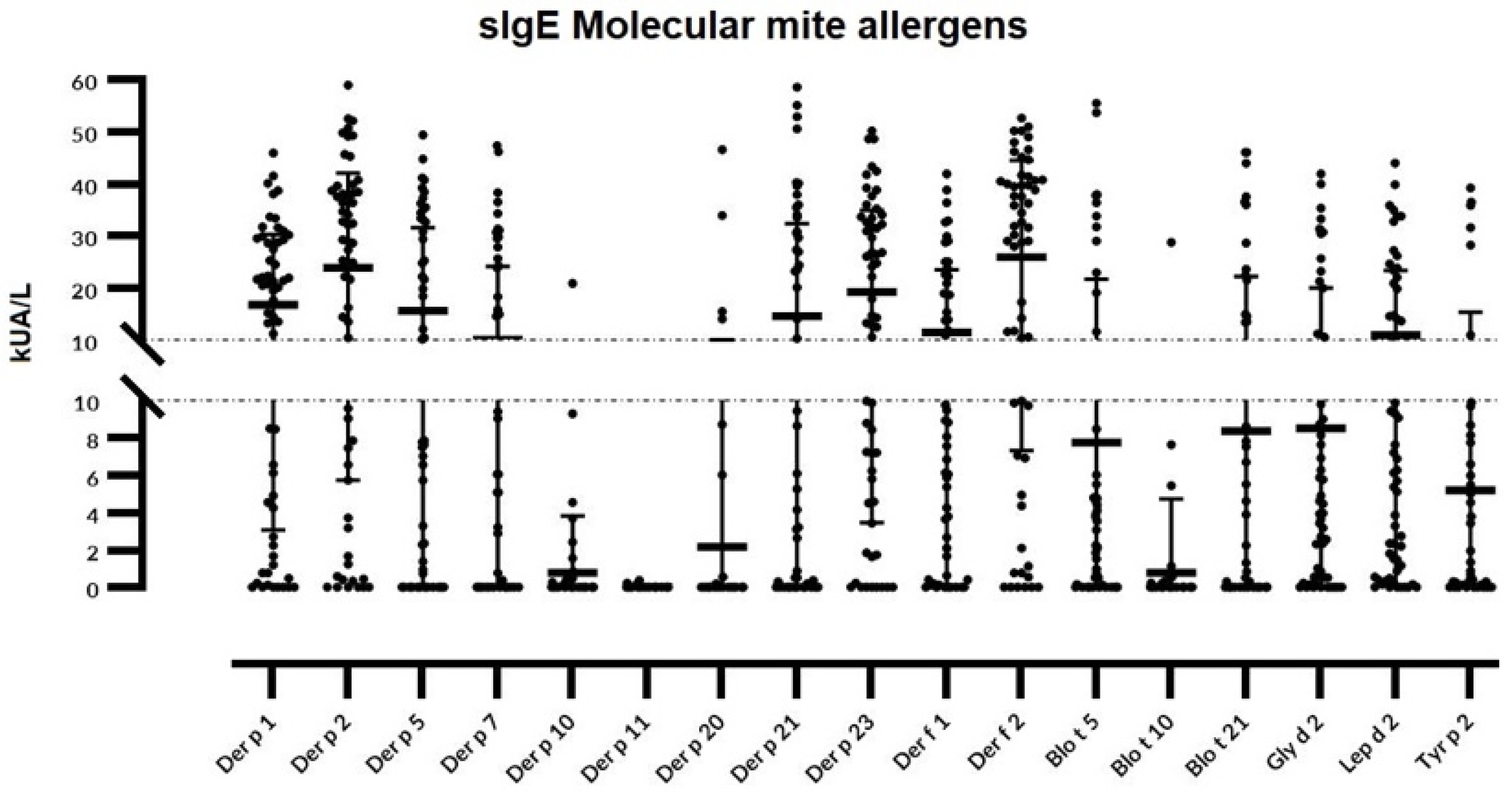
3. Results
3.1. Study Population
3.2. sIgE Reactivity and Individual Molecular Profiles
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Durham, S.R.; Shamji, M.H. Allergen immunotherapy: Past, present and future. Nat. Rev. Immunol. 2023, 23, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Dramburg, S.; Hilger, C.; Santos, A.F.; Vecillas, L.d.L.; Aalberse, R.C.; Acevedo, N.; Aglas, L.; Altmann, F.; Arruda, K.L.; Asero, R.; et al. EAACI Molecular Allergology User’s Guide 2.0. Pediatr. Allergy Immunol. 2023, 34, e13854. [Google Scholar] [CrossRef] [PubMed]
- Mahler, V.; Mentzer, D.; Bonertz, A.; Muraro, A.; Eigenmann, P.; Bousquet, J.; Halken, S.; Pfaar, O.; Jutel, M.; Wahn, U.; et al. Allergen Immunotherapy (AIT) in children: A vulnerable population with its own rights and legislation—Summary of EMA-initiated multi-stakeholder meeting on Allergen Immunotherapy (AIT) for children, held at Paul-Ehrlich-Institut, Langen, Germany, 16.1.2019. Clin. Transl. Allergy 2020, 10, 28. [Google Scholar] [CrossRef] [PubMed]
- Bonertz, A.; Roberts, G.C.; Hoefnagel, M.; Timon, M.; Slater, J.E.; Rabin, R.L.; Bridgewater, J.; Pini, C.; Pfaar, O.; Akdis, C.; et al. Challenges in the implementation of EAACI guidelines on allergen immunotherapy: A global perspective on the regulation of allergen products. Allergy 2018, 73, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Canonica, G.W.; Bachert, C.; Hellings, P.; Ryan, D.; Valovirta, E.; Wickman, M.; De Beaumont, O.; Bousquet, J. Allergen Immunotherapy (AIT): A prototype of Precision Medicine. World Allergy Organ. J. 2015, 8, 31. [Google Scholar] [CrossRef]
- Alvaro-Lozano, M.; Akdis, C.A.; Akdis, M.; Alviani, C.; Angier, E.; Arasi, S.; Arzt-Gradwohl, L.; Barber, D.; Bazire, R.; Cavkaytar, O.; et al. EAACI Allergen Immunotherapy User’s Guide. Pediatr. Allergy Immunol. 2020, 31 (Suppl. S25), 1–101. [Google Scholar] [CrossRef] [PubMed]
- González-Pérez, R.; Poza-Guedes, P.; Pineda, F.; Galán, T.; Mederos-Luis, E.; Abel-Fernández, E.; Martínez, M.J.; Sánchez-Machín, I. Molecular Mapping of Allergen Exposome among Different Atopic Phenotypes. Int. J. Mol. Sci. 2023, 24, 10467. [Google Scholar] [CrossRef]
- Bousquet, J.; Schünemann, H.J.; Togias, A.; Bachert, C.; Erhola, M.; Hellings, P.W.; Klimek, L.; Pfaar, O.; Wallace, D.; Ansotegui, I.; et al. Next-generation Allergic Rhinitis and Its Impact on Asthma (ARIA) guidelines for allergic rhinitis based on Grading of Recommendations Assessment, Development and Evaluation (GRADE) and real-world evidence. J. Allergy Clin. Immunol. 2020, 145, 70–80.e3. [Google Scholar] [CrossRef]
- 2022 GINA Main Report. Available online: https://ginasthma.org/gina-reports/ (accessed on 23 September 2023).
- Eichenfield, L.F.; Ahluwalia, J.; Waldman, A.; Borok, J.; Udkoff, J.; Boguniewicz, M. Current guidelines for the evaluation and management of atopic dermatitis: A comparison of the Joint Task Force Practice Parameter and American Academy of Dermatology guidelines. J. Allergy Clin. Immunol. 2017, 139, S49–S57. [Google Scholar] [CrossRef]
- Heinzerling, L.; Mari, A.; Bergmann, K.C.; Bresciani, M.; Burbach, G.; Darsow, U.; Durham, S.; Fokkens, W.; Gjomarkaj, M.; Haahtela, T.; et al. The skin prick test—European standards. Clin. Transl. Allergy 2013, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- COULTER®. LH SlideMaker Operator’s Guide; PN 4277299D; Beckman Coulter Inc.: Fullerton, CA, USA, 2006. [Google Scholar]
- Lis, K.; Bartuzi, Z. Selected Technical Aspects of Molecular Allergy Diagnostics. Curr. Issues Mol. Biol. 2023, 45, 5481–5493. [Google Scholar] [CrossRef] [PubMed]
- Custovic, A.; Sonntag, H.-J.; Buchan, I.E.; Belgrave, D.; Simpson, A.; Prosperi, M.C.F. Evolution pathways of IgE responses to grass and mite allergens throughout childhood. J. Allergy Clin. Immunol. 2015, 136, 1645–1652.e8. [Google Scholar] [CrossRef] [PubMed]
- Hatzler, L.; Panetta, V.; Lau, S.; Wagner, P.; Bergmann, R.L.; Illi, S.; Bergmann, K.E.; Keil, T.; Hofmaier, S.; Rohrbach, A.; et al. Molecular spreading and predictive value of preclinical IgE response to Phleum pratense in children with hay fever. J. Allergy Clin. Immunol. 2012, 130, 894–901.e5. [Google Scholar] [CrossRef]
- Weghofer, M.; Grote, M.; Dall’Antonia, Y.; Fernández-Caldas, E.; Krauth, M.T.; Van Hage, M.; Horak, F.; Thomas, W.R.; Valent, P.; Keller, W.; et al. Characterization of folded recombinant Der p 5, a potential diagnostic marker allergen for house dust mite allergy. Int. Arch. Allergy Immunol. 2008, 147, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Weghofer, M.; Dall’Antonia, Y.; Grote, M.; Stöcklinger, A.; Kneidinger, M.; Balic, N.; Krauth, M.T.; Fernández-Caldas, E.; Thomas, W.R.; Hage, M.V.; et al. Characterization of Der p 21, a new important allergen derived from the gut of house dust mites. Allergy 2008, 63, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Trompette, A.; Divanovic, S.; Visintin, A.; Blanchard, C.; Hegde, R.S.; Madan, R.; Thorne, P.S.; Wills-Karp, M.; Gioannini, T.L.; Weiss, J.P.; et al. Allergenicity resulting from functional mimicry of a Toll-like receptor complex protein. Nature 2009, 457, 585–588. [Google Scholar] [CrossRef]
- Thomas, W.R.; Hales, B.J.; Smith, W.A. Structural biology of allergens. Curr. Allergy Asthma Rep. 2005, 5, 388–393. [Google Scholar] [CrossRef]
- Mueller, G.A.; Gosavi, R.A.; Krahn, J.M.; Edwards, L.L.; Cuneo, M.J.; Glesner, J.; Pomés, A.; Chapman, M.D.; London, R.E.; Pedersen, L.C. Der p 5 Crystal Structure Provides Insight into the Group 5 Dust Mite Allergens. J. Biol. Chem. 2010, 285, 25394–25401. [Google Scholar] [CrossRef] [PubMed]
- Resch, Y.; Michel, S.; Kabesch, M.; Lupinek, C.; Valenta, R.; Vrtala, S. Different IgE recognition of mite allergen components in asthmatic and nonasthmatic children. J. Allergy Clin. Immunol. 2015, 136, 1083–1091. [Google Scholar] [CrossRef] [PubMed]
- Weghofer, M.; Grote, M.; Resch, Y.; Casset, A.; Kneidinger, M.; Kopec, J.; Thomas, W.R.; Fernández-Caldas, E.; Kabesch, M.; Ferrara, R.; et al. Identification of Der p 23, a Peritrophin-like Protein, as a New Major Dermatophagoides pteronyssinus Allergen Associated with the Peritrophic Matrix of Mite Fecal Pellets. J. Immunol. 2013, 190, 3059–3067. [Google Scholar] [CrossRef] [PubMed]
- Celi, G.; Brusca, I.; Scala, E.; Villalta, D.; Pastorello, E.; Farioli, L.; Cortellini, G.; Deleonardi, G.; Galati, P.; Losappio, L.; et al. House dust mite allergy in Italy—Diagnostic and clinical relevance of Der p 23 (and of minor allergens): A real-life, multicenter study. Allergy 2019, 74, 1787–1789. [Google Scholar] [CrossRef] [PubMed]
- Mueller, G.A.; Randall, T.A.; Glesner, J.; Pedersen, L.C.; Perera, L.; Edwards, L.L.; DeRose, E.F.; Chapman, M.D.; London, R.E.; Pomés, A. Serological, genomic and structural analyses of the major mite allergen Der p 23. Clin. Exp. Allergy 2016, 46, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Curin, M.; Huang, H.-J.; Garmatiuk, T.; Gutfreund, S.; Resch-Marat, Y.; Chen, K.-W.; Fauland, K.; Keller, W.; Zieglmayer, P.; Zieglmayer, R.; et al. IgE Epitopes of the House Dust Mite Allergen Der p 7 Are Mainly Discontinuous and Conformational. Front. Immunol. 2021, 12, 687294. [Google Scholar] [CrossRef] [PubMed]
- Muddaluru, V.; Valenta, R.; Vrtala, S.; Schlederer, T.; Hindley, J.; Hickey, P.; Larché, M.; Tonti, E. Comparison of house dust mite sensitization profiles in allergic adults from Canada, Europe, South Africa and USA. Allergy 2021, 76, 2177–2188. [Google Scholar] [CrossRef] [PubMed]
- Demoly, P.; Passalacqua, G.; Pfaar, O.; Sastre, J.; Wahn, U. Management of the polyallergic patient with allergy immunotherapy: A practice-based approach. Allergy Asthma Clin. Immunol. 2016, 12, 2. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Domínguez, A.; Berings, M.; Rohrbach, A.; Huang, H.-J.; Curin, M.; Gevaert, P.; Matricardi, P.M.; Valenta, R.; Vrtala, S. Molecular profiling of allergen-specific antibody responses may enhance success of specific immunotherapy. J. Allergy Clin. Immunol. 2020, 146, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.-W.; Zieglmayer, P.; Zieglmayer, R.; Lemell, P.; Horak, F.; Bunu, C.P.; Valenta, R.; Vrtala, S. Selection of house dust mite–allergic patients by molecular diagnosis may enhance success of specific immunotherapy. J. Allergy Clin. Immunol. 2018, 143, 1248–1252.e12. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Yang, Y.; Xu, Q.; Zhang, W.; Jiang, Q.; Li, W.; Wang, Y.; Ma, D.; Lin, X.; Sun, B.; et al. Specific IgE and IgG4 Profiles of House Dust Mite Components in Allergen-Specific Immunotherapy. Front. Immunol. 2022, 12, 786738. [Google Scholar] [CrossRef]
- Caraballo, L.; Valenta, R.; Puerta, L.; Pomés, A.; Zakzuk, J.; Fernandez-Caldas, E.; Acevedo, N.; Sanchez-Borges, M.; Ansotegui, I.; Zhang, L.; et al. The allergenic activity and clinical impact of individual IgE-antibody binding molecules from indoor allergen sources. World Allergy Organ. J. 2020, 13, 100118. [Google Scholar] [CrossRef]
- Eržen, R.; Košnik, M.; Silar, M.; Korošec, P. Basophil response and the induction of a tolerance in venom immunotherapy: A long-term sting challenge study. Allergy 2012, 67, 822–830. [Google Scholar] [CrossRef]
- Eberlein, B. Basophil Activation as Marker of Clinically Relevant Allergy and Therapy Outcome. Front Immunol. 2020, 11, 1815. [Google Scholar] [CrossRef] [PubMed]
- Wise, S.K.; Damask, C.; Roland, L.T.; Ebert, C.; Levy, J.M.; Lin, S.; Luong, A.; Rodriguez, K.; Sedaghat, A.R.; Toskala, E.; et al. International consensus statement on allergy and rhinology: Allergic rhinitis—2023. Int. Forum Allergy Rhinol. 2023, 13, 293–859. [Google Scholar] [CrossRef] [PubMed]
- Halken, S.; Larenas-Linnemann, D.; Roberts, G.; Calderón, M.A.; Angier, E.; Pfaar, O.; Ryan, D.; Agache, I.; Ansotegui, I.J.; Arasi, S.; et al. EAACI guidelines on allergen immunotherapy: Prevention of allergy. Pediatr. Allergy Immunol. 2017, 28, 728–745. [Google Scholar] [CrossRef] [PubMed]
- Ridolo, E.; Nicoletta, F.; Barone, A.; Ottoni, M.; Senna, G.; Canonica, G.W. Causes of Non-Adherence to Allergen-Specific Immunotherapy: A Foundation towards a Patient-Personalized Approach. J. Pers. Med. 2023, 13, 1206. [Google Scholar] [CrossRef] [PubMed]
- Izmailovich, M.; Semenova, Y.; Abdushukurova, G.; Mukhamejanova, A.; Dyussupova, A.; Faizova, R.; Gazaliyeva, M.; Akhvlediani, L.; Glushkova, N.; Kalmakhanov, S.; et al. Molecular Aspects of Allergen-Specific Immunotherapy in Patients with Seasonal Allergic Rhinitis. Cells 2023, 12, 383. [Google Scholar] [CrossRef]
- Stringari, G.; Tripodi, S.; Caffarelli, C.; Dondi, A.; Asero, R.; Businco, A.D.R.; Bianchi, A.; Candelotti, P.; Ricci, G.; Bellini, F.; et al. Italian Pediatric Allergy Network (I-PAN). The effect of component-resolved diagnosis on specific immunotherapy prescription in children with hay fever. J. Allergy Clin. Immunol. 2014, 134, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Caraballo, L.; Valenta, R.; Acevedo, N.; Zakzuk, J. Are the Terms Major and Minor Allergens Useful for Precision Allergology? Front. Immunol. 2021, 12, 651500. [Google Scholar] [CrossRef]
- Miller, J.D. The Role of Dust Mites in Allergy. Clin. Rev. Allergy Immunol. 2019, 57, 312–329. [Google Scholar] [CrossRef]
- González-Pérez, R.; Poza-Guedes, P.; Pineda, F. Comment on Platts-Mills et al. Pediatr. Allergy Immunol. 2023, 34, e13986. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, J.; Bonertz, A.; Vieths, S. Quality requirements for allergen extracts and allergoids for allergen immunotherapy. Allergol. Immunopathol. 2017, 45, 4–11. [Google Scholar] [CrossRef]
| Allergic Rhinitis | Allergic Asthma | Atopic Dermatitis | |
|---|---|---|---|
| n = 60 | 20 | 20 | 20 |
| Age (y.o.) median (IQR) | 14.5 (7) | 16 (5.25) | 15 (5.25) |
| Sex (F/M) | 10/10 | 11/9 | 10/10 |
| Severe atopic disease (%) | 12/20 (%) | 11/20 (%) | 7/20 (%) |
| Food allergy (%) | 3/20 (15%) | 4/20 (20.0%) | 6/20 (30%) |
| Drug allergy (%) | 0/20 (0%) | 2/20 (10%) | 1/20 (5%) |
| SPT+ any aeroallergen (%) | 20 (100%) | 20 (100%) | 20 (100%) |
| Total IgE (IU/mL) median (IQR) | 73.15 (232.02) | 551 (999.5) | 965 (1962.7) |
| Blood eosinophils/mm3 median (IQR) | 310 (329.3) | 380 (421.5) | 373 (474.01) |
| Family history of atopy (%) | 15/20 (75%) | 16/20 (80%) | 18/20 (90%) |
| Mite Allergen | Mean sIgE in AR (M ± SD) | % of Sensitized Patients (AR) | Mean sIgE in Asthma (M ± SD) | % of Sensitized Patients (A) | Mean sIgE in Atopic Dermatitis (M ± SD) | % of Sensitized Patients (AD) |
|---|---|---|---|---|---|---|
| Der p 1 | 12.63 ± 0.12 | 75 | 16.69 ± 11.66 | 94.44 | 17.8 ± 0.97 | 83.33 |
| Der f 1 | 4.36 ± 3.21 | 54.16 | 17.21 ± 9.08 | 94.44 | 15.07 ± 6.69 | 83.33 |
| Der p 2 | 13.13 ± 9.7 | 79.66 | 33.55 ± 11.66 | 100 | 28.34 ± 18.82 | 88.88 |
| Der f 2 | 15.39 ± 6.2 | 83.33 | 35.33 ± 11.39 | 100 | 30.35 ± 19.23 | 88.88 |
| Der p 5 | 7.82 ± 4.91 | 52 | 18.08 ± 7.23 | 88.88 | 24.04 ± 16.84 | 83.33 |
| Der p 7 | 3.76 ± 2.44 | 22.72 | 12.63 ± 3.35 | 72.22 | 16.31 ± 3.71 | 66.66 |
| Der p 10 | 1.39 ± 0.65 | 18.18 | 0.6 ± 0.27 | 16.16 | 0.4 ± 0.14 | 16.16 |
| Der p 11 | <0.35 | 0 | <0.35 | 0 | 0.36 ± 0.03 | 5.55 |
| Der p 20 | 2.06 ± 1.94 | 16.66 | 0.43 ± 0.31 | 16.66 | 3.85± 1.23 | 16.66 |
| Der p 21 | 7.29 ± 3.64 | 58.33 | 16.46 ± 7.23 | 77.77 | 24.53 ± 4.32 | 77.77 |
| Der p 23 | 14.61 ± 9.12 | 70.83 | 20.40 ± 3,28 | 94.44 | 24.07 ± 8.01 | 88.88 |
| Number of Identified HDM Allergens | Allergic Rhinitis (n = 20) | Asthma (n = 20) | Atopic Dermatitis (n = 20) |
|---|---|---|---|
| 0 | 0 | 0 | 1 |
| 1 | 3 | 0 | 1 |
| 2 | 3 | 0 | 0 |
| 3 | 1 | 1 | 0 |
| 4 | 3 | 0 | 0 |
| 5 | 6 | 1 | 0 |
| 6 | 1 | 2 | 3 |
| 7 | 3 | 1 | 2 |
| 8 | 3 | 11 | 9 |
| 9 | 0 | 1 | 3 |
| 10 | 1 | 1 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Pérez, R.; Poza-Guedes, P.; Pineda, F.; Sánchez-Machín, I. Advocacy of Precision Allergy Molecular Diagnosis in Decision Making for the Eligibility of Customized Allergen Immunotherapy. Curr. Issues Mol. Biol. 2023, 45, 9976-9984. https://doi.org/10.3390/cimb45120623
González-Pérez R, Poza-Guedes P, Pineda F, Sánchez-Machín I. Advocacy of Precision Allergy Molecular Diagnosis in Decision Making for the Eligibility of Customized Allergen Immunotherapy. Current Issues in Molecular Biology. 2023; 45(12):9976-9984. https://doi.org/10.3390/cimb45120623
Chicago/Turabian StyleGonzález-Pérez, Ruperto, Paloma Poza-Guedes, Fernando Pineda, and Inmaculada Sánchez-Machín. 2023. "Advocacy of Precision Allergy Molecular Diagnosis in Decision Making for the Eligibility of Customized Allergen Immunotherapy" Current Issues in Molecular Biology 45, no. 12: 9976-9984. https://doi.org/10.3390/cimb45120623
APA StyleGonzález-Pérez, R., Poza-Guedes, P., Pineda, F., & Sánchez-Machín, I. (2023). Advocacy of Precision Allergy Molecular Diagnosis in Decision Making for the Eligibility of Customized Allergen Immunotherapy. Current Issues in Molecular Biology, 45(12), 9976-9984. https://doi.org/10.3390/cimb45120623







