Blocking TRPA1 in Respiratory Disorders: Does It Hold a Promise?
Abstract
:1. TRPA1 Receptor
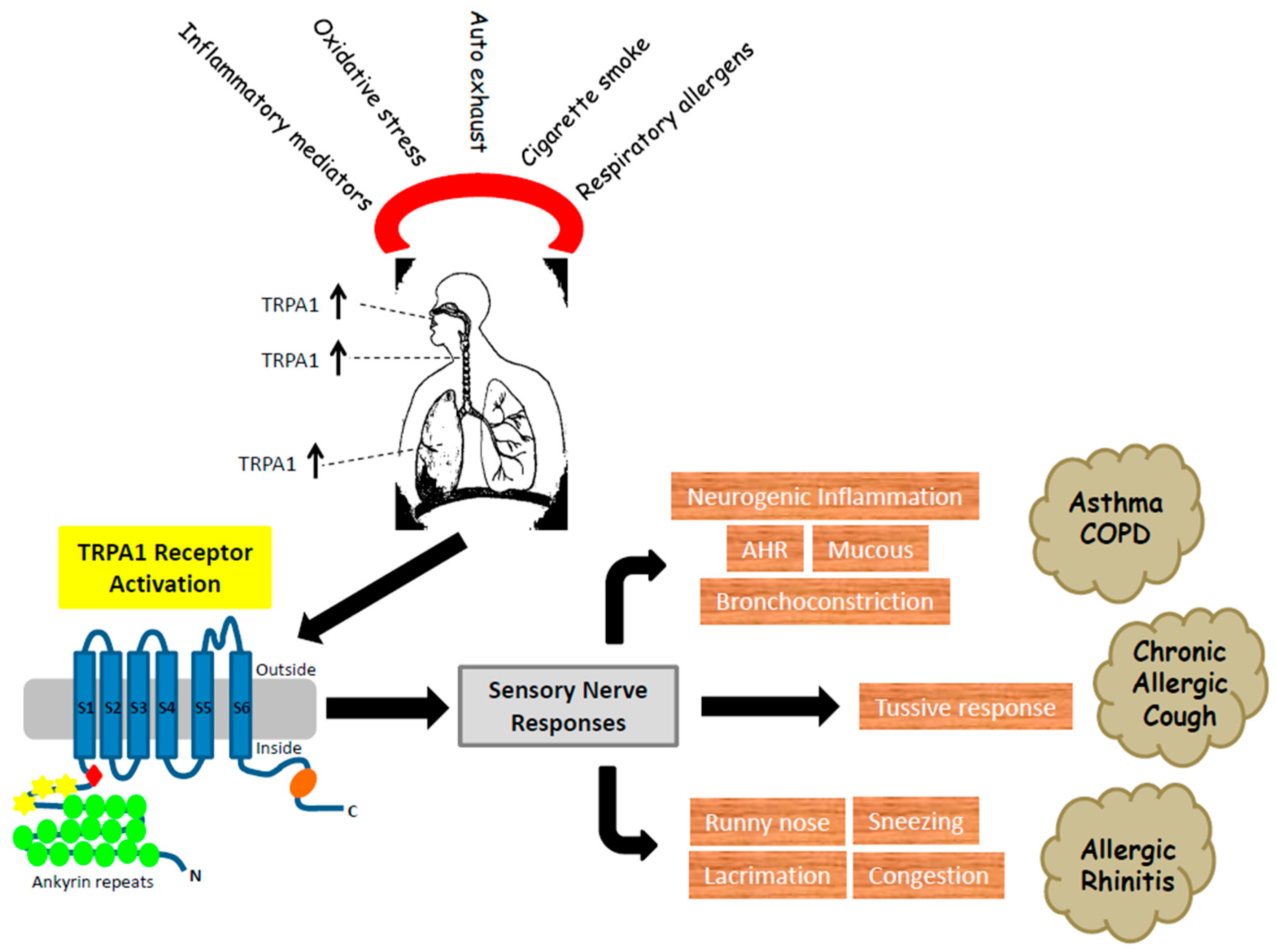
2. TRPA1—Expression and Activation in Airways
3. TRPA1 in Chronic Cough
4. TRPA1 and Asthma
5. Role of TRPA1 in COPD
6. Role of TRPA1 in Allergic Rhinitis
7. Role of TRPA1 in Cystic Fibrosis
8. TRPA1 Antagonists: What’s the Status?
9. TRPA1 Antagonists: Any Safety Concerns?
10. Summary
Conflicts of Interest
Abbreviations
| TRPA1 | Transient Receptor Potential Ankyrin 1 |
| COPD | Chronic obstructive pulmonary disease |
| TRPA1 KO | TRPA1 knockout |
| H2O2 | Hydrogen peroxide |
| 4-HNE | 4-Hydroxynonenal |
| CNS | Central nervous system |
| HEK293 | Human embryonic kidney cells |
| AITC | Allyl isothiocyanate |
| PGE2 | Prostaglandin E2 |
| PNDS | Post nasal drip syndrome |
| ROS | Reactive oxygen species |
| RNS | Reactive nitrogen species |
| 15d-PGJ2 | 15-deoxy-delta-12,14-prostaglandin J2 |
| GERD | Gastroesophageal reflux disease |
| TLRs | Toll like receptors |
| RADS | Reactive airways dysfunction syndrome |
| NGF | Nerve growth factor |
| OVA | Ovalbumin |
| NAPQ1 | N-acetyl-p-benzoquinoneimine |
| CSE | Cigarette smoke extract |
| CS | Cigarette smoke |
| IL8 | Interleukin 8 |
| CCD19-Lu | Human lung fibroblast cells |
| A549 | Human pulmonary alveolar epithelial cell line |
| HBEC | Human bronchial epithelial cells |
| KC | Keratinocyte chemoattractant |
| BALf | Bronchoalveolar lavage fluid |
| WSPM | Wood smoke particulate matter |
| TDI | Toluene diisocyanate |
| CF | Cystic fibrosis |
| CFTR | Cystic fibrosis transmembrane conductance regulator |
| TNFα | Tumor necrosis factor α |
| CuFi-1 | Human cystic fibrosis cell line |
| LPS | Lipopolysaccharide |
| CGRP | Calcitonin gene-related peptide |
| WT | Wild type |
| SNL | Spinal nerve ligation |
References
- Jaquemar, D.; Schenker, T.; Trueb, B. An ankyrin-like protein with transmembrane domains is specifically lost after oncogenic transformation of human fibroblasts. J. Biol. Chem. 1999, 274, 7325–7333. [Google Scholar] [CrossRef]
- Kunert-Keil, C.; Bisping, F.; Krüger, J.; Brinkmeier, H. Tissue-specific expression of TRP channel genes in the mouse and its variation in three different mouse strains. BMC Genom. 2006, 7, 159. [Google Scholar] [CrossRef]
- Nassenstein, C.; Kwong, K.; Taylor-Clark, T.; Kollarik, M.; Macglashan, D.M.; Braun, A.; Undem, B.J. Expression and function of the ion channel TRPA1 in vagal afferent nerves innervating mouse lungs. J. Physiol. 2008, 586, 1595–1604. [Google Scholar] [CrossRef]
- Bautista, D.M.; Jordt, S.E.; Nikai, T.; Tsuruda, P.R.; Read, A.J.; Poblete, J.; Yamoah, E.N.; Basbaum, A.I.; Julius, D. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell 2006, 124, 1269–1282. [Google Scholar] [CrossRef]
- Trevisani, M.; Siemens, J.; Materazzi, S.; Bautista, D.M.; Nassini, R.; Campi, B.; Imamachi, N.; Andrè, E.; Patacchini, R.; Cottrell, G.S.; et al. 4-Hydroxynonenal, an endogenous aldehyde, causes pain and neurogenic inflammation through activation of the irritant receptor TRPA1. Proc. Natl. Acad. Sci. USA 2007, 104, 13519–13524. [Google Scholar] [CrossRef]
- Bessac, B.F.; Sivula, M.; von Hehn, C.A.; Escalera, J.; Cohn, L.; Jordt, S.E. TRPA1 is a major oxidant sensor in murine airway sensory neurons. J. Clin. Invest. 2008, 118, 1899–1910. [Google Scholar] [CrossRef]
- Andrè, E.; Campi, B.; Materazzi, S.; Trevisani, M.; Amadesi, S.; Massi, D.; Creminon, C.; Vaksman, N.; Nassini, R.; Civelli, M.; et al. Cigarette smoke-induced neurogenic inflammation is mediated by α,β-unsaturated aldehydes and the TRPA1 receptor in rodents. J. Clin. Invest. 2008, 118, 2574–2582. [Google Scholar] [CrossRef]
- Matta, J.A.; Cornett, P.M.; Miyares, R.L.; Abe, K.; Sahibzada, N.; Ahern, G.P. General anesthetics activate a nociceptive ion channel to enhance pain and inflammation. Proc. Natl. Acad. Sci. USA 2008, 105, 8784–8789. [Google Scholar] [CrossRef]
- Andrè, E.; Gatti, R.; Trevisani, M.; Preti, D.; Baraldi, P.G.; Patacchini, R.; Geppetti, P. Transient receptor potential ankyrin receptor 1 is a novel target for pro-tussive agents. Br. J. Pharmacol. 2009, 158, 1621–1628. [Google Scholar] [CrossRef]
- Hu, H.; Bandell, M.; Petrus, M.J.; Zhu, M.X.; Patapoutian, A. Zinc activates damage-sensing TRPA1 ion channels. Nat. Chem. Biol. 2009, 5, 183–190. [Google Scholar] [CrossRef]
- Taylor-Clark, T.E.; Ghatta, S.; Bettner, W.; Undem, B.J. Nitrooleic acid, an endogenous product of nitrative stress, activates nociceptive sensory nerves via the direct activation of TRPA1. Mol. Pharmacol. 2009, 75, 820–829. [Google Scholar] [CrossRef]
- Nagata, K.; Duggan, A.; Kumar, G.; García-Añoveros, J. Nociceptor and hair cell transducer properties of TRPA1, a channel for pain and hearing. J. Neurosci. 2005, 25, 4052–4061. [Google Scholar] [CrossRef]
- Mukhopadhyay, I.; Gomes, P.; Aranake, S.; Shetty, M.; Karnik, P.; Damle, M.; Kuruganti, S.; Thorat, S.; Khairatkar-Joshi, N. Expression of functional TRPA1 receptor on human lung fibroblast and epithelial cells. J. Recept. Signal Transduct. 2011, 31, 350–358. [Google Scholar] [CrossRef]
- Nassini, R.; Pedretti, P.; Moretto, N.; Fusi, C.; Carnini, C.; Facchinetti, F.; Viscomi, A.R.; Pisano, A.R.; Stokesberry, S.; Brunmark, C.; et al. Transient receptor potential ankyrin 1 channel localized to non-neuronal airway cells promotes non-neurogenic inflammation. PLoS ONE 2012, 7, e42454. [Google Scholar] [CrossRef]
- Büch, T.R.; Schäfer, E.A.; Demmel, M.T.; Boekhoff, I.; Thiermann, H.; Gudermann, T.; Steinritz, D.; Schmidt, A. Functional expression of the transient receptor potential channel TRPA1, a sensor for toxic lung inhalants, in pulmonary epithelial cells. Chem. Biol. Interact. 2013, 206, 462–471. [Google Scholar] [CrossRef]
- Mihara, S.; Shibamoto, T. The role of flavor and fragrance chemicals in TRPA1 (transient receptor potential cation channel, member A1) activity associated with allergies. Allergy Asthma Clin. Immunol. 2015, 11. [Google Scholar] [CrossRef]
- Kichko, T.I.; Kobal, G.; Reeh, P.W. Cigarette smoke has sensory effects through nicotinic and TRPA1 but not TRPV1 receptors on the isolated mouse trachea and larynx. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 309, L812–L820. [Google Scholar] [CrossRef]
- Shapiro, D.; Deering-Rice, C.E.; Romero, E.G.; Hughen, R.W.; Light, A.R.; Veranth, J.M.; Reilly, C.A. Activation of transient receptor potential ankyrin-1 (TRPA1) in lung cells by wood smoke particulate material. Chem. Res. Toxicol. 2013, 26, 750–758. [Google Scholar] [CrossRef]
- Grace, M.S.; Dubuis, E.; Birrell, M.A.; Belvisi, M.G. Pre-clinical studies in cough research: Role of Transient Receptor Potential (TRP) channels. Pulm. Pharmacol. Ther. 2013, 26, 498–507. [Google Scholar] [CrossRef]
- Birrell, M.A.; Belvisi, M.G.; Grace, M.; Sadofsky, L.; Faruqi, S.; Hele, D.J.; Maher, S.A.; Freund-Michel, V.; Morice, A.H. TRPA1 agonists evoke coughing in guinea pig and human volunteers. Am. J. Respir. Crit. Care Med. 2009, 180, 1042–1047. [Google Scholar] [CrossRef]
- Kistner, K.; Siklosi, N.; Babes, A.; Khalil, M.; Selescu, T.; Zimmermann, K.; Wirtz, S.; Becker, C.; Neurath, M.F.; Reeh, P.W.; et al. Systemic desensitization through TRPA1 channels by capsazepine and mustard oil—A novel strategy against inflammation and pain. Sci. Rep. 2016, 6, 28621. [Google Scholar] [CrossRef]
- Mukhopadhyay, I.; Kulkarni, A.; Aranake, S.; Karnik, P.; Shetty, M.; Thorat, S.; Ghosh, I.; Wale, D.; Bhosale, V.; Khairatkar-Joshi, N. Transient receptor potential ankyrin 1 receptor activation in vitro and in vivo by pro-tussive agents: GRC 17536 as a promising anti-tussive therapeutic. PLoS ONE 2014, 9, e97005. [Google Scholar] [CrossRef]
- Aparici, M.; Tarrasón, G.; Jover, I.; Carcasona, C.; Fernández-Blanco, J.; Eichhorn, P.; Gavaldà, A.; De Alba, J.; Roberts, R.; Miralpeix, M. Pharmacological profile of a novel, potent and oral TRPA1 antagonist. Characterization in a preclinical model of induced cough. Eur. Respir. J. 2015, 46, PA3948. [Google Scholar] [CrossRef]
- Canning, B.J. Afferent nerves regulating the cough reflex: Mechanisms and mediators of cough in disease. Otolaryngol. Clin. North Am. 2010, 43, 15–25. [Google Scholar] [CrossRef]
- Bezemer, G.F.G.; Sagar, S.; van Bergenhenegouwen, J.; Georgiou, N.A.; Garssen, J.; Kraneveld, A.D.; Folkerts, G. Dual Role of Toll-Like Receptors in Asthma and Chronic Obstructive Pulmonary Disease. Pharmacol. Rev. 2012, 64, 337–358. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, J.; Bai, J.; Wang, X.; Li, Y.; Jiang, P. Comparative expression of Toll-like receptors and inflammatory cytokines in pigs infected with different virulent porcine reproductive and respiratory syndrome virus isolates. J. Virol. 2013, 10, 135. [Google Scholar] [CrossRef]
- Qi, J.; Buzas, K.; Fan, H.; Cohen, J.I.; Wang, K.; Mont, E.; Klinman, D.; Oppenheim, J.J.; Howard, O.M. Painful pathways induced by TLR stimulation of dorsal root ganglion neurons. J. Immunol. 2011, 186, 6417–6426. [Google Scholar] [CrossRef]
- Belvisi, M.G.; Dubuis, E.; Birrell, M.A. Transient receptor potential A1 channels: Insights into cough and airway inflammatory disease. Chest 2011, 140, 1040–1047. [Google Scholar] [CrossRef]
- Lavinka, P.C.; Dong, X. Molecular signaling and targets from itch: Lessons for cough. Cough 2013, 9, 8. [Google Scholar] [CrossRef]
- Caceres, A.I.; Brackmann, M.; Elia, M.D.; Bessac, B.F.; del Camino, D.; D’Amours, M.; Witek, J.S.; Fanger, C.M.; Chong, J.A.; Hayward, N.J.; et al. A sensory neuronal ion channel essential for airway inflammation and hyperreactivity in asthma. Proc Natl Acad Sci USA 2009, 106, 9099–9104. [Google Scholar] [CrossRef]
- Facchinetti, F.; Patacchini, R. The rising role of TRPA1 in asthma. Open Drug Discov. J. 2010, 2, 71–80. [Google Scholar] [CrossRef]
- Raemdonck, K.; de Alba, J.; Birrell, M.A.; Grace, M.; Maher, S.A.; Irvin, C.G.; Fozard, J.R.; O'Byrne, P.M.; Belvisi, M.G. A role for sensory nerves in the late asthmatic response. Thorax 2012, 67, 19–25. [Google Scholar] [CrossRef]
- Anupindi, R.; Mukhopadhyay, I.; Thomas, A.; Kumar, S.; Chaudhari, S.S.; Kulkarni, A.; Gudi, G.; Khairatkar-Joshi, N. GRC 17536, a novel, selective TRPA1 antagonist for potential treatment of respiratory disorders. Eur. Respir. J. 2010, 36, E5645. [Google Scholar]
- Camino, D.; Chong, J.A.; Hayward, N.J.; Monsen, J.; Moran, M.M.; Curtis, R.; Murphy, C.; Mortin, L.I.; Abraham, W.M. Effects of blocking TRPA1 in a sheep model of asthma. In Proceedings of the International Workshop on Transient Receptor Potential Channels, a TRiP to Spain, Valencia, Spain, 12–14 September 2012; p. 68.
- Beasley, R.; Clayton, T.; Crane, J.; von Mutius, E.; Lai, C.K.; Montefort, S.; Stewart, A. Association between paracetamol use in infancy and childhood, and risk of asthma, rhinoconjunctivitis, and eczema in children aged 6–7 years: Analysis from phase three of the ISAAC programme. Lancet 2008, 372, 1039–1048. [Google Scholar] [CrossRef]
- Nassini, R.; Materazzi, S.; Andre`, E.; Sartiani, L.; Aldini, G.; Trevisani, M.; Carnini, C.; Massi, D.; Pedretti, P.; Carini, M.; et al. Acetaminophen, via its reactive metabolite N-acetyl-pbenzo-quinoneimine and transient receptor potential ankyrin-1 stimulation, causes neurogenic inflammation in the airways and other tissues in rodents. FASEB J. 2010, 24, 4904–4916. [Google Scholar] [CrossRef]
- Liu, S.; Zhou, Y.; Wang, X.; Wang, D.; Lu, J.; Zheng, J.; Zhong, N.; Ran, P. Biomass fuels are the probable risk factor for chronic obstructive pulmonary disease in rural South China. Thorax 2007, 62, 889–897. [Google Scholar] [CrossRef]
- Facchinetti, F.; Amadei, F.; Geppetti, P.; Tarantini, F.; Serio, C.; Dragotto, A.; Gigli, P.M.; Catinella, S.; Civelli, M.; Patacchini, R. α,β-Unsaturated aldehydes in cigarette smoke release inflammatory mediators from human macrophages. Am. J. Respir. Cell Mol. Biol. 2007, 37, 617–623. [Google Scholar] [CrossRef]
- Lin, A.H.; Liu, M.H.; Ko, H.K.; Perng, D.W.; Lee, T.S.; Kou, Y.R. Lung epithelial TRPA1 transduces the extracellular ROS into transcriptional regulation of lung inflammation induced by cigarette smoke: The role of influxed Ca2+. Mediat. Inflamm. 2015, 2015, 148367. [Google Scholar] [CrossRef]
- Dupont, L.L.; Alpizar, Y.A.; Brusselle, G.G.; Bracke, K.R.; Talavera, K.; Joos, G.F.; Maes, T. Expression of transient receptor potential (TRP) channels in a murine model of cigarette smoke exposure. In Proceedings of the C34. Insights into COPD Pathogenesis from Pre-Clinical Studies, San Diego, CA, USA, 16–21 May 2014; Volume 189, p. A4286.
- Hu, G.; Zhou, Y.; Tian, J.; Yao, W.; Li, J.; Li, B.; Ran, P. Risk of COPD from exposure to biomass smoke: A metaanalysis. Chest 2010, 1381, 20–31. [Google Scholar] [CrossRef]
- Hur, G.Y.; Kohw, D.H.; Choi, G.S.; Park, H.J.; Choi, S.J.; Ye, Y.M.; Kimw, K.S.; Park, H.S. Clinical and immunologic findings of methylene Diphenyl diisocyanate-induced occupational asthma in a car upholstery factory. Clin. Exp. Allergy 2008, 38, 586–593. [Google Scholar] [CrossRef]
- Fisseler-Eckhoff, A.; Bartsch, H.; Zinsky, R.; Schirren, J. Environmental isocyanate-induced asthma: Morphologic and pathogenetic aspects of an increasing occupational disease. Int. J. Environ. Res. Public Health 2011, 8, 3672–3687. [Google Scholar] [CrossRef]
- Johnson, V.J.; Yucesoy, B.; Reynolds, J.S.; Fluharty, K.; Wang, W.; Richardson, D.; Luster, M.I. Inhalation of toluene diisocyanate vapor induces allergic rhinitis in mice. J. Immunol. 2007, 179, 1864–1871. [Google Scholar] [CrossRef]
- Taylor-Clark, T.E.; Kiros, F.; Carr, M.J.; McAlexander, M.A. Transient receptor potential ankyrin 1 mediates toluene diisocyanate–evoked respiratory irritation. Am. J. Respir. Cell Mol. Biol. 2009, 40, 756–762. [Google Scholar] [CrossRef]
- Tourangeau, L.M.; Christiansen, S.C.; Herschbach, J.; Brooks, S.M.; Eddleston, J.; Zuraw, B. Nasal mucosal TRPA1 and TRPV1 levels in human rhinitis. J. Allergy Clin. Immunol. 2011, 127, AB52. [Google Scholar] [CrossRef]
- Prandini, P.; De Logu, F.; Fusi, C.; Provezza, L.; Nassini, R.; Montagner, G.; Materazzi, S.; Munari, S.; Gilioli, E.; Bezzerri, V.; et al. TRPA1 channels modulate inflammatory response in respiratory cells from cystic fibrosis patients. Am. J. Respir. Cell. Mol. Biol. 2016, 55. [Google Scholar] [CrossRef]
- Chen, J.; Hackos, D.H. TRPA1 as drug target-Promise and challenges. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2015, 388, 451–463. [Google Scholar] [CrossRef]
- Meseguer, V.; Alpizar, Y.A.; Luis, E.; Tajada, S.; Denlinger, B.; Fajardo, O.; Manenschijn, J.A.; Fernández-Peña, C.; Talavera, A.; Kichko, T.; et al. TRPA1 channels mediate acute neurogenic inflammation and pain produced by bacterial endotoxins. Nat. Commun. 2014, 5, 3125. [Google Scholar] [CrossRef]
- Tandon, M.; Jain, S.M.; Balamurugan, R.; Koslowski, M.; Keohane, P. Treatment of pain associated with diabetic peripheral neuropathy with the novel TRPA1 antagonist GRC 17536 in patients with intact peripheral nerve function. In Proceedings of the Fifth International Meeting of The Special Interest Group on Neuropathic Pain (NeuPSIG), Nice, France, 14–17 May 2015.
- Ryan, N.M.; Birring, S.S.; Gibson, P.G. Gabapentin for refractory chronic cough: A randomised, double-blind, placebo-controlled trial. Lancet 2012, 380, 1583–1589. [Google Scholar] [CrossRef]
- Kwan, K.Y.; Allchorne, A.J.; Vollrath, M.A.; Christensen, A.P.; Zhang, D.S.; Woolf, C.J.; Corey, D.P. TRPA1 contributes to cold, mechanical, and chemical nociception but is not essential for hair-cell transduction. Neuron 2006, 50, 277–289. [Google Scholar] [CrossRef]
- Chen, J.; Joshi, S.K.; DiDomenico, S.; Perner, R.J.; Mikusa, J.P.; Gauvin, D.M.; Segreti, J.A.; Han, P.; Zhang, X.F.; Niforatos, W.; et al. Selective blockade of TRPA1 channel attenuates pathological pain without altering noxious cold sensation or body temperature regulation. Pain 2011, 152, 1165–1172. [Google Scholar] [CrossRef]
- de Oliveira, C.; Garami, A.; Lehto, S.G.; Pakai, E.; Tekus, V.; Pohoczky, K.; Youngblood, B.D.; Wang, W.; Kort, M.E.; Kym, P.R.; et al. Transient receptor potential channel ankyrin-1 is not a cold sensor for autonomic thermoregulation in rodents. J. Neurosci. 2014, 34, 4445–4452. [Google Scholar] [CrossRef]
- Curtis, R.; Coleman, S.; Camino, D.D.; Hayward, N.J.; Moran, M.M.; Bokesch, P. Transient Receptor Potential A1 (TRPA1) is not involved in thermoregulation in dogs and humans. In Proceedings of the A Trip to Spain: International workshop on transient receptor potential channels, Valencia, Spain, 12–14 September 2012; p. 65.
- Obata, K.; Katsura, H.; Mizushima, T.; Yamanaka, H.; Kobayashi, K.; Dai, Y.; Fukuoka, T.; Tokunaga, A.; Tominaga, M.; Noguchi, K. TRPA1 induced in sensory neurons contributes to cold hyperalgesia after inflammation and nerve injury. J. Clin. Invest. 2005, 115, 2393–2401. [Google Scholar] [CrossRef]
- Bodkin, J.V.; Thakore, P.; Aubdool, A.A.; Liang, L.; Fernandes, E.S.; Nandi, M.; Spina, D.; Clark, J.E.; Aaronson, P.I.; Shattock, M.J.; et al. Investigating the potential role of TRPA1 in locomotion and cardiovascular control during hypertension. Pharmacol. Res. Perspect. 2014, 2, e00052. [Google Scholar] [CrossRef]
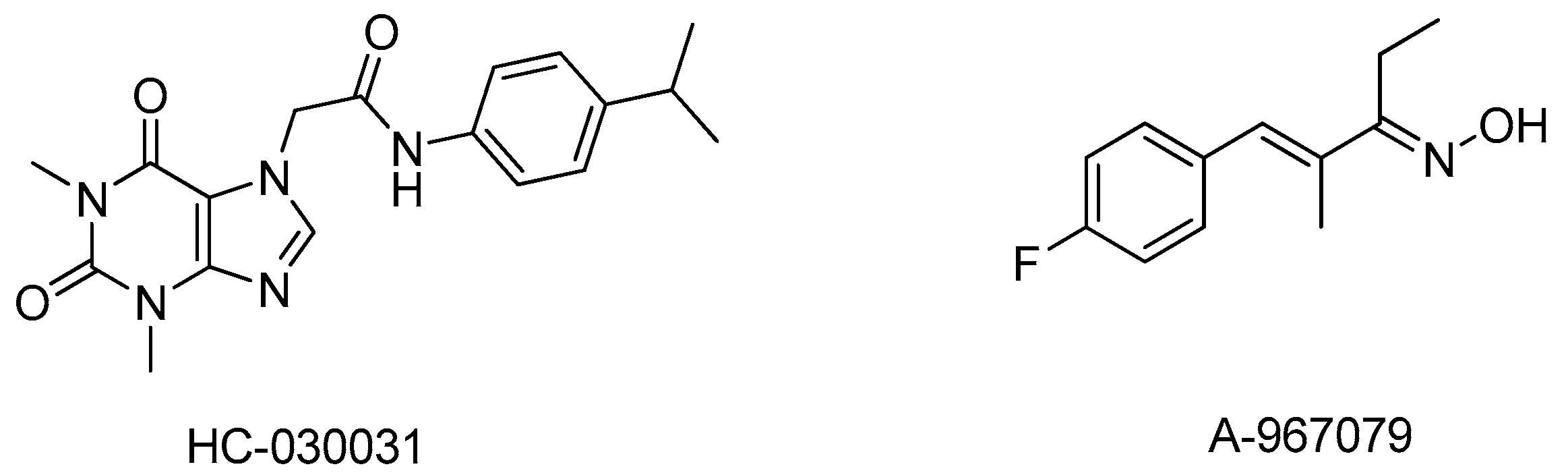
| Patent Number | Owner Companies | Indications |
|---|---|---|
| WO2013084153; WO2013014597; WO2012176143; WO2012172475; WO2012176105; WO2011132017; WO2014203210; WO2010125469; WO2010004390; WO2010109287; WO2013183035; WO2009118596; WO2012085662; WO2009144548; WO2011114184 | Glenmark Pharmaceuticals SA | Asthma; COPD; Bronchitis, COPD, Cough, Respiratory disorder |
| WO2009140517; WO2010132838; WO2007073505; WO2010039289; WO2015164643; WO2016044792; WO2010036821 | Hydra Biosciences Inc. | Asthma, Cough; Respiratory disease; Asthma; COPD; Lung injury |
| WO2009071631; WO2010141805 | Janssen Pharmaceutica NV | COPD; Lung disease; Cough |
| WO2015052264; WO2014060341; WO2014056958; WO2014049047; WO2014072325 | F Hoffmann-La Roche AG; Hoffmann-La Roche Inc; Roche Holding AG Genentech Inc. | Asthma; COPD; Cough; Allergic rhinitis; Respiratory disease; Bronchospasm |
| WO2015144976; WO2012152983; WO2014053694; WO2015144977 | Orion Corp | Asthma; COPD; Cough |
| WO2013023102; WO2014113671 | Cubist Pharmaceuticals Inc.; Hydra Biosciences Inc. | Asthma; COPD; Respiratory disease |
| WO2015115507; WO2014098098; WO2013108857 | Ajinomoto Co. Inc. | Asthma; COPD; Cough; Lung disease |
| JP2014024810 | Kao Corp | Respiratory failure |
| WO2009089082 | AbbVie Deutschland GmbH & Co KG; Abbott Laboratories | Lung disease |
| WO2016028325 | Duke University; University of California | Fibrosis |
| WO2015155306 | Almirall Prodesfarma SA | Respiratory disease |
| WO2014135617 | Ario Pharma Ltd; PharmEste SRL | Asthma; COPD; Cough |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mukhopadhyay, I.; Kulkarni, A.; Khairatkar-Joshi, N. Blocking TRPA1 in Respiratory Disorders: Does It Hold a Promise? Pharmaceuticals 2016, 9, 70. https://doi.org/10.3390/ph9040070
Mukhopadhyay I, Kulkarni A, Khairatkar-Joshi N. Blocking TRPA1 in Respiratory Disorders: Does It Hold a Promise? Pharmaceuticals. 2016; 9(4):70. https://doi.org/10.3390/ph9040070
Chicago/Turabian StyleMukhopadhyay, Indranil, Abhay Kulkarni, and Neelima Khairatkar-Joshi. 2016. "Blocking TRPA1 in Respiratory Disorders: Does It Hold a Promise?" Pharmaceuticals 9, no. 4: 70. https://doi.org/10.3390/ph9040070
APA StyleMukhopadhyay, I., Kulkarni, A., & Khairatkar-Joshi, N. (2016). Blocking TRPA1 in Respiratory Disorders: Does It Hold a Promise? Pharmaceuticals, 9(4), 70. https://doi.org/10.3390/ph9040070





