Abstract
Maurocalcine is a highly potent cell-penetrating peptide isolated from the Tunisian scorpion Maurus palmatus. Many cell-penetrating peptide analogues have been derived from the full-length maurocalcine by internal cysteine substitutions and sequence truncation. Herein we have further characterized the cell-penetrating properties of one such peptide, MCaUF1-9, whose sequence matches that of the hydrophobic face of maurocalcine. This peptide shows very favorable cell-penetration efficacy compared to Tat, penetratin or polyarginine. The peptide appears so specialized in cell penetration that it seems hard to improve by site directed mutagenesis. A comparative analysis of the efficacies of similar peptides isolated from other toxin members of the same family leads to the identification of hadrucalcin’s hydrophobic face as an even better CPP. Protonation of the histidine residue at position 6 renders the cell penetration of MCaUF1-9 pH-sensitive. Greater cell penetration at acidic pH suggests that MCaUF1-9 can be used to specifically target cancer cells in vivo where tumor masses grow in more acidic environments.
1. Introduction
Maurocalcine (MCa) is a 33 amino acid residue peptide that was isolated in 2000 from the venom of the Tunisian chactid scorpion Scorpio maurus palmatus [1]. It folds according to an ‘Inhibitor Cystine Knot’ (ICK) motif [2] and contains three disulfide bridges connected by the following pattern: C1-C4, C2-C5 and C3-C6 [3]. Based on high amino acid sequence and pharmacological target similarities, MCa belongs to a larger family of scorpion toxins that also includes imperatoxin A (from Pandinus imperator) [4], opicalcine 1 and opicalcine 2 (from Opistophthalmus carinatus) [5], hemicalcin [6] and hadrucalcin [7]. All these peptides act on ryanodine receptors resulting in pharmacological activation. These receptors are calcium channels located in the membrane of the endoplasmic reticulum. They control Ca2+ release from internal stores and therefore a large number of cell functions [7,8,9,10]. Binding of MCa on the ryanodine receptor type 1 occurs on protein cytoplasmic domains. Because MCa acts within seconds, once applied to the extracellular medium, it was soon obvious that it had to cross the plasma membrane very efficiently in order to activate the ryanodine receptor [11]. An additional curiosity of MCa lies into the fact that there is an intriguing sequence homology with a domain of the L-type voltage-gated calcium channel from the skeletal muscle (domain A). This channel lies in the plasma membrane, while domain A is found underneath the membrane in the cytoplasm within a loop that has been recognized as extremely important for the process of excitation-contraction coupling [12,13]. While the role of domain A in excitation-contraction coupling is still unclear, it is however surprising and stimulating to observe that peptides containing domain A sequence act with quite a lot of similarities to MCa on the ryanodine receptor type 1 [14,15]. According to the 1H-NMR solution structure, MCa is rigidly structured by the three disulfide bridges and contains three β-strands, comprising the following stretches of amino acid residues: 9–11 (strand 1), 20–23 (strand 2), and 30–33 (strand 3). MCa has an incredibly stable structure since it cannot be denatured, even at high temperatures up to 100 °C or extreme pH values [16]. Interestingly, the peptide is highly enriched in basic amino acid residues, more than a third of the amino acids being either lysine (seven out of 33) or arginine residues (four out of 33). The histidine residue at position 6 is susceptible, depending of the environmental pH, to introduce an additional positive charge to the peptide by protonation. Interestingly, stretches of positively charged residues seem to confound with the three MCa β-strands. The 3D structure of MCa also strikingly highlights the asymmetrical distribution of the positive charges at its surface: one face is highly basic, while the opposite face is rather hydrophobic. If, in addition, one depicts the amino acid residues important for the ryanodine receptor activation [17], then it appears that the peptide can be schematically represented with three domains: one hydrophobic head that tops the peptide, a larger second face, mainly basic, and a third side domain that contains the pharmacophore (Figure 1A).
Besides its Ca2+ channel activity, MCa is also used as a cell-penetrating peptide (CPP). Its properties have been investigated in detail. It was soon discovered that MCa could act as vector for the cell penetration of a variety of cargoes, including proteins [11], peptides [18], small dyes [16,19], drugs [20,21,22,23] or nanoparticles [24,25]. The mechanism of cell penetration most likely includes a combination of membrane translocation (direct passage to the cytoplasm) and endocytosis, mostly macropinocytosis (indirect access to the cytoplasm through leakage from late endosomes) [16,19,26,27].
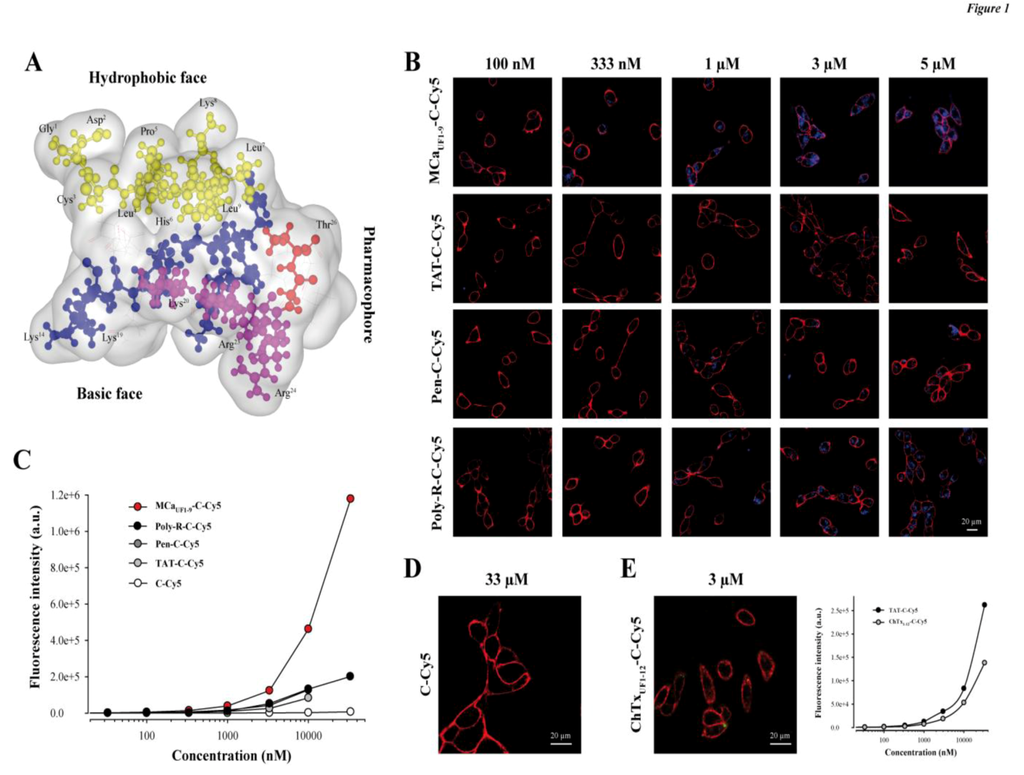
Figure 1.
The hydrophobic domain of MCa is an efficient CPP. (A) Schematic representation of MCa 3D structure. The hydrophobic domain (from amino acid residue 1 to 9) is shown on top. Residues are in yellow. Shown also are the basic face (basic amino acid residues are in blue or in pink) and the pharmacophore (residues identified as interacting with the ryanodine receptor are in red or in pink). Pink residues belong both to the pharmacophore and the basic face. (B) Confocal microscopy images illustrating the penetration of four different peptides labeled with Cy5 at various concentrations into glioma F98 cells (blue color). Incubation times were 2 h for each peptide/concentration. Images were taken immediately after washout of the extracellular peptide. The plasma membrane is labeled with concanavalin-A-rhodamine (red color). (C) Dose-dependent penetration of each peptide-cargo complex in F98 cells as assessed by flow cytometry. A control Cys-Cy5 is also provided. (D) Absence of cell penetration of 33 µM Cys-Cy5 (a single Cys residue linked to Cy5—abbreviated C-Cy5) evaluated by confocal microscopy. (E) Lack of cell penetration of 3 µM ChTxUF1-12-C-Cy5 as determined by confocal microscopy (right panel). Quantitative analysis of F98 ChTxUF1-12-C-Cy5 fluorescence as determined by flow cytometry. Internalization of the ChTxUF1-12-C-Cy5 peptide is lower than TAT-C-Cy5 but not negligible.
Even if the mechanism(s) of penetration of CPP are subject to debate [28,29], because of the rapidity of action of MCa on the ryanodine receptor, it appears clearly that one way of penetration of MCa can be membrane translocation. Furthermore, MCa has the ability to bind onto glycosaminoglycans, including heparin and heparan sulfates, with micromolar affinity. However, cell penetration still occurs according to both mechanisms of penetration in cells devoid of glycosaminoglycans, suggesting that glycosaminoglycans do not preferentially direct MCa’s cell penetration towards endocytosis [26]. In contrast, these cell surface receptors appear to be helpful as peptide sinks for increased peptide delivery into cells. More relevant to cell penetration is the fact that MCa binds to a number of membrane lipids, mostly negatively charged ones [26,27], as observed for other CPP [28]. Binding seems to occur with higher affinity (100 nM) in close agreement with the concentration of MCa required for cell penetration. In addition, MCa analogues that penetrate better than wild-type MCa also exhibit a greater affinity for membrane lipids and vice versa. Endocytosis becomes predominant with some large cargoes. Conversely, small cargoes seem to have less interference with a translocation mode of entry of CPP. These observations tend to indicate that endocytosis may become the preferential mode of cell entry of MCa if coupled to bulky cargoes that are expected to increase the duration of residency at the plasma membrane. However, although MCa is an extremely efficient vector for cell penetration of impermeable cargoes, it is of limited usefulness in its native conformation. Indeed, complex disulfide bridging may hamper the attachment of some cargoes. The size of the CPP is greater than those used in the literature. Finally, the native pharmacological activity is generally undesirable for most applications, so like other CPPs before, modification of the native MCa was made in order to ease its use in vivo as a new delivery system [30]. Canceling MCa’s pharmacological activity turned out to be quite simple due to the fact that structural requirements involved in binding onto the ryanodine receptor are more stringent than for cell penetration. Several chemical strategies turned out to be successful including point mutations [17], blocking MCa’s folding by preventing disulfide bridge formation [18], and producing a D-diastereomer MCa [16]. The second strategy had the advantage to produce a MCa analogue that was simpler to produce since the oxidation/folding step was no longer necessary. However, the resulting peptide turned out to be slightly less efficient in cell penetration than the folded/oxidized MCa, indicating that the correct positioning in space of the various structural determinants of MCa is important to optimize cell penetration. In addition, the unfolded MCa CPP was still thirty three amino acid residues in length. Quite recently, in an attempt to further delimitate the cell penetrating properties of MCa to smaller sequences, a number of unfolded truncated MCa-derived peptides were synthesized and assessed for cell penetration properties [19]. Surprisingly, all truncated peptides turned out to be more efficient than the unfolded full-length MCa for cell penetration, suggesting that each structured domain within the folded/oxidized MCa may provide a specific contribution to the cell penetration of the wild-type peptide. The shortest peptides were nine residues in length and included both the N-terminal and the C-terminal sequences. One of the peptides, MCaUF1-9, stood out as atypical since the net charge of the peptide was 0 and its cell penetration properties differed to some extent from the significantly more basic other MCa-derived truncated peptides. Penetration of this peptide occurred at polarized ends of CHO cells. The peptide also showed greater residency times within the plasma membrane. Its penetration did not rely at all on macropinocytosis for cell entry (at least when coupled to a dye as cargo). Finally, penetration of this peptide occurs at lower extracellular concentrations than the more basic peptides derived from MCa [19]. Altogether the properties of MCaUF1-9 seemed interesting enough to warrant a more in-depth investigation of its cell penetration properties. We therefore compare herein the properties of this peptide to well-reputed CPP (Tat, penetratin and poly-R) or analogous peptides derived from other toxins of the calcin family. We investigated the properties of a number of point mutated MCaUF1-9 analogues, and more specifically the pH-sensitivity of its penetration in order to design pH-sensitive CPP. The data point to new very powerful CPP with unprecedented efficacies and demonstrate the pH-sensitivities of several of our analogues for cell penetration.
2. Results and Discussion
2.1. A Peptide Derived from the Hydrophobic Face of MCa Behaves as a Highly Competitive CPP
A schematic representation of MCa illustrates that the amino acid sequence 1 to 9 tops the rest of the peptide (yellow residues) and defines an independent more hydrophobic face (Figure 1A). The opposite side of the peptide is highly basic (blue and pink residues) and defines therefore a basic face. Some of the residues involved in binding onto the ryanodine receptor have been defined in the past [17]. They include important residues such as Arg23 and Arg24 and define the pharmacophore side (residues in red and in pink). Therefore the pharmacophore and basic regions overlap to some degree.
We compared the efficacy of MCaUF1-9 for cell penetration within the glioma rat cell line F98 to other very popular peptides, comprising TAT, penetratin (Pen) and poly-Arg (poly-R). All peptides included an additional C-terminal Cys residue that was labeled with the Cy5 fluorochrome which served the purpose of cargo in this study. Of note, the extra Cys residue in MCaUF1-9-C-Cy5 is at its natural position as in the folded/oxidized MCa (Cys10) where it is linked to Cys21 by a disulfide bridge. In contrast, Cys3 of MCaUF1-9 was replaced by an isosteric 2-aminobutyric acid (Abu) residue to avoid mislabeling by Cy5 in the middle of the sequence. A series of confocal microscopy images were taken immediately after a 2 h incubation of F98 cells with various concentrations of the four peptides tested (MCaUF1-9-C-Cy5, TAT-C-Cy5, Pen-C-Cy5 and poly-R-C-Cy5). As shown, cell penetration of MCaUF1-9-C-Cy5 is perceptible at concentrations as low as 100 nM, whereas none of the other peptides showed penetration at this concentration (Figure 1B). Penetration was then dose-dependent for all peptides. It was more marked at 333 nM for MCaUF1-9-C-Cy5 and started to show up for Poly-R-C-Cy5, while still absent for TAT-C-Cy5 and Pen-C-Cy5. At 1 µM all peptides showed some degree of penetration, the least efficient peptide being TAT-C-Cy5. At 5 µM, F98 cells incubated with TAT-C-Cy5 showed levels of fluorescence that were more or less comparable to those obtained with MCaUF1-9-C-Cy5 at 333 nM. These data qualitatively indicated that MCaUF1-9-C-Cy5 behaved better than those three popular CPP as far as F98 cells are concerned. Similar results were obtained with CHO cells indicating that these differences in performances between the four peptides do not depend on the cell type studied (data not shown). With regard to cell distribution of the peptides, we did not notice any obvious differences in distribution suggesting that the peptides may all borrow the same mechanisms of cell penetration. However, a more complete investigation on this issue is needed before one comes to a firm conclusion. To more quantitatively compare the CPP, we investigated fluorescence levels by flow cytometry (Figure 1C). The data confirmed the confocal microscopy analyses showing that MCaUF1-9-C-Cy5 behaves more potently than other CPP. The following order of penetration efficiency was observed: MCaUF1-9-C-Cy5 >> Poly-R-C-Cy5 = Pen-C-Cy5 > TAT-C-Cy5. At 10 µM, MCaUF1-9-C-Cy5 penetrates 5.5-fold better than TAT-C-Cy5, 3.6-fold better than Pen-C-Cy5, and 3.5-fold better than Poly-R-C-Cy5.
2.2. Randomly Defined Control Peptides Delimit the Threshold Level of an Acceptable Cell Penetration
Our data also illustrate that the control linker-cargo, a single Cys residue linked to Cy5 (C-Cy5), does not penetrate at all into F98 cells (at concentrations up to 33 µM), demonstrating the peptide specificity of cell entry (Figure 1C and D). Another control was also tested, based on a fragment of charybdotoxin (ChTx), a voltage-gated potassium channel blocker, not known previously for cell penetration aptitude. The peptide encompasses the 12 first amino acids of ChTx. Internal Cys residues were replaced by Abu, an additional Cys residue was added at the C-terminus and the resulting peptide labeled with Cy5 as well to yield ChTxUF1-12-C-Cy5. Confocal microscopy images do not show any evidence of cell penetration if F98 cells are incubated 2 h with 3 µM ChTxUF1-12-C-Cy5 (Figure 1E). However, if a more sensitive and quantitative approach is taken to examine the levels of fluorescence, it becomes obvious that F98 cells take up a defined amount of ChTxUF1-12-C-Cy5. This is unlikely to represent the level of binding of this peptide to some cell-surface potassium channels as this fragment is not known to bind potassium channels. In contrast, it may indicate the propensity of some cell types to internalize peptides that present even low affinity for the plasma membrane. A comparison with our least-performing peptide TAT-C-Cy5 indicates that ChTxUF1-12-C-Cy5 is less efficient than TAT-C-Cy5, although significant. At 10 µM, the cell entry of this non-conventional CPP is 1.57-fold less than TAT. These findings may question the relevance of some studies reporting the discovery of “new” CPP or alternatively may suggest that ChTxUF1-12 can also be considered as a poorly performing CPP.
2.3. Pharmacological Blockade of Endocytosis in F98 Cells Affects Poorly MCaUF1-9 Cell Entry
Punctiform distribution of MCaUF1-9-C-Cy5 may be interpreted as a cell entry that is mainly based on a form of endocytosis. This point was assessed by FACS analyses using various drugs at the 2 h cell entry timepoint of 1 µM MCaUF1-9-C-Cy5 in F98 cells. We tested amiloride, a macropinocytosis inhibitor, methyl-β-cyclodextrin to deplete membrane cholesterol and inhibit lipid raft-dependent pathways, nocodazole to prevent microtubule formation, and cytochalasin D to stop F-actin elongation, required for macropinocytosis and clathrin-dependent endocytosis [31]. Amiloride only affected MCaUF1-9-C-Cy5 cell entry in F98 cells very mildly, with an average inhibition of 27% indicating an entry partly based on macropinocytosis (Figure 2).
The 18.5% inhibition observed by cytochalasin D, while being non-significant, was in agreement with the contribution of macropinocytosis to the cell entry of the peptide. Quite surprisingly, both methyl-β-cyclodextrin and nocodazole produced 24.7 and 48.9% increases in the cell entry of MCaUF1-9-C-Cy5. The lack of inhibition by methyl-β-cyclodextrin indicates that caveolae-mediated endocytosis is not involved in the entry of this peptide. The observed increase in cell penetration may indicate on the contrary that one preferential route of cell entry by translocation may be in lipid rafts. Preventing the loss of lipid rafts by blocking endocytosis at this level would increase the surface area devoted to lipid rafts and hence peptide entry.
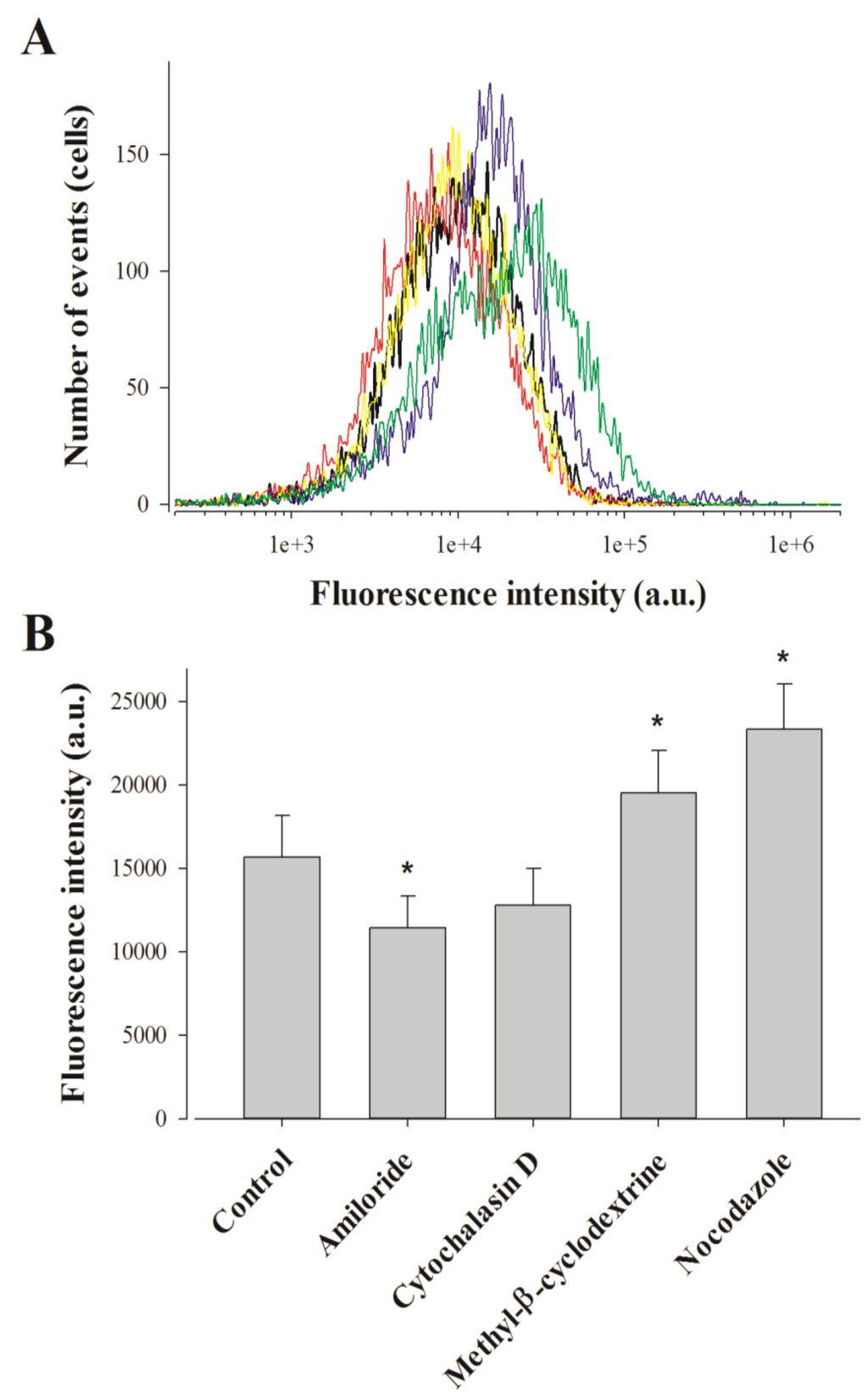
Figure 2.
Effect of endocytosis inhibitors on MCaUF1-9 peptide penetration in F98 cells. (A) Representative FACS data showing the effect of amiloride (red curve), cytochalasine D (yellow curve), methyl-β-cyclodextrine (blue curve) and nocodazole (green curve) on 1 µM MCaUF1-9-C-Cy5 cell entry (black curve). (B) Average fluorescence intensities for the cell entry of 1 µM MCaUF1-9-C-Cy5 in F98 cells without and with endocytosis inhibitors. * p≤ 0.1.
Similarly, it is possible that microtubules hinder cell penetration explaining why blocking their formation may help peptide entry to this large extent. These observations demonstrate that while the intracellular distribution of MCaUF1-9-C-Cy5 in F98 cells looks punctiform, this is not necessarily the consequence of a cell entry by endocytosis.
2.4. Point Mutation of MCaUF1-9 Fails to Optimize The Cell Penetrating Properties of This Peptide
Next, we attempted to design a number of MCaUF1-9 peptide analogues in order to get some hints on what structural determinants may be important for the efficacy of this peptide in cell penetration. At the N-terminal side of Cys3 (replaced by Abu) of MCaUF1-9, there are two residues that appear of minor importance (Gly1 and Asp2). Removing these two residues yields MCaUF3-9. As shown, MCaUF3-9-C-Cy5 still accumulates very well in F98 cells (Figure 3A,B).
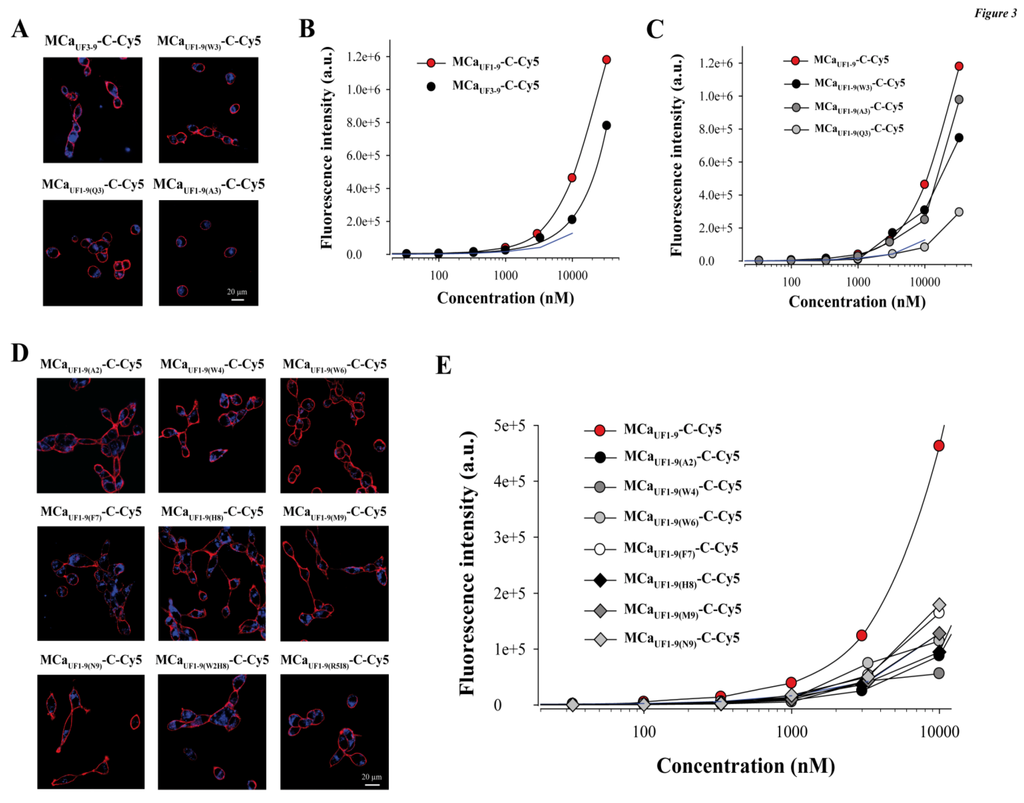
Figure 3.
Structural determinants of MCaUF1-9 peptide penetration. (A) Confocal images illustrating the penetration of 3 µM MCaUF3-9-C-Cy5, MCaUF1-9(W3)-C-Cy5, MCaUF1-9(Q3)-C-Cy5 or MCaUF1-9(A3)-C-Cy5 into F98 cells. 2 h incubation time before washout and imaging. (B) Effect of N-terminal peptide truncation on cell penetration efficacy of MCaUF1-9-C-Cy5 as assessed by flow cytometry. (C) Effect of point mutation at position 3 on the cell penetration efficacy of MCaUF1-9-C-Cy5. (D) Confocal images illustrating the penetration of 3 µM single or double point mutated MCaUF1-9-C-Cy5 peptide into F98 cells. (E) Effect of single point mutations on the cell penetration efficacy of MCaUF1-9-C-Cy5 as assessed by flow cytometry.
At the quantitative level, there was a 1.25-decrease in cell penetration efficacy at 3 µM, indicating that these two residues were most likely not essential for cell penetration. This deletion brings the size of this efficient CPP down to seven amino acid residues which is remarkably short. At position 3 of the wild-type MCa there is normally a Cys residue that engages itself into a disulfide bridge. The lateral chain of this residue is therefore not exposed towards the outside face of the molecule and is unlikely to play a role in cell penetration. MCaUF1-9 contains an Abu residue instead of a Cys residue and the peptide is quite efficient for cell penetration. We nevertheless probed this position by replacing the Abu residue by Trp, Gln or Ala. As shown, none of these replacements at position 3 within MCaUF1-9-C-Cy5 hindered the cell penetration of these analogues (Figure 3A). At 3 µM, MCaUF1-9(W3)-C-Cy5 penetrated 1.36-fold better than MCaUF1-9-C-Cy5, while MCaUF1-9(A3)-C-Cy5 penetrated 1.08-fold less well, indicating little variations (Figure 3C). In contrast, when Gln was put at position 3 in the sequence, the resulting MCaUF1-9(Q3)-C-Cy5 peptide behaved similarly to TAT but 2.94-fold less well than MCaUF1-9-C-Cy5 suggesting that a Gln may hinder the cell penetration process. Next, we made a series of single (seven peptide) or double (two peptide) point mutated analogues to probe the functional importance of these MCa residues. As shown, all mutated MCaUF1-9-C-Cy5 analogues produced evident cell penetration at 3 µM, indicating that none of the substitutions were powerful enough to fully prevent cell penetration (Figure 3D). However, according to flow cytometry analyses, none of the mutated peptides performed better than MCaUF1-9-C-Cy5 (Figure 3E). Taking TAT-C-Cy5 as a standard, MCaUF1-9(A2)-C-Cy5, MCaUF1-9(W4)-C-Cy5 and MCaUF1-9(H8)-C-Cy5 behaved slightly less well. In contrast, MCaUF1-9(F7)-C-Cy5 and MCaUF1-9(M9)-C-Cy5 still behaved better than TAT-C-Cy5. Double mutants (MCaUF1-9(W2H8)-C-Cy5 and MCaUF1-9(R5I8)-C-Cy5 were also closely similar to TAT-C-Cy5 (not shown). Overall, these data indicate that MCaUF1-9 peptide has been optimized for cell penetration with many amino acid residues playing an important role for cell entry. At this stage it would be difficult to point to one single residue as being more important than another one within the sequence.
2.5. Analogous Hydrophobic Domains of Other Toxin Members of the Calcin Family Are Also Excellent CPP
The inability to produce MCaUF1-9 peptides with greater cell penetration efficacies tends to indicate that sequence variation of this hydrophobic domain needs to be considered more cautiously to design optimized peptides. Interestingly, MCa belongs to a larger family of peptides that have, most of them, never been assessed for cell penetration. Results have been presented indicating that imperatoxin A also behaves as a CPP [32]. All these peptides are structured similarly to MCa, with a similar hydrophobic face topping a more basic face (Figure 4A). All peptides that have been tested are also active on the ryanodine receptor [6,7,33], indicating the presence of a similar pharmacophore. A close examination of the amino acid sequence of the hydrophobic domain reveals only minor sequence diversity among these peptides (Figure 4B). The nine first residues of hemicalcin are identical to MCa. Imperatoxin A, opicalcin 1 and opicalcin 2 differ from MCa only by residue 9 (an arginine instead of a leucine residue).
The most differing peptide sequence is the one derived from hadrucalcin with two extra N-terminal residues. Interestingly, the sequence SerGluLys replaces the Gly1 residue of MCa, but Asp2 of MCa is conserved. Also, four internal substitutions are noticeable. Leu4, Pro5, Lys8 and Leu9 of MCa are substituted by Iso, Lys, Gln and Arg, respectively. Finally, HadUF1-11 has an additional basic amino acid compared to MCaUF1-9. We next evaluated the ability of these peptides to accumulate into F98 cells. At 1 µM, it was obvious, according to confocal imaging, that ImpUF1-9-C-Cy5 (equivalent to Opi1UF1-9-C-Cy5 or Opi2UF1-9-C-Cy5) is less efficient for cell penetration than MCaUF1-9-C-Cy5 and HadUF1-11-C-Cy5 (Figure 4D). Interestingly, a similar deletion of the two first amino terminal residues of HadUF1-9-C-Cy5 yielded a peptide, MCaUF3-11-C-Cy5 with excellent penetration capabilities. Evaluation of dose-dependent penetration by flow cytometry demonstrated that ImpUF1-9-C-Cy5 penetrates quantitatively in a similar way than TAT-C-Cy5. For the first time, we also show that HadUF1-11-C-Cy5 and HadUF3-11-C-Cy5 both penetrate better than MCaUF1-9-C-Cy5 (Figure 4C).
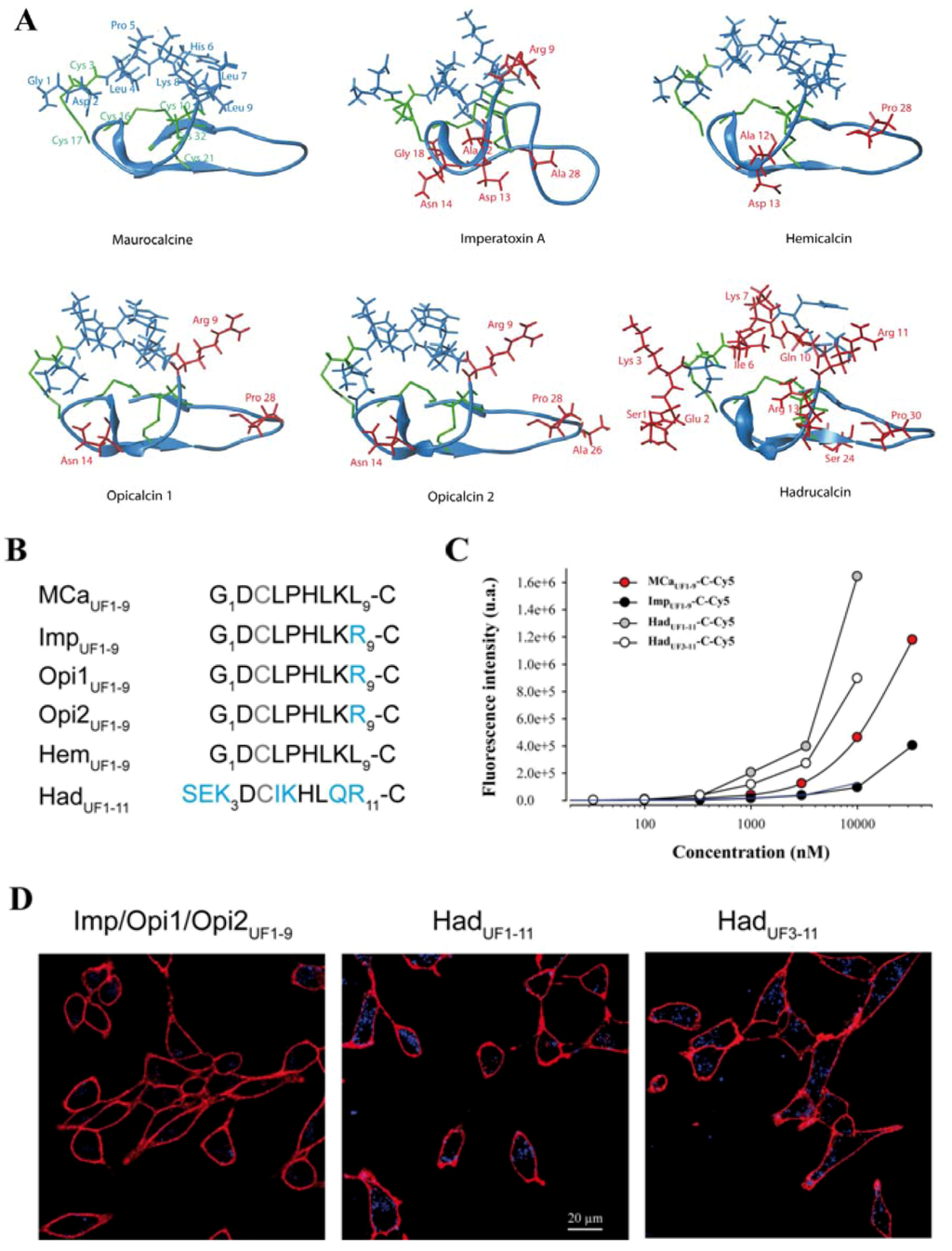
Figure 4.
Cell penetration properties of the hydrophobic domains of toxins from the calcin family. (A) Real or modeled 3D structures of MCa (PDB access code 1C6W), imperatoxin A (access code 1IE6), hemicalcin (model), opicalcin 1 (model), opicalcin 2 (model) and hadrucalcin (model). Residues in blue describe the hydrophobic domain investigated in this study. Red residues are all amino acids that differ from MCa’s amino acid sequence. Residues in green are cystine residues. (B) Amino acid sequences of the hydrophobic domains of each member of the calcin family. The third Cys residue is systematically replaced by Abu in our synthetic peptides and is represented in grey color. Residues in blue are those that differ from MCaUF1-9 amino acid sequence. This sequence alignment defines three groups of peptides with similar N-terminal sequences. (C) Comparative cell penetration efficacies of the peptides derived from members of the calcin family as assessed by dose-response curves from flow cytometry data. TAT-C-Cy5 data are indicated by a blue dashed line for comparison. (D) Confocal images illustrating the cell penetration of each peptide of the calcin family after incubation of 1 µM of the peptides with F98 cells during 2 h. A significantly lower cell penetration is observed for ImpUF1-9-C-Cy5 compared to the two hadrucalcin-derived peptides (HadUF1-11-C-Cy5 and HadUF3-11-C-Cy5).
This indicates that a series of very selective set of amino acid substitutions are required to improve MCaUF1-9 cell penetration properties. They also demonstrate that the calcin family can accommodate some variation in cell penetration efficacy for the activation of the ryanodine receptor. Nevertheless, it remains to be investigated whether the additional basic face and pharmacophore region are necessary to improve the characteristics of cell penetration of imperatoxin A, opicalcin 1 or opicalcin 2. In any case, it is obvious that HadUF1-11 is a remarkable cell penetration peptide by the extent of its efficacy compared to the popular peptides challenged in Figure 1.
2.6. Cell Penetration of MCaUF1-9 is pH-sensitive Owing to the Presence of an His Residue in its Amino Acid Sequence
Close examination of the amino acid sequence of MCa reveals that it contains a histidine residue at position 6. This residue is therefore also present in MCaUF1-9. According to Figure 2 data, this histidine residue contributes to some extent to the cell penetration efficacy of MCaUF1-9-C-Cy5. The imidazole sidechain of histidine has a pKa of approximately 6.0, while overall the pKa of the amino acid is 6.5. However, this value is susceptible to be influenced by the direct amino acid environment of this residue. In any case, it can be considered that at physiological conditions, relatively small changes in pH value is susceptible to alter the average charge of MCaUF1-9. At a pH value lower than 6, the imidazole ring is essentially protonated. Protonation of the His residue at position 6 may affect the cell penetration efficacy of MCaUF1-9-C-Cy5. To test this idea, the extracellular pH value was varied between 5.0 and 8.0 during the 2 h incubation of F98 cells with MCaUF1-9-C-Cy5 and the total accumulated fluorescence level evaluated by FACS. The data were normalized to the value at pH 5.0. As shown, decreasing pH values results in higher fluorescence values and therefore greater accumulation of the peptide in F98 cells (Figure 5A).
A fit of the data with a decreasing exponential suggests that the peptide enters into F98 cells with a 2.8-fold lower efficacy than at acidic pH values. Half of this decrease in efficacy occurs for a variation in pH from 5.0 to 5.7 indicating that the pKa value of this histidine residue within MCaUF1-9-C-Cy5 may be close to 5.7. The involvement of His6 in this pH-dependence of the cell penetration of the peptide is demonstrated by the lack of pH-dependence in cell penetration of the mutant peptide MCaUF1-9(W6)-C-Cy5 in which His6 is replaced by Trp6 (Figure 5B). Overall, these data indicate that protonation of His6, provided by acidic environments, results in improved MCaUF1-9-C-Cy5 cell delivery. In that respect, it is important to note that F98 cells are from rat glioma and that the extracellular pH within the glioma tumors masses in the brain has been predicted to be acidic [34]. Our finding therefore suggests that the MCaUF1-9 peptide may be useful to more specifically deliver anti-tumor drugs within glioma.
2.7. Long-Lasting Cell Retention of MCaUF1-9
One property of CPP is that they enter quite rapidly into cells. However, the persistence of their intracellular accumulation is seldom looked after. We investigated this question with three peptides. We used MCaUF1-9-C-Cy5, a non or poorly charged peptide depending on the protonation level of His6, MCaUF11-33-C-Cy5, a highly charged peptide mainly encompassing the two C-terminal thirds of MCa, and HadUF3-11-C-Cy5 which is less hydrophobic than MCaUF1-9-C-Cy5. One could expect that hydrophobic peptides may more readily escape from the cell interior than charged peptides. To test this idea, F98 cells were loaded 2 h with 1 µM of each of these peptides, extensively washed, maintained in culture for a variable duration (between 0 and 34 h, washout time), treated with trypsin and the fluorescence level remaining in the cells estimated by FACS (Figure 5C).
As shown and quite remarkably, the fluorescence levels of the accumulated peptides fade away only slowly with time indicating that the cell entry of the peptides are faster than their cell exit (Figure 5C,D). Time constant of half exit were estimated to be 5 h for MCaUF1-9-C-Cy5, 8 h and 20 min for MCaUF11-33-C-Cy5 and 16 h and 40 min for HadUF3-11-C-Cy5. After 34 h, cells still contained 27, 21 and 23% of MCaUF1-9-C-Cy5, MCaUF11-33-C-Cy5 and HadUF3-11-C-Cy5 fluorescence, respectively. These values are suspected to be higher in fact since 34 h is enough to register at least one cell division cycle. This property of persistence indicates that MCaUF1-9, but also MCaUF11-33 and HadUF3-11, can be used both as CPP and retention agents for drugs that may freely enter into cells but would also freely escape from them. We have previously used such a property to fight against chemo-resistance of the breast tumor cell line MDA-MB-231 by coupling doxorubicin to a non-folded version of MCa [20,22,23]. Confocal microscopy images of the cell distribution of both peptides tested indicate that the distribution of the peptides does not evolve with washout time (Figure 5D). The distribution remains mostly punctiform (with an evolution towards what may seem smaller dots), internal and hardly invades the nucleus.
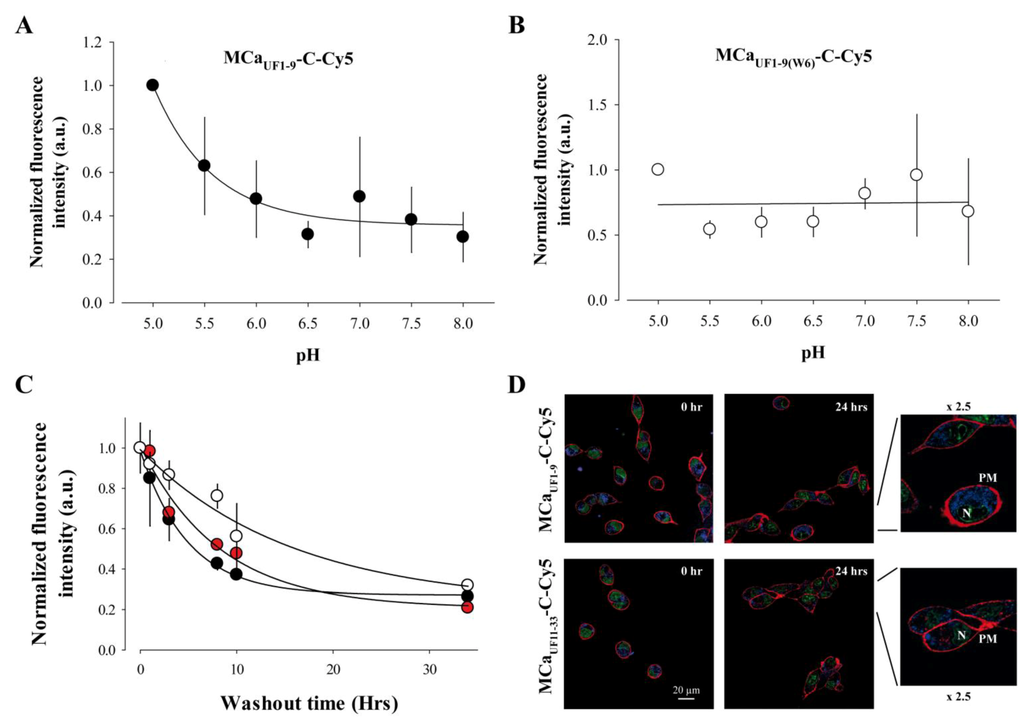
Figure 5.
pH-dependence and persistence of the cell penetration of MCaUF1-9-C-Cy5. (A) Effect of extracellular pH variation on the cell penetration efficacy of 1 µM MCaUF1-9-C-Cy5 in F98 cells (2 h incubation). Mean of n = 3 experiments ± S.D. Mean FACS results were normalized before averaging. Data were fitted by a decreasing exponential of the type y = y0 + a.e−bx where y0 = 0.36 ± 0.05, a = 0.64 ± 0.09 and b = 1.76 ± 0.61. (B) Effect of extracellular pH variation on the cell penetration of 1 µM MCaUF1-9(W6)-C-Cy5 in F98 cells (2 h incubation). The pH-insensitive Trp6 replaces the pH-sensitive His6 in this mutant peptide. (C) Persistence of the fluorescence signal in F98 cells preincubated 2 h with 1 µM MCaUF1-9-C-Cy5 (net charge 0 if His6 is unprotonated, black symbol), the positively charged MCaUF11-33-C-Cy5 (net charge +7, red symbol) or the mildly charged HadUF3-11-C-Cy5 peptide (net charge +2 if His8 is unprotonated, white symbol). FACS results were normalized to 1 at t=0 min at the start of the washout of the CPP. Data were fitted with a decreasing exponential function with 1/τ = 0.20 ± 0.01 h−1 (MCaUF1-9-C-Cy5), 0.12 ± 0.03 h−1 (MCaUF11-33-C-Cy5) et 0.06 ± 0.02 h−1 (HadUF3-11-C-Cy5), where τ is the time constant of the decrease in the mean cell fluorescence level. The non-decreasing fraction of fluorescence was equal to 0.27 ± 0.14 (MCaUF1-9-C-Cy5), 0.21 ± 0.07 (MCaUF11-33-C-Cy5) and 0.23 ± 0.13 (HadUF3-11-C-Cy5). (D) Corresponding confocal images illustrating that the intracellular distribution of the peptides did not change with time. The 2.5-fold image enhancement also shows a close to complete lack of nucleus invasion by the peptides. The images also show that the two peptides do not differ in the type of intracellular distribution 3.5 h after washout of the extracellular peptides.
2.8. Discussion
While all MCa truncations and/or disruptions of disulfide bridges produce a loss of the 3D structure, MCaUF1-9 is more susceptible than other truncated peptides to preserve some of the structural characteristics that it may possess within the full-length MCa as it encloses by itself the entire hydrophobic face of MCa. At this stage, one may only speculate as to why MCa presents one hydrophobic face diametrically opposite to a highly charged basic face. It may present a strong advantage for the peptide if it has to deal with both a hydrophilic environment (extracellular space and cytoplasm) and a hydrophobic one (membrane lipids). Besides it may be essential to the mechanism of cell translocation if the peptide needs to cope with the amphiphilic nature of membrane lipids. The strong dipole moment of the peptide, resulting from the existence of these two different faces, probably orients the peptide in its interaction with the plasma membrane. The highly basic nature of MCa should also speed up the peptide entry through electrochemical attraction if one considers that it diffuses freely through the plasma membrane, in a similar way that Na+ or Ca2+ ions would do when permeating through adequate ion channels while attracted by the inside negative membrane potential. Such a mechanism, if proven, would result in peptide accumulation against the concentration gradient. What the effect of a physical separation between these two peptide entities (hydrophobic face and basic face) might be on the cell penetration mechanism remains however unclear. One may notice at this point that MCaUF1-9-C-Cy5 and MCaUF11-33-C-Cy5 produce quite similar intracellular punctiform distributions in F98 cells arguing that they share nonetheless similar mechanisms of cell penetration. This was also the case for the other CPP investigated in this study (poly-R-C-Cy5, Tat-C-Cy5 and Pen-C-Cy5). It is of interest to note however that the cell distribution of fluorescent D-MCa, a full-length and well-structured analogue of MCa, is mostly diffuse [16] suggesting that combining the hydrophobic and basic faces of the molecule to shorten the residency time in the plasma membrane may represent a significant advantage in cell penetration.
The most interesting information that we could gather from our mutagenesis program of MCaUF1-9-C-Cy5 was that the two first amino acid residues were the most dispensable for its penetration properties. This truncation approach led to the design of a 7-mer CPP that has greater potency than most popular CPPs on the market. Investigating the cell penetrating properties of peptides derived from the hydrophobic face of other peptides members of the calcin family turned out as a more interesting approach than mutagenesis. Most of these peptides differ by only a few amino acid residues, with the notable exception of hadrucalcin. Coherent with the mono-substitution we performed at amino acid 9 of MCaUF1-9-C-Cy5, the cell penetration properties of ImpUF1-9-C-Cy5, Opi1UF1-9-C-Cy5 and Opi2UF1-9-C-Cy5, all presenting Arg9 instead of Leu9, were reduced to some extent compared to MCaUF1-9-C-Cy5. Remarkably, multiple amino acid substitution as demonstrated within HadUF1-11-C-Cy5 resulted in an important and unexpected improvement of cell penetration. Similarly to MCaUF1-9-C-Cy5, removing the two first N-terminal amino acid residues of HadUF1-11-C-Cy5 produced a peptide with quite significant levels of cell penetration. The core of the sequence, the one we assume to be important for cell penetration (after Abu3 in MCaUF1-9), contains no less than 4 substitutions (Leu4 by Ile, Pro5 by Lys, Lys8 by Gln, and Leu9 by Arg). This indicates that quite elaborate alterations need to be done to MCaUF1-9 sequence to further improve it cell penetrating properties. In any case, these findings (i) define a novel CPP with unprecedented efficacy, and (ii) open the door for the design of hadrucalcin/MCa chimeras 1 µM MCaUF1-9-C-Cy5 fluorescence at any given concentration and never in terms of starting concentration at which cell entry was observed. These observations suggest that the affinity of these peptides for plasma membrane components remains unaltered.
Since we are interested in developing a number of applications in oncology for MCa analogues, we also investigated the role of His6 in cell entry into the glioma F98 cells. Of great interest for future applications, we found that protonation of His6, occurring at more acidic pH, but in a range compatible with pH values observed in glioma, produced a three-fold more potent peptide for cell penetration. We assume that protonated His6 may form a salt bridge with Asp2 in the non-structured MCaUF1-9. It is likely that, without this salt bridge, the negative charge carried by Asp2 may disfavor cell penetration of MCaUF1-9. This observation on the importance of protonation in peptide cell penetration will be useful for the future design of new MCa analogues in which important basic amino acid residues may be substituted by His residues in order to further improve the tumor-selectivity of these potent CPP. Interesting positions will include Lys19, Lys20, Lys22 and Arg24 all shown to contribute to the cell penetration efficacy of the full length MCa [17].
3. Experimental
3.1. Reagents
N-α-Fmoc-l-aminoacids, Wang-Tentagel resin and reagents used for peptide syntheses were obtained from Iris Biotech (Marktredwitz, Germany). Solvents were analytical grade products from Acros Organics (Geel, Belgium). Cy5 maleimide mono-reactive dye was purchased from GE Healthcare (Saclay, France).
3.2. Peptide Syntheses
Chemical syntheses of truncated toxin peptides were performed as previously described [16]. Briefly, peptides were chemically synthesized by the solid-phase method [35] using an automated peptide synthesizer (CEM© Liberty 12, Matthews, NC, USA). Peptide chains were assembled stepwise on 0.24 meq of Fmoc-D-Arg-Pbf-Wang-Tentagel resin using 0.24 mmol of N-α-fluorenylmethyloxycarbonyl (Fmoc) L-amino-acid derivatives. The side-chain protecting groups were: trityl for Cys and Asn, tert-butyl for Ser, Thr, Glu and Asp, Pbf for Arg and tert-butylcarbonyl for Lys. Reagents were at the following concentrations: Fmoc-amino-acids [0.2 M Fmoc-AA-OH in dimethylformamide (DMF)], activator (0.5 M 2-(1H-benzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate in DMF), activator base [2 M diisopropylethylamine in N-methylpyrrolidone (NMP)] and deprotecting agent (5% piperazine/0.1 M 1-hydroxybenzotriazole in DMF), as advised by PepDriver (CEM©). After peptide chain assembly, resins were treated 4 h at room temperature with a mixture of trifluoroacetic acid/water/triisopropylsilane (TIS)/dithiothreitol (DTT) (92.5/2.5/2.5/2.5). The peptide mixtures were then filtered and the filtrates were precipitated by adding cold tert-butylmethyl ether. The crude peptides were pelleted by centrifugation (10,000 × g, 15 min) and the supernatants were discarded. Truncated toxin analogues were purified by HPLC using a Vydac C18 column (218TP1010, 25 × 10 cm). Elutions of the peptides were performed with a 10–60% acetonitrile linear gradient containing 0.1% trifluoroacetic acid. The purified fractions were analyzed by analytical RP-HPLC (Vydac C18 column 218TP104, 25 × 4.6 cm). All analogues were characterized by MALDI-TOF mass spectrometry.
3.3. Peptide Labeling With Cy5
Each peptide was labeled with Cy5 according to the manufacturer’s protocol (GE Healthcare). Peptides were dissolved at 200 µg/mL in 1 M NaHCO3 buffer, pH 9.3. 500 µL of solubilized peptides were added to Cy5-maleimide containing tubes. The mixtures were incubated during 2 h at room temperature and then purified by HPLC using an analytical Vydac C18 column. Elution of the Cy5-labeled peptides was performed with a 5–90% acetonitrile linear gradient containing 0.1% trifluoroacetic acid. The pure peak fractions were lyophilized and peptides quantified by UV spectrophotometer at 650 nm.
3.4. Cell Culture
Undifferentiated malignant glioma rat (F98) cell line (from ATCC) was maintained at 37 °C in 5% CO2 in DMEM/F-12 nutrient medium (Invitrogen, Cergy Pontoise, France) supplemented with 2% (v/v) heat-inactivated fetal bovine serum (Invitrogen) and 100 µg/mL streptomycine and 100 units/mL penicillin (Invitrogen).
3.5. Confocal Microscopy
For analysis of the cell entry of Cy5-labeled-truncated toxin peptides in living cells, cell cultures were incubated with the fluorescent peptides (in DMEM/F-12 nutrient medium only) for 2 h, and then washed twice with phosphate-buffered saline (PBS) alone. The plasma membrane was stained with 50 μg/mL rhodamine-conjugated concanavalin A (Invitrogen, Cergy Pontoise, France) for 5 min. Cells were washed once more. Live cells were then immediately analyzed by confocal laser scanning microscopy using a Zeiss LSM operating system. Rhodamine (561 nm) and Cy5 (633 nm) were sequentially excited and emission fluorescence were collected.
3.6. Fluorescence Activated Cell Sorting Analyses
F98 cells were incubated with various concentrations of Cy5-labeled peptides in DMEM/F-12 culture medium without serum at 37 °C for 2 h. The cells were then washed with PBS to remove excess extracellular peptide and treated with 0.48 mM versene (Invitrogen) for 5 min at 37 °C to detach cells from the surface, and centrifuged at 200 g in DMEM/F-12 culture medium before suspension in PBS. For experiments concerning endocytosis inhibitors, F98 cells were initially preincubated in DMEM/F-12 culture medium without serum for 30 min at 37 °C with different inhibitors of endocytosis: (i) 100 µM amiloride, (ii) 5 µg/mL cytochalasin D (10 µM), (iii) 20 µM nocodazole, or (iv) 5 mM methyl-β-cyclodextrin (all from Sigma, Lyon, France). The cells were then incubated for 2 h at 37 °C with 1 µM MCaUF1-9-C-Cy5 in the presence of each drug. Flow cytometry analyses were performed with live cells using an Accuri C6 flow cytometer (BD Biosciences, Le Pont de Claix, France). Data were obtained and analyzed using CFlow Sampler (BD Biosciences). Live cells were gated by forward/side scattering from a total of 10,000 events.
3.7. Molecular Modeling
Using Sybyl X 1.3 (Tripos Inc., St. Louis, MO, USA) and PDB structure of MCa (code 1C6W) and imperatoxin A (code 1IE6), we generated 3D models of opicalcin 1 and 2, hemicalcin and hadrucalcin. There is a sequence homology (76% up to 91%) between these proteins. Based on previous reports [6,7], we replaced some amino acid of MCa sequence to obtain the corresponding ones for the four different proteins. Several steps of minimization and control of the stereochemistry were performed to obtain a model for each molecule.
4. Conclusions
In this manuscript, we have demonstrated that MCaUF1-9-C-Cy5 starts to show detectable penetration in glioma F98 cells at concentrations as low as 33 nM (5-fold increase over control) as detected by FACS. The process is visible at 100 nM by confocal microscopy and a comparative analysis reveals that it is highly competitive compared to TAT, penetratin or Poly-R CPP. One analogue turns out to be extremely competitive, MCaUF3-9, owing to its performance, length and ease of synthesis. Nevertheless, we also demonstrate that engineered optimization of its cell penetrating properties is hard to achieve but that Mother Nature has provided an elegant solution to this problem by selecting itself the best amino acid substitutions under the form of new calcin analogues. In that respect, the hydrophobic domain of hadrucalcin outperforms that of MCa. We evidence for the first time the possibility to modulate peptide cell penetration by external pH provided that His residues are strategically incorporated within the amino acid sequence. This finding enlarges the potential application of these peptides to the treatment of glioma. Additionally, the observation that the residency time of these peptides in glioma F98 cells is quite long suggest that these peptides may be best used when injected once inside a solid tumor rather than by intravenous route.
Acknowledgements
We acknowledge financial support to MDW by Inserm and by the Nanofret grant and LabEx ICST of the Agence Nationale de la Recherche (ANR). MDW is a recipient of a contrat d’interface from Inserm and Grenoble Hospital. EB has a fellowship from ANR. We also thank the Région Rhône Alpes for the financial support of CT by an Emergence grant.
References
- Fajloun, Z.; Kharrat, R.; Chen, L.; Lecomte, C.; di Luccio, E.; Bichet, D.; El Ayeb, M.; Rochat, H.; Allen, P.D.; Pessah, I.N.; et al. Chemical synthesis and characterization of maurocalcine, a scorpion toxin that activates Ca2+ release channel/ryanodine receptors. FEBS Lett. 2000, 469, 179–185. [Google Scholar] [CrossRef]
- Mouhat, S.; Jouirou, B.; Mosbah, A.; de Waard, M.; Sabatier, J.M. Diversity of folds in animal toxins acting on ion channels. Biochem J. 2004, 378, 717–726. [Google Scholar] [CrossRef]
- Mosbah, A.; Kharrat, R.; Fajloun, Z.; Renisio, J.G.; Blanc, E.; Sabatier, J.M.; El Ayeb, M.; Darbon, H. A new fold in the scorpion toxin family, associated with an activity on a ryanodine-sensitive calcium channel. Proteins 2000, 40, 436–442. [Google Scholar] [CrossRef]
- Zamudio, F.Z.; Gurrola, G.B.; Arevalo, C.; Sreekumar, R.; Walker, J.W.; Valdivia, H.H.; Possani, L.D. Primary structure and synthesis of imperatoxin A (iptx(a)), a peptide activator of Ca2+ release channels/ryanodine receptors. FEBS Lett. 1997, 405, 385–389. [Google Scholar] [CrossRef]
- Zhu, S.; Darbon, H.; Dyason, K.; Verdonck, F.; Tytgat, J. Evolutionary origin of inhibitor cystine knot peptides. FASEB J. 2003, 17, 1765–1767. [Google Scholar]
- Shahbazzadeh, D.; Srairi-Abid, N.; Feng, W.; Ram, N.; Borchani, L.; Ronjat, M.; Akbari, A.; Pessah, I.N.; de Waard, M.; El Ayeb, M. Hemicalcin, a new toxin from the iranian scorpion hemiscorpius lepturus which is active on ryanodine-sensitive Ca2+ channels. Biochem. J. 2007, 404, 89–96. [Google Scholar] [CrossRef]
- Schwartz, E.F.; Capes, E.M.; Diego-Garcia, E.; Zamudio, F.Z.; Fuentes, O.; Possani, L.D.; Valdivia, H.H. Characterization of hadrucalcin, a peptide from hadrurus gertschi scorpion venom with pharmacological activity on ryanodine receptors. Br. J. Pharmacol. 2009, 157, 392–403. [Google Scholar] [CrossRef]
- Altafaj, X.; France, J.; Almassy, J.; Jona, I.; Rossi, D.; Sorrentino, V.; Mabrouk, K.; de Waard, M.; Ronjat, M. Maurocalcine interacts with the cardiac ryanodine receptor without inducing channel modification. Biochem. J. 2007, 406, 309–315. [Google Scholar] [CrossRef]
- Szappanos, H.; Smida-Rezgui, S.; Cseri, J.; Simut, C.; Sabatier, J.M.; de Waard, M.; Kovacs, L.; Csernoch, L.; Ronjat, M. Differential effects of maurocalcine on Ca2+ release events and depolarization-induced Ca2+ release in rat skeletal muscle. J. Physiol. 2005, 565, 843–853. [Google Scholar] [CrossRef]
- Gurrola, G.B.; Arevalo, C.; Sreekumar, R.; Lokuta, A.J.; Walker, J.W.; Valdivia, H.H. Activation of ryanodine receptors by imperatoxin A and a peptide segment of the II-III loop of the dihydropyridine receptor. J. Biol. Chem. 1999, 274, 7879–7886. [Google Scholar]
- Esteve, E.; Mabrouk, K.; Dupuis, A.; Smida-Rezgui, S.; Altafaj, X.; Grunwald, D.; Platel, J.C.; Andreotti, N.; Marty, I.; Sabatier, J.M.; et al. Transduction of the scorpion toxin maurocalcine into cells. Evidence that the toxin crosses the plasma membrane. J. Biol. Chem. 2005, 280, 12833–12839. [Google Scholar]
- Tanabe, T.; Beam, K.G.; Adams, B.A.; Niidome, T.; Numa, S. Regions of the skeletal muscle dihydropyridine receptor critical for excitation-contraction coupling. Nature 1990, 346, 567–569. [Google Scholar] [CrossRef]
- Tanabe, T.; Beam, K.G.; Powell, J.A.; Numa, S. Restoration of excitation-contraction coupling and slow calcium current in dysgenic muscle by dihydropyridine receptor complementary DNA. Nature 1988, 336, 134–139. [Google Scholar] [CrossRef]
- Altafaj, X.; Cheng, W.; Esteve, E.; Urbani, J.; Grunwald, D.; Sabatier, J.M.; Coronado, R.; de Waard, M.; Ronjat, M. Maurocalcine and domain a of the II-III loop of the dihydropyridine receptor Cav1.1 subunit share common binding sites on the skeletal ryanodine receptor. J. Biol. Chem. 2005, 280, 4013–4016. [Google Scholar]
- Chen, L.; Esteve, E.; Sabatier, J.M.; Ronjat, M.; de Waard, M.; Allen, P.D.; Pessah, I.N. Maurocalcine and peptide a stabilize distinct subconductance states of ryanodine receptor type 1, revealing a proportional gating mechanism. J. Biol. Chem. 2003, 278, 16095–16106. [Google Scholar]
- Poillot, C.; Dridi, K.; Bichraoui, H.; Pecher, J.; Alphonse, S.; Douzi, B.; Ronjat, M.; Darbon, H.; de Waard, M. D-maurocalcine, a pharmacologically inert efficient cell-penetrating peptide analogue. J. Biol. Chem. 2010, 285, 34168–34180. [Google Scholar] [CrossRef]
- Mabrouk, K.; Ram, N.; Boisseau, S.; Strappazzon, F.; Rehaim, A.; Sadoul, R.; Darbon, H.; Ronjat, M.; de Waard, M. Critical amino acid residues of maurocalcine involved in pharmacology, lipid interaction and cell penetration. Biochim. Biophys. Acta 2007, 1768, 2528–2540. [Google Scholar] [CrossRef]
- Ram, N.; Weiss, N.; Texier-Nogues, I.; Aroui, S.; Andreotti, N.; Pirollet, F.; Ronjat, M.; Sabatier, J.M.; Darbon, H.; Jacquemond, V.; et al. Design of a disulfide-less, pharmacologically-inert and chemically-competent analog of maurocalcine for the efficient transport of impermeant compounds into cells. J. Biol. Chem. 2008, 283, 27048–27056. [Google Scholar] [CrossRef]
- Poillot, C.; Bichraoui, H.; Tisseyre, C.; Bahemberae, E.; Andreotti, N.; Sabatier, J.M.; Ronjat, M.; de Waard, M. Small efficient cell-penetrating peptides derived from scorpion toxin maurocalcine. J. Biol. Chem. 2012, 287, 17331–17342. [Google Scholar] [CrossRef]
- Aroui, S.; Brahim, S.; de Waard, M.; Breard, J.; Kenani, A. Efficient induction of apoptosis by doxorubicin coupled to cell-penetrating peptides compared to unconjugated doxorubicin in the human breast cancer cell line MDA-MB 231. Cancer Lett. 2009, 285, 28–38. [Google Scholar] [CrossRef]
- Aroui, S.; Brahim, S.; de Waard, M.; Kenani, A. Cytotoxicity, intracellular distribution and uptake of doxorubicin and doxorubicin coupled to cell-penetrating peptides in different cell lines: A comparative study. Biochem. Biophys. Res. Commun. 2010, 391, 419–425. [Google Scholar] [CrossRef]
- Aroui, S.; Brahim, S.; Hamelin, J.; de Waard, M.; Breard, J.; Kenani, A. Conjugation of doxorubicin to cell penetrating peptides sensitizes human breast MDA-MB 231 cancer cells to endogenous trail-induced apoptosis. Apoptosis 2009, 14, 1352–1365. [Google Scholar] [CrossRef]
- Aroui, S.; Ram, N.; Appaix, F.; Ronjat, M.; Kenani, A.; Pirollet, F.; de Waard, M. Maurocalcine as a non-toxic drug carrier overcomes doxorubicin resistance in the cancer cell line MDA-MB 231. Pharm. Res. 2009, 26, 836–845. [Google Scholar] [CrossRef]
- Ram, N.; Texier-Nogues, I.; Pernet-Gallay, K.; Poillot, C.; Ronjat, M.; Andrieux, A.; Arnoult, C.; Daou, J.; de Waard, M. In vitro and in vivo cell delivery of quantum dots by the cell penetrating peptide maurocalcine. Int. J. Biomed. Nanosci. Nanotechnol. 2011, 2, 12–32. [Google Scholar] [CrossRef]
- Stasiuk, G.J.; Tamang, S.; Imbert, D.; Poillot, C.; Giardiello, M.; Tisseyre, C.; Barbider, E.L.; Fries, P.H.; de Waard, M.; Reiss, P.; Mazzanti, M. Cell-permeable ln(III) chelate-functionalized InP quantum dots as multimodal imaging agents. ACS Nano 2011, 5, 8193–8201. [Google Scholar] [CrossRef]
- Ram, N.; Aroui, S.; Jaumain, E.; Bichraoui, H.; Mabrouk, K.; Ronjat, M.; Lortat-Jacob, H.; de Waard, M. Direct peptide interaction with surface glycosaminoglycans contributes to the cell penetration of maurocalcine. J. Biol. Chem. 2008, 283, 24274–24284. [Google Scholar] [CrossRef]
- Boisseau, S.; Mabrouk, K.; Ram, N.; Garmy, N.; Collin, V.; Tadmouri, A.; Mikati, M.; Sabatier, J.M.; Ronjat, M.; Fantini, J.; et al. Cell penetration properties of maurocalcine, a natural venom peptide active on the intracellular ryanodine receptor. Biochim. Biophys. Acta 2006, 1758, 308–319. [Google Scholar] [CrossRef]
- Jones, A.T.; Sayers, E.J. Cell entry of cell penetrating peptides: Tales of tails wagging dogs. J. Control. Release 2012, 161, 582–591. [Google Scholar] [CrossRef]
- Lindgren, M.; Hallbrink, M.; Prochiantz, A.; Langel, U. Cell-penetrating peptides. Trends Pharmacol. Sci. 2000, 21, 99–103. [Google Scholar] [CrossRef]
- Lundberg, P.; Langel, U. A brief introduction to cell-penetrating peptides. J. Mol. Recognit. 2003, 16, 227–233. [Google Scholar] [CrossRef]
- Mano, M.; Teodosio, C.; Paiva, A.; Simoes, S.; Pedroso de Lima, M.C. On the mechanisms of the internalization of s4(13)-pv cell-penetrating peptide. Biochem. J. 2005, 390, 603–612. [Google Scholar] [CrossRef]
- Gurrola, G.B.; Capes, E.M.; Zamudio, F.Z.; Possani, L.D.; Valdivia, H.H. Imperatoxin A, a cell-penetrating peptide from scorpion venom, as a probe of Ca-release channels/ryanodine receptors. Pharmaceuticals (Basel) 2010, 3, 1093–1107. [Google Scholar]
- El-Hayek, R.; Lokuta, A.J.; Arevalo, C.; Valdivia, H.H. Peptide probe of ryanodine receptor function. Imperatoxin A, a peptide from the venom of the scorpion pandinus imperator, selectively activates skeletal-type ryanodine receptor isoforms. J. Biol. Chem. 1995, 270, 28696–28704. [Google Scholar]
- Garcia-Martin, M.L.; Herigault, G.; Remy, C.; Farion, R.; Ballesteros, P.; Coles, J.A.; Cerdan, S.; Ziegler, A. Mapping extracellular pH in rat brain gliomas in vivo by 1H magnetic resonance spectroscopic imaging: Comparison with maps of metabolites. Cancer Res. 2001, 61, 6524–6531. [Google Scholar]
- Merrifield, R.B. Solid-phase peptide synthesis. Adv. Enzymol. Relat. Areas Mol. Biol. 1969, 32, 221–296. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).