Role of the G Protein-Coupled Receptor, mGlu1, in Melanoma Development
Abstract
Introduction
GPCRs
Role of GPCRs in Human Diseases
GPCRs as Oncogenes
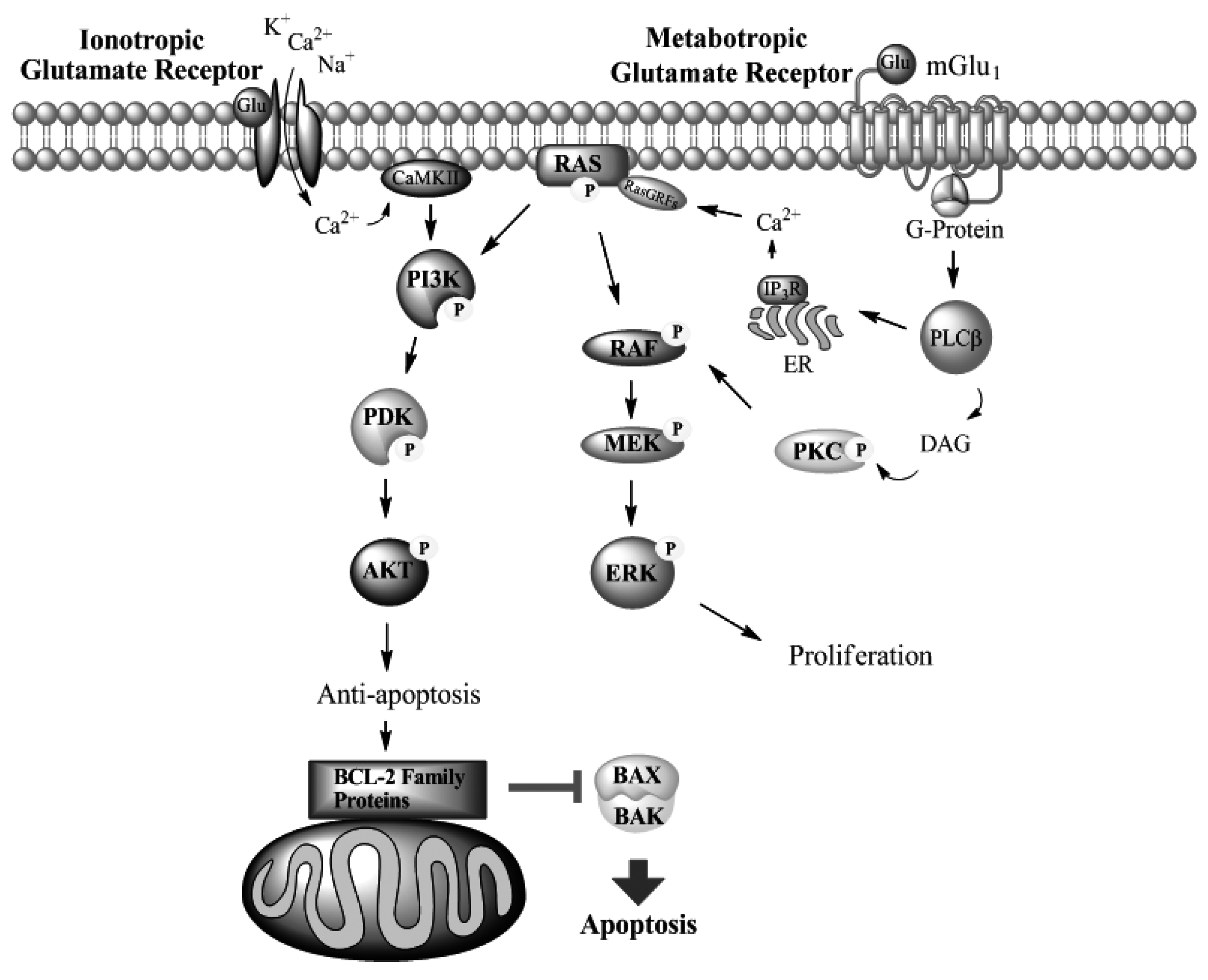
Metabotropic Glutamate Receptors
mGlu1 in Melanoma
Glutamate Receptors in Other Cancers
Conclusions
References
- Rigel, D.S. Malignant melanoma: Perspectives on incidence and its effects on awareness, diagnosis, and treatment. Ca-A Cancer J. Clin. 1996, 46, 195–198. [Google Scholar]
- Chang, A.E.; Karnell, L.H.; Menck, H.R. The National Cancer Data Base report on cutaneous and noncutaneous melanoma: A summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer 1998, 83, 1664–1678. [Google Scholar] [PubMed]
- American Cancer Society, Cancer facts & figures; American Cancer Society: Atlanta, GA, USA, 2010.
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Murray, T.; Thun, M.J. Cancer statistics, 2008. CA-Cancer J. Clin. 2008, 58, 71–96. [Google Scholar] [CrossRef] [PubMed]
- Cannon-Albright, L.A.; Goldgar, D.E.; Meyer, L.J.; Lewis, C.M.; Anderson, D.E.; Fountain, J.W.; Hegi, M.E.; Wiseman, R.W.; Petty, E.M.; Bale, A.E.; et al. Assignment of a locus for familial melanoma, MLM, to chromosome 9p13-p22. Science 1992, 258, 1148–1152. [Google Scholar] [PubMed]
- Quelle, D.E.; Cheng, M.; Ashmun, R.A.; Sherr, C.J. Cancer-associated mutations at the INK4a locus cancel cell cycle arrest by p16INK4a but not by the alternative reading frame protein p19ARF. Proc. Natl. Acad. Sci. USA 1997, 94, 669–673. [Google Scholar]
- Quelle, D.E.; Zindy, F.; Ashmun, R.A.; Sherr, C.J. Alternative reading frames of the INK4a tumor suppressor gene encode two unrelated proteins capable of inducing cell cycle arrest. Cell 1995, 83, 993–1000. [Google Scholar]
- Becker, T.M.; Rizos, H.; Kefford, R.F.; Mann, G.J. Functional impairment of melanoma-associated p16(INK4a) mutants in melanoma cells despite retention of cyclin-dependent kinase 4 binding. Clin. Cancer Res. 2001, 7, 3282–3288. [Google Scholar]
- Sotillo, R.; Garcia, J.F.; Ortega, S.; Martin, J.; Dubus, P.; Barbacid, M.; Malumbres, M. Invasive melanoma in Cdk4-targeted mice. Proc. Natl. Acad. Sci. USA 2001, 98, 13312–13317. [Google Scholar]
- McKenzie, H.; Becker, T.M.; Scurr, L.L.; Kefford, R.F.; Rizos, H. Wild type and melanoma-associated mutant p16(IN4a) proteins do not oligomerize in vivo. Pigment Cell Melanoma Res. 2009, 22, 131–133. [Google Scholar] [PubMed]
- Davies, H.; Bignell, G.R.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.J.; Bottomley, W.; Davis, N.; Dicks, E.; Ewing, R.; Floyd, Y.; Gray, K.; Hall, S.; Hawes, R.; Hughes, J.; Kosmidou, V.; Menzies, A.; Mould, C.; Parker, A.; Stevens, C.; Watt, S.; Hooper, S.; Wilson, R.; Jayatilake, H.; Gusterson, B.A.; Cooper, C.; Shipley, J.; Hargrave, D.; Pritchard-Jones, K.; Maitland, N.; Chenevix-Trench, G.; Riggins, G.J.; Bigner, D.D.; Palmieri, G.; Cossu, A.; Flanagan, A.; Nicholson, A.; Ho, J.W.; Leung, S.Y.; Yuen, S.T.; Weber, B.L.; Seigler, H.F.; Darrow, T.L.; Paterson, H.; Marais, R.; Marshall, C.J.; Wooster, R.; Stratton, M.R.; Futreal, P.A. Mutations of the BRAF gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar]
- Collisson, E.; De, A.; Suzuki, H.; Gambhir, S.; Kolodney, M. Treatment of metastatic melanoma with an orally available inhibitor of the Ras-Raf-MAPK cascade. Can. Res. 2003, 63, 5669–5673. [Google Scholar]
- Libra, M.; Malaponte, G.; Navolanic, P.M.; Gangemi, P.; Bevelacqua, V.; Proietti, L.; Bruni, B.; Stivala, F.; Mazzarino, M.C.; Travali, S.; McCubrey, J.A. Analysis of BRAF mutation in primary and metastatic melanoma. Cell Cycle 2005, 4, 1382–1384. [Google Scholar]
- Dahl, C.; Guldberg, P. The genome and epigenome of malignant melanoma. APMIS 2007, 115, 1161–1176. [Google Scholar]
- Tuveson, D.A.; Weber, B.L.; Herlyn, M. BRAF as a potential therapeutic target in melanoma and other malignancies. Cancer Cell 2003, 4, 95–98. [Google Scholar]
- Pollock, P.; Harper, U.; Hansen, K.; Yudt, L.; Stark, M.; Robbins, C.; Moses, T.; Hostetter, G.; Wagner, U.; Kakareka, J.; Salem, G.; Pohida, T.; Heenan, P.; Duray, P.; Kallioniemi, O.; Hayward, N.; Trent, J.; Meltzer, P. High frequency of BRAF mutations in nevi. Nat. Genet. 2003b, 33, 19–20. [Google Scholar] [PubMed]
- Whitwam, T.; Vanbrocklin, M.W.; Russo, M.E.; Haak, P.T.; Bilgili, D.; Resau, J.H.; Koo, H.M.; Holmen, S.L. Differential oncogenic potential of activated RAS isoforms in melanocytes. Oncogene 2007, 26, 4563–4570. [Google Scholar]
- Barbacid, M. Ras oncogenes: Their role in neoplasia. Eur. J. Clin. Invest. 1990, 20, 225–235. [Google Scholar]
- Pollock, P.M.; Cohen-Solal, K.; Sood, R.; Namkoong, J.; Martino, J.J.; Koganti, A.; Zhu, H.; Robbins, C.; Makalowska, I.; Shin, S.S.; Marin, Y.; Roberts, K.G.; Yudt, L.M.; Chen, A.; Cheng, J.; Incao, A.; Pinkett, H.W.; Graham, C.L.; Dunn, K.; Crespo-Carbone, S.M.; Mackason, K.R.; Ryan, K.B.; Sinsimer, D.; Goydos, J.; Reuhl, K.R.; Eckhaus, M.; Meltzer, P.S.; Pavan, W.J.; Trent, J.M.; Chen, S. Melanoma mouse model implicates metabotropic glutamate signaling in melanocytic neoplasia. Nat. Genet. 2003, 34, 108–112. [Google Scholar]
- Gudermann, T.; Nurnberg, B.; Schultz, G. Receptors and G-Proteins as primary components of transmembrane signal transduction. G-protein-coupled receptors -structure and function. J. Molec. Med.-JMM 1995, 73, 51–63. [Google Scholar] [CrossRef]
- Bourne, H.R.; Sanders, D.A.; McCormick, F. The GTPase superfamily: a conserved switch for diverse cell functions. Nature 1990, 348, 125–132. [Google Scholar]
- Bourne, H.R.; Sanders, D.A.; McCormick, F. The GTPase superfamily: conserved structure and molecular mechanism. Nature 1991, 349, 117–127. [Google Scholar]
- Fredriksson, R.; Schioth, H.B. The repertoire of G-protein-coupled receptors in fully sequenced genomes. Mol. Pharmacol. 2005, 67, 1414–1425. [Google Scholar]
- Insel, P.A.; Tang, C.M.; Hahntow, I.; Michel, M.C. Impact of GPCRs in clinical medicine: Monogenic diseases, genetic variants and drug targets. Biochim.Biophys. Acta 2007, 1768, 994–1005. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.D.; Burnham, W.M.; Cole, D.E. The G protein-coupled receptors: Pharmacogenetics and disease. Crit. Rev. Clin. Lab. Sci. 2005, 42, 311–392. [Google Scholar]
- Spiegel, A.M.; Weinstein, L.S. Inherited diseases involving g proteins and g protein-coupled receptors. Annu.Rev. Med. 2004, 55, 27–39. [Google Scholar]
- Muradov, K.G.; Artemyev, N.O. Loss of the effector function in a transducin-alpha mutant associated with Nougaret night blindness. J. Biol. Chem. 2000, 275, 6969–6974. [Google Scholar]
- Wuller, S.; Wiesner, B.; Loffler, A.; Furkert, J.; Krause, G.; Hermosilla, R.; Schaefer, M.; Schulein, R.; Rosenthal, W.; Oksche, A. Pharmacochaperones post-translationally enhance cell surface expression by increasing conformational stability of wild-type and mutant vasopressin V2 receptors. J. Biol. Chem. 2004, 279, 47254–47263. [Google Scholar]
- Small, K.M.; Wagoner, L.E.; Levin, A.M.; Kardia, S.L.; Liggett, S.B. Synergistic polymorphisms of beta1- and alpha2C-adrenergic receptors and the risk of congestive heart failure. N. Engl. J. Med. 2002, 347, 1135–1142. [Google Scholar]
- Na, G.Y.; Lee, K.H.; Kim, M.K.; Lee, S.J.; Kim, D.W.; Kim, J.C. Polymorphisms in the melanocortin-1 receptor (MC1R) and agouti signaling protein (ASIP) genes in Korean vitiligo patients. Pigment Cell Res. 2003, 16, 383–387. [Google Scholar]
- Liu, R.; Paxton, W.A.; Choe, S.; Ceradini, D.; Martin, S.R.; Horuk, R.; MacDonald, M.E.; Stuhlmann, H.; Koup, R.A.; Landau, N.R. Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell 1996, 86, 367–377. [Google Scholar]
- Marinissen, M.J.; Gutkind, J.S. G-protein-coupled receptors and signaling networks: emerging paradigms. Trends Pharmacol.Sci. 2001, 22, 368–376. [Google Scholar]
- Rozengurt, E. Early signals in the mitogenic response. Science 1986, 234, 161–166. [Google Scholar]
- Gutkind, J.S.; Novotny, E.A.; Brann, M.R.; Robbins, K.C. Muscarinic acetylcholine receptor subtypes as agonist-dependent oncogenes. Proc. Natl. Acad. Sci. USA 1991, 88, 4703–4707. [Google Scholar]
- Gutkind, J.S. Cell growth control by G protein-coupled receptors: From signal transduction to signal integration. Oncogene 1998, 17, 1331–1342. [Google Scholar]
- Cuttitta, F.; Carney, D.N.; Mulshine, J.; Moody, T.W.; Fedorko, J.; Fischler, A.; Minna, J.D. Bombesin-like peptides can function as autocrine growth factors in human small-cell lung cancer. Nature 1985, 316, 823–826. [Google Scholar]
- Cardona, C.; Rabbitts, P.H.; Spindel, E.R.; Ghatei, M.A.; Bleehen, N.M.; Bloom, S.R.; Reeve, J.G. Production of neuromedin B and neuromedin B gene expression in human lung tumor cell lines. Cancer Res. 1991, 51, 5205–5211. [Google Scholar]
- Namkoong, J.; Shin, S.S.; Lee, H.J.; Marin, Y.E.; Wall, B.A.; Goydos, J.S.; Chen, S. Metabotropic glutamate receptor 1 and glutamate signaling in human melanoma. Cancer Res. 2007, 67, 2298–2305. [Google Scholar]
- Hoogduijn, M.J.; Hitchcock, I.S.; Smit, N.P.; Gillbro, J.M.; Schallreuter, K.U.; Genever, P.G. Glutamate receptors on human melanocytes regulate the expression of MiTF. Pigment Cell Res. 2006, 19, 58–67. [Google Scholar]
- Young, D.; Waitches, G.; Birchmeier, C.; Fasano, O.; Wigler, M. Isolation and characterization of a new cellular oncogene encoding a protein with multiple potential transmembrane domains. Cell 1986, 45, 711–719. [Google Scholar]
- Julius, D.; Livelli, T.J.; Jessell, T.M.; Axel, R. Ectopic expression of the serotonin 1c receptor and the triggering of malignant transformation. Science 1989, 244, 1057–1062. [Google Scholar]
- Sodhi, A.; Montaner, S.; Patel, V.; Zohar, M.; Bais, C.; Mesri, E.A.; Gutkind, J.S. The Kaposi's sarcoma-associated herpes virus G protein-coupled receptor up-regulates vascular endothelial growth factor expression and secretion through mitogen-activated protein kinase and p38 pathways acting on hypoxia-inducible factor 1alpha. Cancer Res. 2000, 60, 4873–4880. [Google Scholar]
- Bais, C.; Santomasso, B.; Coso, O.; Arvanitakis, L.; Raaka, E.G.; Gutkind, J.S.; Asch, A.S.; Cesarman, E.; Gershengorn, M.C.; Mesri, E.A. G-protein-coupled receptor of Kaposi's sarcoma-associated herpesvirus is a viral oncogene and angiogenesis activator. Nature 1998, 391, 86–89. [Google Scholar]
- Montaner, S.; Sodhi, A.; Pece, S.; Mesri, E.A.; Gutkind, J.S. The Kaposi's sarcoma-associated herpesvirus G protein-coupled receptor promotes endothelial cell survival through the activation of Akt/protein kinase B. Cancer Res. 2001, 61, 2641–2648. [Google Scholar]
- Mazzuco, T.L.; Chabre, O.; Feige, J.J.; Thomas, M. Aberrant GPCR expression is a sufficient genetic event to trigger adrenocortical tumorigenesis. Mol. Cell Endocrinol. 2007, 265-266, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Mazzuco, T.L.; Chabre, O.; Sturm, N.; Feige, J.J.; Thomas, M. Ectopic expression of the gastric inhibitory polypeptide receptor gene is a sufficient genetic event to induce benign adrenocortical tumor in a xenotransplantation model. Endocrinology 2006, 147, 782–790. [Google Scholar]
- Sladeczek, F.; Pin, J.P.; Recasens, M.; Bockaert, J.; Weiss, S. Glutamate stimulates inositol phosphate formation in striatal neurones. Nature 1985, 317, 717–719. [Google Scholar]
- Tanabe, Y.; Masu, M.; Ishii, T.; Shigemoto, R.; Nakanishi, S. A family of metabotropic glutamate receptors. Neuron 1992, 8, 169–179. [Google Scholar]
- Masu, M.; Tanabe, Y.; Tsuchida, K.; Shigemoto, R.; Nakanishi, S. Sequence and expression of a metabotropic glutamate receptor. Nature 1991, 349, 760–765. [Google Scholar]
- Houamed, K.M.; Kuijper, J.L.; Gilbert, T.L.; Haldeman, B.A.; O'Hara, P.J.; Mulvihill, E.R.; Almers, W.; Hagen, F.S. Cloning, expression, and gene structure of a G protein-coupled glutamate receptor from rat brain. Science 1991, 252, 1318–1321. [Google Scholar] [PubMed]
- Conn, P.J.; Pin, J.-P. Pharmacology and functions of metabotropic glutamate receptors. Annu.Rev. Pharmacol. Toxicol. 1997, 37, 205–237. [Google Scholar]
- Hermans, E.; Challiss, R.A. Structural, signalling and regulatory properties of the group I metabotropic glutamate receptors: Prototypic family C G-protein-coupled receptors. Biochem J 2001, 359, 465–484. [Google Scholar]
- Goudet, C.; Magnaghi, V.; Landry, M.; Nagy, F.; Gereau, R.W.t.; Pin, J.P. Metabotropic receptors for glutamate and GABA in pain. Brain Res. Rev. 2009, 60, 43–56. [Google Scholar]
- Pin, J.P.; Kniazeff, J.; Goudet, C.; Bessis, A.S.; Liu, J.; Galvez, T.; Acher, F.; Rondard, P.; Prezeau, L. The activation mechanism of class-C G-protein coupled receptors. Biol. Cell 2004, 96, 335–342. [Google Scholar]
- Bradl, M.; Klein-Szanto, A.; Porter, S.; Mintz, B. Malignant melanoma in transgenic mice. Proc. Natl. Acad. Sci. USA 1991, 88, 164–168. [Google Scholar]
- Broome Powell, M.; Gause, P.R.; Hyman, P.; Gregus, J.; Lluria-Prevatt, M.; Nagle, R.; Bowden, G.T. Induction of melanoma in TPras transgenic mice. Carcinogenesis 1999, 20, 1747–1753. [Google Scholar]
- Chin, L.; Merlino, G.; DePinho, R.A. Malignant melanoma: Modern black plague and genetic black box. Genes. Dev. 1998, 12, 3467–3481. [Google Scholar]
- Iwamoto, T.; Takahashi, M.; Ito, M.; Hamatani, K.; Ohbayashi, M.; Wajjwalku, W.; Isobe, K.; Nakashima, I. Aberrant melanogenesis and melanocytic tumour development in transgenic mice that carry a metallothionein/ret fusion gene. EMBO J. 1991, 10, 3167–3175. [Google Scholar]
- Otsuka, T.; Takayama, H.; Sharp, R.; Celli, G.; LaRochelle, W.J.; Bottaro, D.P.; Ellmore, N.; Vieira, W.; Owens, J.W.; Anver, M.; Merlino, G. c-Met autocrine activation induces development of malignant melanoma and acquisition of the metastatic phenotype. Cancer Res. 1998, 58, 5157–5167. [Google Scholar]
- Sharpless, N.E.; Bardeesy, N.; Lee, K.H.; Carrasco, D.; Castrillon, D.H.; Aguirre, A.J.; Wu, E.A.; Horner, J.W.; DePinho, R.A. Loss of p16Ink4a with retention of p19Arf predisposes mice to tumorigenesis. Nature 2001, 413, 86–91. [Google Scholar]
- Krimpenfort, P.; Quon, K.C.; Mooi, W.J.; Loonstra, A.; Berns, A. Loss of p16Ink4a confers susceptibility to metastatic melanoma in mice. Nature 2001, 413, 83–86. [Google Scholar]
- Zhu, H.; Reuhl, K.; Zhang, X.; Botha, R.; Ryan, K.; Wei, J.; Chen, S. Development of heritable melanoma in transgenic mice. J. Invest. Dermatol. 1998, 110, 247–252. [Google Scholar]
- Colon-Teicher, L.; Wise, L.S.; Martino, J.J.; Baskin, L.; Sakoulas, G.; Pollack, R.; Chen, S. Genomic sequences capable of committing mouse and rat fibroblasts to adipogenesis. Nucl. Acid. Res. 1993, 21, 2223–2228. [Google Scholar]
- Chen, S.; Teicher, L.C.; Kazim, D.; Pollack, R.E.; Wise, L.S. Commitment of mouse fibroblasts to adipocyte differentiation by DNA transfection. Science 1989, 244, 582–585. [Google Scholar]
- Zhu, H.; Reuhl, K.; Botha, R.; Ryan, K.; Wei, J.; Chen, S. Development of early melanocytic lesions in transgenic mice predisposed to melanoma. Pigm.Cell Res. 2000, 13, 158–164. [Google Scholar]
- Trent, J.; Stanbridge, E.; McBride, H.; Meese, E.; Casey, G.; Araujo, D.; Witkowski, C.; Nagle, R. Tumorigenicity in human melanoma cell lines controlled by introduction of human chromosome 6. Science 1990, 247, 568–571. [Google Scholar]
- Thompson, F.H.; Emerson, J.; Olson, S.; Weinstein, R.; Leavitt, S.A.; Leong, S.P.; Emerson, S.; Trent, J.M.; Nelson, M.A.; Salmon, S.E.; et al. Cytogenetics of 158 patients with regional or disseminated melanoma. Subset analysis of near-diploid and simple karyotypes. Cancer Genet. Cytogenet. 1995, 83, 93–104. [Google Scholar] [PubMed]
- Ohtani, Y.; Harada, T.; Funasaka, Y.; Nakao, K.; Takahara, C.; Abdel-Daim, M.; Sakai, N.; Saito, N.; Nishigori, C.; Aiba, A. Metabotropic glutamate receptor subtype-1 is essential for in vivo growth of melanoma. Oncogene 2008, 27, 7162–7170. [Google Scholar]
- Funusaka, Y.; Harada, T.; Aiba, A.; Nishigori, C. Expression of metabotropic glutamate receptor 1 and phosphorylated extracellular signal-regulated kinase 1/2 proteins in human melanocytic lesions. Pigm. Cell Res. 2006, 19, 256. [Google Scholar]
- Marin, Y.E.; Namkoong, J.; Cohen-Solal, K.; Shin, S.S.; Martino, J.J.; Oka, M.; Chen, S. Stimulation of oncogenic metabotropic glutamate receptor 1 in melanoma cells activates ERK1/2 via PKCepsilon. Cell Signal 2006, 18, 1279–1286. [Google Scholar]
- Ishiuchi, S.; Tsuzuki, K.; Yoshida, Y.; Yamada, N.; Hagimura, N.; Okado, H.; Miwa, A.; Kurihara, H.; Nakazato, Y.; Tamura, M.; Sasaki, T.; Ozawa, S. Blockage of Ca(2+)-permeable AMPA receptors suppresses migration and induces apoptosis in human glioblastoma cells. Nat. Med. 2002, 8, 971–978. [Google Scholar]
- Rzeski, W.; Turski, L.; Ikonomidou, C. Glutamate antagonists limit tumor growth. Proc. Natl. Acad. Sci. USA 2001, 98, 6372–6377. [Google Scholar]
- Otsuka, T.; Takayama, H.; Sharp, R.; Celli, G.; LaRochelle, W.J.; Bottaro, D.P.; Ellmore, N.; Vieira, W.; Owens, J.W.; Anver, M.; Merlino, G. c-Met autocrine activation induces development of malignant melanoma and acquisition of the metastatic phenotype. Cancer Res. 1998, 58, 5157–5167. [Google Scholar]
- Bensimon, G.; Lacomblez, L.; Meininger, V. A controlled trial of riluzole in amyotrophic lateral sclerosis. ALS/Riluzole Study Group. N. Engl. J. Med. 1994, 330, 585–591. [Google Scholar] [PubMed]
- Van Den Bosch, L.; Van Damme, P.; Bogaert, E.; Robberecht, W. The role of excitotoxicity in the pathogenesis of amyotrophic lateral sclerosis. Biochim. Biophys. Acta 2006, 1762, 1068–1082. [Google Scholar] [PubMed]
- Bryson, H.M.; Fulton, B.; Benfield, P. Riluzole. A review of its pharmacodynamic and pharmacokinetic properties and therapeutic potential in amyotrophic lateral sclerosis. Drugs 1996, 52, 549–563. [Google Scholar] [PubMed]
- Doble, A. The pharmacology and mechanism of action of riluzole. Neurology 1996, 47, S233–S241. [Google Scholar] [PubMed]
- Kretschmer, B.D.; Kratzer, U.; Schmidt, W.J. Riluzole, a glutamate release inhibitor, and motor behavior. Naunyn Schmiedebergs Arch Pharmacol 1998, 358, 181–190. [Google Scholar] [PubMed]
- Wang, S.J.; Wang, K.Y.; Wang, W.C. Mechanisms underlying the riluzole inhibition of glutamate release from rat cerebral cortex nerve terminals (synaptosomes). Neuroscience 2004, 125, 191–201. [Google Scholar]
- Yip, D.; Le, M.N.; Chan, J.L.; Lee, J.H.; Mehnert, J.A.; Yudd, A.; Kempf, J.; Shih, W.J.; Chen, S.; Goydos, J.S. A phase 0 trial of riluzole in patients with resectable stage III and IV melanoma. Clin. Cancer Res. 2009, 15, 3896–3902. [Google Scholar]
- Wilhelm, S.M.; Carter, C.; Tang, L.; Wilkie, D.; McNabola, A.; Rong, H.; Chen, C.; Zhang, X.; Vincent, P.; McHugh, M.; Cao, Y.; Shujath, J.; Gawlak, S.; Eveleigh, D.; Rowley, B.; Liu, L.; Adnane, L.; Lynch, M.; Auclair, D.; Taylor, I.; Gedrich, R.; Voznesensky, A.; Riedl, B.; Post, L.E.; Bollag, G.; Trail, P.A. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004, 64, 7099–7109. [Google Scholar]
- Lee, H.; Wall, B.; Wangari-Talbot, J.; Shin, S.; Rosenberg, S.; Chan, J.-K.; Namkoong, J.; Goydos, J.; Chen, S. Targeting Metabotropic Glutamate Signaling in Melanoma; Single Agent and Combinatorial Therapies. 2010; in review. [Google Scholar]
- Kalariti, N.; Pissimissis, N.; Koutsilieris, M. The glutamatergic system outside the CNS and in cancer biology. Expert. Opin. Investig. Drugs 2005, 14, 1487–1496. [Google Scholar]
- Aronica, E.; Yankaya, B.; Jansen, G.H.; Leenstra, S.; van Veelen, C.W.; Gorter, J.A.; Troost, D. Ionotropic and metabotropic glutamate receptor protein expression in glioneuronal tumours from patients with intractable epilepsy. Neuropathol.Appl. Neurobiol. 2001, 27, 223–237. [Google Scholar]
- Naarala, J.; Nykvist, P.; Tuomala, M.; Savolainen, K. Excitatory amino acid-induced slow biphasic responses of free intracellular calcium in human neuroblastoma cells. FEBS Lett. 1993, 330, 222–226. [Google Scholar]
- Stepulak, A.; Luksch, H.; Gebhardt, C.; Uckermann, O.; Marzahn, J.; Sifringer, M.; Rzeski, W.; Staufner, C.; Brocke, K.S.; Turski, L.; Ikonomidou, C. Expression of glutamate receptor subunits in human cancers. Histochem. Cell Biol. 2009, 132, 435–445. [Google Scholar]
- Iacovelli, L.; Arcella, A.; Battaglia, G.; Pazzaglia, S.; Aronica, E.; Spinsanti, P.; Caruso, A.; De Smaele, E.; Saran, A.; Gulino, A.; D'Onofrio, M.; Giangaspero, F.; Nicoletti, F. Pharmacological activation of mGlu4 metabotropic glutamate receptors inhibits the growth of medulloblastomas. J. Neurosci. 2006, 26, 8388–8397. [Google Scholar]
- de Groot, J.F.; Piao, Y.; Lu, L.; Fuller, G.N.; Yung, W.K. Knockdown of GluR1 expression by RNA interference inhibits glioma proliferation. J. Neurooncol. 2008, 88, 121–133. [Google Scholar]
- Chang, H.J.; Yoo, B.C.; Lim, S.B.; Jeong, S.Y.; Kim, W.H.; Park, J.G. Metabotropic glutamate receptor 4 expression in colorectal carcinoma and its prognostic significance. Clin. Cancer Res. 2005, 11, 3288–3295. [Google Scholar]
- Yoo, B.C.; Jeon, E.; Hong, S.H.; Shin, Y.K.; Chang, H.J.; Park, J.G. Metabotropic glutamate receptor 4-mediated 5-Fluorouracil resistance in a human colon cancer cell line. Clin. Cancer Res. 2004, 10, 4176–4184. [Google Scholar]
- Park, S.Y.; Lee, S.A.; Han, I.H.; Yoo, B.C.; Lee, S.H.; Park, J.Y.; Cha, I.H.; Kim, J.; Choi, S.W. Clinical significance of metabotropic glutamate receptor 5 expression in oral squamous cell carcinoma. Oncol. Rep. 2007, 17, 81–87. [Google Scholar] [Green Version]
- Liu, J.W.; Kim, M.S.; Nagpal, J.; Yamashita, K.; Poeta, L.; Chang, X.; Lee, J.; Park, H.L.; Jeronimo, C.; Westra, W.H.; Mori, M.; Moon, C.; Trink, B.; Sidransky, D. Quantitative hypermethylation of NMDAR2B in human gastric cancer. Int. J. Cancer 2007, 121, 1994–2000. [Google Scholar]
- Abdul, M.; Hoosein, N. N-methyl-D-aspartate receptor in human prostate cancer. J. Membr. Biol. 2005, 205, 125–128. [Google Scholar]
- Luikart, S.D.; Kennealey, G.T.; Kirkwood, J.M. Randomized phase III trial of vinblastine, bleomycin, and cis-dichlorodiammine-platinum versus dacarbazine in malignant melanoma. J. Clin. Oncol. 1984, 2, 164–168. [Google Scholar] [PubMed]
- Huncharek, M.; Caubet, J.F.; McGarry, R. Single-agent DTIC versus combination chemotherapy with or without immunotherapy in metastatic melanoma: a meta-analysis of 3273 patients from 20 randomized trials. Melanoma. Res. 2001, 11, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, J.M.; Strawderman, M.H.; Ernstoff, M.S.; Smith, T.J.; Borden, E.C.; Blum, R.H. Interferon alfa-2b adjuvant therapy of high-risk resected cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J. Clin. Oncol. 1996, 14, 7–17. [Google Scholar]
- Kirkwood, J.M.; Ibrahim, J.G.; Sosman, J.A.; Sondak, V.K.; Agarwala, S.S.; Ernstoff, M.S.; Rao, U. High-dose interferon alfa-2b significantly prolongs relapse-free and overall survival compared with the GM2-KLH/QS-21 vaccine in patients with resected stage IIB-III melanoma: results of intergroup trial E1694/S9512/C509801. J. Clin. Oncol. 2001, 19, 2370–2380. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wangari-Talbot, J.; Goydos, J.; Chen, S. Role of the G Protein-Coupled Receptor, mGlu1, in Melanoma Development. Pharmaceuticals 2010, 3, 2821-2837. https://doi.org/10.3390/ph3092821
Wangari-Talbot J, Goydos J, Chen S. Role of the G Protein-Coupled Receptor, mGlu1, in Melanoma Development. Pharmaceuticals. 2010; 3(9):2821-2837. https://doi.org/10.3390/ph3092821
Chicago/Turabian StyleWangari-Talbot, Janet, James Goydos, and Suzie Chen. 2010. "Role of the G Protein-Coupled Receptor, mGlu1, in Melanoma Development" Pharmaceuticals 3, no. 9: 2821-2837. https://doi.org/10.3390/ph3092821
APA StyleWangari-Talbot, J., Goydos, J., & Chen, S. (2010). Role of the G Protein-Coupled Receptor, mGlu1, in Melanoma Development. Pharmaceuticals, 3(9), 2821-2837. https://doi.org/10.3390/ph3092821



